Abstract
Background:
The aim of this study was to test the ability of a low-pressure, low-flow, heliox-based rebreathing system to reduce work of breathing and conserve gas while preserving CO2 concentration, temperature, and humidity at physiological levels in a bench study.
Methods:
We performed a bench study of a novel low-pressure, low-flow, noninvasive heliox rebreathing system with CO2 scrubber that was connected to an artificial lung simulator with careful monitoring of flow, pressure, work of breathing, O2, CO2, temperature, and humidity levels. Multiple runs of breathing were performed while manipulating levels of resistance (5–30 cm H2O/L/s), gas mixtures (room air, 79% helium [He] and 21% O2, and 70% He and 30% O2), and leak levels (ultralow, low, and high).
Results:
We found significant reductions in work of breathing (up to 64%) while conserving gas with estimates of up to 10- to 54-fold reduction in medical gas wastage (P < .001). Specifically, at resistances of 5, 10, 20, and 30 cm H2O/L/s, we demonstrated 64, 57, 36, and 7% reduction in work of breathing (P < .001). Gas wastage was reduced by 10- to 54-fold while the end-tidal CO2 concentration, humidity, and temperature were maintained by the device at physiological levels.
Conclusions:
In a bench test, a low-pressure, low-flow, noninvasive heliox rebreathing system with CO2 scrubber reduced work of breathing and conserved gas while preserving CO2 concentration, temperature, and humidity at physiological levels in a lung model. Future studies in human subjects need to be performed to determine whether reduction of work of breathing and gas conservation can be achieved.
Introduction
Heliox has been used alone or in combination with noninvasive ventilation (NIV) for the treatment of acute respiratory failure due to asthma exacerbation, upper-airway obstruction, bronchiolitis, and COPD. 1 –5 One of the major barriers for the utilization of heliox gas in the clinical setting is the cumbersome nature of administering the gas. 6 Specifically, the need for transferring large tanks into the ICU rooms and storage needs are both labor intensive and time consuming for the respiratory therapist. Additionally, the respiratory therapists need to transport new tanks to replenish the supply every 8–12 h in the ICU from the storage area. Moreover, helium (He) is a natural resource, and gas wastage due to leaks incurred by using a simple non–rebreather or air-entrainment mask does not capture the exhaled or leaked gas and thereby increases costs and wastes a finite resource. 7 A system that uses a rebreathing circuit and low-pressure, low-flow system can reduce the magnitude of air leak and also helps improve tolerability of the ventilatory support by avoiding a high-flow system that uses a conventional loose-fitting face mask. There is a knowledge gap, however, as to whether such a rebreather system (CO2 scrubber and rebreathing circuit) with heliox and low-pressure, low-flow state can reduce work of breathing and conserve gas.
To address the aforementioned knowledge gap, we devised and bench tested a prototype device of a heliox rebreathing system with CO2 scrubber that conserves gas and assessed the impact on work of breathing with an artificial lung system. Specifically, our aims were to test the ability of a low-pressure, low-flow, heliox-based rebreathing system to (1) reduce work of breathing (primary aim); and (2) conserve gas while preserving CO2 concentration, temperature, and humidity at physiological levels (secondary aim) in a bench study. If our aims are achieved, and testing in simulated airway obstruction is successfully performed to demonstrate efficacy, 2 then the device can be further tested and made available in various clinical applications.
QUICK LOOK
Current knowledge
Current methods of administering heliox in clinical settings suffer from barriers to delivery such as large and difficult-to-transport tanks needed due to a high-flow delivery system that is associated with gas wastage. Gas waste can be reduced by rebreathing systems, but such a rebreathing system with CO2 scrubbers can create an exothermic reaction with heat and be adversely affected by moisture.
What this paper contributes to our knowledge
In a laboratory bench evaluation using an artificial lung simulator, a novel low-pressure, low-flow, noninvasive heliox rebreathing system with CO2 scrubber that reduced work of breathing and conserved gas while preserving CO2 concentration, temperature, and humidity at physiological levels was tested. The system conserved gas by reducing waste while preventing
Methods
A bench study experimental setup is shown in the schematic diagram in Figure 1. The device was tested with the mask interface attached to a manikin head with realistic upper-airway structure. The dead space of the manikin airway is 150 mL with an airway resistance is 2.4 cm H2O/L/s. The furthest end of the manikin’s tracheal airway was connected to a lung simulator (ASL 5000, IngMar Medical, Pittsburgh, Pennsylvania) that was programmed to simulate spontaneous breathing with various additional resistances. 8,9 Measurements of flow (V̇ in Fig. 1) (heated pneumotachograph, Fleisch, Lausanne, Switzerland) and airway pressure (Paw in Fig. 1) (Validyne Engineering, Canoga Park, California) were made at the corresponding locations between the manikin mask interface and the device. 8,10,11 Data were collected and stored on a laptop after analog to digital conversion and signal conditioning (WinDaq, DATAQ Instruments, Akron, Ohio) for subsequent review and analysis. An adjustable leak aperture (L in Fig. 1) was placed between the mask interface and the device to facilitate known levels of leak. 8 CO2 was entrained at the location (CO2 in Fig. 1) indicated between the lung simulator and manikin head as indicated. Temperature was measured at 2 locations: A temperature probe was placed at a side port near the mask (T in Fig. 1), and an additional temperature probe was placed downstream to the CO2 scrubber (T’ in Fig. 1).
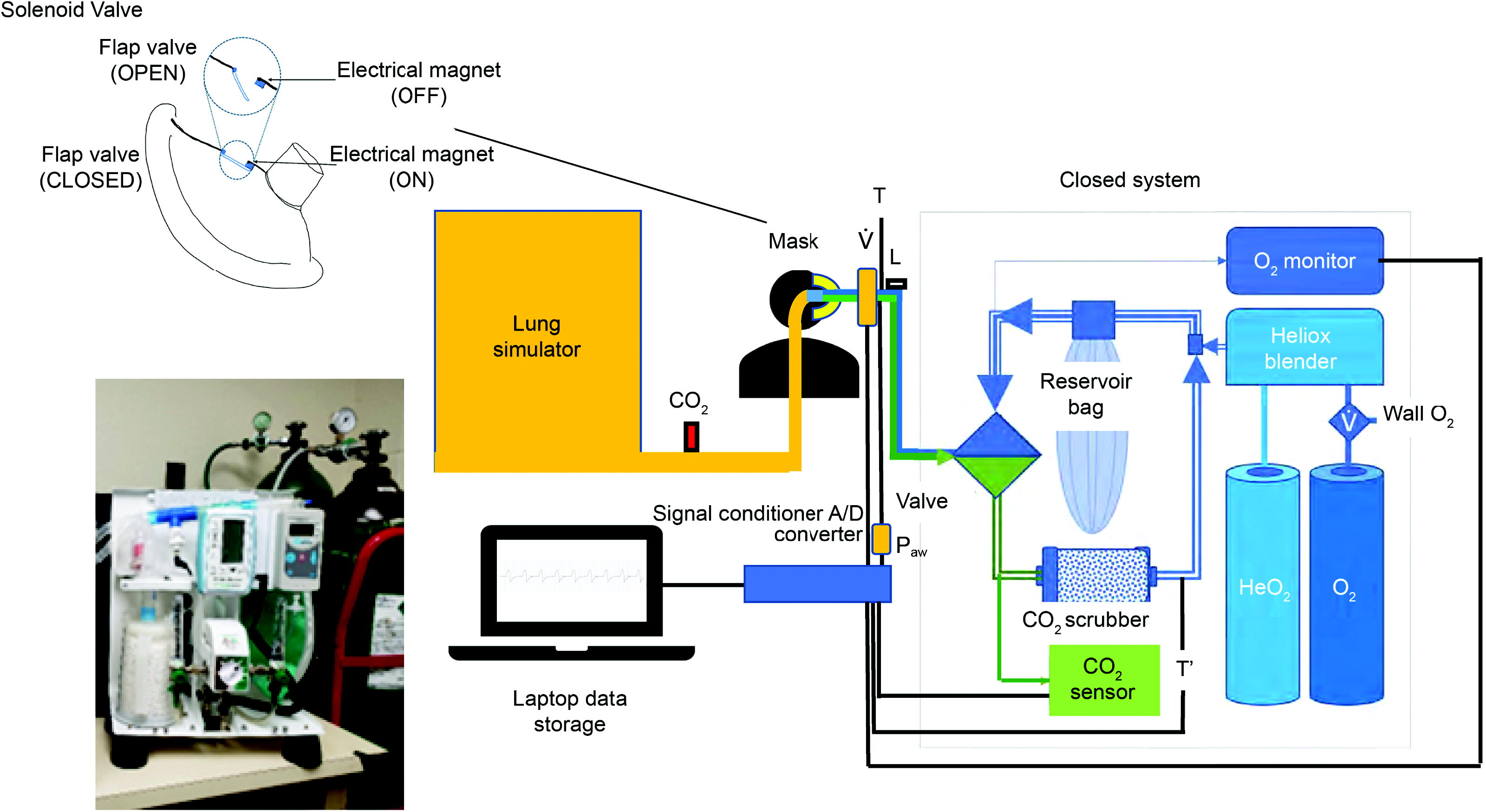
The bench setup of the device, mask interface, and the manikin head with realistic upper-airway structure is shown. The manikin’s tracheal airway was connected to a lung simulator programmed to simulate spontaneous breathing with various additional resistances. Measurements of flow (V̇), airway pressure (Paw), temperature near the mask (T) and downstream to the CO2 scrubber (T’), CO2, and O2 levels were made, and the data were collected and stored on a laptop after analog-to-digital conversion (A/D converter) for analysis. An adjustable leak aperture (L) was placed between the mask interface and the device to facilitate known levels of leak. CO2 was entrained at the location (CO2) indicated between the lung simulator and manikin head as indicated. The device involved a one-way valve, reservoir bag, CO2 scrubber, heliox blender, and medical gas sources (heliox; at 80% He and 20% O2 and 100% O2) as well as a wall source for O2 at 100%. The upper-left inset reveals the mask with a solenoid valve that would automatically open if there were an electrical failure due to demagnetization of an electrical magnet. An image of the compact device without the shell cover is shown in the lower-bottom inset.
The device configuration is shown in Figure 1. The gas source was a tank containing mixed gas (80% He and 20% O2) and another tank with 100% O2. The device has an option for a wall source for 100% O2 in addition to the tank. A heliox blender enabled appropriate admixture of gases to the desired He:O2 concentration levels. A closed circuit with CO2 scrubber and monitoring devices for CO2 and O2 concentration was included (capnograph, Nonin Medical, Plymouth, Minnesota). A reservoir bag enabled appropriate gas inspiratory flow and thereby prevented any situation during which a negative airway pressure—that can increase work of breathing—would be created. A one-way valve enabled directionality of the flow as indicated in Figure 1.
Prevention of asphyxiation
To prevent asphyxiation, there are 3 features. We incorporated both an O2 sensor (blue box on the inspiratory limb) and CO2 sensor (green box on the expiratory limb) with preset limits that would alarm if either or both tanks (HeO2 or O2 tanks) were to become empty or if the CO2 scrubber were to malfunction (Fig. 1). Second, both tanks that serve as gas source contain O2 (100% O2 and an admixture of 80% He and 20% O2) (Fig. 1). Such a dual tank system is aimed at reducing chance for asphyxiation if either one of the tanks runs out of gas. Third, a solenoid valve that is on the face mask is shown in the inset, which, in the event of a system electrical failure, will immediately open a trapdoor on the full face mask (due to loss of electricity) and allow the patient to breathe room air and thereby prevent asphyxiation.
Work of breathing
During the simulation runs, the airway resistance was varied between 4 different levels—5, 10, 20, or 30 cm H2O/L/s. At each resistance level, the muscle pressure was set on the lung simulator to achieve a tidal volume of 500 mL for each run. In addition to varying the resistance level, there were 3 different gas admixtures of He:O2 concentrations that were delivered: 0:21 (room air), 79:21 heliox, and 70:30 heliox. In all, 4 runs were performed for 3 min each at each resistance and gas admixture level for a total of 48 runs. The rationale for the 3-min runs was based upon our prior study in which we found that 3-min runs of stable conditions in a bench testing model demonstrated reliable results and allowed stable and representative data of work of breathing and corresponding findings. 11 The inspiratory and expiratory work of breathing generated by the lung simulator device was recorded at the end of each 3-min run.
Gas leak and gas conservation
Multiple simulation runs (no. = 3) were performed at different leak levels at the low-pressure state (mean airway pressure of 0.5 cm H2O and range of 0–2 cm H2O)—ultralow (mean ± SD, 0.53 ± 0.35 L/min), low (0.85 ± 0.14 L/min), and high (1.30 ± 0.02 L/min)—set by an adjustable aperture to mimic real-world air leak from the face-mask interface with a diameter range of 10.7-, 12.0-, and 13.4-mm range, respectively. 12 Such apertures at a conventional (higher) airway pressure setting of 15 cm H2O would have resulted in air leaks of 16, 25, and 40 L/min, respectively. Considering that exhaled gas rich in oxygen is recirculated through the device CO2 scrubber, the FIO2 was maintained with very little loss of gas outside the rebreather system. A continuous background flow of gas of ∼1–2 L/min as measured during the expiratory phase constituted the low-flow state. When the lung simulator switches to inspiratory cycle, a sinusoidal inspiratory flow with a peak of 60 L/min was programmed into the lung simulator to mimic the human condition. In contrast, the flow during exhalation for NIV ranges much higher (∼40 L/min) for a full face mask with bleeder valves at a pressure setting of 15 cm H2O. 13 Three runs for 8 h each were performed at each leak level (ultralow, low, or high) and for each gas admixture concentration of heliox (70:30 and 79:21) for a total of 18 runs.
CO2 elimination
CO2 gas was entrained at 2.0–2.5 L/min between the lung simulator and the manikin to simulate CO2 gas production and to artificially raise the end-tidal CO2 concentration in the breathing circuit to 46 mm Hg (which is higher than normal range of 38–42 mm Hg). Subsequently, an 8-h run was performed to assess whether the exhaled CO2 levels were normalized and maintained at normal levels. This run was repeated 3 times at a He:O2 blend of 70/30 and ultralow leak level.
Humidity
An in-line humidifier and de-humidifier were introduced in the inspiratory and expiratory limbs of the device, respectively, to humidify and de-humidify the gas within the circuit at an He:O2 blend of 70/30 and ultralow leak level. The exhaled humidity levels when compared to the ambient humidity levels in the laboratory were measured and reported over 8-h period (3 runs).
Comparisons for work of breathing across various resistance levels and medical gas admixtures were performed using 2-way analysis of variance (ANOVA) with repeated measures (SPSS, IBM, Armonk, New York). Means and SDs are reported unless otherwise specified. Coefficient of variation was measured as the proportion of SD of mean values and expressed as percentage. All values are reported as statistically significant if P values are < .05.
Results
Work of breathing
Work of breathing was consistently reduced at both heliox concentrations and is shown in Table 1 and Figure 2. The work of breathing was reduced by 64, 57, 36, and 7% at resistances of 5, 10, 20, and 30 cm H2O/L/s, respectively, (P < .001, 2-way ANOVA with repeated measures). There was negligible difference in reduction of work of breathing between 70/30 and 79/21 heliox gas mixtures. There were very small differences in variability across the various runs, and hence the SD bars are not seen in Figure 2. The coefficient of variation was < 0.02%.
Effect of heliox on work of breathing
Data are presented as mean ± SD.
Work of breathing was reduced to various extent by the heliox gas admixtures and at various settings of resistance.
He, helium.
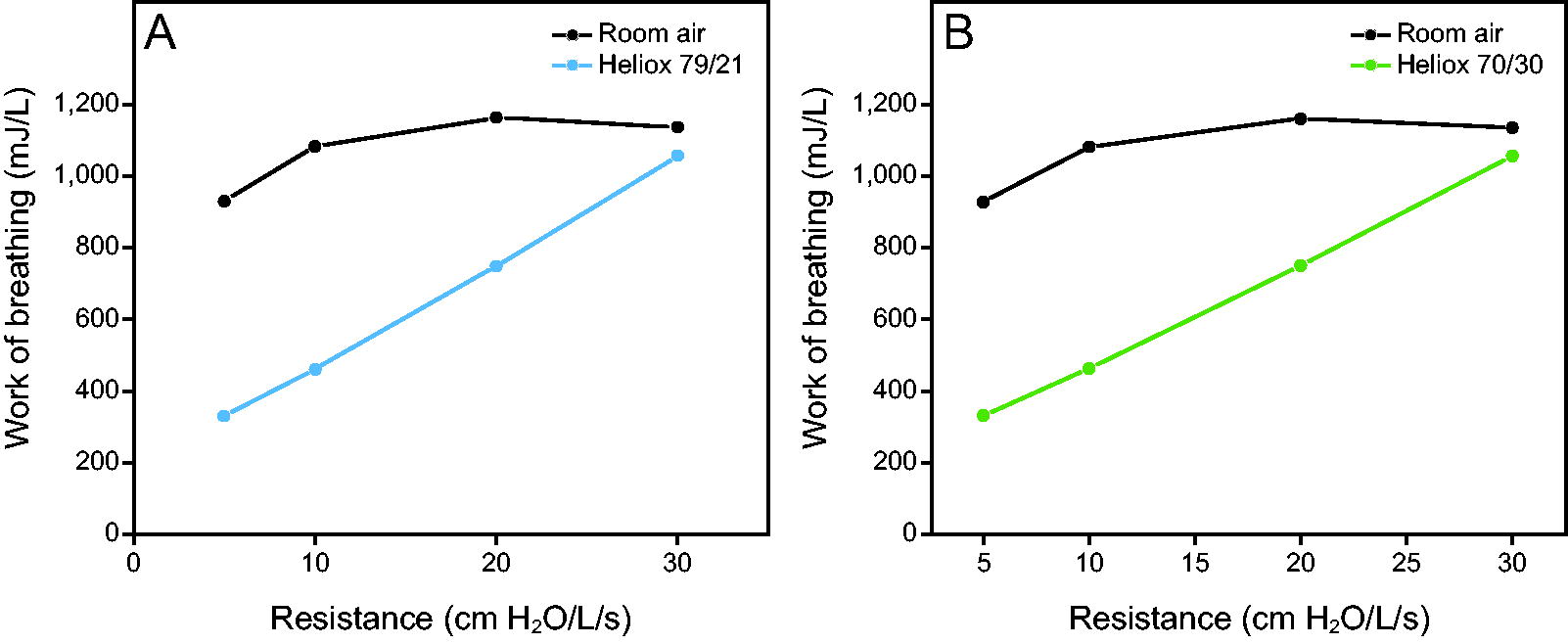
Work of breathing is plotted for various levels of resistance for 79%:21% helium (He):O2 (heliox 79/21; A) and 70%:30% He:O2 (heliox 70/30, B). There is significant reduction in work of breathing that ranges from 7–64%. The SDs across multiple runs are not apparent as the coefficient of variance (proportion of SD of the mean values) is < 0.02%.
Gas leak and conservation
Multiple simulation runs (no. = 3) were performed at different leak levels, and the remaining time that was left in the tank at a certain flow (gas leak rate) was estimated for an M-tank (3,200 L) using the remaining pressure (pressure gauge) (Table 2). The gas loss was then used against the lifespan of an M-tank without the conservation system at 6 L/min to provide meaningful estimates of fold increase in tank lifespan (Table 2). Depending on leak levels and heliox concentration chosen, the tank would last from 5–27 d. A fold change for increase in time based upon such an assumption is provided in Table 2 for each heliox admixture level and leak level. All fold changes were statistically significant (P < .001).
Estimations of tank lifespan at various leak levels
Assumptions are based upon a standard M-tank lasts for 0.5 d in a hospital setting without the rebreathing system. Ultralow (mean ± SD, 0.53 ± 0.35 L/min), low (0.85 ± 0.14 L/min), and high (1.3 ± 0.02 L/min) were preset by adjusting the leak aperture in the circuit.
CO2 elimination, temperature, and humidity
Considering that the system assumes adequate CO2 elimination and that the CO2 scrubber eliminates CO2 through an exothermic reaction, the end-tidal CO2 levels and temperature levels were also measured at the level of the mask. An additional temperature probe was placed near the CO2 scrubber. After entraining 3% CO2 gas, over an 8-h run, following an initially high CO2 level there was an immediate and persistent normalization of the CO2 levels in Figure 3. Moreover, the mask temperature (Fig. 3) remained constant whereas the temperature at the CO2 scrubber was mildly elevated and remained elevated over an 8-h period (P = .01). The humidity levels remained normal between 43–47% (laboratory humidity level was 38–42%).
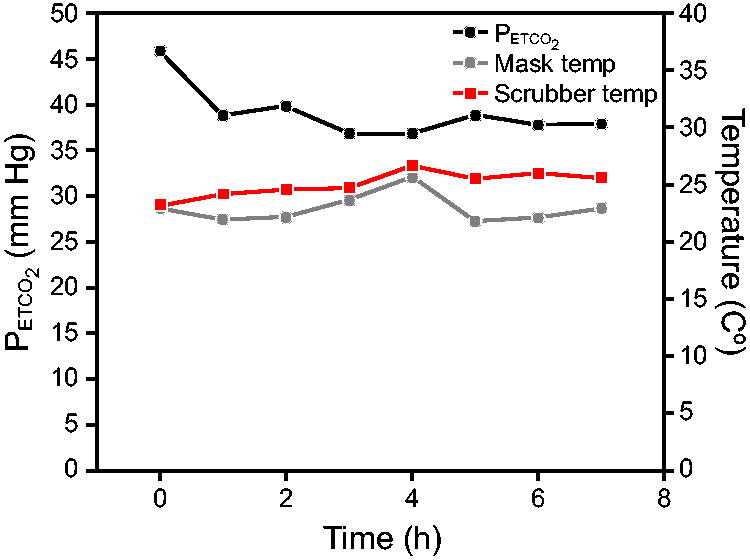
Breathing runs that lasted for 8 h demonstrate that the device was able to maintain CO2 and temperature levels at the site of the mask at normal physiological levels. Measurement of temperature distal to the CO2 scrubber is also shown revealing the exothermic reaction of CO2 removal. PETCO2 , end-tidal CO2 pressure.
Discussion
In a bench testing experiment, we have demonstrated that a novel rebreather system (CO2 scrubber and rebreathing circuit) with heliox and a low-pressure, low-flow state can reduce work of breathing. We have also shown that the system can significantly conserve gas by reducing waste while preventing hypercapnia or adverse alterations in airway temperature and humidity levels.
Our system provides a novel approach at providing low-pressure, low-flow system that can alleviate the work of breathing due to the reduced density of the gas. We report our system as a low-flow system in comparison to the high-flow nasal oxygen systems that are currently used in patients with acute respiratory failure. 14 Novel systems that do not use high-flow or high-pressure systems for reducing risk for intubation and mechanical ventilation when compared to conventional O2 therapy are needed considering the morbidity associated with invasive mechanical ventilation. However, human studies aimed at demonstrating the ability of our device to reduce work of breathing need to be performed.
Our gas conservation approach prolonged the lifespan of an M-size tank significantly and reduced wasteful exhalation of medical gases (He and O2) (see Table 2). Such an approach aimed at conserving heliox can save time for the busy respiratory therapists and also enable smaller tanks that can facilitate ease of transport, storage, and occupy less space in the ICU room. Moreover, when used purely with 100% O2 alone, and without any heliox, our proposed system can significantly conserve precious oxygen supplies, although it may not reduce work of breathing due to lack of high airway pressure assistance or low-density gas (heliox). Efforts need to be undertaken to bring such simple technology to a strained infrastructure with lifesaving implications in both high- and low-resource countries. 15
Bench tests of our device demonstrated that our gas conservation approach with the heliox rebreathing system with CO2 scrubber would prolong the life of each tank to 27 d when compared to the life of a full tank of conventional heliox systems of just 12 h (half a day) (Table 2). This is a 54-fold improvement in gas conservation that can, in turn, facilitate implementation of this intervention in both the hospital and possibly, in the future, even in the home setting for stable severe COPD with huge cost savings (Table 2). The gas cost of our proposed system is only $650–1,680/y if this device were used only nocturnally for 8 h in hospitalized patients.
Conclusions
In summary, we performed a bench test of a low-pressure, low-flow, noninvasive heliox rebreathing system with CO2 scrubber that reduced work of breathing and conserved gas while preserving CO2 concentration, temperature, and humidity at physiological levels in a bench model. This low-pressure, low-flow, system needs further testing in humans.
Footnotes
Authors' Contributions
Dr. Parthasarathy has a patent for a home-based breathing system (US20160213879A1) that has been licensed by SaiOx. Mr. Morton has disclosed no conflicts of interest.
Author Disclosure Statement
Study was performed at University of Arizona, Tucson, Arizona.
Funding Information
This study was funded by Tech Launch Arizona of the University of Arizona, Tucson, Arizona. During the writing of this manuscript, Dr. Parthasarathy was also supported by R01-AG080609, 5R01-AG065346, R33-HL151254, R21HD109777, OT2HL161847, OT2HL156812, C06OD028307, OT2HL158287, and R25-HL126140 from National Institutes of Health and PCORI-CER-2018C2-13262 from Patient-Centered Outcomes Research Institute.
