Abstract
The rhizomes of
Introduction
Ginger (
Ginger Chemistry and Pharmacological Effects
The rhizome of ginger contains a wide variety of biologically active secondary metabolites. The rhizome comprises 1%–4% of volatile oils and an oleoresin.
6
The distinctive odor and flavor of ginger are due to these volatile oils and also nonvolatile phenolic compounds, which have pungent properties.
7
The volatile (steam extracted) oils consist mainly of sequiterpene hydrocarbons, predominantly zingiberol, which gives rise to the characteristic aroma of ginger. The nonvolatile phenolic phytochemicals of ginger consist of gingerols, shogaols, paradols, and zingerone, and more than 30 gingerol-related compounds can be fractionated from crude ginger.
8
Gingerols correspond to a series of chemical homologs differentiated by the length of their unbranched alkyl chains (
The major pharmacological activity of ginger appears to be attributed to gingerols and shogaols, which are the dehydrated products of gingerols. Consequently, gingerols are the major components in the fresh ginger rhizome, whereas shogaols, especially 6-shogaol, are the most abundant polyphenolic constituents of dried ginger. 7
In relation to its antiemetic properties, ginger (and its constituents) acts peripherally, within the gastrointestinal tract, by increasing the gastric tone and motility due to anticholinenergic and antiserotonergic actions.9,10 It is also reported to increase gastric emptying. 11 This combination of functions explains the widely accepted ability of ginger to relieve symptoms of functional gastrointestinal disorders, such as dyspepsia, abdominal pain, and nausea, which is often associated with decreased gastric motility. Although the exact mode of action of ginger in relation to its antiemetic properties is still being unraveled, three recent studies have investigated the action of ginger on serotonin (5-hydroxytryptamine, 5-HT3, and 5-HT4) and cholinergic (M3) receptor activities.12–14 Working on the evidence that emetogenic chemotherapeutic drugs increase 5-HT concentration and activate visceral vagal afferent nerve activity, Jin et al used patch-clamp methods to study the effects of ginger and its pungent constituents on 5-HT-evoked inward currents in rat nodose ganglia neurons. Results showed that 6-shogaol, 6-gingerol, and zingerone could inhibit the 5-HT response in a concentration-dependent manner, with 6-shogaol exhibiting the greatest potency. 12 Furthermore, the inhibition of 5-HT activity occurred in a noncompetitive manner, validating the earlier work. 10 Using a different methodological approach (calcium influx and radioligand-binding assays), Walstab et al. 13 used heterologous expression to demonstrate, for the first time, the inhibitory effect of 6-shogaol and 6-gingeral on recombinant human 5-HT3 receptors and also native receptors from human gut enteric neurons. This inhibition was found to be noncompetitive since a 5-HT3 receptor antagonist, GR65630, was not displaced by the ginger extract. Interestingly, both studies posited that since binding of ginger to 5-HT receptors occurs at a site other than the orthosteric-binding site of competitive 5-HT antagonists, combination therapy with known pharmaceutical 5-HT antagonists might increase the antiemetic efficacy. Additionally, using a bioassay for contractile (M) 3 receptors (guinea pig ileum), Pertz et al. 14 demonstrated that 6-, 8-, and 10-gingerol and 6-shogaol could slightly but significantly depress carbachol-induced contractions. Collectively, these studies provide molecular evidence that ginger antagonizes activation of (M) 3 and 5-HT3 receptors, thereby inhibiting afferent inputs to the central nervous system that are stimulated by specific neurotransmitters, such as serotonin, released from the gastrointestinal tract.
Ginger has also been studied extensively
Presentations of Ginger
Ginger is used in numerous forms, including fresh, dried, pickled, preserved, crystallized, candied, and powdered or ground. Presentations can include capsules, tablets, tinctures, teas, and liquid extracts. Evidently, the concentrations of active ingredients (gingerols and shogaols) will differ between the different preparations and the processing steps involved. Indeed, gingerols are thermally labile, and the extent of gingerol-to-shogaol conversion will likely impact significantly on the medicinal benefits since the two compounds vary in their bioavailability and pharmacological properties. 18 A recent methodological analysis using high-performance liquid chromatography (HPLC) coupled to time-of-flight mass spectrometry demonstrated that dried ginger powder products contained the highest quantity of gingerol-related compounds (7–14 mg/g), followed by fresh ginger (2–2.8 mg/g) and powdered ginger tea products (∼0.8 mg/g). 19 Attempts to assess the efficacy of ginger in many clinical trials might have been conceivably weakened by the inconsistency in the form of ginger used (fresh or dried) and also the dosing regimen. Of the 12 studies reviewed in a recent meta-analysis on the use of ginger in NVP, various preparations were described, including ginger biscuits, ginger powder capsules, ginger essence capsules, ginger extract capsules, and ginger syrup in water. 20 Also, the daily dosage varied from 600 to 2500 mg. Similarly, in a recent systematic review on the use of ginger in CINV, typical dosing regimens were 1–2 g of ginger. 21 To obtain patient compliance, it would be necessary to formulate ginger into the dosage forms that are practical to use, while retaining the active components. In this respect, capsules are the common dosage form considered for many oral drugs and different methodologies exist for the preparation of active gingerols (and shogaols) in capsule form. 22 In the above meta-analysis of NVP, 10 out of 12 studies used ginger in a capsule form and 7 out of 7 the studies reviewed for CINV used encapsulated ginger. 20 Considering the dosage, there is a remarkable lack of information on the concentration of active ingredients in the various preparations used in clinical trials; none of the NVP studies reviewed above described any form of chemical analysis to quantify the concentration of active ingredients, and only 2 out of 7 CINV studies did so. This is obviously essential information as commercial preparations of ginger may have widely different concentrations of gingerols. In a study of dietary ginger root supplements, Schwertner et al used HPLC to measure the concentrations of active ingredients in locally purchased ginger capsules. Results ranged from 0.0 to 9.43 mg/g for 6-gingerol, 0.16 to 2.18 mg/g for 6-shogaol, 0.00 to 1.1 mg/g for 8-gingerol, and 0.00 to 1.40 mg/g for 10-gingerol, and somewhat worryingly, the suggested daily dose varied from 250 mg to nearly 5 g. 23 The absence of standardized ginger constituents has also been highlighted in a recent study protocol to assess ginger in the setting of chemotherapy-induced nausea. 24 In this study design, the authors have chosen to use a commercially prepared ginger extract capsule that has been standardized to contain 5% gingerols (referring to the total gingerol strength), which is equivalent to 15 mg of active ingredient per 300 mg ginger extract and is an amount utilized in some previous clinical trials. 24
In addition to differences in the quantity of ginger used between studies, the dosing intervals also vary between clinical trials. In this regard, two recent clinical studies have investigated the pharmacokinetics of different ginger preparations in humans.25,26 In the first study, healthy volunteers were given a single oral dose of ginger, ranging from 100 mg to 2 g, and the blood samples were periodically taken up to 72 hours. Results showed that no free gingerols or shogaol could be detected in the plasma; however, these analytes were readily detected as predominantly glucuronide and sulfate conjugates in serum, indicating that gingerols undergo oxidation of their phenolic side chain. 25 Extending this analysis, the same authors developed a more sensitive technique and established that free forms of 10-gingerol and 6-shogaol, as well as glucuronide metabolites of 6-, 8-, and 10-gingerol and 6-shogaol could be identified one hour after oral dosing with 2 g of the ginger extract. 26 Interestingly, the half-life of these compounds and their metabolites was determined to be between one and three hours in human plasma, and multiple dosing experiments established that no accumulation of metabolites occurred. Given these results, it may be prudent to extend the frequency of dosing, within the expectations of patient compliance.
Although there is no consensus agreement on the correct dosage of ginger, most clinical studies recommend a safe daily dose of 1000 mg, at least in the setting on NVP.27–29 Accordingly, 7 out of 12 studies described in the Viljoen et al.
20
meta-analysis used this final amount, and a subgroup analysis in this report favored the lower daily dosage of <1500 mg for nausea relief. As a demonstration, Ding et al.
30
calculated that 1000 mg is equivalent to one teaspoon (5 g) of freshly grated ginger extract, 2 mL of liquid ginger extract, four cups (237 mL each) of prepackaged ginger tea, two teaspoons of ginger syrup (10 mL), or two pieces of crystallized ginger (1 in
2
). The
Clinical Effectiveness of Ginger
As stated earlier, the most common and well-established use of ginger is undoubtedly its utilization in alleviating symptoms of nausea and vomiting. In this sense, it is perhaps appropriate to mention the differences between nausea and vomiting. Nausea is characterized by an uncomfortable sensation experienced in the throat and epigastrum that may or may not result in the expulsion of contents from the stomach, while vomiting is the involuntary, forceful expulsion of contents from the stomach. Nausea and vomiting can occur separately, although since vomiting is nearly always preceeded by nausea, they are often considered components of a single entity. 32 Nausea is a nonobservable phenomenon, while vomiting is objective, and the occurrence and the frequency of vomiting may be measured. In the clinical setting, various instruments are used to assess nausea and vomiting based on self-reporting. 33 The INV-2 or Rhodes Index of Nausea, Vomiting, and Retching is an eight-item self-report questionnaire that measures nausea and vomiting as separate entities and is used frequently as an outcome measure in controlled studies. 34 Another common tool to measure nausea is a visual analog scale (VAS) of 0–10 cm to score severity.
Several medications are currently available for the treatment of NVP.43,44 Emesis can be treated with drugs known as antiemetics, most notably serotonin (5-HT3) receptor antagonists. However, many women are cautious of medicines for fear of harming the fetus, especially given that NVP usually occurs during the vulnerable period of embryonic organogenesis. Accordingly, the popularity of CAM, including nonpharmacological medicines and herbal extracts, has grown considerably in recent years, and a high frequency of CAM use during pregnancy has been noted. 45 A recent multinational study on the prevalence of herbal medicine use in pregnancy found that over 28% of participating women used herbals (2735/9459). 46 Of the 134 different herbs used, ginger and cranberry accounted for the majority of herbals (23.5% and 22.7%, respectively), with valerian and raspberry being also popular choices.
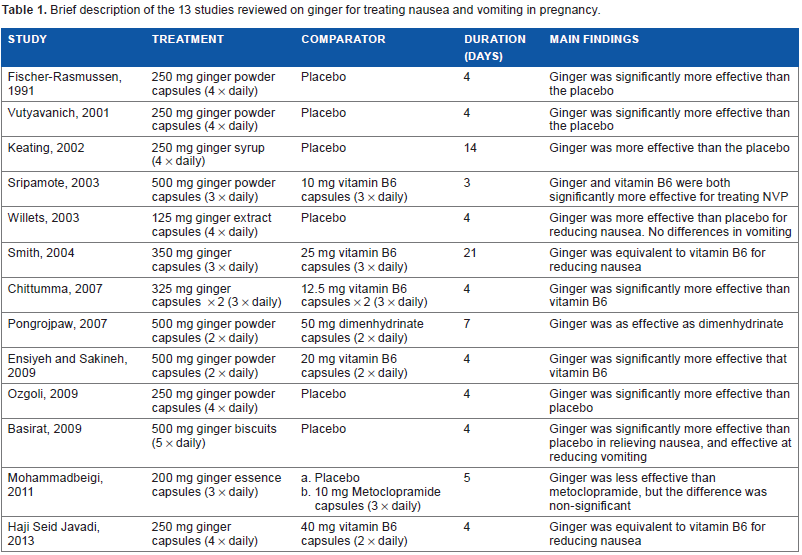
The impact of the use of ginger as an antiemetic in NVP has been extensively investigated in clinical studies for at least 30 years. 47 Because of the heterogeneity inherent in the study design and occasional problems with quality, not all randomized clinical trials can be incorporated into a meta-analysis. Nonetheless, two meta-analyses of randomized clinical trials (Level I evidence) have been published very recently.20,48 In the smaller of the two meta-analyses, six studies conducted from 1991 to 2009 fulfilled the inclusion criteria, and 508 subjects were randomly assigned to receive ginger (∼1000 mg daily) or placebo. 48 Predictably, these studies varied in the formulation and dosage of ginger: three studies administered 250 mg ginger capsules four times daily28,29,49; one study used 350 mg ginger capsules four times daily, 50 one study administered 250 mg ginger syrup four times daily, 51 and one study administered 500 mg ginger powder in biscuit, five biscuits daily. 52 Although the duration of study also varied (between four days and three weeks), using an end point of improvement of early NVP, the meta-analysis demonstrated that ginger was better than placebo in improving NVP when given at a dose of ∼1000 mg/d for at least four days. The authors of the meta-analysis concluded that ginger was an effective nonpharmacological option for NVP and was better than placebo. In the second systematic review and meta-analysis, Viljoen et al. 20 studied the efficacy of orally administered ginger as treatment for NVP in pregnant women at any stage of pregnancy and reviewed randomized studies from 1991 to 2011. From 302 records identified through database searching, 12 studies met the criteria established by the authors, involving 1278 pregnant women and included the six studies reviewed by Thomson et al. 3 The six additional studies used ginger capsules of different dosages: 125 mg four times daily, 53 200 mg three times daily, 54 325 mg (x 2) three times daily, 55 and 500 mg two times56,57 or three times 58 daily. Ginger versus placebo was assessed in 7 out of 12 studies.51–54 Individual results from all seven studies concluded that ginger was more effective than placebo in relieving the intensity of NVP in general; however, only three from the seven studies concluded that ginger was more effective in reducing the number of vomiting episodes (although there was a trend for improvement).28,29,48 In four studies assessing ginger versus vitamin B6 supplementation, a common first-line treatment for nausea, three studies reported no difference between the two groups,50,57,58 and one study showed that ginger significantly improved nausea and vomiting symptoms. 55 Besides this meta-analysis, a recently published study also found no significant differences between the ginger group (47 patients treated with 250 mg ginger four times daily) and the vitamin B6 group (40 mg twice daily). 59 One study assessed the efficacy of ginger against the antihistaminic drug dimenhydrinate and found ginger to be just as effective, with fewer side effects. 56 Lastly, one study used metoclopramide, a dopamine receptor antagonist, as comparator. The effects of ginger were not significantly different from those obtained to metoclopramide 54 In conclusion, Viljoen et al acknowledged the limited number of studies and low quality of evidence, but based on the evidence, ginger could be a possibly effective option for women with NVP, although large standardized trials are necessary. A brief description of the studies is given in Table 1.
Brief description of the 13 studies reviewed on ginger for treating nausea and vomiting in pregnancy.
Safety Issues of Ginger
Adverse effects after ingestion of ginger are uncommon but can include mild gastrointestinal complications, such as pyrosis (heartburn or reflux) 71 and eructation (belching). 50 In a study of 27 healthy volunteers who were given a single oral dose of ginger (ranging from 100 mg to 2 g), minor gastrointestinal upsets were the major treatment associated toxicities. 25 Despite previous studies indicating that ginger could interfere with platelet aggregation and cause excessive bleeding, 72 in a randomized crossover study of 12 healthy volunteers taking 1.2 g of dried rhizome three times daily for two weeks, ginger did not affect platelet aggregation and had no effect on the pharmacokinetics or pharmacodynamics of a single 25 mg dose of warfarin taken on day 7. 73 Of note, the meta-analysis of NVP by Viljoen et al also reviewed the safety of ginger as a secondary objective. The authors found that ginger did “[not] pose a risk for side-effects or adverse events during pregnancy.” 20
Aside from Level I evidence, safety of ginger in NVP has been studied in at least two Level II (nonrandomized or cohort) studies. In the first prospective study, the pregnancy outcome of 187 women from Toronto who were exposed to ginger during the first trimester of pregnancy was compared with women who had been exposed to nonteratogenic drugs that were not antiemetics. 74 There were no statistically significant differences between the two groups in terms of live births, spontaneous abortions, therapeutic abortions, birth weight, or gestational age. A more recent and larger population-based cohort study in Norway (68,522 women) found that the use of ginger during pregnancy (1020 women, 1.5%) was not associated with an increased risk of congenital malformations, still birth/perinatal birth, low birth weight, or preterm birth. 75
Conclusion
Ginger is an ancient herb used widely in history for its many natural medicinal properties and particularly as an antiemetic. The best available evidence demonstrates that ginger is an effective and inexpensive treatment for nausea and vomiting and is safe. Given the attainability of ginger preparations with known active ingredients, it would be interesting to perform preclinical studies to understand the efficacy of principal ginger constituents, including gingerols and shogaols. Dose-finding studies using varied standardized extracts should also be undertaken to accurately determine the effective dose and preparation of ginger. The results from these studies could be used to optimize the design of clinical trials to enhance the efficacy of ginger in nausea and vomiting.
Author Contributions
Conceived the concept: IL, JA. Analyzed the data: IL, JA. Wrote the first draft of the manuscript: IL, JA. Contributed to the writing of the manuscript: IL, JA. Agreed with manuscript results and conclusions: IL, JA. Jointly developed the structure and arguments for the article: IL, JA. Made critical revisions and approved the final version: IL, JA. Both authors reviewed and approved the final manuscript.
Footnotes
Acknowledgments
The authors are grateful to Adamed Farma for the unrestricted grant that made this article possible and to Kenneth McCreath and Nature Publishing Group for the editorial support.
