Abstract
Tryptophan metabolism has been extensively studied in humans as well as in soil. Its metabolism takes place mainly through kynurenine pathway yielding hydroxylated, deaminated and many other products of physiological significance. However, tryptophan metabolism has not been studied in an isolated thermophilic bacterium.
Introduction
Tryptophan (Trp) is an important essential aromatic amino acid with a daily recommended requirement of 4 mg/kg body weight in humans with clinical implications.1,2 L-Trp is a precursor for many compounds such as niacin, and actinomycins
3
and its metabolism leads to many metabolites that may be physiologically valuable or even toxic.
4
These molecules may include quinolinic acid, indoleacetic acid, indole, indoxyl, skatole and numerous other compounds.5–7 Metabolic products of tryptophan such as serotonin upregulates appetite, sleep pattern and mood.
8
Its degradation via the kynurenine is the major degradative pathway leading to
Soil bacterial metabolism of Trp has also been studied for its conversion to auxins such as indole 3-acetic acid (IAA) for plant growth and development.19,20 Although many Trp metabolites have been identified in soil, 21 IAA is the only metabolite that retains its indole structure. 22 In soil bacteria, it is not only the L-Trp that is catabolized into IAA, but many researchers have suggested that D-Trp may also be converted into auxin IAA by microbiota22–25 where L-Trp aminotransferase (L-TAT) plays a major role in the transformation of Trp to IAA. 26 However, for the transformation of D-Trp, it requires TAT with a special D-stereospecificity to produce IAA. It is known that D-amino acids are a part of the bacterial cell wall and antibiotics elaborated by actinomyces. 27 Martens and Frankenberger 28 have indicated that 80% of the L-Trp carbon added to the soil is evolved as CO2 after 5 days of exposure suggesting the majority of L-Trp added to the soil is utilized by the kynurenine pathway (KP). This may be through degradation of Trp catalyzed by indoleamine 2,3-dioxygenase resulting in the formation of intermediates such as kynurenine, anthranilic acid, 3-hydroxyanthranilic acid, and NAD+.15,16
It has been reported that the ability of different soils to catabolize Trp into IAA may be different. 29 Kuwait desert soil bacteria have not been studied before for catabolism of Trp into IAA, its hydroxylated products and other metabolites. A study of this transformation is of particular interest because of the harsh climatic nature of Kuwait desert where most bacteria are thermophiles rather than mesophiles. Our objective was to study a thermophile, isolated from Kuwait soil contaminated with petroleum hydrocarbons, for its potential to catabolize Trp into IAA and other metabolites. The second objective of this study, besides the identification of metabolites, was to unravel the Trp metabolic profile using a purified thermophile avoiding any synergistic effect of other soil microbes. 30
Materials and Methods
L-trytophan, L-tryptophan ethyl ester and various KP metabolites were obtained from Sigma Chemical Co. (St. Louis, Mo, USA). All solvents and buffer chemicals were of HPLC grade purchased from Fluka and Riedel-de Haën. (Switzerland). Inorganic salts were purchased from Fluka and Riedel-de Haën (Germany) and Merck (Darmstadt, Germany). Tryptone, yeast extract and agar were from Difco Becton Dickinson and company (Sparks USA) and Scharlau (Barcelona, Spain). Kieselgel-60 F254 fluorescent thin layer chromatographic plates (TLC) were obtained from Merck (Darmstadt, Germany).
Organism
The thermophilic Bacilus identified as
Tryptophan transformation studies
The strain was maintained at 4 °C on tryptone and yeast extract agar plates as described previously.31–36 Starter cultures, (50 mL) of
Extraction and isolation of transformation products
The incubated cultures were freeze dried after adjusting the pH to 6.8. The freeze dried material was extracted with acetonitrile by stirring it overnight at 40 °C and then with acetonitrile:water (70:30 V/V). The pooled extract, after filtration, was evaporated under reduced pressure on a rotary evaporator. The concentrated extract was re-dissolved in acetonitrile:water (70:30 V/V) and chromatographed on TLC plates using benzene:ethyl acetate:acetone:methanol:water (22:20:3:3:2 V/V) and also butanol:acetic acid:water (2:1:1 V/V) as mobile phase. Two well resolved bands, as visualised under UV light (λ 254 nm), were marked and a two inch side strip of the 20 × 20 cm chromatograms was stained with ninhydrin solution made in aqueous acetone. The TLC strip, after heating, developed colored bands corresponding to amino group bearing components. Corresponding bands from the TLC plates were scratched, pooled and re-extracted into acetonitrile:water (70:30 V/V). The concentrated extracts were re-chromatographed on TLC till both compounds were obtained in pure state. Most other metabolites could not be resolved on TLC plates in the above mentioned mobile phases. Although many metabolites, present in the crude mixture could not be separated by liquid chromatography by the procedure described by Martens and Frankenberger, 38 all metabolites could be resolved by GC as shown in Figure 1.

Gas Chromatogram of tryptophan metabolites.
Analytical methods
1 H and 13 CNMR spectral data were used when sufficient material was available for spectroscopy. The data were collected using a 600 MHz NMR (Bruker AC 600) instrument as methanol-d4 solution. Two GC/MS systems were used for analyses of L-Trp metabolites and instrumentation and experimental conditions are given in Table 1.
Details of the instrument and experimental conditions used in the analyses.

A pre-column derivatization of amino acids was obtained with phenyl isothiocyanate (PITC) according to the recommended procedure of Waters Pico-Tag method. Derivatization of amino acids was also carried out with [N, O-bis(trimethylsilyl) acetamide] BSA; [N, O-bis(trimethylsilyl) trifluoroacetamide] BSTFA; and trimethylchlorosilane, BSTFA + TMCS.
ESI-LC/MS spectra were obtained using Thermo LCQ Fleet LC/MS equipped with a Thermo Hypersil Gold column 10 cm × 2.1 mm, particle size of 5 μ. Linear gradient elution was carried out using acetonitrile changing over to acetonitrile:water (90:10 V/V) in 45 minutes at a flow rate of 0.8 mLl/minute.
Results and Discussion
For optimizing the incubation time, a time course study was initiated by incubating Trp with
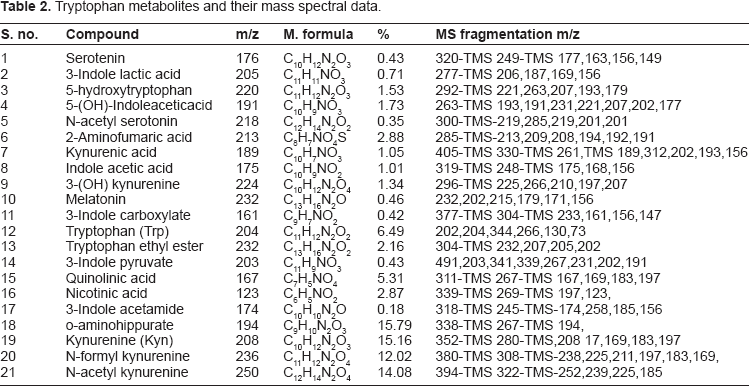
Elemental composition was calculated for the molecular or pseudo molecular ion as well as for fragment ions and neutral losses observed in the ESI-MS spectra. Further information, about the number of double bonds and rings, was obtained from the elemental composition of the compounds. Mass spectral fragments of silylated compounds and their relative% yields are given in Table 2.
Tryptophan metabolites and their mass spectral data.
Time course studies showed that the formation of IAA could not be increased with an increase in incubation time, or a change in its media pH. Our results indicate that kynurenine pathway (KP) may be the major route for conversion of Trp to IAA as observed by Martens and Frankenberger who studied the metabolism of Trp, 28 where Trp is reported to be the primary precursor of IAA in soil39,40 and Trp transaminase is the major enzyme involved in its conversion into IAA. 41 The enzyme, L-Trp aminotransferase (L-TAT) which is known to play a major role in the initial transformation of L-Trp into auxins in plant kingdom 41 is an α-ketoglutarate-dependent conversion enzyme and indole-3-pyruvic acid is the product of this catalysis. In summary, for the first time, our work demonstrates that a single thermophile is responsible for transformation of L-Trp into IAA and many other metabolites.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgment
This work was supported by research grants # SL04/05 and SL03/09 from Kuwait University Research Adminstration (KURA) and are thankfully acknowledged. The investigators also thankfully acknowledge the assistance of Science Analytical Facilities (SAF) (General Facility Project # GS01/01, GS03/01, GS01/03) for spectral data.
