Abstract
Lung perfusion/ventilation was introduced as a means to minimize cardiopulmonary (CPB)-related pulmonary ischemic injury. Current results in the literature are divergent, and the role of gas exchange during lung perfusion/ventilation during CPB, remains undefined. This report details a) the technique of continuous lung perfusion/ventilation during CPB, b) provides initial observations, and c) discusses gas exchange during CPB.
Introduction
Prolonged cardiopulmonary bypass (CPB) causes severe lung injury, manifested by poor oxygenation, pulmonary edema and, even acute respiratory distress syndrome. 1 Lung perfusion/ventilation was recently reported to minimize CPB-related pulmonary ischemic injury in animal models.2,3 Proposed to achieve more favorable clinical outcomes, the technique aims at promoting better pulmonary gas exchange, with prevention of atelectasis and ischemic injury. Experimental and clinical studies assessed (A-a) O2 gradients during coronary artery bypass grafting with and without CPB. Results were unclear and divergent.4,5 We herein, describe the technique of lung/perfusion ventilation in three patients undergoing valve replacements, and review the collected data in order to better understand the dynamics of alveolar gas exchange during CPB.
Case Reports
Institutional Review Board (IRB) approval for this study was obtained, and patients signed informed consents for the operation and for the study.
Patient #1
A 65 year-old man presented with aortic bio-prosthetic valve MRSA endocarditis. He was in critical condition, with embolic ischemia in the splanchnic and peripheral vascular beds. Transthoracic echocardiography showed multiple vegetations of the prosthesis, abscesses near the left ostium, and combined aortic stenosis and insufficiency. Preoperative PaO2 was 320 mmHg on 100% O2. Beating heart redo-valve replacement was performed, as previously reported.6,7 After cross-clamping the aorta, warm blood retrograde perfusion was initiated (mean coronary sinus pressure of 55 mmHg, flows of 300 ml/min). After transverse aortotomy, both the left and right coronary ostia were perfused via a dual perfusion system, previously reported, 8 with blood flow derived from the aortic cannula. Normal sinus rhythm was maintained throughout the procedure. A 14-gauge catheter, derived from the aortic cannula, was inserted into the main pulmonary artery, which was perfused with flows of 400 ml/min. During CPB, the lungs were ventilated with air (tidal volume of 350 ml, and frequency 6 breaths/min) (Table 1). A vent was inserted into the right superior pulmonary vein. The aortic prosthesis was removed, the abscesses were drained, and the area was debrided. The abscess cavity posterior to the left coronary ostium was patched with bovine pericardium. A #23 Carpentier Edwards Pericardial Magna® prosthesis was inserted. The patient required minimal inotropic support to separate from CPB. Arterial blood gases post CPB had pO2 of 380 mmHg. The patient was extubated 8 hours later. He had a stormy course, with systemic fungal septicemia and necrosis of the right leg below the knee, requiring amputation. He remained hemodynamically stable throughout hospitalization.
Intraoperative parameters.
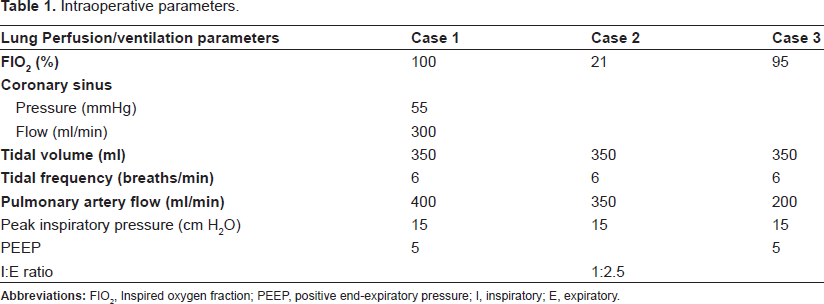
Patient #2
A 26 year-old man with incompetent bicuspid aortic valve, with aneurysmal dilation of the ascending aorta, underwent aortic valve replacement (mechanical prosthesis), with ascending aortic aneurysm repair with graft interposition. After induction of anesthesia, controlled ventilation with FiO2 of 1.0 was instituted with peak inspiratory pressure of 15 cm H2O (plateau close to 12 cm H2O) without positive end-expiratory pressure (PEEP), with inspiratory pause with I:E ratio 1:2.5. Isoflurane was added to the inflow limb of the CPB circuit via a vaporizer, at 1 vol%. Priming of CPB machine was with crystalloid solution, with aminocaproic acid added. After median sternotomy, the ascending aorta and right atrium were cannulated. CPB was instituted at temperature of 34 °C. Antegrade/retrograde cold blood cardioplegia was administered. Continuous perfusion of the pulmonary artery was achieved with mean flows of 350 ml/min, through a #14-gauge catheter derived from the aortic cannula. The lungs were ventilated with air, tidal volume of 350 ml, frequency 6 breaths/min. (Table 1). End-tidal CO2 (ETCO2) inspired and expired concentrations of O2 and anesthetic were continuously measured throughout the operation (Figs. 1, 2 and 3, respectively). Afferent and efferent pulmonary blood gases (pulmonary artery (PA) and the left atrium (LA)) are listed in Figure 7. The aortic valve was replaced with #23 St. Jude® mechanical bioprosthesis, with replacement of the ascending aorta with #28 Hemashield® graft. The patient was separated from CPB without difficulty. He was extubated on POD #1, and was discharged home on postoperative day 15.
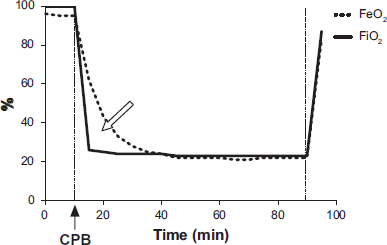
Expired (dashed lines) and inspired (solid lines) O2 fractions during CPB at FIO2 21% (room air). Solid arrow indicates onset of CPB. Blank arrow indicates the period of time which reversal of O2 flow from capillaries to alveoli, reflected by higher expiratory concentrations. (Patient #2).
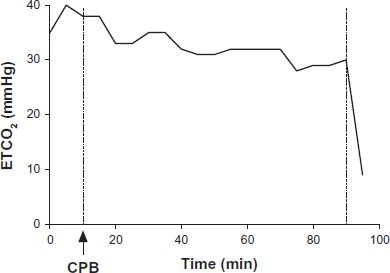
End-tidal CO2 during CPB FIO2 21% (room air). At room air, ETCO2 is practically stable during the period of CPB. Arrow indicates onset of CPB. (Patient #2).
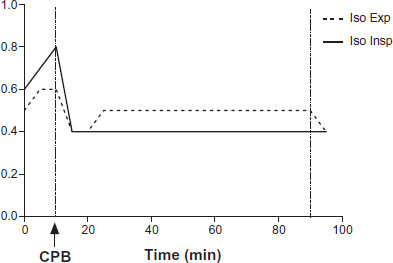
Expired (dashed line) and inspired (solid line) isoflurane anesthetic during CPB at FIO2 21% (room air). Arrow indicates the onset of CPB. Changes of inspired and expired anesthetic concentrations reflect the described significant and variable dead space ventilation. Practically no anesthetic uptake takes place during CPB, due to profound V/Q mismatch. Higher expiratory anesthetic concentrations reflect the settings of isofluorane vaporizer in the CPB manifold. (Patient #2).
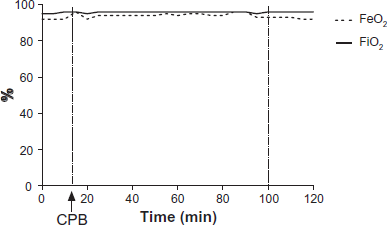
Expired (dashed lines) and inspired (solid lines) O2 fractions during CPB at FIO2 95%. Arrow indicates the onset of CPB. (Patient #3).
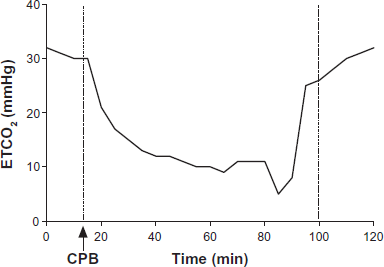
ETCO2 during valve surgery, at FIO2 95%. At FIO2 95%, ETCO2 showed significant and progressive decrease during CPB. Arrow indicates onset of CPB. (Patient #3).
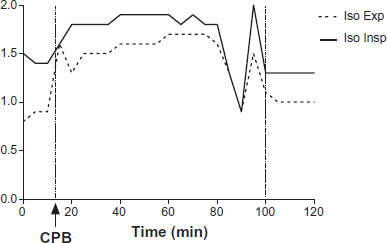
?Expired (dashed line) and inspired (solid line) isoflurane anesthetic during CPB at FIO2 95%. Arrow indicates the onset of CPB. Changes of inspired and expired anesthetic concentrations reflect the described significant, and variable, dead space ventilation. Practically no anesthetic uptake takes place during CPB, due to profound V/Q mismatch. (Patient # 3).
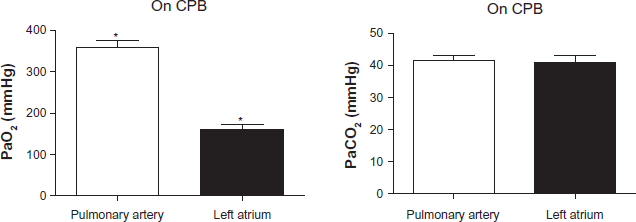
Blood gas analyses on afferent and efferent pulmonary circulation. *Mann-Whitney test p < 0.05. (Patient # 2)
Patient #3
A 71 year-old African American female, in pulmonary edema secondary to severe aortic stenosis, underwent urgent aortic valve replacement with a #23 St. Jude Biocor® prosthesis. After median sternotomy, the ascending aorta and right atrium were cannulated. CPB was instituted at temperature of 34 °C. Antegrade/retrograde cold blood cardioplegia was administered. Continuous perfusion of the pulmonary artery was achieved with mean flows of 250 ml/min, through a #14-gauge catheter derived from the aortic cannula. The lungs were ventilated with 95% O2, tidal volume of 350 ml, frequency 6 breaths/min (Table 1). ETCO2, inspired and expired concentrations of O2, and anesthetic (isoflurane), were continuously measured throughout the operation (Figs. 4, 5 and 6). PA and LA blood gases are tabulated (Fig. 8). The aortic valve was replaced with #21 St. Jude® mechanical bioprosthesis. This patient developed cardiac tamponade six hours postoperatively and was returned to the OR for mediastinal re-exploration. Bleeding was generalized and was not related to the pulmonary cannulation. She was discharged home on day 6.
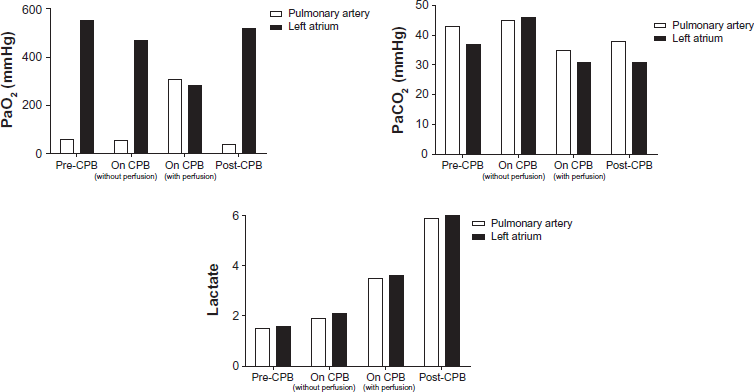
Blood gas analysis on the afferent and efferent pulmonary circulation. Pre-CPB = time 0; On CPB (without perfusion) = 15 min; On CPB (with perfusion) = 42 min; Post-CPB = 157 min. (Patient #3)
Discussion
Pulmonary complications due to CPB remain an important clinical problem. Early pulmonary dysfunction and ARDS occur in 12% and 1.3% of patients undergoing surgery with CPB, respectively 9 This pulmonary dysfunction is thought to be multi-factorial, and is usually traced to atelectasis with its attendant hypoxemia and shunting,10,11 interstitial fluid accumulation resulting in pulmonary edema, and systemic inflammatory response due to surgical trauma and CPB. 12
We strongly believe that the pulmonary ischemiareperfusion injury associated with current technique of CPB is often overlooked. This is nevertheless very important and potentially preventable. During CPB, pulmonary blood flow to the lungs ceases. In most instances, the lungs are neither ventilated or perfused via the pulmonary artery. Blood flow to the lungs is mainly via the bronchial circulation. However, bronchial artery blood flow decreases immediately with the onset of CPB, and may become inadequate for the maintenance of normal alveolar perfusion. 13 When the pulmonary circulation is restored, ischemiareperfusion injury occurs, contributing to post-CPB pulmonary dysfunction. This might contribute to ischemic injury at the blood-gas interface in the alveolus.
Pulmonary perfusion/ventilation during CPB is not a new concept. Low-frequency ventilation with air during CPB in pigs has been shown to decrease atelectasis, prevents water accumulation, and promotes intraalveolar capillary flow. 2 These authors also showed that low-frequency ventilation in pigs is beneficial especially when combined with ultra-filtration. 2 Siepe et al demonstrated lowered inflammatory response during CPB with pulsatile lung perfusion, compared to non-pulsatile group. 14 Animal models of CPB without lung perfusion show increased pulmonary vascular resistance, A-a gradient, and lower pulmonary compliance. 14 Gabriel et al demonstrated pulmonary protection in pigs subjected to prolonged circulatory arrest with lung perfusion/ventilation. 3 Even a single-shot of hypothermic blood into the pulmonary artery at the beginning of CPB may be protective. 15 Whether this would improve clinical outcome during prolonged CPB remains to be determined.
Other studies found no effect on (A-a) O2 gradient attributable to CPB in patients who had undergone CABG with or without CPB. 4 Cox et al 5 did not find significant lung dysfunction secondary to CPB, other than that caused by operative and anesthetic techniques themselves. Loer et al 16 found higher PaCO2, PaO2 and higher thromboxane B2 in the collapsed lung, compared to the contra-lateral ventilated lung. However, those differences did not translate into increased injury markers. In this study, CPB times were short, and pulmonary venous blood samples were obtained before separation from CPB. Not only these data may not reflect pulmonary dysfunction due to longer CPB times, 10 but we believe that the samples should have been taken after restoration of the pulmonary circulation, in order to detect any changes in ischemia markers in efferent pulmonary flow.
This report reflects our technique of lung perfusion/ventilation during CPB, and presents our preliminary results. We have used this technique in all valve patients operated upon over the last 8 months. We found no adverse effects of perfusion/ventilation of the lungs in any of the patients, and no evidence of cerebral embolization of air to date, as the right superior pulmonary vein were vented and separated from the systemic circulation.
Pulmonary artery perfusion flow depends on the systemic perfusion pressure. At systemic flow of 5 L/min, and mean pressure of 60 mmHg, measured Doppler flow in the PA was close to 300 ml/min. In this preliminary study, pulmonary blood flow was not controlled or varied. Therefore, one cannot predict the actual correlation between pulmonary perfusion and ETCO2 during CPB. We intend to perfuse the pulmonary artery with venous blood via separate line and pump, at higher flows, in order to decrease V/Q mismatch during CPB.
Alveolar ventilation was maintained during CPB to reduce atelectasis and to promote alveolar capillary flow. Ventilatory parameters were as indicated: tidal volume 300 ml, frequency 6 breaths/min, and PEEP 5 cm H2O. In patient #2, the FIO2 was 21% (room air), and in case #3, it was 95%. Inspired and expired tidal flows were continuously analyzed with continuous side-stream capnography.
During CPB, blood gas analysis was obtained in the pulmonary afferent and efferent blood (from PA and LA). In patient #2, with the alveolar ventilation on CPB maintained with air, the partial pressure gradient of oxygen from capillary to alveolus (PaO2 in PA flow was 339–410 mmHg, and calculated PAO2 was 150 mmHg), resulted in “reversed” oxygen flow from capillary to alveolus. That was reflected in higher expiratory than inspiratory concentrations of oxygen, and in lower PaO2 in left atrium (LA), compared to PA. Nevertheless, the calculated oxygen content was not significantly different in pulmonary inflow and effluent flows. When the lungs were ventilated with 95% O2 (patient #3), no difference in oxygen concentrations was observed in inspired and expired flows, and in pulmonary afferent and efferent O2 contents. (Fig. 4)
Analysis of expired CO2 in both cases (Figs. 2 and 5) indicates widely variable ETCO2. Since ventilator settings were similar in both cases, it is thought to reflect changes in alveolar dead space ventilation. As alveolar ventilation during CPB was close to 1.8 L/min, and pulmonary flow was 300 ml/min at full calculated systemic flows, the baseline ventilation/perfusion mismatch is significant (V/Q = 1.8/0.3 = 60). This results in reflects a significant dead space ventilation. ETCO2 in this circumstances reflects significant solubility of CO2, which can be eliminated even in the presence of such profound V/Q mismatch; however, with further decrease in PA flows (secondary to temporary decreases in systemic flows), elimination of CO2 becomes compromised, despite high CO2 solubility. Although decreased V/Q mismatch was not the primary goal of this study, we used V/Q matching to illustrate the rationale behind poor correlation between the numeric values of capnographic ETCO2, end-tidal oxygen concentration and expiratory isofluorane and their respective arterial partial pressures values. In order to improve correlation, increased perfusion flows to the pulmonary artery are needed to decrease intrapulmonary shunting and wasted ventilation (dead-space).
However, the magnitude of the perfusing pulmonary artery flow during CPB, in order to achieve sufficient parenchymal perfusion and decrease ischemiareperfusion injury, has to be restricted to avoid “luxury” excess perfusion with resultant pulmonary edema. We propose to match perfusion to ventilation. It is imprecise, since we normally decrease the amount of ventilation on CPB, but there is no better option at this time.
The alveoli must be kept open to ensure gas exchange across the alveolar membrane. Perfused, but collapsed alveoli contribute to shunting and alveolar edema. Ventilated and un-perfused alveoli result in dead-space ventilation and alveolar baro-trauma. We used pressure-controlled ventilation (15 cm H2O, achieving tidal volumes of 350 ml). Another option is to provide continuous PEEP (10 cm H2O). This is work in progress, aimed at better understanding the coupling regional ventilation to perfusion. At this moment, V/Q matching remains a goal to be achieved. Regional energetics and radioactive glucose PET MRI, might shed light into these questions.
Conclusions
There is profound V/Q mismatch during CPB with lung perfusion/ventilation. Even though the number of observations is small, we believe this to be an important, albeit unrecognized, and poorly understood, phenomenon that may account for postoperative pulmonary dysfunction due to CPB. It is possible that current clinical indicators are too insensitive to assess pulmonary ischemia, in the context of relatively short CPB.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
