Abstract
Nausea and occasional vomiting in early pregnancy (NVP) are common. When vomiting is severe or protracted, it is referred to as hyperemesis gravidarum (HG). HG affects up to 3% of pregnancies and is characterized by weight loss, dehydration, electrolyte imbalance, and the need for hospital admission. HG has significant consequences for maternal well-being, is associated with adverse birth outcomes, and leads to major health care costs. Treatment options are symptomatic, hampered by the lack of evidence-based options including studies on nutritional interventions. One of the reasons for this lack of evidence is the use of a broad range of definitions and outcome measures. An internationally accepted definition and the formulation of core outcomes would facilitate meta-analysis of trial results and implementation of evidence in guidelines to ultimately improve patient care.
Introduction
Progress in the understanding of the etiology and treatment of hyperemesis gravidarum (HG) has been slow in recent decades.1–3 This might be due to difficulty in pinpointing a cause and the presumed psychogenic etiology, the latter remaining unsupported by evidence. 4 Clinicians may find it challenging to differentiate HG from “physiological” nausea and vomiting in pregnancy (NVP) and consider HG a self-limiting, non-life-threatening affliction, lacking curative treatment options. “Therapeutic nihilism” among caregivers may lead to poor care for HG patients. 5 Research is limited to single/small study groups with no or minimal funding, often yielding underpowered studies. Meta-analysis has the ability to increase power and is considered to provide the highest level of evidence. 6 It is therefore unfortunate that attempts to perform meta-analyses on HG etiology, diagnosis, and treatment options are hampered by the lack of consensus on the exact definition of HG and most relevant outcomes.7,8
In addition to the forthcoming Cochrane review on the treatment of HG, 9 several systematic reviews on HG have been published, most recently in 2014. 1 Here, we therefore will not attempt to present an extensive overview of all evidence but will summarize briefly where HG research stands today and suggest ways to bring HG research forward.
What is Hyperemesis?
NVP in early pregnancy is common, affecting 50%-80% of pregnant women during the first half of gestation,10,11 with major impact on maternal well-being and quality of life. 12 When vomiting is severe or protracted, it is often referred to as hyperemesis gravidarum (HG). 13 HG is much less common than NVP, affecting 0.2%-3.6% of pregnant women.14,15 Despite this relatively low incidence, HG is the single most frequent reason for hospital admission in the first half of pregnancy, 16 with substantial health care costs. In 2012, the costs per HG patient in the USA were estimated at $47,351. 17
Little is known about the pathophysiology of HG. Proposed underlying causes are related to maternal endocrine function, placental growth and function, and gastrointestinal conditions. Rise of human chorionic gonadotrophin (hCG) during early pregnancy coincides with the occurrence of HG, and several conditions associated with increased hCG are more prevalent in HG patients. 18 It is thought that rising hCG levels may cause nausea via several pathways, including through rises of other hormones such as thyroid hormone and estradiol. Although the majority of studies on hCG and HG describe a positive association, heterogenic use of definitions and outcome measures limit the possibility of meta-analysis, and findings remain inconclusive. 7 High hCG levels in the second trimester may be caused by abnormal placentation. This is in line with the study of Bolin et al, who found placental dysfunction disorders to be associated with HG. 19 Colonization with the gastric bacterium Helicobacter pylori is associated with an increased risk of HG, but the strength and size of these associations vary among different populations and countries. 20 Despite many causes for HG having been proposed, no single mechanism has yet been identified, making a multifactorial origin more likely. 1
Large registry studies have identified several risk factors. The recurrence rate of HG after a first pregnancy complicated by HG ranges from 15% to 81% and may vary with HG definition based on symptoms or hospital admission and study methodology.21,22 In addition to having had HG in a previous pregnancy, having a mother or sister who had HG is an important risk factor. This suggests that yet-unidentified genetic factors are involved in HG etiology21,23 Furthermore, young age, nulliparity, non-Western ethnicity, low socioeconomic status, diabetes, hypertensive disorders, psychopathology, assisted reproductive techniques, female fetus, and multiple gestation are all associated with HG. 15 In the past, psychopathology has been suggested to play a causative role, 24 but convincing evidence is lacking. 4
How Do We Diagnose HG?
HG is a clinical diagnosis, made after other causes of nausea and vomiting have been excluded. Commonly used criteria include nausea and vomiting before 20 weeks of gestation, the need for hospitalization, weight loss, dehydration, and electrolyte imbalances.13,25 Patients generally undergo a diagnostic work-up, including laboratory testing and ultrasonography to rule out molar pregnancy or multiple gestation. However, such a work-up is not based on reliable data from the literature. A recent review showed that, despite its widespread use as a major diagnostic criterion, there is little evidence to support the use of ketonuria in HG diagnosis due to its lack of association with symptom severity, admission duration, or readmission rates. 7 There is no international consensus on the exact definition of HG.
Do We Have Treatment Options?
Management of care
Hospital admission, intravenous rehydration, and antiemetic medication are often applied. 13 The only randomized controlled trial (RCT) investigating rehydration for HG did not show any benefits after 24 hours of treatment when 5% dextrose was added to 0.9% saline solution in terms of ketonuria, nausea, and vomiting symptoms. 26 Early thiamine (vitamin B1) supplementation before the administration of dextrose is mandatory to prevent the development of Wernicke encephalopathy. 13 It could be debated whether HG should always be treated in hospital. McCarthy et al recently showed that day care management of HG reduced total days of inpatient stay without affecting patient satisfaction. 27 Another trial on the effectiveness of outpatient management of HG in reducing symptoms is still ongoing (ISRCTN24659467).
Pharmacologic interventions
According to the most recent Cochrane review on interventions for NVP, evidence on the effectiveness of pyridoxine (vitamin B6), ginger, and antiemetic medication is inconsistent. 3 Pyridoxine mainly reduces nausea but not vomiting. However, pyridoxine in combination with antihistamines (H1-receptor blockers including doxylamine and meclizine) significantly reduces nausea and vomiting symptoms and is not associated with teratogenic effects. 28 Phenothiazines (eg, phenergan) and dopamine antagonists (eg, metoclopramide) may be used as second-line treatments, followed by 5-hydroxytryptamine-3 receptor antagonists (eg, ondansetron). They are all described to reduce nausea and vomiting symptoms but might have maternal side effects, and potential teratogenic effects have been less studied. 13 The use of corticosteroids (CCS) has been advocated. 29 CCS are often used for chemotherapy-induced nausea and vomiting, but strong evidence for their effectiveness in the management of HG is missing. 30 Several case studies have reported that H. pylori eradication can effectively relieve symptoms in women with persistent HG who are unresponsive to conventional treatment, although an RCT is yet to be conducted.31–33
Nonpharmacologic interventions
Mindfulness-based cognitive therapy has just been proved to effectively reduce symptoms in women with moderate NVP, 34 but it has not yet been studied in women with HG. Dietary advice based on anecdotal evidence includes frequent small meals and avoidance of spicy, fatty, and odorous foods. 35 Nutritional interventions for HG have never been studied in clinical trials. This is surprising because poor nutritional intake36,37 and maternal weight loss37,38 are important features of HG, and substantial evidence indicates that poor maternal nutrition in early gestation has permanent detrimental effects on short- and long-term health of the offspring. 39 Small case series40,41 indicated that enteral tube feeding alleviated symptoms and was well tolerated when continued in a home setting. Furthermore, a retrospective cohort study by Stokke et al 42 suggested that enteral tube feeding improved maternal weight gain during pregnancy in women with severe HG and substantial early pregnancy weight loss. Currently, the first RCT on enteral tube feeding in HG patients is being performed (NTR4197; www.studies-obsgyn.nl/mother), to investigate the extent to which enteral tube feeding can affect birth outcomes, as well as reducing nausea and vomiting symptoms or time in hospital.
Are there Adverse Maternal Outcomes?
Wernicke encephalopathy is a very rare but potentially life-threatening complication of HG, caused by thiamine deficiency. 13 Patients are also at risk of developing refeeding syndrome when nutrition is not introduced carefully after a period of starvation. 43 A much more common consequence of HG with great impact is the psychological distress experienced by patients. This might result in termination of pregnancy 44 and increases the risk of depression, anxiety, and posttraumatic stress disorder.45,46 No studies thus far have yet addressed whether potential treatments have any beneficial effects on maternal well-being.
Are there Adverse Offspring Outcomes?
HG is repeatedly associated with adverse birth outcomes. A systematic review and meta-analysis on offspring consequences of HG showed increased risk of low birth weight, small for gestational age, and prematurity. 47 However, not all studies found adverse outcomes as a consequence of HG, 48 which might be due to heterogeneous patient populations. Maternal characteristics partly determine pregnancy outcome in HG patients, 15 but it is also likely that other factors associated with HG, such as insufficient weight gain during pregnancy, 42 maternal stress responses during pregnancy, 49 or H. pylori infection involving the placenta, 50 could each contribute to adverse birth outcomes associated with HG.
Thus far, reports on possible long-term consequences of HG beyond the perinatal period are limited. One study 51 reported the association of HG and testicular cancer, but a large registry study 52 found no increased cancer risk in HG offspring. Two studies53,54 reported on neurodevelopment in children, with conflicting findings, although there are some indications that risk of psychological and behavioral problems in adulthood is increased. 55 Lastly, Ayyavoo et al 56 recently showed that insulin sensitivity in children of hyperemetic mothers is decreased, and others found that early pregnancy weight loss was associated with increased blood pressure in offspring, 57 suggesting that risk for cardiometabolic disease in later life might be increased.
Can We Improve HG Research?
HG severely affects the lives of many women and their families, and better understanding of causes, consequences, and treatment options is needed. The lack of guidelines for the treatment of HG might have its origin in the inability to synthesize results across studies in systematic reviews. It is clear that the lack of consensus on a definition, diagnostic criteria, and classification of HG can hamper progress in the context of both clinical trials and clinical practice. Primary outcomes for HG are numerous, often differentially reported and performed in small trials.
Tables 1 and 2 illustrate the variations in HG-defining characteristics and outcome measures in eight clinical trials investigating the effectiveness of metoclopramide and promethazine.58–65 We chose these studies because they were specifically designed to study the effectiveness of antiemetic medication in patients with HG (and not NVP). Although there are similarities, only the trials by Abas 58 and Tan 60 used the same set of defining characteristics and outcome measures (Table 1). Both trials were conducted by the same research group. Gestational age58–62,64,65 and ketonuria58–61,63,64,66 were included in the definition by all but one trial. Sullivan 63 did not include gestational age, while Neri 63 did not include ketonuria. Weight loss,59,61–65 vomiting episodes59,61,62,64,65 and hospitalisation for HG58,60,61,64 were used by at least four studies but these did not completely overlap. In addition, the definition of each criterion varied widely. Regarding trial outcomes (Table 2), five studies58–60,62,65 reported vomiting episodes and nausea severity, of which four58,60,62,65 also included some, but not identical, measure of well-being. Interestingly, only one study 62 included birth outcomes.
HG-defining characteristics in published randomised controlled trials on metoclopramide and/or promethazine for HG.
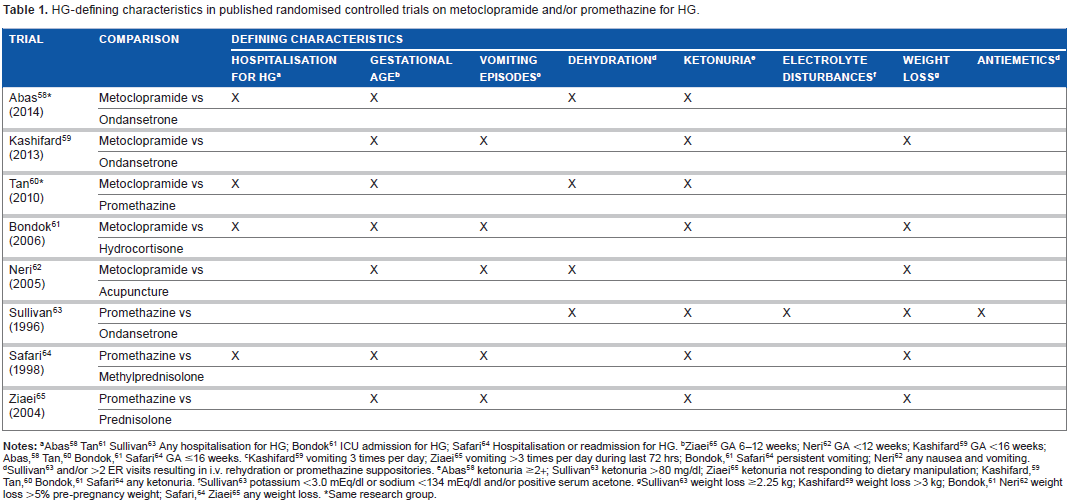
Abas 58 Tan 61 Sullivan 63 Any hospitalisation for HG; Bondok 61 ICU admission for HG; Safari 64 Hospitalisation or readmission for HG.
Ziaei 65 GA 6-12 weeks; Neri 62 GA <12 weeks; Kashifard 59 GA <16 weeks; Abas, 58 Tan, 60 Bondok, 61 Safari 64 GA ≤16 weeks.
Kashifard 59 vomiting 3 times per day; Ziaei 65 vomiting >3 times per day during last 72 hrs; Bondok, 61 Safari 64 persistent vomiting; Neri 62 any nausea and vomiting.
Sullivan 63 and/or >2 ER visits resulting in i.v. rehydration or promethazine suppositories.
Abas 58 ketonuria ≥2+; Sullivan 63 ketonuria >80 mg/dl; Ziaei 65 ketonuria not responding to dietary manipulation; Kashifard, 59 Tan, 60 Bondok, 61 Safari 64 any ketonuria.
Sullivan 63 potassium <3.0 mEq/dl or sodium <134 mEq/dl and/or positive serum acetone.
Sullivan 63 weight loss ≥2.25 kg; Kashifard 59 weight loss >3 kg; Bondok, 61 Neri 62 weight loss >5% pre-pregnancy weight; Safari, 64 Ziaei 65 any weight loss.
Same research group.
Outcome measures in published randomised controlled trials on metoclopramide and/or promethazine for HG.

Abas, 58 Tan, 60 Ziaei 65 number of episodes; Kashifard 59 VAS-score; Neri 62 vomiting episodes improvement; Safari 64 symptom improvement based on vomiting episodes, ability to tolerate fluids and well-being.
Abas, 58 Kashifard, 59 Tan, 60 Sullivan, 63 Ziaei 65 VAS-score; Neri 62 nausea severity improvement.
Sullivan 63 daily ketonuria.
Length of hospital stay following the intervention.
Bondok 61 ICU readmission within 2 weeks of study completion; Safari 64 hospital readmission within 2 weeks after start intervention.
Sullivan 63 daily weight.
Abas, 58 Tan, 60 Sullivan 63 treatment failure; Abas, 58 Tan, 60 Sullivan 63 use of antiemetics; Tan 60 duration of i.v. rehydration; Bondok 61 serum albumin; Neri 62 food intake improvement; Neri 62 birth outcomes.
Same research group.
Not only varying outcome measures, but also the diversity of interventions studied, limit the possibility of meta-analysis. Communicating study protocols might overcome this problem. Furthermore, the patient perspective is hardly recognized to play a role in HG management, 66 although patient involvement could help prioritize the research agenda. In other fields of medicine, this problem has long been acknowledged, and methods to reach consensus on definition and core outcome sets (COS) have been developed.67,68 The Delphi process is a method that can be used to reach consensus among relevant stakeholders, including patients, to formulate a definition and a COS that should be reported in clinical trials 69 to ultimately improve implementation of research findings in clinical practice. Currently, a Delphi process to formulate a definition and a COS for HG is in preparation (www.comet-initiative.org).
Conclusion
If clinicians do not know how to approach and treat HG patients, this is likely to affect patient care. Although an internationally accepted definition and the formulation of a COS is no panacea, we believe researchers, clinicians, and patients would benefit greatly. Bringing stakeholders together could further increase collaborative research on HG and enable prioritizing research questions that are relevant not just to researchers and clinicians but also to patients.
Author Contributions
Conceived and designed the manuscript: IJG, RCP, TJR. Analyzed the data: IJG. Wrote the first draft of the manuscript: IJG. Contributed to the writing of the manuscript: RCP, TJR. Agree with manuscript results and conclusions: IJG, RCP, TJR. Jointly developed the structure and arguments for the paper: IJG, RCP, TJR. Made critical revisions and approved final version: RCP, TJR. All authors reviewed and approved the final manuscript.
