Abstract
Apoptosis is an important cell death mechanism by which multicellular organisms remove unwanted cells. It culminates in a rapid, controlled removal of cell corpses by neighboring or recruited viable cells. Whilst many of the molecular mechanisms that mediate corpse clearance are components of the innate immune system, clearance of apoptotic cells is an anti-inflammatory process. Control of cell death is dependent on competing pro-apoptotic and anti-apoptotic signals. Evidence now suggests a similar balance of competing signals is central to the effective removal of cells, through so called ‘eat me’ and ‘don't eat me’ signals. Competing signals are also important for the controlled recruitment of phagocytes to sites of cell death. Consequently recruitment of phagocytes to and from sites of cell death can underlie the resolution or inappropriate propagation of cell death and inflammation. This article highlights our understanding of mechanisms mediating clearance of dying cells and discusses those mechanisms controlling phagocyte migration and how inappropriate control may promote important pathologies.
Introduction
Apoptosis, also known as programmed cell death, allows controlled removal of aged, damaged, infected or unwanted cells, maintaining homeostasis in multicellular organisms. This process would be functionally ineffective without clearance of resulting apoptotic corpses, as without disposal, they would progress to secondary necrosis and drive unwanted inflammation and autoimmune disease. 1 Effective clearance of dying cells is orchestrated by a balance of pro- and anti-clearance mediators that, under normal physiological conditions, skew towards phagocytic clearance as apoptosis proceeds. This process typically results in an overall anti-inflammatory phenotype. 2 However, if this balance is altered, clearance mechanisms can fail and pathology may ensue.
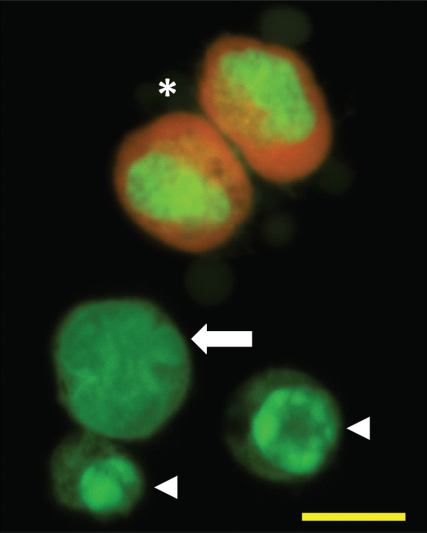
It is likely that much, and perhaps a majority of, apoptotic cell clearance occurs before dying cells show ‘classical’ morphological features of apoptosis (such as the characteristic nuclear changes associated with in vitro apoptotic death of lymphocytes—Fig. 1). Such death may be cleared by viable neighboring cells (e.g., epithelial cells), phagocytes that have often been called ‘amateur’ phagocytes, as their primary function is not phagocytosis of material (e.g., bacteria and cell debris). 3 However, it has been suggested that perhaps these local cells may in fact be the ‘professional’ phagocytes of early apoptotic cells, and that recruited phagocytes (e.g., macro-phages, whose primary function is phagocytosis of such material) are the professional scavengers of dying cells that have failed to be cleared sooner. 4 This latter situation will occur at sites of high level apoptosis 5 and will often be associated with pathological situations. 1
Effective clearance of dying cells requires the orchestration of a complex multi-stage process (Fig. 2). An early event, often neglected as it is not seen in simple in vitro culture models of apoptosis, is the recruitment of phagocytes to sites of cell death. This recruitment requires the release of attractants from dying cells and the consequent attraction of phagocytes. As the recruited phagocyte recognizes the ‘scent’ of dying cells it must migrate to and bind (i.e. tether) apoptotic cells prior to engulfing and degrading the cell corpse. Dead cell removal alone at inflammatory sites is insufficient to ensure resolution of cell death—emigration of phagocytes must also occur to restore the tissue to its previous state.
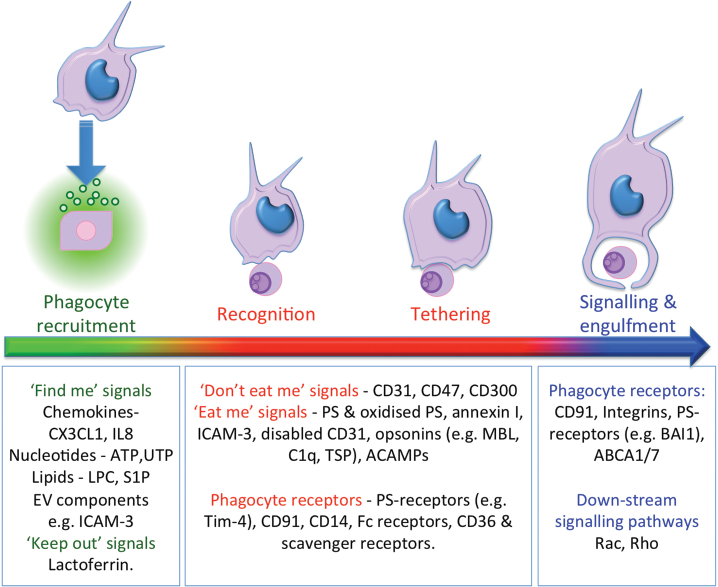
Ineffective clearance of apoptotic cells may thus arise from a defect in any one, or more, of these stages. Defects in genes related to clearance of apoptotic cells are associated with a variety of disease conditions, though it is not always clear whether the pathology arises directly from a failure to remove apoptotic cells. However, failed clearance of apoptotic cells is well-established in the pathogenesis of autoimmune disease, as persistent apoptotic cells ultimately lose their membrane integrity, thus promoting the production of autoantibodies to usually sequestered intracellular antigens that are released from necrotic cells. For example, defects in C1q expression (a soluble complement component known to opsonise apoptotic cells and mediate efficient corpse clearance) can drive systemic lupus erythematosus. 6 Furthermore, increased incidence of apoptotic cells is also noted in range of inflammatory lung diseases (reviewed in 7 ). Failed clearance of dying cells has also been implicated in inflammation associated with aging 8 and a number of age-related conditions, including arthritis, neuropathy and atherosclerosis (reviewed in 1 ). Atherosclerosis, the major pathology focus of this review, represents an important in vivo example of a site of high level cell death that is associated clearly with defective apoptotic cell clearance and failed emigration of phagocytes.
Within developing atherosclerotic plaques, large numbers of monocytes/macrophages are recruited to remove dying ‘foam’ cells (lipid-laden macrophages). However, with a failure in clearance and lack of phagocyte egress, further phagocyte recruitment exacerbates the problem. Collectively these events drive this important pathology,9,10 which is, perhaps, a key target for therapeutic intervention based on a detailed understanding of the mechanisms associated with apoptotic cell clearance. As risk factors for atherosclerosis, such as aging and obesity, are on the increase in the general population, it is imperative to gain a full understanding of cell clearance mechanisms, which may aid future management and treatment of these conditions. 11
This article will outline the molecular and cellular mechanisms proposed to function in concert for the efficient removal of apoptotic cells in vivo by monocytes and macro-phages, and will highlight areas that require further study to address the role of apoptotic cell removal in important pathological situations such as cardiovascular disease.
Dealing with Death—the Janitors
Macrophages are inherently plastic, switching phenotype in response to the local microenvironment. Classically-activated ‘M1’ macrophages are associated with a pro-inflammatory phenotype; for example, in response to pathogenic stimuli, whereas alternatively-activated ‘M2’ macrophages have a more reparative role, with functions in apoptotic cell removal and resolution of inflammation. 12 Researchers have tried to designate macrophage phenotypes according to characteristic responses and receptor profiles, but it is becoming clear that rather than definitive subtypes, macrophages exhibit a spectrum of activity that can continually alter, depending upon their microenvironment (reviewed in 13 ). Consequently, the macrophage phenotype will likely be crucial to the ultimate outcome arising from sites of cell death, with non-resolving chronic inflammatory sites supporting inflammatory phenotypes. Apoptotic cells are known to modulate the phenotype of phagocytes towards an anti-inflammatory phenotype. 2
Macrophages are often recruited from circulating blood monocytes rather than long-lived tissue-resident macro-phages. This is particularly true of macrophages infiltrating atheromatous plaques. Monocyte subtypes are highly studied, with surface receptors (CD14, CD16 and chemokine receptors e.g. CX3CR1) utilized to identify distinct phenotypic subgroups. 14 Expression of classical chemokine receptors may define those monocyte populations recruited to sites of cell death but detailed studies of recruited monocytes are still to be fully undertaken. This may be crucially important to outcome. CX3CL1, released in association with apoptotic cell-derived extracellular vesicles, has been shown to function as a chemoattractant for phagocytes. 15 This identifies CX3CR1high monocytes as an interesting research target in the field of apoptotic cell clearance, especially as CX3CR1 and CX3CL1 are both expressed on foam cells 16 and CX3CR1 is involved in monocyte recruitment to the atherosclerotic plaque, 17 a site of defective cell clearance (reviewed in 18 ).
Phagocyte Recruitment—'Find me’ versus ‘Keep out'
In the case of professional phagocyte (i.e. monocyte/macrophage) recruitment, evidence indicates clearly that ‘find me’ signals are released by apoptotic cells, either as soluble molecules or as part of complex extracellular vesicles. This relatively new area of apoptotic cell research addresses phagocyte migration to dying cells in a manner that is relevant to the in vivo clearance of apoptotic cells. In the past, apoptotic cell clearance studies have often relied upon a simple assay system where apoptotic cells were ‘fed’ to phagocytes in the presence or absence of putative blocking antibodies. Whilst fruitful, current in vitro studies are now directed to address the full extent of the processes within the apoptotic cell clearance program. Several different classes of attractant have now been identified as ‘find me’ signals released by apoptotic cells. Deficiencies, outlined below, in some of these signals or their receptors have been linked to inflammatory conditions.
CX3CL1 (fractalkine) is released by apoptotic human B cells in association with extracellular vesicles (often referred to as microparticles) and was, until recently, the only classical chemokine implicated in the recruitment of phagocytes to dying cells. It ligates the receptor CX3CR1 on the phagocyte surface to promote directional migration.15,19 CX3CL1 also increases expression of milk fat globule-endothelial growth factor 8 (MFG-E8), an identified bridging molecule between apoptotic cells and phagocytes, thus enhancing clearance. 20 More recently, however, cells induced to apoptosis through ligation of Fas/CD95 have been shown to release an array of cytokines and chemokines including MCP-1 and IL-8 that were demonstrated to be attractive to THP-1 monocytes and primary human neutrophils respectively. 21
Lysophosphatidylcholine (LPC) is a lipid mediator associated with phagocyte recruitment to dying cells 22 via interaction with the G-protein-coupled receptor (GPCR) receptor G2A.23,24 Whilst its precise role has been questioned, mice defective in the ‘find me’ signal receptor G2A develop an autoimmune syndrome. 25 It may be most important as an autocrine effector molecule, as it has been shown to induce expression of monocyte, neutrophil and lymphocyte chemoattractants monocyte chemotactic protein-1 (MCP-1), interleukin-8 (IL-8) and chemokine (C-C motif) ligand 5 (CCL5). 26 Sphingosine-1-phosphate (S1P) is another lipid mediator with roles as both a chemoattractant and immune-modulator. Interacting with GPCRs on the phagocyte, S1P also induces IL-8 and IL-10 release, whilst reducing tumor necrosis factor-α (TNF-α) and IL-12p70 production. 27 29
Nucleotides such as ATP and UTP have been proposed to be short-range chemoattractants, interacting with purinoreceptor-2 (P2Y2) on the phagocyte surface. 30 The possible short range of action of these attractants highlights the likelihood that multiple attractants will work together in effecting the efficient homing of phagocytes to sites of cell death. Whether some attractants are more active than others on different sub-populations of phagocytes remains to be determined, but these studies are necessary to provide a full picture of the mechanisms at work within the clearance of apoptotic cells. Recent work has also suggested that oxidative stress, even without cell death, may mediate selective recruitment of monocytes. 31 Such attraction to stressed, but not dying cells, may be a physiologically relevant mechanism by which phagocytes are deployed in readiness for cell deaths, though this may also promote disease.
Many of these chemoattractants, e.g., CX3CL1, are packaged in association with extracellular vesicles released following zeiosis, or blebbing, of the apoptotic cell membrane. Thus, extracellular vesicles play a key role in phagocyte recruitment.15,32 Torr et al (2012) demonstrated that the reduction in apoptotic cell surface intercellular adhesion molecule (ICAM)-3 was the result of its shedding on extracellular vesicles. 33 These vesicles were shown to be attractive to phagocytes in an ICAM-3-dependent manner. This effect was demonstrated using a vertical migration chamber where gravity exerts its effects in ‘driving’ phagocytes towards apoptotic cells in a lower chamber. Chemotaxis is modeled with varying success in vitro. Models are often highly simplified and results confounded by poor chemotactic gradients (e.g., ‘all or nothing’ step gradients in transwell-based assays) and the impact of gravity. Routinely now, horizontal migration chambers with time-lapse video microscopy can be used that rely on true chemotactic gradients and remove the impact of gravity on the assay. In these assay systems, phagocyte migration towards apoptotic cells can be clearly revealed and measures of phagocyte speed and direction can be made (Fig. 3).
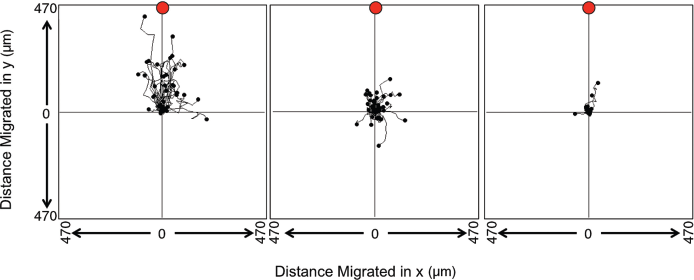
A limited number of molecules (e.g., ICAM-3 and CX3CL1) on apoptotic cell-derived extracellular vesicles have been identified to promote phagocyte attraction, and a full analysis of the physicochemical characteristics and molecular constitution (proteome, lipidome and glycome) has yet to be undertaken. This approach will likely yield important information that will pave the way to manipulating phagocyte recruitment. Furthermore, little is known of the cargo associated with the extracellular vesicles, though cargo carried by extracellular vesicles from other sources are known to have profound biological effects (reviewed in 34 ).
The nature of recruited phagocytes must also be managed carefully at sites of cell death due to the inflammatory nature of some phagocyte populations, and is a key stage with regards to immunological outcome. Some apoptotic cell-derived ‘find me’ signals are capable of recruiting neutrophils, which would result in an undesirable phlogistic reaction to apoptotic cells. Some examples of these signals are IL-8 released from Fas-mediated apoptosis of epithelial cells or LPC-induced IL-8 production in endothelial cells.21,26 Lactoferrin may provide an answer as to why neutrophilic infiltrates are not found at sites of persistent apoptosis. Lactoferrin acts as a ‘keep out’ signal, deterring granulocyte migration.35,36 This highlights again the need for balance between the mixed messages being relayed by apoptotic cells in order to maintain effective, immunologically-silent clearance, and further ‘keep out’ signals could yet be discovered.
The lineage of recruited phagocytes (e.g., granulocyte versus mononuclear) and their phenotype may be central to the elicited response at sites of cell death and the balance of ‘find me’ and ‘keep out’ signals will control this. Thus the ability to tip the balance in favor of ‘keep out’ signals represents an attractive therapeutic option to inhibit recruitment of phagocytes where this is not beneficial. For example, lactoferrin is known to reduce the migration of neutrophilic 35 and eosinophilic 36 granulocytes, and thus may be developed for the therapy of inflammatory conditions where granulocyte recruitment causes more harm than good. 37 For example, targeting of eosinophil apoptosis and clearance has been discussed in a recent review as possible intervention for asthma 38 and prevention of granulocyte recruitment may represent a therapeutic strategy. 36
Despite recruitment of phagocytes to apoptosis, there are sites of cell death that do not resolve (i.e., where the focus of cell death is not removed). The atherosclerotic plaque is a prime example of one such non-resolving inflammation. This pathological situation highlights how continued phagocyte recruitment may not be beneficial; phagocytes enter the plaque and remove dying cells, but this phagocytic clearance has been suggested to be defective.18,39 The phagocytes also become trapped at this site such that they ultimately die by apoptosis, leading to further monocyte recruitment.10,40 It has been suggested that oxidized LDL within the plaque environment may upregulate CX3CL1 expression, preventing macrophage egress, 16 whilst macrophage emigration from the plaque is also prevented by netrin-1. 10 Thus, when balance in phagocyte recruitment fails, it may lead to pathologies such as those in atherosclerosis, where either over-recruitment or a lack of cell egress contribute to pathology (reviewed in 40 ). Consequently, this site could benefit from modulation of the monocyte recruitment phase of apoptotic cell clearance. This may occur through recruitment of alternative phagocyte subsets—evidence suggests that monocyte sub-populations may influence disease, with sub-population counts being predictors of disease. 41 43 It may also be through blockade of recruitment such that the pathology, whilst not resolved, is halted in its progression. Targets for effecting such modulation might include CX3CL1, ICAM-3 and other ‘find me’ signals or ‘keep out’ signals. In support of this, mice defective in CX3CR1 show both reduced plaque size and phagocyte accumulation. 44
Phagocyte recruitment to important pathological sites (e.g., the atherosclerotic plaque) still requires further research. Sites such as these are difficult to model in vitro, with much of the work done in animal models; however, mechanisms between species are not always conserved, leaving gaps in research areas where murine models are prominent. For example, ICAM-3, which is released from apoptotic cells in association with extracellular vesicles and promotes macrophage recruitment to apoptotic cells, is absent from rodents. It is also unclear whether ‘find me’ signals and/or apoptotic cell-derived extracellular vesicles from foam cell death activate endothelium to recruit leukocytes, and which attractants are key to monocytes finding apoptotic cells within the plaque following transmigration.
Recognition and Tethering—'Eat me’ versus ‘Don't Eat me'
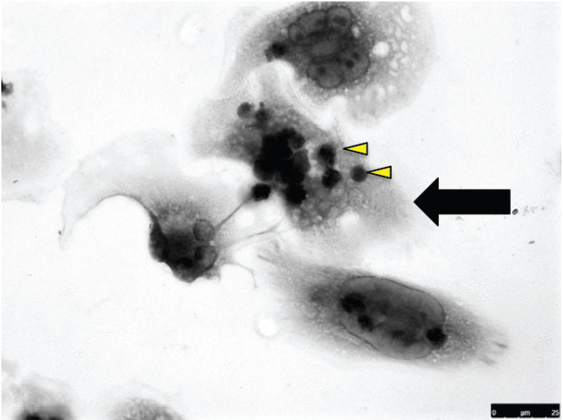
In order for phagocytes to begin the clearance process, target cells must be recognized as apoptotic. Recognition is the first stage of a 4-stage model, 4 followed by tethering (binding), tickling (signaling) and finally phagocytosis. Figure 4 shows a typical presentation of a human monocyte-derived macro-phage associated with apoptotic cells. The molecular players involved in these processes have been extensively reviewed elsewhere4,9,45 and so will be covered relatively briefly in this review. However it is clear that the apoptotic cell ligands and their cognate phagocyte receptors work as a team (in a phagocytic synapse) 46 to effect these different stages of the apoptotic cell clearance pathway, with some players solely involved in tethering and others involved in multiple stages (Fig. 2). Figure 5 depicts major molecular players underpinning the clearance of apoptotic cells.
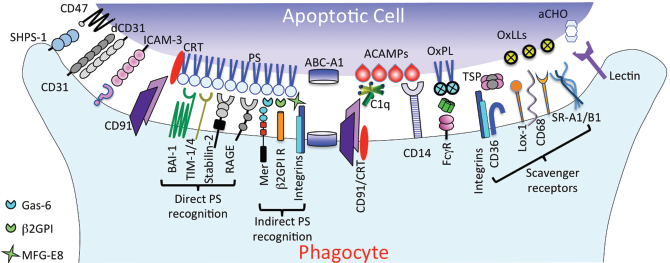
Molecular associations that drive apoptotic cell clearance.
As cells enter into apoptosis, ‘eat me’ flags are presented on the cell surface and are released on extracellular vesicles. The flags so far identified encompass exposure of intracellular molecules (e.g., phosphatidylserine (PS)47,48 and annexin I), 49 modification, redistribution and removal of cellular proteins, lipids and glycoproteins (e.g. modification of ICAM-3, 50 PS oxidation, 51 removal of sialic acid) 52 and binding of opsonins (e.g. mannan-binding lectin (MBL), C1q).53,54 Often the precise nature of molecular modifications that may occur during apoptosis remains ill-defined.
PS is the most extensively studied ‘eat me’ marker, which in viable cells is restricted to the inner leaflet of the plasma membrane and is redistributed during apoptosis.48,55 This exposure of PS through loss of phospholipid asymmetry is required for phagocytic clearance of apoptotic cells.47,48 It seems likely that this redistribution of membrane lipids also permits the movement of other surface molecules to indirectly promote apoptotic cell clearance. PS has been shown to bind to the phagocyte directly through phagocyte receptors T cell immunoglobulin mucin-1 (TIM-1) and TIM-4, 56 brain-specific angiogenesis inhibitor-1 (BAI1), 57 stabilin-2 58 and the Receptor for Advanced Glycation Endproducts (RAGE). 59 These receptors are not ubiquitously expressed indicating that different phagocytes for apoptotic cells will use a different set of receptors for clearance and they likely have different functions, as Tim-4 appears to solely tether apoptotic cells via exposed PS. 60 PS on the apoptotic cell may also bind to phagocyte receptors indirectly through molecular bridges: Gas-6 and protein S bridge to MerTK whilst MFG-E8 bridges to αv integrins. 61 The phagocyte receptor CD91 (LDL-receptor-related protein (LRP)) has been shown to bind PS when colocalized with redistributed calreticulin. 62 Calreticulin, upregulated during apoptosis, also associates with bridging molecules C1q and MBL for recognition by LRP. 54 Modification of PS, by mechanisms such as oxidation, also allows binding to phagocytic scavenger receptors such as CD36. 63 Scavenger receptors CD68, SRA-1, SRB-1 and LOX-1 also have roles in apoptotic cell recognition. 64 67
Mechanisms for the recognition of PS are clearly important and highlight, once again, the benefits to efficient clearance of dying cells, as defects in PS recognition genes are associated with important pathologies. For example in experimental atherosclerosis, a defect in Gas-6 promotes more stable plaque development with reduced phagocyte presence, 68 possibly suggesting a role for Gas-6 in phagocyte recruitment/retention as well as in apoptotic cell clearance. However, defects in Mer, to which Gas-6 bridges PS, result in defective apoptotic cell clearance and support atherogenesis.69,70 Further work is required to explain this apparent paradox. Defects in Mer and Tim-4 are also associated with reduced apoptotic cell clearance and development of autoimmunity in mice.70,71 PS recognition via Gas-6 and MFG-E8 is also implicated in the efficient clearance of photoreceptor fragments, a homeostatic process central to retinal maintenance. Failed clearance leads to death of photoreceptors and degenerative eye conditions. 71 Similarly, Mer kinase is implicated in the pathology of retinitis pigmentosa, where patients may carry a natural mutation in Mer and thus have defective clearance of photoreceptor fragments. 72 Furthermore, reduced recognition of PS has also been implicated in Alzheimer's disease, where both increased neuronal death and reduced clearance, possibly from reduced MFG-E8 expression, 73 may contribute to the increased levels of apoptosis detectable in the Alzheimer's disease brain (reviewed in 71 ). Loss of integrins has also been associated with chronic ulcerative colitis and autoimmunity. 74 It is clear that efficient clearance of dying cells is essential to prevention of a range of important inflammatory diseases, but it is interesting to note that pathology tends to follow disruption of those clearance pathways that function to mediate phagocytosis and cell responses. 1
Other important bridging molecules in clearance have been identified, including collectins (MBL/SP-A/SP-D), complement components (C1q, C3b), pentraxins (PTX3, SAP and CRP), and TSP-1. 75 83 Soluble CD14 has also been observed to bind PS and AC, but a clear role in promoting removal of AC has proved elusive. 84 Antibodies also have a role in opsonising/bridging of ACs, with a recent study detailing thousands of IgG autoantibodies that are ubiquitous in human serum. 85
ICAM-3 expressed specifically on human leukocytes is another cell surface receptor that becomes modified, at least functionally, during apoptosis, providing a ligand for an as yet unidentified phagocyte receptor.33,50 It has been proposed that ICAM-3 may bind the pattern-recognition receptor (PRR) CD14, 50 though evidence of a direct interaction is lacking. The precise molecular changes that underpin the role of ICAM-3 as an apoptotic cell-associated ligand and promoter of phagocyte recruitment remain ill-defined, but may (as with other apoptotic cell ligands) lie in changes of its location on viable versus apoptotic cells.
Numerous PRR, which have a role in innate immunity/host defense, have been shown to mediate apoptotic cell clearance, prompting the hypothesis that some apoptotic cell ‘eat me’ markers are structurally analogous to PAMPs (pathogen-associated molecular patterns; e.g., LPS). These have been termed apoptotic cell-associated molecular patterns (ACAMPs).86,87 Recently anti-PAMP antibodies have been used to probe apoptotic cells to identify ACAMPs. 88 Scavenger receptors are a group of PRRs with identified roles in apoptotic cell recognition, with CD14 (the prototypic PRR) having a well-established role in responses to LPS through its functional association with signalling partner toll-like receptor 4 (TLR4). 89 However, CD14 also mediates tethering of apoptotic cells to phagocytes,84,90 though there has currently been no noted involvement of TLR4 in CD14-mediated binding of AC.
A role for immunoglobulin in dead cell removal through opsonisation of dying cells has been demonstrated.91,92 Polyreactive IgM antibodies play a role in this by binding to ligands on dead cells and much of the focus of such studies have addressed the role of IgM as a mediator of complement opsonisation to promote AC clearance.91,93 However, IgM has also been suggested to promote AC clearance in the absence of complement activation. 94 These studies have also tended to focus on the clearance of late apoptotic cells/secondary necrotic cells, highlighting how different clearance pathways may be tailored to a certain phase of cell death. IgM has also been recently reported to promote the clearance of extracellular vesicles (microparticles) released by apoptotic cells, 95 possibly implicating IgM in phagocyte responses to these (e.g., in chemoattraction). IgG-based immune complexes have also been suggested to opsonise apoptotic neutrophils to promote their clearance. 96
It has been shown that many viable cells constitutively express ‘eat me’ markers without being engulfed by phagocytes, suggesting that they are not sufficient for clearance, and highlight other mechanisms must be in play to tip the balance towards recognition.97,98 This counterbalance comes through the alteration or down regulation of ‘don't eat me’ signals, expressed on viable cells. These include CD300a, 68 CD31 69 and CD47, 45 which all vary in mechanism of function. CD300a is newly identified as an inhibitor of AC engulfment via competitive phospholipid binding on apoptotic cells.99,101 CD31 binds homophilically to prevent ingestion of viable cells, but its function is altered when CD31 on the phagocyte binds disabled CD31 on AC, as this leads to AC tethering. 100 Mechanisms of CD47 action are more fully defined. Found on viable cells, CD47 exerts its inhibitory effect via its receptor SIRPα, and clearance in vitro only occurs with CD47 disruption. 62 Changes in these inhibitory molecules allows ‘eat me’ signals to mediate apoptotic cell uptake though inappropriate function of inhibitory signals, which may lead to failed clearance and subsequent disease; for example, immune evasion via CD47 over-expression in leukemic cells. 102
Signalling and Engulfment
Once the balance of molecules tips towards recognition and tethering of the apoptotic cell, several downstream signalling pathways can be activated. This depends largely on the specific receptor complexes activated in the phagocytic synapse where a combination of receptor classes is engaged. The precise constitution of a phagocytic synapse on any given cell is yet to be defined. It seems likely that this will be variable depending upon the phagocyte and the target cell, but will comprise a sub-group of the array of molecules implicated in clearance.
Many apoptotic cell receptors co-localize with others for effective clearance, as some function only to tether apoptotic cells, and of particular interest are the receptors that recognize multiple ligands but with opposing immunological consequences. For example, CD14 responds in an inflammatory manner to ligation with LPS; however, it also functions to tether apoptotic cells in a non-inflammatory manner.84,90 The precise mechanism by which receptors such as CD14 elicit ligand-dependent responses has yet to be fully elucidated, but altered signalling partners may provide one mechanism. 103
Activation of αv integrins, 104 the Tyro3, Axl, Mer (TAM) family receptor Mer, 105 and brain-specific angiogenesis inhibitor 1 (BAI1) 57 which all recognize PS either directly or indirectly, activates CrkII-Dock180-ELMO complex, which initiates Rac activation via GDP-GTP exchange. 106 This results in Scar/WAVE mediated cytoskeletal rearrangement.107,108
LRP-1 and Stabilin-2 binding initiates a second pathway via adaptor protein enGULfment adaPter protein (GULP) interaction, which activates ABCA1 and/or ABCA7. 109 111 Though the understanding of ABC-binding cassette transporters ABCA1 and ABCA7, involvement is unclear, there is evidence that this pathway converges with the CrkII-Dock180-ELMO pathway at the equivalent of Rac, CED-10, in C. elegans. 112 Further signalling pathways have been put forward, though research is so far restricted to C. elegans models. 113
Following engulfment, the phagosome becomes acidic and fuses with lysosomes, 114 resulting in apoptotic cell digestion. This process is key to regulating future events including further engulfment potential, 115 cytokine release2,116 and self-antigen presentation. Even at this late stage, defects in clearance mechanisms can have detrimental effects. 117
What Next for the Phagocyte?
The immunological consequences of apoptotic clearance are key to the success of the apoptosis program. The benefits of ordered cell deletion by apoptosis are evident when contrasted to the devastating consequences associated with cell necrosis. There seems little logic to expending energy to push a cell through apoptosis if the net effect is cell lysis and immune activation. Thus, the benefit of apoptosis is realized through its associated immune modulation. Apoptotic cell clearance promotes a non-inflammatory or actively anti-inflammatory response whilst neglect of apoptotic cells allows development of secondary necrotic bodies, and unwanted inflammation will ensue. 118 Full understanding of the balance of interactions that mediates the overall anti-inflammatory nature of apoptotic cell clearance could have implications for treatment of inflammatory conditions.
Resolution of inflammation is an active process, rather than the ebbing of an inflammatory response. The release of anti-inflammatory mediators has been reported in response to apoptotic ells, including TGF-β1, IL-10, PGE2, and PAF, along with suppression of mediators associated with inflammation, including TNF-α, IL-1, IL-12 and IL-8.2,116 Results also showed that apoptotic cells were able to dampen a pro-inflammatory response to LPS. 2 Other factors also play a role, including lipoxins, resolvins and protectins (reviewed in 119 ). At sites of inflammation, where large-scale clean-up of apoptotic cells can be expected, resolution is followed by immune cell egress into the lymphatic system and accumulation in the lymph nodes. 40 It has been proposed that defective cell egress at inflammatory sites may be exacerbated by factors within the local microenvironment, 40 e.g., secretion of netrin-1 by macrophages in the atherosclerotic plaque may cause leukocyte trapping. 10 Targeted netrin-1 deletion in murine macrophages promoted macrophage egress and reduced atherosclerosis. 10
Conclusions and Future Directions
An almost bewildering array of molecular players have been implicated in the recognition and removal of apoptotic cells though some functional redundancy is apparent. 120 In reality, the mix of molecules involved in clearance of any given apoptotic target cells by a phagocyte will be a simpler subset of those outlined above.
Effective and timely clearance of cells dying by apoptosis is a powerful mechanism by which inflammation and auto-immunity are avoided, despite the constant death of cells in vivo. Understanding the balance of molecules and mechanisms that underpin this control is the key to understanding many inflammatory disease states. Furthermore, this may lead to targeted interventions for the control of these diseases and the identification of novel anti-inflammatory therapies. Studies have shown that immune-modulation can be uncoupled from clearance.84,121 Understanding the fine details of phagocyte recruitment and the roles of individual mediators within the phagocytic synapse may provide direction for future treatments for pathologies including cancers, arthritis and cardiovascular disease. Novel treatment strategies will become more important as the population ages.
Author Contributions
Conceived and designed the experiments: LAH, AD. Analyzed the data: LAH, AD. Wrote the first draft of the manuscript: LAH. Contributed to the writing of the manuscript: LAH, AD. Agree with manuscript results and conclusions: LAH, AD. Jointly developed the structure and arguments for the paper: LAH, AD. Made critical revisions and approved final version: LAH, AD. All authors reviewed and approved of the final manuscript.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests. Provenance: the authors were invited to submit this paper.
Footnotes
Acknowledgements
The authors are grateful for the expert microscopy support provided by Miss Charlotte Bland of the Aston Research Centre for Healthy Ageing Imaging Facility and expert technical support provided by Miss Parbata Chauhan.
