Abstract
Purpose
To report a case of corneal melting in a patient with fornix foreshortening being treated with reformulated generic diclofenac.
Methods
Case report.
Results
An asymptomatic 76-year old man presented with central corneal melting while being treated with reformulated generic diclofenac. This formulation did not contain the vitamin E derivative, tocophersolan, which has been felt to contribute to corneal melting associated with generic diclofenac in the past. Other factors for corneal melting included multiple medication use and altered forniceal architecture, but did not include previous corneal or cataract surgery. Following discontinuation of the topical medications and temporary tarsorrhapy, the defect reepithelialized.
Conclusion
In patients with a compromised ocular surface, reformulated generic diclofenac has the potential to cause corneal melting without prior cataract or refractive surgery. Use should not be indiscriminate or without close supervision.
Introduction
Nonsteroidal anti-inflammatory drugs (NSAIDs), including diclofenac, are inhibitors of the enzyme cyclooxygenase, which is required for the synthesis of prostaglandins. Prostaglandins are known to be mediators of inflammation, including ocular inflammation. 1 Topical NSAIDs are commonly used in ophthalmology for decreasing pain after cataract and refractive surgery, treatment and prophylaxis of cystoid macular edema following cataract surgery, as well as for ocular inflammation such as episcleritis. However, many times, topical NSAIDs are used by eye care practitioners for unclear reasons for extended periods of time without a clear diagnosis or indication. 2
In 1999, in response to media reports, the American Society of Cataract and Refractive Surgeons surveyed its members about the incidence of ocular surface complications due to NSAID use following cataract and refractive surgery. At least 200 cases of corneal toxicity were reported, the severity of which ranged from superficial keratitis to corneal melting. Generic diclofenac (Falcon, Ft. Worth, Texas) was reported as the offending agent in most of the cases, along with some reports of complications due to Voltaren (Cibavision, Duluth, Georgia) and a handful of cases due to Acular (Allergan, Irvine, California). 3 11 cases of corneal melting were reported, with 7 cases associated with generic diclofenac and 4 associated with Voltaren. Most of these patients had recently undergone cataract or refractive surgery. A review by Flach found that many of these eyes had comorbid conditions and lacked a clinical diagnosis supporting the use of NSAIDS. 2
In September of 1999, Alcon, Falcon's parent company, voluntarily recalled generic diclofenac. 4 One theory postulated that an inactive ingredient in the generic diclofenac formulation, tocophersolan, a Vitamin E derivative, was responsible for a large number of melts associated with generic diclofenac. This hypothesis was based upon the observation that Vitamin E derived compounds have been shown to inhibit proliferation and migration of retinal pigment epithelial cells in vitro. 5 However, in the last decade, there have been additional reports of post surgical corneal melting involving NSAIDs that did not contain tocophersolan. 6 8
In the following case, a patient without previous refractive or cataract surgery, but with abnormal inferior forniceal architecture, sustained a corneal melt after treatment with reformulated generic diclofenac no longer containing tocophersolan.
Case Report
An asymptomatic 76-year old man being treated chronically with generic diclofenac (Falcon, Fort Worth, TX) twice daily presented to his optometrist for a routine glaucoma follow-up, and was found to have a significant central corneal ulcer (Figure 1). He had been seen 3 months prior to this and was noted only to have a corneal scar in the left eye. He was referred to the Ophthalmology clinic the same day.

Left eye at presentation showing a central corneal melt.
On examination, he had 3+ conjunctival injection with significant shortening of the inferior conjunctival fornix (Figure 2). There was no entropion or trichiasis, however. Examination of the cornea revealed a central 2.0 × 1.0 mm oval corneal ulcer, with 60% corneal thinning. Fluorescein staining prior to arrival in the clinic prevented accurate assessment of whether there was an infiltrate. The right eye had no corneal or conjunctival abnormalities.
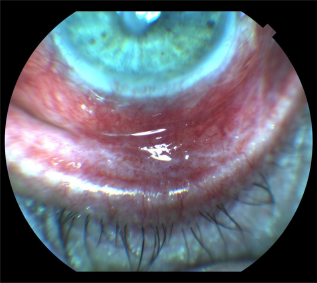
Foreshortening of the patient's inferior fornix.
The patient stated that he had undergone successful surgical treatment for trichiasis by an outside ophthalmologist approximately one year earlier and was placed on bromfenac at that time. He thought that his conjunctival scarring and trichiasis may have been due to a trauma in his youth from a stick injury to the eye. He was on this medication until he moved to our region 7 months ago, at which time his primary care doctor substituted generic diclofenac for the bromfenac. Review of the patient's records showed use of timolol and brimonidine in that eye as well.
Since infectious keratitis could not be ruled out at presentation, the cornea was cultured and the patient was started on fortified vancomycin and tobramycin. The timolol, brimonidine, and diclofenac were discontinued. In follow-up treatment it was apparent that there was no corneal infiltrate. The culture was negative. The patient was changed to prophylactic moxifloxacin and placed on doxycyline and vitamin C to help prevent further corneal melting. A temporary tarsorrhaphy was also performed. By two weeks post presentation the corneal ulcer had healed.
Discussion
This case is important for several reasons. First, it confirms that reformulated generic diclofenac carries similar risks to other NSAIDS. Although bromfenac, which the patient was taking initially, has also been reported to cause corneal melting, 7 the patient was changed to diclofenac seven months prior to presentation. This indicates that it was diclofenac, not bromfenac, which precipitated this episode. When generic diclofenac was initially withdrawn from the market, it contained tocophersolan, a vitamin E based solubilizer which was not contained in Voltaren. It was hypothesized that this compound inhibited epithelial proliferation and upregulated matrix metalloproteinases (MMPs), which predisposed patients to corneal melting. 5 Tocophersolan may, indeed, have been a contributing factor. However, if it was the only agent causing corneal melting, we should not see melting with other NSAIDs, including Voltaren and reformulated generic diclofenac. There have been cases of post surgical corneal melting reported with diclofenac formulations which do not contain tocophersolan, including preservative free diclofenac. 6 This points to the NSAID itself as a cause of corneal melting. This view is given further support by a study by Gabison et al, which found upregulation of MMP-9 and MMP-3 by immunohistochemistry in a patient requiring penetrating keratoplasty for a perforated corneal ulcer after being treated with diclofenac for two months following photorefractive keratectomy. The French version of diclofenac (Novartis International AG, Basel, Switzerland), which that patient received, did not contain tocophersolan. 9
Secondly, to the author's knowledge, since the recall of the original formulation of diclofenac, all reported cases of corneal melting associated with diclofenac have occurred after refractive or cataract surgery.6,9,10 This is the first reported case of corneal melting with reformulated generic diclofenac occurring in an eye without previous corneal surgical manipulation. It is felt that corneal melting requires an epithelial defect to occur. This case illustrates that a condition such as post-surgical or post-traumatic shortening of the inferior fornix can predispose a patient to NSAID associated corneal melting due to a compromised ocular surface and high risk of mild epithelial defects. In these patients, the epithelium is more vulnerable due to reduction of the protective function of the lids, even without noticeable lagophthalmos. A case of corneal melting associated with bromfenac in a patient with ocular surface compromise due to severe dry eye after Stevens Johnson Syndrome has also been reported with a much shorter, two week, duration of treatment. 11 Other comorbid conditions in the patient's past ocular history which could have contributed to the patient's corneal melt include trichiasis and glaucoma. Trichiasis was unlikely to have contributed because it had been surgically corrected and was no longer present on presentation. Since the patient's intraocular pressure was normal at presentation, there was no contributing factor such as microcystic edema which could have compromised the patient's epithelium. However, his glaucoma could have contributed to this disease process in that both generic timolol and brimonidine contain the preservative benzalkonium chloride (BAK), which has significant epithelial toxicity. Additionally, timolol can decrease corneal sensation, which along with the analgesic effect of diclofenac, increased the patient's risk of neurotrophic keratopathy. 12
Finally, this case is an important illustration of the possible consequences when topical NSAIDs are used in patients with compromised ocular surfaces for non-indicated reasons for extended periods of time. This patient had the risk factors of abnormal eyelid function and multiple topical medications. However, he was on indefinite, unmonitored, NSAID treatment without a clear indication. A. Flach, in a letter to the editor of Cornea in 2006 described this problem. 13 5 years later the problem continues to persist. The author has noted several occasions in his own practice when a NSAID has been prescribed by an Ophthalmology resident with “as needed” refills. By reporting this patient, the hope is that more thoughtfulness will be encouraged in the practice of prescribing topical NSAIDs and that eye care professionals will recognize that these medications are not completely benign, especially in patients with ocular surface comorbidities.
Disclosures
This manuscript has been read and approved by the author. This paper is unique and not under consideration by any other publication and has not been published elsewhere. The author and peer reviewers report no conflicts of interest. The author confirms that he has permission to reproduce any copyrighted material. Written consent was obtained from the patient for publication of this study.
Footnotes
Acknowledgements
Supported in part by an unrestricted grant from Research to Prevent Blindness. Publication of this article was funded in part by the University of Florida Open-Access Publishing Fund.
