Abstract
Essential tremor (ET) is the most common pathological tremor characterized by upper limb action—postural tremor (PT)/kinetic tremor (KT). There are no specific neuropathological or biochemical abnormalities in ET. The disability is consequent to amplitude of KT, which may remain mild without handicap or may become disabling. The most effective drugs for sustained tremor control are propranolol and primidone. Symptomatic drug treatment must be individualized depending on the circumstances that provoke the tremor-related disability. Broad guidelines for treatment are discussed in this review. Patients may be treated intermittently only on stressful occasions with propranolol, clonazepam, or primidone monotherapy, or an alcoholic drink. Those with persistently disabling tremor need continued treatment.
Introduction
Tremor is an involuntary oscillation—to and fro—movement of a body part. 1 It is a rhythmic, regular, repetitive movement, 2 and can involve any part of the body. Tremor is the most common involuntary movement, and essential tremor (ET) is the most prevalent pathological tremor in human adults. 3 Familial tremor was first described in the English literature by C.L. Dana in 1887.4,5 The first detailed description of all familial and sporadic ET cases was provided by L. Minor in 1920s,4,5 and the condition soon became known as Minor's disease. 6 Some physicians started referring to it as “minor tremor” or “minor cerebellar tremor,” which created confusion. Therefore, these terms were abandoned. 6 Over the years, the terms “essential familial tremor,” “benign essential tremor,” and “heredo-familial tremor” were used but they have all been discontinued. The term “benign” is not warranted as in some ET cases, the tremor produces significant disability.7–10 Because all cases do not have a family history of tremor, the term “heredofamilial tremor” is not justified. Based on onset age, the terms “infantile,” “juvenile,” “presenile,” or “senile tremor” that were used at one time have also been discontinued.5,11 The current terminology for this disorder is thus “essential tremor.”
Diagnosis of ET
The diagnosis of ET is based on clinical findings a lone, as there is no known biological marker. While different clinical criteria for ET diagnosis have been used,1,12,13 the differences between the sets of criteria are minor. They all include the presence of action—postural tremor (PT) and/or kinetic tremor (KT)—as the essential feature. The Movement Disorder Society consensus for ET diagnosis is the bilateral upper limb action tremor—PT and/or KT—though one may be more pronounced than the other.1,14 The tremor must not be consequent to any metabolic or toxic cause or to another neurological disease.1,12,14,15 These diagnostic criteria are applicable to ET in the adults. Although ET may manifest differently in children, by the time they reach adult age they develop typical ET. 6
Some experts recommend that the tremor be present for three to five years before ET diagnosis is made, and others suggest that the tremor must be present on both sides after three to five years of onset, 15 but it was regarded as arbitrary and is no longer used. 1 Neither of these is critical, as by the time a neurologist sees an ET patient, the duration of tremor is usually longer than three years and the tremor is almost always bilateral. 12 Although family history of tremor is present in a large proportion of ET cases, this is not a requirement for the diagnosis. 14
Onset and Course of ET
ET can begin at any age, though it manifests differently in the pediatric population. In infants, ET may manifest as shuddering attacks or generalized tremulousness. In children, it may be present as jerky and choreic movements, but by the second decade of life, the tremor becomes more sinusoidal (rhythmic). 6 The onset of tremor is insidious. Most patients recall retrospectively having had tremor under stressful circumstances for a long time, before the symptoms become more persistent. Physiological tremor, which is present in all healthy persons and may become visible under stress, does not transform into ET. 16 The most common site of tremor onset is the upper limbs, followed by simultaneous head and upper limb onset. 12 Even though ET may first manifest in only one upper limb, detailed neurophysiological studies show that there is a subclinical tremor in the other upper limbs as well. 2
The course of ET varies widely. In some patients the tremor remains mild, causing no functional disability, whereas in others it becomes progressively disabling. In those cases where the tremor is progressive, the evolution is characterized by the tremor involving more anatomical sites, 12 the tremor amplitude increasing,2,12,17 and the tremor frequency declining. 18 Approximately one-third of ET cases also develop resting tremor (RT), usually later in the course.3,12,19,20 RT in such cases is typically in the upper limbs, though at times it may involve the lower limbs as well.
The natural course in the ET cases, whose tremor worsens, is a slow progression, and ET does not resolve spontaneously. 6 There is no sudden worsening of ET without other cause. The functional disability in ET is related to the amplitude of upper limb KT.8,12,21–23 The life expectancy in ET patients is normal.4,24
Classical ET
Classical ET in the adults is bilateral, largely symmetrical, PT, and/or KT—although one may be more pronounced than the other. It involves the hands and forearms, with or without involvement of the head, and is visible to the naked eye. 1 The tremor frequency varies from 4 to 8 Hz. 3 PT is typically elicited when the arms are extended in front at right angle to the body. However, the tremor may manifest more prominently when the shoulders are abducted, elbows flexed, and hands held under the chin in a winged position. KT is typically tested by asking the patient to go back and forth, between touching the examiner's finger held in front of the patient (such that the patient would fully extend the arm at the elbow) and the patient's chin or the nose. If the tremor accentuates or appears only when nearing the target, it is called the intention tremor (IT) or terminal tremor, which is a well-known feature of ET. 1 Other methods for testing action tremor are drawing Archimedes spiral or a maze where the patient traces between the lines, 22 and during writing, drinking from a full cup, or pouring liquid from a pitcher.
Differential Diagnoses of ET
Many ET patients seen at specialty clinics come with an incorrect referral diagnosis.3,25,26 In our ET autopsy series, most cases were referred with Parkinson's disease (PD) diagnosis (unpublished). Following are common tremor disorders that need to be distinguished from ET.
Dystonic Tremor
When the tremor is present at the same body part that is affected by dystonia, it is classified as dystonic tremor. An example of this is cervical dystonia and associated head tremor. Dystonic tremor is typically irregular and jerky. It is most prominent when the body part is positioned in the direction opposite to the direction of the (abnormal) dystonic posture. It is a PT and/or KT, 1 but there is no RT in this case. While the most common site of ET is the upper limbs, dystonic tremor is relatively uncommon in the upper limbs but is most common in the head. A sensory trick (touching chin), which alleviates dystonia and dystonic tremor in many cases, does not improve the ET. 1 Isolated voice tremor may be part of dystonic tremor or of ET diathesis.
Physiological Tremor
It is either not visible or barely visible to naked eye. 3 When under stressful situation the physiological tremor becomes visible, it is called enhanced physiological tremor. It may be symptomatic, only with precise task under extreme stress. The tremor frequency ranges from 8 to 12 Hz, and the tremor is non-progressive. There is usually an identifiable reversible underlying cause (such as caffeine, sleep deprivation, hunger, or stress) for enhanced physiological tremor. Physiological tremor does not evolve into ET. 16
Drug-Induced Tremor and Metabolic Tremor
Many drugs including lithium, valproic acid, and antidepressants can produce ET-like tremor. Metabolic disorders like hyperthyroidism also produce tremor similar to ET. 1 Such causes of tremor can be excluded by history and by appropriate laboratory tests. Toxic/metabolic tremor is more rapid and is of smaller amplitude than ET, but clinically some of these cases may be indistinguishable from ET. It is best treated by stopping the offending drug (wherever possible) or correcting the underlying metabolic problem. Beta blockers are the choice for symptomatic treatment of toxic/metabolic tremor if necessary. 3
Writing Tremor
Writing tremor is divided into two types: (i) Type A—when the tremor occurs only during writing and (ii) Type B—if there is tremor when the hand adopts the writing position regardless of whether the individual actually writes or not. 1 In some cases writing tremor is a feature of ET, but in others additional dystonic posturing of the hand is evident, and as such they are classified as dystonic tremor. 1 We have followed some such patients with writing tremor, who responded to anticholinergic drugs.
Parkinsonian Tremor
Typically, Parkinsonian patients have RT at 4–6 Hz frequency. Some patients during early course of PD may only have action tremor without evidence of RT, bradykinesia, or rigidity, thus making it difficult to distinguish early PD from ET. 27 In the fully developed PD cases, KT is more common than is generally recognized.22,27 In PD patients, where the PT is prominent, the frequency of the PT is the same as of the RT.1,28
RT, which is typical of PD, is also present in approximately one-third of the ET patients seen in neurology clinics.3,12,19,20 Resting limb tremor should be verified with the patient lying on the table and the upper and lower limbs fully supported against gravity. Some experts consider the presence of upper limb tremor during walking as a fair representation of RT. We have observed that some patients manifesting upper limb RT while sitting in a chair have no RT in the supine position, when the limbs are fully supported against gravity. Thus for definite RT, the tremor should be evident when the patient is lying on examining table and arms and legs are fully supported.
It may take several seconds of rest before the RT emerges in the supine position. RT can be brought out by physical or emotional stress, such as counting backwards from 100 by serial 7's or activity with the other limb. For example, to elicit upper limb RT the patient is asked to forcefully dorsiflex the feet against the examiner's hands. Squeezing the examiner's finger with either hand can trigger lower limb RT, although lower limb RT is less common in ET than the upper limb RT. In PD, the postural upper limb tremor usually manifests several seconds after the arm is held in that position and is known as re-emergent tremor. But in the ET, the PT may manifest immediately on positioning the arms in front.
RT was first reported in the ET cases by Critchley in 1949, 4 but the first autopsy verification of RT in ET patients in the English literature was reported by Rajput et al. in 1993. 19 Typically, RT manifests late in the course of ET.3,12,19,20 If it is suspected that a patient with well-documented ET of long duration may have developed PD, all three cardinal parkinsonian motor features—RT, bradykinesia, and rigidity (preferably asymmetrical)—must be detectable to make the second PD diagnosis. 19
Cerebellar Tremor
Cerebellar tremor is slower (2–4 Hz) than the ET. The amplitude is variable and the rhythm is irregular. 3 In the upper limbs, it is primarily a proximal tremor in contrast to the distal tremor of ET. 1 Cerebellar tremor is evident with activity, and accentuates on reaching the target (IT). PT, which is a diagnostic feature of classical ET, 1 is not a part of cerebellar dysfunction. 1 In fact, cerebellar lesions may relieve PT in ET patients.29–31 Although RT is present in nearly one-third of ET cases, it is not a feature of cerebellar disorders. In the cerebellar disease there is ataxia and dysmetria, as well as rebound phenomenon. To test the upper limb PT in the sitting position, it is important to achieve steady upper limb position. In some cerebellar disease patients, truncal ataxia may be transmitted to the arms held perpendicular to the body, and may thus simulate postural upper limb tremor. In such cases, the examiner should stabilize the trunk by holding it firmly. In cerebellar disease, motor activity, such as finger-to-nose testing, is disrupted by ataxia, producing a slow zigzag pattern rather than a straight trajectory to the intended target—this ataxic movement can be misinterpreted as tremor. During an attempt to reach the target, the patient overshoots (dysmetria) and then compensates back to reach the intended point, and overshoots again. There is also a regular tremor characterized by worsening on attempting to reach the intended target or tremor appearing only when the hand is near the target known as IT. 1 Typical cerebellar tremor is easy to distinguish from ET in most cases. In the cerebellar disorders, there is also evidence of ataxia in the other body parts, such as during heel-to-knee-to-shin testing, where the action tremor is not obvious.
Pathophysiology of ET
The pathophysiology of ET remains unknown. There are no known pathological or biochemical findings that are characteristic of ET, and there is no suitable animal ET model. Most of the current knowledge is based on the studies of ET patients.
There are two components that contribute to the tremor in the ET—a central and a peripheral.3,6,32,33 The frequency of tremor is linked to dysfunction in the central component.6,33,34 Several central lesions—cerebellum,29–31 frontal cortex/subcortex,31,35 pons, 31 internal capsule, 31 and striatum, 36 —modify or alleviate ET. Thalamotomy and deep brain stimulation (DBS) of the ventral intermediate (VIM) nucleus improve ET. DBS of the subthalamic nucleus is also reported to improve ET.13,37 No central tremor pacemaker has been established, but the olivo-cerebello-thalamo-cortical motor circuit function disruption is believed to generate the tremor in ET.3,38,39 The peripheral component of ET modifies the tremor amplitude and is linked to adrenergic mechanisms in the muscle spindle. 38
Standard neuropathology studies have reported no consistent pathology in ET.11,12,40–43 Some recent morphometric studies have reported that all ET cases have brain histological changes—24% had lower brainstem Lewy body (LB) inclusions and 76% had cerebellar pathology, 44 notably Purkinje cell loss.44,45 Based on these studies, the terms “Lewy body variant of ET” and “cerebellar ET” were coined. 46 Other independent studies did not find a link between lower brainstem LB inclusions and ET.42,43,47,48 In addition, studies by others could not verify the Purkinje cell loss in ET.42,48–51 Some recent studies have reported altered gamma amino-butyric acid (GABA) function in ET brains.52,53 Yet these are early observations and need confirmation. There is also one report of excessive noradrenaline levels in the red nucleus and the locus caeruleus. 54 Thus, a definite anatomical or biochemical basis of ET remains to be established.
While the ET clinically manifests as action tremor, the pathogenesis of this clinical feature is likely heterogenous. 55 Some experts contend that there are abnormal central oscillations in the olivo-cerebello-thalamo-cortical loop that trigger as yet, unknown biochemical changes.3,38,56
Consideration of ET Treatment
ET is much more prevalent than the PD. In one study of 40 years and older community residents in Finland, the prevalence of ET was 5.6% whereas the prevalence of PD was 0.29%. 57 Thus, ET was 20 times as prevalent as the PD. 57 In a community study of elderly in Saskatchewan, we found ET to be five times more prevalent than the PD. 58 However, the needs for medical treatment are vastly different in the two disorders. Nearly every PD patient consults a physician and nearly everyone diagnosed as PD receives appropriate treatment. In the vast majority of ET cases, however, patients do not consult a physician for tremor and are therefore undiagnosed and untreated. A German study found that 27% of ET cases reported tremor to physicians, whereas a Finnish study reported only 11%.13,57 Possible explanations for this patient under-reporting include a mild and non-disabling tremor, the patient's interpretation that it is a familial trait rather than a disease, or the patient's belief that tremor is an age-related condition. In a study of 70 community residents of Saskatchewan aged 65 years and older who had detailed neurological examination, 10 (14%) had ET. 58 In another study of 67 institutional residents aged 65 and older, 7 (10%) had ET. 59 None of these cases were aware of their diagnosis.58,59 A community-based population study from Turkey noted that 93% of ET cases were unaware of their diagnosis, 60 and a population study of the elderly in Brazil noted that 91% of ET cases were undiagnosed. 61 In one community-based study from the USA, only 8% of ET cases were being treated for that disorder. 62 Another population study from Turkey found only 7% of the ET cases were diagnosed and treated. 60 Thus, there is major dissociation between the prevalence of ET in the general population and those who consult a physician and are diagnosed with ET and receive treatment. Those ET patients who consult a neurologist, we suspect, are an even smaller proportion than those who consult any physician for the tremor.
ET Patient Consulting Neurologist for Tremor
Most ET patients come to neurologists with specific questions in mind. Some may already have been diagnosed with ET and want to discuss treatment options, some suspect that they have ET but are concerned that they may have another condition (eg, PD), and others wish to find out what is causing their tremor. Misdiagnosis of ET is common. 11 Clinical diagnostic errors occur in both directions as there is no biological marker to make accurate diagnosis of ET.
At a clinic that has special interest in ET, 37% of the patients referred with the diagnosis of ET had another disorder. 25 In a large number of ET cases seen at specialty clinics, either a physician has made the diagnosis of PD or the patient suspects of having PD. 3 In our autopsy-verified ET cases (where the patients were mostly referred by family physicians), about 75% of cases were either taking antiparkinsonian drugs or had the referring physician diagnosis of PD (unpublished). When an ET patient who has an outside diagnosis of PD is told that she/he does not have PD and the ET diagnosis is explained, there is often a great sense of relief. 3 Many of those cases choose not to pursue drug therapy at that time.
Medical Treatment of ET
Medical treatment of ET is based on the severity of tremor, tremor-related disability, and the patient's perception of the quality of life. Most cases fall in the following four categories:
Mild tremor, which produces no functional or psychological disability/handicap and does not require treatment Mild to moderate tremor-producing disability only where there is tremor exacerbation in stressful situations such as social occasions or public speaking. These patients can be treated intermittently as necessary for these occasions. ET cases with persistent disability/handicap because of tremor. These patients need continued therapy to improve daily life function. ET cases that have persistent handicap but whose tremor persists despite appropriate pharmacotherapy. Alternatives to conventional pharmacotherapy should be considered in these cases.
The decision to initiate therapy is largely up to the patient, based on his or her perception of quality of life. The facts are that there is no cure for ET, nothing available slows its progression, the symptomatic drug benefit declines with time, and the life expectancy in ET is normal.3,13,24 These factors should play a role in the patient's decision to start symptom-controlling drugs.
ET drug therapy is broadly divided into intermittent and continued treatment categories as discussed later.
ET Drugs’ Evolution History and Current Status
The scientific basis of pharmacotherapy of ET is limited, as the underlying biochemical abnormality has not been established. It is well known that there is no drug that benefits all patients. Serendipity, studies of other forms of tremor, and experience with other disorders have contributed to the currently available drugs for ET.
Some patients who on their own discover that certain everyday consumed foods or drinks benefit their tremor may share that information with their physicians. Once the physician confirms such a benefit in some other cases, it becomes a treatment option. In this context, the most likely scenario is that the first agent reported to benefit ET cases was the socially consumed alcohol.4,11 In the 1950s and 1960s, it was widely held that improvement of tremor after alcohol intake was diagnostic of ET. We reported a study on the effect of alcohol on the ET and action tremor in several other disorders in 1973, 21 and published a more detailed paper in 1975 on 39 cases. 22 Included in that study were 21 ET, 15 PD, and isolated cases of cervical spinal cord injury, olivopontocerebellar degeneration, and ataxia telangiectasias with action tremor. 22 The patients were assessed by handwriting, maze tracing, and stringing beads at baseline and 30 minutes after ingesting one ounce (30 mL) of absolute alcohol mixed with water (60 mL). At final clinical assessment, blood alcohol levels were measured. In all, 62% of the ET and 47% of PD cases had improvement of action tremor after 30 minutes of alcohol intake. 22 The lone cases of ataxia telangiectasias and a case of C6 spinal cord injury resulting in paraplegia and upper limb weakness, manifesting as action tremor, also improved with alcohol. Figures 1 and 2 illustrate two such cases. Our studies established that alcohol-related benefit on action tremor is neither invariable nor specific for ET.21,22 Thus, we concluded that improvement on alcohol is not diagnostic of ET. Koller and Biary reported that the action tremor in 15 ET cases improved but the IT in 5 cerebellar disorder cases did not improve on intravenous alcohol infusion. 63 To our knowledge there are no other studies on the effect of alcohol on the action tremor in cerebellar disease, PD, or other neurological disorders.

Maze tracing by a 64-year-old female with ET of long duration: (

Maze tracing by a 52-year-old male suffering from ataxia telangiectasias: (
Blood alcohol level did not correlate with the tremor benefit in our study—some individuals with a trace or undetectable alcohol levels had tremor improvement. 22 Another study of five ET cases confirmed that there was no threshold of blood alcohol level for the symptomatic benefit in ET. 64 There is individual sensitivity for alcohol benefit in ET and other forms of action tremor.21,22,64 Alcohol benefit is characterized by reduction in the tremor amplitude but not in the tremor frequency. 63 One study, which used sufficient oral alcohol dose to attain serum level of 0.08%, noted that the tremor improved in ET after 10 minutes but the benefit faded 60–90 minutes later, and after three hours, there was tremor rebound. 65 Rebound was also detected in the morning after alcohol consumption during the preceding evening. 65
Alcohol is known to modify the abnormal central tremor oscillations in ET,33,64,65 presumably by its action on GABA receptors. 65 Central oscillators are autonomous neural networks, whose frequencies are independent of the reflex arc length, mechanics of the limb, or the sensory feedback. 33 We found that cases whose action tremor benefited from 30 mL oral absolute alcohol mixed with 60 mL of water—regardless of the cause of tremor—also benefited from propranolol for three to six months or longer21,22 (Figs. 1 and 2). However, propranolol benefited a larger proportion of cases than the single alcoholic drink did. 22 Another study included those ET cases that had a known history of alcohol improving their tremor. 63 Intravenous 200 mL of 10% alcohol infusion produced improvement in all ET cases but propranolol did not benefit all those cases. 63 As intravenous and intra-arterial alcohol uses are laboratory procedures, these observations have limited application to the practice of neurology.63,64 Considering that most ET patients who benefited from oral alcohol also improved on propranolol, which is a non-selective beta-adrenergic blocker,22,63 the pharmacological action of alcohol may be more complex than only the modification of central GABA receptor function.
Because of the short duration of symptomatic benefit on alcohol, the rebound of tremor, and the adverse effects of long-term alcohol consumption, it is recommended that alcohol be used in ET, for tremor relief, only occasionally. Our observations 22 indicate that more than half of the ET cases have temporary tremor improvement even with orally 30 mL of absolute alcohol.21,22 In spite of the commonly recognized symptomatic benefit on alcohol, ET patients do not consume alcohol more than the general population does.12,44,66,67
Currently Used Pharmacological Agents for ET
The common pharmacological agents used for ET are summarized in Table 1. The two most effective drugs to date are primidone and propranolol.13,68
Pharmacological agents for sustained control of essential tremor.

Current drug therapy for ET began with the discovery of barbiturates. These drugs have sedative and hypnotic action. Phenobarbital, the most common modern day barbiturate, was first introduced in 1912. 6 Barbiturates act by enhancing central GABAergic inhibition and, as other tranquilizers and sedatives including phenobarbital, are effective in some ET cases. 6 They may act by reducing anxiety and thereby reducing physiological tremor, but most likely, they have a direct effect on the central oscillator in ET. 6
We will briefly discuss these and other available drugs, and provide broad guidelines on the use of drugs for ET in the clinical practice. This classification is not precise as some drugs could fit in multiple classes. For purposes of this review, we classify the agents as (1) anti-convulsants, (2) beta-adrenergic blockers, (3) GABAergic agents, (4) calcium channel blockers, (5) atypical neuroleptics, and (6) botulinum toxin. We will focus on the most effective drug in each class.13,68
Anti-Convulsants
The most effective anti-convulsant drug for ET control is primidone. It was found to be effective in epilepsy in 1949 and in 1952 was approved for patients who did not benefit from the other available drugs. Its mechanism of action in ET is not fully understood. Primidone is metabolized into phenobarbital and phenylethylmalonamide (PEMA). These two main metabolites of primidone have very limited anti-tremor efficacy, 6 and thus primidone compound itself seems to directly suppress tremor. Primidone can be used once, twice, or three times a day on a regular basis. Its action is slow and the duration of benefit is long. Although some observers note that it is not suitable for intermittent use, we have observed intermittent use of primidone to be effective in some ET cases where tremor exacerbation by situational anxiety needed control. Primidone reduces tremor amplitude by 50%. 68 Although some benzodiazepines have major antiepileptic effect, we have classified them separately for use in ET. Other antiepileptic agents are noted in Table 1.
Beta-Adrenergic Blockers
In this class, propranolol is the most effective drug. 68 In 1968 while studying physiological tremor, Marshall suggested that beta-adrenergic blockers should be tried in ET. 2 Isolated ET cases benefiting from propranolol were reported in 1972 69 and in a large series in 1973. 21 In 1974, Winkler and Young reported on 24 ET patients treated with propranolol. 70 In more than half of the patients, the tremor improved on propranolol. 70 Propranolol is a non-selective, beta-adrenergic antagonist that has both peripheral and central effect. It reduces the tremor amplitude by 50%, 68 on both the accelerometry and clinical rating scale. The dosing regimen is individualized, based on the tremor control and the adverse effects. The details are noted in Table 1. It can be used intermittently, for situations that increase tremor, as well as on an ongoing basis. Other drugs in this class are also noted in Table 1.
Benzodiazepines/GABAergic Agents
Gabapentin and clonazepam are nearly equally effective in the short term. 68 However because of concern for drug dependence on continued use of clonazepam, gabapentin is the preferred drug in this class (Table 1). These can be used intermittently or regularly for longer term. The dosing regimen is noted in the table.
Calcium Channel Blockers
Nimodipine is the most effective drug in this class.71–73 Efficacy of nimodipine in ET was demonstrated by Biary et al. in a placebo-controlled study. 74 They used dose of 30 mg four times daily but a lower dose may also be effective. In all, 50% of their ET cases had tremor improvement. 74 It is a recommended treatment option by the practice parameters of American Academy of Neurology (AAN). 68 The dosing regimen is noted in Table 1.
Atypical Neuroleptics
Clozapine was reported to be effective in ET by Ceravolo et al. 75 and by Pakkenberg and Pakkenberg. 76 It is one of the drug treatment options reported by AAN practice parameters. 68 Clozapine is the most effective drug in this category but side effects include drowsiness, dizziness, and the rare but serious risk of agranulocytosis, which requires careful monitoring. 68 Other drugs are noted in Table 1.68,77
Botulinum Toxin A
Botulinum toxin is used by intramuscular injection. The upper limb injection improves PT but not the KT. However, there is no functional benefit to ET cases. 78 Hand weakness is a major adverse effect. 78 It is therefore not recommended for upper limb ET. The most effective use of botulinum toxin is for the head and the voice tremor. 68
Medical Management of Individual Cases
Medical treatment of ET must take into consideration several factors: (1) ET can begin at any age, (2) there are wide variations in tremor severity and tremor-related disability, (3) individual cases have varying abilities to cope with tremor, (4) treatment response including the benefit and development of the adverse effects is variable, and treatment failure is common, and (5) there is no cure for ET. The drugs are effective only for symptomatic control. Some patients may live more than 70 years after the ET onset and thus may need treatment for several decades.12,79 As noted above, the reasons why patients consult physicians vary widely. Treatment should therefore be individualized after consultation with the patient.
Figure 3 shows an algorithm of diagnosis and medical treatment of ET cases.
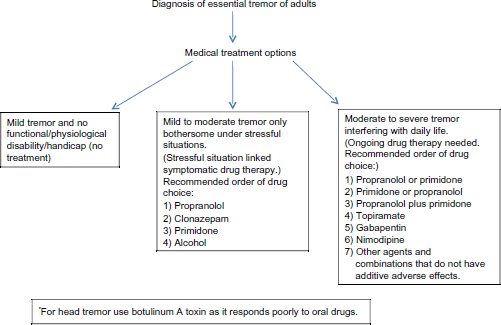
A summary of medical treatment options in patients with different severity of ET.
To Treat or Not to Treat
Once the diagnosis of ET diagnosis is made and the pertinent information is provided, the patient decides whether to pursue drug treatment. When there is minimal functional disability, many patients elect to forgo drug treatment.
Intermittent Treatment
Some patients are only concerned about the tremor becoming worse during stressful situations, such as public speaking, social gatherings, etc. Such cases need a short duration symptomatic tremor relief. In these cases, intermittent drug use is recommended. The choice of drugs is between propranolol, clonazepam, primidone, or a small amount of alcohol. The patient should be recommended to first experiment with the drug of choice at home, before trying it in public. The smallest possible dose is tried initially, which can then be adjusted upwards as needed. For example, the patient is recommended to take 10 mg of propranolol at home and make notes on the effect on tremor and any adverse effects (not to rely on memory). This should be tried twice again—each trial separated by three days. If there is insufficient benefit but no adverse effects on initial dose, the propranolol dose could be increased, eg, 20 mg, etc., as needed and tolerated. Similarly, 0.25 mg of clonazepam should be tried initially on three separate occasions at home, each separated by three days, and the patient should make notes on the response to treatment. With primidone, depending on the formulation available, 25–31.25 mg dose should be tried at home as above and the dose can be adjusted upwards if necessary. Standard one ounce (30 mL) absolute alcohol equivalent, one or at the most two drinks, for intermittent tremor control is another alternative. Once the patient has verified the pattern of response to a drug at home—latency, duration of tremor benefit, and the adverse effects—that drug can then be used in public, in a predictable fashion.
Continued Medical Therapy
The general principle is that the patient should be given a small dose as needed to control the tremor, without producing side effects. The dosing frequency and the dose of major drugs in each class is noted in Table 1.
There are very few studies of continued long-term medical treatment benefits in ET. However, it is known that on every drug, the symptomatic benefits decline with time. The two most effective currently available drugs are primidone and propranolol. Each can maintain anti-tremor efficacy in ET for more than one year. 68
Long-term continued anti-tremor drug treatment profile has been reported in one large study. 80 About one-third of the ET cases that were once started on drug therapy discontinued the treatment. 80 The lowest failure rate was with propranolol and primidone indicating that these drugs have a long-term sustained benefit.
Surprisingly, the discontinuation of the medication did not depend on the tremor severity, age, or gender. 80 Neither the duration of drug treatment before discontinuing the medications was clarified in this study nor were the reasons to do so identified. 80 No doubt the reasons included loss of efficacy and adverse effects of drugs.
Other Considerations
Most studies deal with reducing the upper limb KT amplitude, which is the primary cause of disability in the activities of daily living in ET cases. 23 Head tremor and voice tremor do not respond well to most of the oral medications, but it is worth trying different drugs. Some head tremor cases however benefit from propranolol, primidone, and topiramate. Botulinum toxin injection is effective in reducing head and vocal cord tremor. Rational polypharmacy where drugs from different classes are used in combination, for instance primidone and propranolol, is also useful in cases that are resistant to monotherapy.
It is not uncommon to switch from one agent to another for the control of tremor where necessary. The drugs noted below as well as others listed in Table 1 should be considered when the two primary drugs—primidone and propranolol—are ineffective or are not tolerated.
Different studies have come to different conclusions on the reported efficacy of some of the drugs in ET.13,37,68,77 The following represents a list of less commonly used drugs and drugs not listed in Table 1 that have been found effective in some studies of ET: sotalol, levetiracetam, flunarizine, zonisamide, mirtazapine, nifedipine, verapamil, phenobarbital, clonidine, olanzapine, and metoprolol.
The following drugs have been tried in isolated cases or small series and found ineffective: amantadine, glutethimide, L-tryptophan/pyridoxine, quetiapine, nicardipine, theophylline, pindolol, methazolamide, trazodone, isoniazid, and acetazolamide.
Authors’ Contributions
Analyzed the data: AHR, AR. Wrote the first draft of the manuscript: AHR. Contributed to the writing of the manuscript: AR. Agreed with manuscript results and conclusions, jointly developed the structure and arguments for the paper, and made critical revisions and approved the final version: AHR, AR. All authors reviewed and approved the final manuscript.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests. Provenance: the authors were invited to submit this paper.
Footnotes
Acknowledgements
We are grateful to Greystone Golf Classic, Regina Curling Classic and Royal University Hospital Foundation for their ongoing financial support of movement disorders research, and to Ms. L. Beatty and Dr. M. Rajput for their support in preparing this manuscript.
