Abstract
Objective
Aim of the study was to analyze changes over a ten years observation period regarding demography, indication for support and clinical outcome in patients treated with short-term mechanical circulatory support (MCS) for cardiogenic failure.
Methods
39 patients treated with MCS between 2006–2008 (2008 group) were analyzed, assessing demography, complication rates and survival. Results were compared with 36 consecutive patients that had received centrifugal MCS between 1996–1998 (1998 group) at our institution.
Results
Mean age was 59.9 ± 12.9 (1998 group) and 60.9 ± 13.9 years (P = 0.74). Mean logEuroScores rose from 12.0% ± 14.6% (1998 group) to 26.9% ± 20.5% (2008 group); P < 0.001. Postcardiotomy low output syndrome was the main cause for MCS. However the percentage of patients in cardiogenic shock prior to surgery increased from 19.4% (1998 group) to 33.3% (2008 group); P = 0.17. Complexity and urgency of the primary surgical procedure increased significantly. 16.7% (1998 group) vs. 41.0% (2008 group); P = 0.02 of interventions were classified “salvage/emergent”. Mean duration of support was 2.9 ± 1.9 days (1998 group) and 3.8 ± 3.1 days (2008 group); P = 0.14. Significantly more biventricular support was implemented in the 2008 group (23.1% vs. 5.6% in the 1998 group); P = 0.03. The incidence of complications, including device failure, thromboembolism and infection remained the same in both groups. 63.9% (1998 group) and 61.5% (2008 group) of the patients were successfully weaned from the device (P = 0.83), 12% (1998 group) and 3% (2008 group) of the VAD patients were bridged to long-term VAD (P = 0.12). Overall 30-day survival rates were similar (22.2% 1998 group vs. 28.2%; 2008 group); P = 0.55, however, survival rate in BVAD supported patients improved significantly.
Conclusion
These data demonstrate the beneficial effect of MCS to salvage patients with cardiac failure. Taking into consideration that the severity of illness, the complexity and urgency of the primary surgical procedure have steadily increased a comparable improvement in MCS outcome over the past decade was observed. Nevertheless, in-hospital mortality and VAD related complication rates such as bleeding remain high.
Introduction
The two main indications that lead to the need of short-term mechanical circulatory support (MCS) are acute cardiogenic shock due to various causes (eg, myocardial infarction, myocarditis etc.) or postcardiotomy failure after open heart surgery. Postcardiotomy failure is reported with an incidence of up to 6% after cardiac surgical procedures.1,2 About 5% to 25% of these patients are refractory to conventional treatments of high dose inotropics and intraaortic balloon pump (IABP) counterpulsation and require additional mechanical circulatory VAD support. 2 Today, short-term mechanical circulatory support is usually performed using centrifugal blood pumps. Despite this treatment, the outcome of this patient population has been persistently poor.
Over the past decade, there have been significant advances in pump technology, surgical experience as well as in anesthesia and critical care. Our main objectives were to characterize 1) trends in the use of centrifugal VADs, 2) changes in the clinical characteristics and demography of patients, and 3) outcome in patients treated with short-term mechanical circulatory support between a ten year period at our institution.
Methods
Study design
All patients (n = 39) that received short-term centrifugal mechanical support at our institution between 2006–2008 (2008 group) were analyzed in this study. Clinical outcome and complication rates were assessed using a common clinical protocol. To evaluate changes in centrifugal MCS outcome that might have occurred in the past decade, all consecutive patients treated with MCS between 1996 and 1998 (1998 group) at our institution (n = 36) were also analyzed by retrospective chart review. Data of both groups were compared as described in detail below.
In all patients, intention of support was “bridge-to-recovery”. Mechanical assisted circulation was implemented in case of low cardiac output syndrome (cardiac index CI < 2.0 l/min/m2) and/or the impossibility to wean from cardiopulmonary bypass despite high dose inotropic- and IABP support. Devices used were the Capiox® (Terumo Cardiovascular Systems, Ann Arbor, Michigan, USA) and Bio-medicus® (Medtronic, Minneapolis, Minnesota, USA) centrifugal blood pumps. In the 2008 group, the majority of patients (59%) was supported utilizing the magnetically levitated Centrimag® (Levitronix, Waltham, Massachusetts, USA) centrifugal blood pump.
Assessment of Preoperative Symptom Status and co-Morbid Risk factors
Several important variables including ie, age, extracardiac arteriopathy, renal function, and left ventricular ejection fraction were analyzed to assess the preoperative symptom status and co-morbid risk factors of the patients. Each of these risk factors were taken together in the European system for cardiac operative risk evaluation score (EuroSCORE) method. 3 EuroSCORE stands for European System for Cardiac Operative Risk Evaluation. It is a method of predicting the mortality during or shortly after undergoing open heart surgery. This method allows the logistic calculation of risk for open heart surgery. The log EuroScore has been shown to be a valuable measure for prediction of immediate postoperative death after adult cardiac surgery.4–6
Assessment of Operative mortality
The primary outcome was operative mortality. Operative mortality was defined according to the definition of the Joint EACTS-STS Congenital Database Committee as any death, regardless of cause occurring 1) within 30 days after surgery in or out of the hospital, and 2) after 30 days during the same hospitalization subsequent to the operation. 7 Patient status (dead or alive) was determined through the examination of medical records.
Assessment of Vad-Related Postoperative events
The three major complications that are associated with mechanical assisted circulation (bleeding, thromboembolism, and infection) were examined.
VAD-related bleeding complications were evaluated through the documented usage of fresh frozen plasma (FFP), units of packed red blood cell concentrates (RBC), and thrombocyte concentrates (PC) during VAD support and up to three days after pump explantation.
Thromboembolic complications were any documented persistent central neurological deficit >72 hours or peripheral embolization under MCS and up to three days after VAD explantation. All clinical records and microbiological results were reviewed to assess any infection.
Statistical analysis
Binary and nominal factors were expressed as the percentage of patients and compared by using the Chi-square test. For categorical variables, results were additionally crosschecked by using Fisher's exact test. Variables recorded on a continuous scale were analyzed by using the Student's t-test and in addition by the Wilcoxon rank-sum test. The values are expressed as mean ± standard deviation. Survival data were analyzed by using the Kaplan-Meier method and compared by using log-rank statistics.
Predictors of mortality were analyzed for pooled data by using crosstabs and the Odds ratio was computed. Results were compared by using the Chi-square test and crosschecked by using Fisher's exact test. A P-value of <0.05 was considered statistically significant. All data were analyzed by using the SPSS software version 11.0.4 (SPSS Inc, Chic, IL, USA).
Results
Demography
Mean age and sex was almost identical in both groups (59.9 ± 12.9 years [range 21.4–75.1 years] in the 1998 group vs. 60.9 ± 13.9 years [range 24.8–75.6 years] in the 2008 group; P = 0.74). 75% of patients were male in the 1998 group vs. 72% in the 2008 group (P = 0.75).
Operative Characteristics and procedures
The mean logistic EuroScore prior to surgery increased significantly from 12.2% in the 1998 group to 26.9% in the 2008 group (P < 0.001). Figure 1 illustrates the classification of the primary surgical procedure according to the following definitions: emergent/salvage procedure, a procedure performed in the setting of acute ischemia, infarction, or hemodynamic compromise; urgent procedure, a procedure judged by the attending cardiac surgeon to be required within 24 hours from presentation; elective surgery, a planned, non-emergency surgical procedure. Figure 2 illustrates the performed primary surgical procedures in the two groups. In these procedures centrifugal VAD implantation was always “unplanned”.
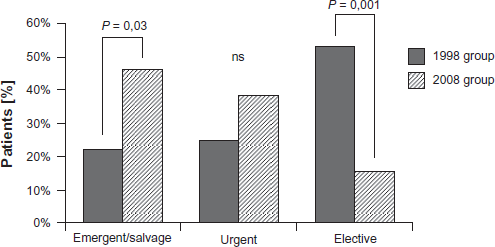
Status of the surgical procedures.
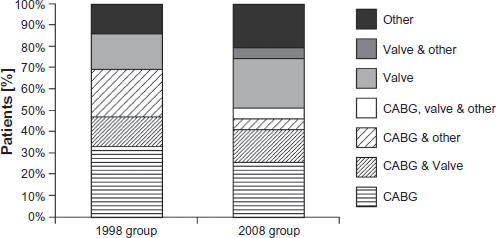
Characteristics of the surgical procedures.
Indication for MCS
Postcardiotomy low output syndrome was the primary indication for MCS. The incidence of postcardiotomy low output syndrome decreased from 80.6% in the 1998 group to 66.7% in the 2008 group. However, this trend was statistically insignificant (P = 0.17).
Acute myocardial infarction was the leading cause for cardiac insufficiency in patients presenting in shock prior to surgery. The percentage of patients with cardiogenic shock prior to surgery increased from 19.4% in the 1998 group to 33.3% in the 2008 group (P = 0.07).
Duration of support
Mean support length was equal in both groups with 2.9 ± 1.9 days of support (range 0–7 days) in the 1998 group and 3.8 ± 3.1 days (0–13 days) in the 2008 group (P = 0.14).
Mode of support
The centrifugal VADs were implanted in LVAD, RVAD or BVAD configurations. Most of the centrifugal VADs were left sided (88.9% in the 1998 group vs. 53.8% in the 2008 group; P = 0.001). The observed significant reduction in the percentage of leftventricular support in the 2008 group was due to a significant increase in the use of biventricular support (BVAD). The percentage of biventricular support increased from 5.6% in the 1998 group to 23.1% in the 2008 group (P = 0.03) (Fig. 3).
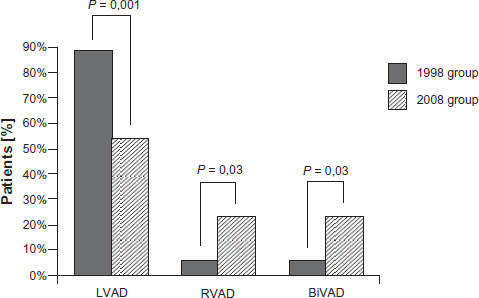
Type of support.
Intra-aortic balloon counterpulsation was frequently needed preoperatively and intraoperatively, reflecting compromised hemodynamics present in the cohort early in the perioperative period. IABPs were all used based on the hypothesis of additional pulsatile flow, reduction of afterload and better coronary flow. While 38.9% of the patients in the 1998 group were treated with an intraaortic ballon pump prior to surgery, the use of preoperative IABP support increased significantly to 66.7% in the 2008 group (P = 0.02). There was also a trend towards more frequent application of IABP counterpulsation in conjunction with VAD support in the 2008 group (61.1% in 1998 vs. 71.8% in 2008). However, this trend was statistically insignificant.
Bleeding complication
Neither in the 1998 group nor in the 2008 group, heparin-coated circuits were used. Systemic heparinization was initiated based on activated clotting time (ACT), which was optimally kept between 140 and 160 seconds. All patients were monitored for the need of blood product transfusion. In our experience, a bleeding tendency such as haematuria or surgical wound oozing was always noted. The number of administered units of RBC were not different between the groups (5.9 ± 4.4 in the 1998 group vs. 5.1 ± 5.5 in the 2008 group; P = 0.51). A significant increase was observed in the 2008 group regarding fresh frozen plasma (FFP)- and platelet concentrate (PC) consumption (4.7 ± 4.0 units FFP in the 1998 group vs. 10.0 ± 11.6 units FFP in the 2008 group; P = 0.02) (0.8 ± 0.7 units PC in the 1998 group vs. 2.0 ± 2.1 units PC in the 2008; P = 0.003).
Thromboembolic complications
Clinically recognized thromboembolic events occurred in both observation periods. We observed thromboembolic complications in three patients (8.3%) in the 1998 group and in six patients (15.4%) in the 2008 group. However, the difference in the incidence of thromboembolic complications between both groups was statistically insignificant (P = 0.35). Two cerebrovascular and one peripheral embolization occurred in the 1998 group. Five cerebrovascular and one peripheral (abdominal) events were observed in the 2008 group.
This results in freedom from thromboembolism under centrifugal mechanical assistance of 91.7% and 84.6% in the 1998 and 2008 group, respectively (P = 0.35).
Infectious complications
Critically-ill patients under MCS are prone to nosocomial and device-related infectious complications. All clinical records and microbiological results were reviewed to assess any infection. An infection was diagnosed in six patients (16.7%) in the 1998 group and in eight patients (20.5%) in the 2008 group. Overall, 85% of all infections were due to postoperative pneumonia. No difference was observed between the two groups in the incidence of pneumonia (80.0% in the 1998 group vs. 87.5% in the 2008 group; P = 0.72).
S. aureus followed by P. aeruginosa, were the most common pathogens. Duration of support and mechanical ventilation showed a trend to be independent risk factors for infection, however, this trend was not significant.
Clinical outcome
Death on device occurred in 36.1% (1998 group) and 38.5% (2008 group) of patients, respectively (P = 0.83). 11.8% (1998 group) and 3% (2008 group) of the VAD patients were bridged to long-term VAD (P = 0.12). No patient was bridged to heart transplantation.
Weaning rates from MCS were similar in both cohorts: in the 1998 group, 63.9% of the VA D patients were successfully weaned from the device compared to 61.5% in the 2008 group (P = 0.83). The leading causes of death after successful VAD weaning were multiorgan failure due to sepsis and nosocomial acquired infection (primarily pneumonia) under prolonged mechanical ventilation.
Overall out-of-hospital survival was similar in both groups (22.2% in the 1998 vs. 28.2% in the 2008 group; P = 0.55 (Fisher's exact test: P = 0.61)). Yet, a difference in out-of-hospital survival was observed depending on the mode of support. Patients requiring right ventricular or biventricular mechanical assistance had a significant poorer prognosis compared to patients treated with univentricular left sided support. Survival of patients supported with univentricular LVAD increased from 21.9% in the 1998 group to 38.1% in the 2008 group. Comparison of outcome in patients treated with biventricular support (BVAD) also showed a trend towards improved survival. In this subgroup, the 30-day survival rate reached 22.2% in the 2008 group, while no BVAD patient survived this time period in the 1998 group (P = 0.46 (Fisher's exact test: P = 0.66)). Nevertheless, Kaplan-Meier analysis of all VAD patients showed congruent values between the two groups (Fig. 4).
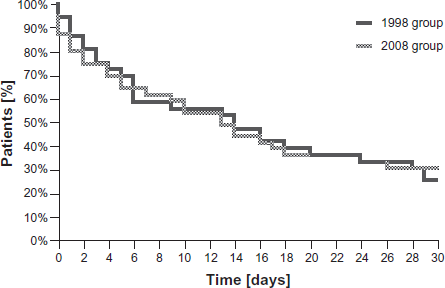
30-day-mortality after VAD implantation.
Risk of in-Hospital mortality
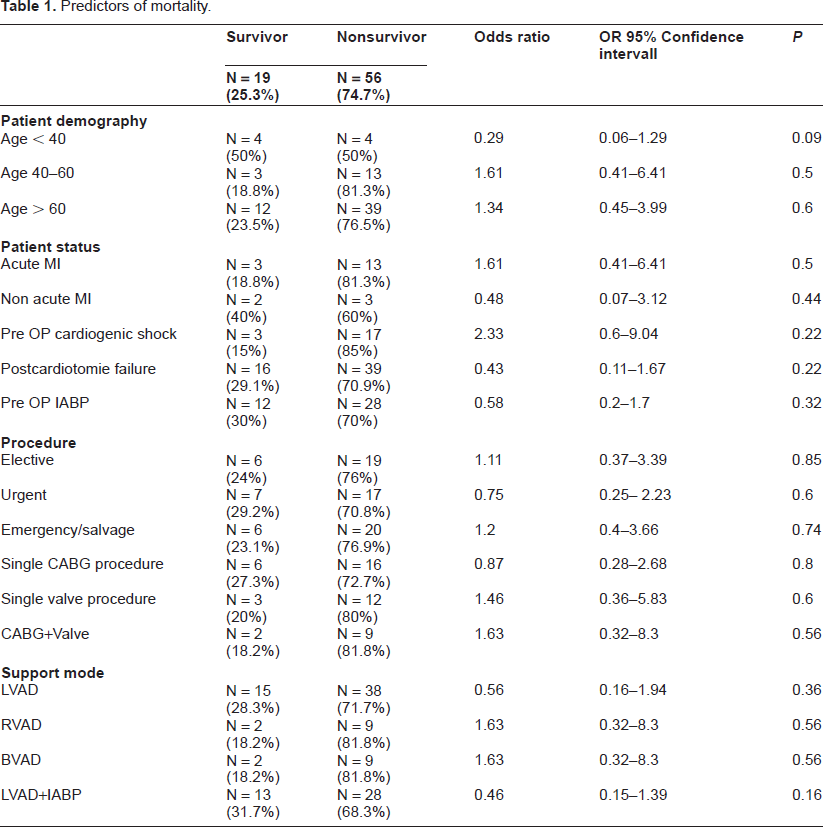
Several preoperative patient characteristics and parameters of the surgical procedure were abstracted from the medical records to evaluate the risk of in-hospital mortality. Correlates of in-hospital mortality are shown for the total population in Table 1. Preoperative cardiogenic shock was the most important predictor of in-hospital mortality, as indicated by the highest odds ratio (OR 2.33). In both groups, variables indicative of higher mortality risk were advanced age, acute myocardial infarction and complexity of the surgical procedure. Single LVAD implantation was associated with lower risk for in-hospital death compared with right ventricular (RVAD) or biventricular (BVAD) mechanical circulatory support. However, these observations were statistically not significant.
Predictors of mortality.
Discussion
The major purpose of the study was to evaluate the use of short-term centrifugal MCS in the management of patients with cardiogenic shock in a recent group of patients (2006–2008) compared to a similar group of patients who had support placed in an era ten years earlier (1996–1998). Although, a constant number of patients received short-term MCS between these two time intervals at our institution, these data were excluded from detailed analysis to identify and accentuate possible differences regarding demography, indication for support and clinical outcome. Interestingly, our results demonstrate that demographic parameters such as age, gender, (and in other series race) 1 have not changed in patients that were treated with MCS. This has to be noticed in contrast to the continued increase in age, observed in patients undergoing cardiac surgery. It seems that the current application of VAD therapy is limited to the same patient population than ten years ago.
Despite the remarkable progress in cardiac surgery, critical care, and assist device design in recent years, our single center analysis demonstrated that mechanical circulatory support continues to have a poor prognosis. Yet, our evaluation yielded almost identical results to recently published studies or series from the past. Evaluations of the Biomedicus bio-pump from the mid 1990s demonstrated a 20% to 30% survival rate. 8 Recently conducted studies by Shuhaiber et al 9 and de Robertis et al 10 also evaluated the latest generation magnetically levitated centrifugal blood pumps. Post-VAD 30-day survival rates of 30% and 33%, respectively were reported, similar to our results.
Three major complications have always been associated with MCS: bleeding, thromboembolism, and infection. We evaluated both groups to assess possible changes in the incidence of these VAD-related postoperative events and evaluated the impact of these complications. Coagulopathy and bleeding necessitated blood product transfusion in up to 95% patients in the mid 1990s.11–13 Our study shows, that this fact has not changed: virtually all patients required blood transfusion in our series. Our data also suggests that the occurrence of bleeding is independent from the type of device employed. Despite several design modifications over the years up to the clinical introduction of magnetically suspended impellers, bleeding episodes remained the major complication. In fact, a significant increase in consumption of fresh frozen plasma and platelet concentrates was observed in the 2008 group. This increase, however, is most likely due to the emerging dual antiplatelet therapy in patients with acute coronary syndrome (glycoprotein IIb/IIIa inhibitors available from 1999), rather than the design of the utilized assist device (bearing type vs. magnetically levitated impeller VAD).
Thromboembolism is the second major threat in MCS patients. The clinically recognized thromboembolism rate under centrifugal VAD support did not show a significant change and was also independent from novel VAD engineering designs. With an average stroke rate of about ten percent, improvement of the centrifugal pump blood biocompatibility is still the major challenge. Mechanical or electrical failures of the assist devices were negligible in both observation periods. Not a single device failure was observed in the 2008 group, demonstrating the safety and reliability of contemporary centrifugal VAD.
It seems remarkable that neither support length nor weaning rate have changed over the years. Weaning rates were equally high in both groups. About two-third of patients were successfully weaned from the assist device. These findings are consistent with other studies.9,10 However, despite successful VAD weaning, many patients die in the course of the hospital stay. In our study, the leading causes of death after successful VAD weaning have not changed. While the leading cause of death under mechanical support remains cardiogenic failure, infection is still the predominant factor for in-hospital mortality after successful VAD weaning. Multiorgan failure due to sepsis and nosocomial acquired infection (primarily pneumonia, representing more than 80% of the diagnosed infectious complications) was noted in both observation periods in spite of extensive use of antibiotics. This demonstrates the tremendous impact of nosocomial infection on survival in patients under assisted circulation. Rigorous infection management and -where possible- early device weaning and closure of the sternum seem to be crucial points for the success of LVAD programs.
Yet, our retrospective analysis revealed remarkable changes in patient and surgical characteristics. Rates of patients in cardiogenic shock referred to surgical therapy increased substantially during the past decade. About one third of all patients receiving centrifugal VAD support in the 2008 group presented with cardiogenic shock prior to surgery. This resulted in the risk-aggravating factor, that the initial surgical procedure was classified “emergent/salvage” in almost half of the patients receiving centrifugal VAD support in the 2008 group. While salvage operations requiring VAD implantation increased significantly, elective procedures declined by factor 3.4 between the two observation periods. Consequently, the calculated perioperative mortality risk has significantly increased, a fact that is supported by the observed two fold rise in the mean log Euro-Score between the two groups.
Considering the facts that the severity of illness, as well as the urgency and complexity of the surgical intervention have apparently increased, a comparative improvement in clinical outcome of MCS can not be dissmissed.
Particulary, survival after biventricular centrifugal support has improved. Right ventricular failure under left ventricular mechanical support has been identified as leading cause of death long time ago. Timely implantation of biventricular support systems has therefore been postulated. 14 Our data demonstrates that not only the rate of BVAD implantations, but also the survival after BVAD support have indeed increased. A changed disposition to use centrifugal MCS at an early stage and -if necessary- consequently apply complex BVAD support is evident.
The efficacy of ventricular assist device support has been clearly demonstrated in chronic heart failure patients. The landmark REMATCH trial 15 gave evidence about the superiority of left ventricular assist device support in terms of survival and quality of life compared with optimal medical management. Furthermore, it demonstrated a remarkable 52 percent rate of survival one year after VAD implantation. Unfortunately, these excellent results of long-term MCS in chronic heart failure patients can not be achieved in patients receiving short-term circulatory support for acute cardiogenic shock. Mechanical circulatory support remains an extraordinarily resource-intensive undertaking. For this reason, concerns have been raised about the appropriateness of such an aggressive strategy, especially in light of the high costs. Recently conducted studies evaluated the cost-utility of salvage cardiac extracorporeal membrane oxygenation and revealed median hospital cost in the range of USD 150,000 to USD 210,000 per patient.16,17 Yet, it was concluded that mechanical circulatory support–-even when used in the setting of salvage therapy–-still falls within the bounds of routinely accepted cost-utility. 16
The decision to implant an assist device in an individual patient can be difficult and is influenced by expected outcomes. We examined pre- and periplacement factors in order to identify parameters indicating patients who would benefit from VAD therapy. Our analysis documented that advanced age, pre-VAD median base deficit value (data not shown) and preoperative cardiogenic shock were the most important predictors of in-hospital mortality, as indicated by the highest odds ratio. Further variables showing a predictive trend towards increased mortality in VAD patients were acute myocardial infarction and complexity of the surgical procedure as well as biventricular failure. However, the small cohort size limits the power of our study. Although, several factors may have been significant if our study size had been large enough, our analysis is not capable of providing a valid concept which patient would benefit from MCS. Within a single center experience, this is difficult to rectify, as fortunately the incidence of MCS (only 75 patients in approximately 8000 patients) has remained low.
In conclusion, our data clearly demonstrate the beneficial effect of MCS for patients who otherwise would decease from cardiac failure. Considering that severity of illness, complexity and urgency of the surgical procedure have steadily increased, an improvement in MCS outcome over the past decade should not be dissmissed. Nevertheless, in-hospital mortality and VAD related complication rates such as bleeding and infection remain high.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
