Abstract
Although dietary supplement use is common, its assessment is challenging, especially among ethnic minority populations such as Hispanics/Latinos. Using the Hispanic Community Health Study/Study of Latinos (HCHS/SOL) (n = 16,415), this report compares two strategies for capturing dietary supplement use over a 30-day period: a medication-based inventory and a nutrition-based dietary supplement interview. Age-standardized prevalence was calculated across multiple dietary supplement definitions, adjusted with survey/nonresponse weights. The prevalence of dietary supplement use was substantially higher as measured in the dietary supplement interview, compared to the medication inventory: for total dietary supplements (39% vs 26%, respectively), for nonvitamin, nonmineral supplements (24% vs 12%), and for botanicals (9.2% vs 4.5%). Concordance between the two assessments was fair to moderate (Cohen's kappa: 0.31–0.52). Among women, inclusion of botanical teas increased the prevalence of botanical supplement use from 7% to 15%. Supplement assessment that includes queries about botanical teas yields more information about patient supplement use.
Introduction
Growth in the number of marketed dietary supplements has been marked, increasing from 4,000 products in 1994 to 55,000 in 2009. 1 Dietary supplements result in >20 billion in sales each year with growth of about 6% per year 2 and prevalence of dietary supplement use is common in the United States (US) general public.3,4 In national studies, among Hispanics/Latinos, the reported prevalence of any dietary supplement use (34%) in the past 30 days was lower as compared with non-Hispanic whites (59%). 3 National studies of botanical and nonvitamin, nonmineral (NVNM) supplement use in the prior year also report lower prevalence among Hispanics/Latinos, 12% compared with 23%, 4 but other studies conducted within Hispanic/Latino populations have resulted in much higher estimates: >60% in the past 12 months,5,6 leading to questions about the comparability of these study designs.
Assessment of supplement use is challenging in any population, but is particularly difficult among ethnic minorities and with regard to botanical and other NVNM supplements. Most observational studies rely on self-completed questionnaires7–10 or telephone surveys 11 to collect dietary supplement data, asking participants to choose from a list of supplements that may or may not be consistent with cultural traditions. Product examination is considered a criterion standard for supplement assessment, 12 but, if not assessed in the home, could miss supplements individuals forget to bring in to the study visit. Moreover, disclosure of supplement use to healthcare providers has been suboptimal, potentially leaving patients open to adverse effects. 13 A more systematic approach to supplement assessment is clearly indicated.
The current study utilizes the baseline data of the Hispanic Community Health Study/Study of Latinos (HCHS/SOL). HCHS/SOL is unique in its supplement assessments in two ways. The study collected supplement data in a sample of US Hispanics/Latinos using both a dietary interview method and a medication-based (product examination inventory) method to assess supplement use over 30 days. In addition, supplement use was also captured in the 24-hour dietary recall data, reflecting immediate use. This analysis categorized dietary supplement types and measured their prevalence among Hispanics/Latinos of diverse Hispanic backgrounds by a medication inventory (MV) and a dietary supplement interview (DI). The analysis also addresses the prevalence of common supplement ingredients.
Methods
Although the Langual system nicely organizes dietary supplement ingredients, definitions of dietary supplement product types have not yet been standardized. In this study, based on the available product information, combination products that contained ≥50% of the recommended daily allowance (RDA) of a vitamin or mineral, and also contained a botanical or other NVNM ingredient were classified as a dietary supplement with NVNM. Those that contained <50% RDA of a vitamin or mineral were classified as a primarily NVNM product. Botanicals were similarly classified. NVNM products included botanicals, but botanical products excluded other NVNM (eg, glucosamine, coenzyme Q10, omega-3 and omega-6 fatty acids, fiber products, enzymes, probiotics, amino acids, and protein supplements) (Fig. 1). Based on these classifications, supplement users were defined as: (1) users of any dietary supplements; (2) users of supplements containing any NVNM (including botanicals); (3) users of supplements consisting of primarily NVNM ingredients (including botanicals); (4) users of supplements containing any botanical ingredients; (5) users of primarily botanical supplements.
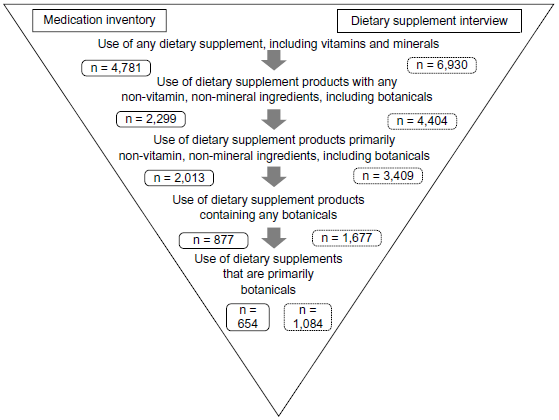
Dietary supplement assessment protocol.
Dietary supplement interview. At the time of the field center clinic visit, immediately following the 24-hour dietary recall, technicians interviewed participants (in Spanish or English, according to their preference) about their recalled dietary supplement use in the 30 days prior to the study visit. The dietary supplements from the 30-day supplement interview were recorded in the Dietary Supplement Assessment Module (DSAM) of the Nutrition Data System for Research database (NDSR). Information for supplements that did not match in the DSAM were updated from the product label (obtained from the manufacturer when possible) and validated against outside resources. DSAM-based product name files were recorded with greater detail than the medication inventory files, enabling refined product coding. The dietary supplement interview data consisted of ∼2,700 different products representing ∼15,000 entries. Calculations of the prevalence of individual botanicals within dietary supplement products depended on the DSAM ingredient-level dataset. Botanical supplement reports were limited to those with a prevalence of at least 0.2%.
Twenty-four-hour dietary recall. The 30-day dietary supplement interview files recorded a few botanical teas. However, botanical teas, other liquid dietary supplement products, and culturally specific botanicals appeared in the two 24-hour dietary recalls enabling the calculation of: (1) estimates of supplement use including botanical teas and (2) estimates also including green tea (Camellia sinensis) and nopal (whole fruit).
Because neither the dietary supplement interview nor the medication inventory could be considered a gold standard measure, comparison of the two was limited to calculation of Cohen's kappa statistics with positive and negative agreements 17 and prevalence and bias-corrected kappa (PABAK). 18 Kappa statistics were interpreted following established criteria. 19 In addition, to look at the sensitivity of dietary supplement prevalence estimates to various assumptions, estimates were calculated based on: (1) the dietary supplement interview updated with botanical teas from the 24-hour dietary recall; (2) the dietary supplement interview with 24-hour recall data, including nopal and green tea; and (3) the combined dietary supplement interview, medication inventory, and dietary recall.
Results
Distribution of sample population characteristics by the use of supplement typesa in HCHS/SOL.

Supplement users were defined as users by either the medication inventory or the dietary supplement interview. Categories presented for comparison of distributions include only users of any dietary supplement, users of primarily NVNM products (including botanicals), and users of botanical supplements (including fiber).
N, number of participants in sample, rather than the representative population.
Means and percentages reflect sample weights and are age standardized to the US population.
Physical activity data were summarized as meeting or not meeting CDC 2008 guidelines for physical activity.
Nutritional data were summarized with the Alternative Healthy Eating Index-2010 (AHEI-2010). The AHEI was developed to update prior healthy eating indices based on US Dietary Guidelines with recommendations derived from scientific studies of diet and health outcomes.33,34 The AHEI assigns values for consumption of vegetables, fruit, whole grains, sugar-sweetened beverages, nuts/legumes, red/processed meats, trans fat, long-chain n-3 fatty acids, total polyunsaturated fatty acids, sodium, and alcohol intake with a maximum value of 110. 34 Healthy individuals aged 55–80 in the Nurses’ Health Study had a mean AHEI-2010 of 53.2. 33
Users of botanical supplements only had a higher education and greater adherence to a higher quality diet as measured by the 2010 Alternative Healthy Eating Index. They were more likely to be of Mexican background and living in San Diego; they were less likely to have insurance or to have access to needed healthcare.
Comparison of the 30-day dietary supplement use assessed through the medication inventory and dietary supplement interview in HCHS/SOL a .
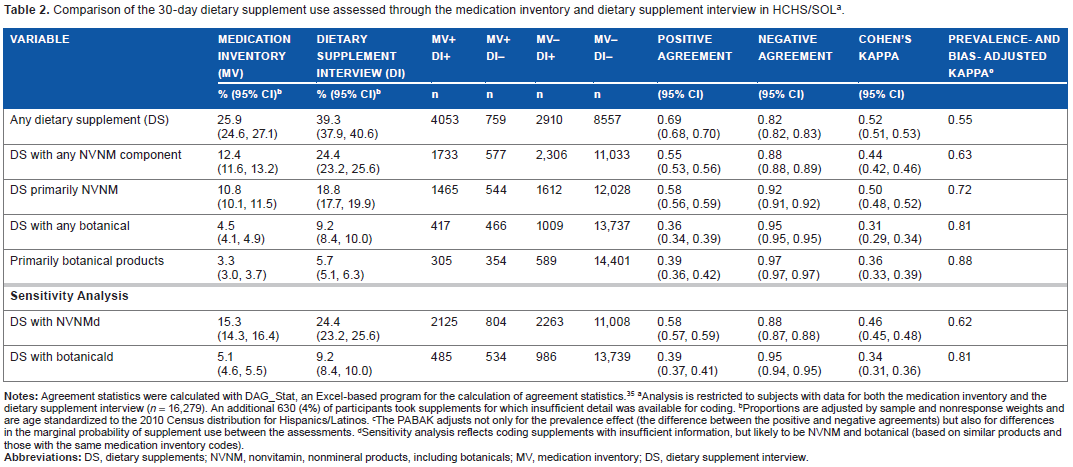
Analysis is restricted to subjects with data for both the medication inventory and the dietary supplement interview (n = 16,279). An additional 630 (4%) of participants took supplements for which insufficient detail was available for coding.
Proportions are adjusted by sample and nonresponse weights and are age standardized to the 2010 Census distribution for Hispanics/Latinos.
The PABAK adjusts not only for the prevalence effect (the difference between the positive and negative agreements) but also for differences in the marginal probability of supplement use between the assessments.
Sensitivity analysis reflects coding supplements with insufficient information, but likely to be NVNM and botanical (based on similar products and those with the same medication inventory codes).
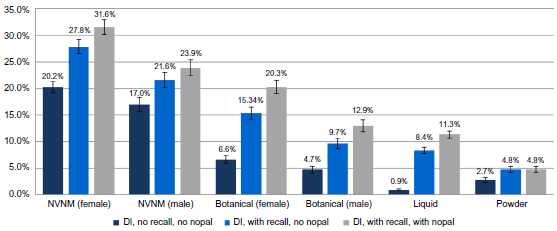
Prevalence of NVNM supplements considering the addition of supplements captured in the dietary recalls. Prevalence estimates are standardized to the 2010 US Census and weighted with sample and nonresponse weights and stratified by gender: ‘DI no recall no nopal’ references the dietary supplement interview data without dietary recall data. “DI with recall no nopal” references the dietary supplement interview data with the addition of dietary recall data, but excluding raw nopal and green tea. “DI with recall with nopal” references the dietary supplement interview data with dietary recall data, including raw/cooked nopal and green tea.
Prevalence of selected supplement ingredients in the 30-day dietary supplement interview in HCHS/SOL.
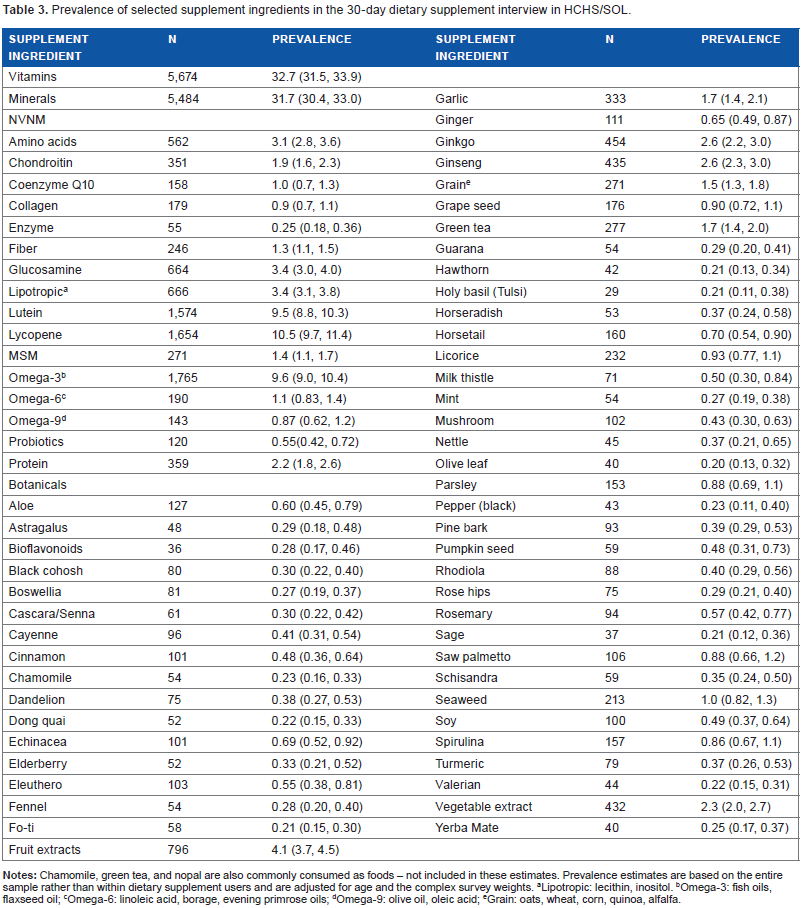
Lipotropic: lecithin, inositol.
Omega-3: fish oils, flaxseed oil;
Omega-6: linoleic acid, borage, evening primrose oils;
Omega-9: olive oil, oleic acid;
Grain: oats, wheat, corn, quinoa, alfalfa.
Many combination products containing vegetable or fruit extracts were counted as botanicals. Over 2% of study participants consumed a supplement with at least one vegetable extract and over 4% consumed a product containing a fruit extract. Culinary herbs, such as parsley, sage, and oregano were occasional constituents of combination products.
Ginkgo and ginseng were the most commonly reported botanicals (2.6%), followed by green tea extracts and garlic (1.7%). Licorice was also a component of botanical products among about 1% of participants. Chamomile at 0.23% and nopal at 0.13% were less common constituents of dietary supplement products – almost 2% of participants reported consuming chamomile tea in the dietary recall (1.9%; 95% CI: 1.6–2.3).
Discussion
Depending on the assessment (medication inventory and/or dietary supplement interview ± dietary recall data), HCHS/SOL estimated that 4.5%–19% of participants reported taking botanical supplements and 12%–32% reported use of NVNM supplements, including botanicals. Results of this study demonstrate the challenges of accurate dietary supplement assessments, particularly with regard to the botanical and other NVNM products. Estimates based on the medication inventory alone differed substantially from those based on the dietary supplement interview and neither assessment appears to have captured the use completely. Some products were reported only with the medication inventory and the others only with the dietary supplement interview. Botanical estimates in particular were markedly sensitive to varying assumptions about what constitutes a botanical product – including all fiber products as botanicals, a reasonable, but not universal assumption, markedly increased estimates, as did the inclusion of botanical teas and other liquid dietary supplements ascertained in the 24-hour dietary recall files.
Kappa statistics in studies comparing questionnaires with label capture have been variable, ranging from 0.46 to 0.92.12,21 Agreement of supplement use as measured by a telephone interview compared to a label capture was somewhat lower–with a kappa as low as 0.14. 20 A fourth study compared vitamin and mineral supplement assessment via a self-completed health questionnaire and two types of dietary assessments, a food frequency questionnaire and a 7-day food diary. 22 Agreement in these studies comparing the three instruments was substantial (k: 0.72–0.81).
In contrast, our agreement statistics are somewhat lower, especially for botanical products. Although every effort was made to classify dietary supplement products similarly in the two datasets, some misclassification was likely, given the amount of missing information in the medication inventory. In addition, a kappa statistic is less reliable in a setting of low prevalence, as seen with botanical supplements 23 and with imbalances in the marginal totals as was seen in both the botanical and NVNM data. 24 However, moderate agreement was seen with the comparisons of overall dietary supplement use and with the comparisons of NVNM supplements and negative agreement statistics were uniformly high.
Low agreement was not surprising, given the very different supplement ascertainment by the two systems. A review of medications is more likely to underestimate dietary supplement use. In clinical situations, accurate capture of dietary supplement use is uncommon: 33% of individuals with chronic disease reported that they had informed their healthcare provider of their supplement use and disclosure rates were lower among Hispanics (22%). 13 When asked why they did not disclose their supplement use, patients reported that they did not think that it was important for their providers to know. 25 A similar mechanism may be driving the lower prevalence in the medication inventory data in this study.
On the other hand, the dietary supplement interview benefited from both a systematic inquiry and the use of DSAM, a detailed database designed specifically for the capture of dietary supplement data. In addition, the dietary supplement interview followed a detailed interview about food intakes in the past 24 hours. Individuals may have been more likely to recall supplements they regarded as enhancements to their diet after a 24-hour dietary recall.
The value of the medication inventory and the dietary supplement interview independently could be called into question. However, together, and when combined with data from the 24-hour dietary recalls, a more complete picture of supplement use emerges. Comparisons of supplement assessments by these different techniques furnish important information about the impact of study design on reports of supplement prevalence in the Hispanic/Latino literature. Wide variations in botanical supplement prevalence may result from differences in assessment strategies as well as supplement definitions (including vs excluding botanical teas). In addition, because of the high probability of under-reporting, asking about supplement use multiple times is more likely to yield accurate estimates. In a study of home remedies for childhood diarrhea, researchers only learned of the botanical remedies after establishing a trusted relationship with participants. 26
Comparable studies were limited to four; most of them targeted an older population. In a home-based interview of dietary supplement use in the past two weeks with label capture among individuals aged ≥77 years in Texas, about 38% of Hispanics reported using vitamin or mineral supplements and 5% reported using botanicals, most commonly garlic, ginkgo, and saw palmetto. 28 Another study employed a similar strategy (home-based interviews, use in the past two weeks) among Mexican Americans aged ≥65 years across the US–Mexican Border States and reported botanical medicine use was about 10%. 29 In this latter study, investigators specifically inquired about the use of herbal teas and the most common botanical supplements were consistent with the previously reported favorites among Hispanics/Latinos: chamomile, mint, and aloe. 29 In another study, using a telephone interview of supplement and medication use in the past seven days 30 in the general US public, the prevalence of NVNM use was 12% among Hispanics; the most common supplements were lutein, ginkgo, garlic, and glucosamine, 30 similar to those reported in HCHS/SOL. Another telephone interview (subjects ≥52) reported Hispanic/Latino use of any supplement at 45% and botanicals at 12%.
Dietary supplement assessment suffers from the same limitations as other data largely dependent on self-report or inventory methods conducted outside the home. It is unlikely that the medication inventory or the dietary supplement interview alone adequately captured supplement use. In the medication inventory, because supplements were spread across the files, instead of receiving a designation as a supplement, some supplements could have been missed, artificially reducing the prevalence estimates resulting from these data. On the other hand, the dietary supplement interview could have been improved by the use of prompts for supplements common among Hispanics/Latinos (eg, chamomile, mint, and aloe) rather than those common in the general population (Echinacea, ginseng, ginkgo, St. John's wort). However, the dietary supplement interview data, using the DSAM, achieved much greater detail than has been reported in prior studies. The ability to add botanical teas from the 24-hour dietary recall data filled in some of the missing information from the dietary supplement interview alone. Moreover, the combination of instruments enriched the picture of dietary supplement use in the population.
An additional limitation of self-reported data is that consistency of use is impossible to determine. This limitation also applies to administrative data: even with prescription fill data, it is unclear whether or not individuals are taking the medications they purchase.
Author Contributions
Conceived and designed the study: AMSR, KRF, EAW, HMG, GAT, SFC, MLD, JB, REG, LVH. Analyzed the data: KRF. Wrote the first draft of the manuscript: KRF, AMSR. Contributed to the writing of the manuscript: KRF, AMSR, PG, JOR, LAY, EAW, CP. Agreed with manuscript results and conclusions: KRF, AMSR, PG, JOR, LAY, CP, EAW, HMG, DACM, GAT, SFC, MLD, JB, REG, LVH. Jointly developed the structure and arguments for the paper: KRF, AMSR, PG, JOR, LAY, EAW, CP. Made critical revisions and approved the final version: KRF, AMSR, PG, JOR, LAY, CP, EAW, HMG, DACM, GAT, SFC, MLD, JB, REG, LVH. All the authors reviewed and approved the final manuscript.
