Abstract
The need to have an improved knowledge on the bioecology of
Introduction
Among these factors, genetic variability (within and without offsprings/progeny) tends to be most important, as the genetic makeup of an individual species serves as a blue print for vectorial competence, although this may be highly modified by the environment. Genetic unevenness has been reported for different species of mosquitoes, 16 even for sibling species, 18 and from one locality to another. 19 Preliminary studies have shown that Minna is rich in mosquitoes species diversity11,20,21 that have shown variations in survivorship and duration of development, 19 ,22–24 teneral reserve component, 17 and vectorial fitness attributes17,19 of the species in Minna. In a bid to harmonize these variations, which will advance present knowledge about the species and fill existing gaps in our knowledge of variability within the species, baseline information needs to be generated and this has informed this study. In addition, gaps still exist in our knowledge of variability of egg rafts among individuals of a species/family for the vector species in Minna, North Central Minna.
Therefore, this study aims to bridge this gap of knowledge. It intends to elucidate inherent variations in the productivity of egg rafts (ie, proxy for reproductive viability), survivorship and duration of development of immature stages, and reproductive and vectorial fitness attributes that exist in the imagines from such egg rafts.
Materials and Methods
Study area
This study was carried out in Minna, the capital of Niger State, North Central Nigeria. Minna is located within longitude 6° 33′E and latitude 9° 27′N, covering a land area of 88 km2 with an estimated human population of 1.2 million. The area is characterized by a tropical climate with mean annual temperature, relative humidity, and rainfall of 30.20°C, 61.%, and 1334.00 mm, respectively. The climate presents two distinct seasons in a year: a rainy season between May and October and a dry season between November and April. The vegetation in the area is typically grass-dominated savannah with scattered trees. 14
Source and incubation of egg rafts
The city was combed thoroughly for egg rafts from conventional breeding habitats; recovered rafts were put in carefully labeled containers and transported to the entomological unit of the Department of Biological Sciences. The egg rafts were screened and identified for those belonging to
Larvae culture
The larvae were reared following standard techniques in plastic bowls (1250 mL capacity) at the rate of 250 larvae/bowl in 1000 mL of borehole water. The larvae were fed with fish feed (TetraMin®), at the rate of 0.32 mg/ larva every other day, sprinkled on the water surface. On every alternate day, the water from the culture bowls was changed till pupation.19,28
Pupae culture
The pupae were separated daily and placed in plastic bowls (5 cm in height and 20 cm in diameter) half filled with borehole water. The plastic bowls with pupae were labeled and placed in adult-holding cages for emergence, and mortality was noted; pupae that were unable to emerge or adults that were unable to break free from the pupal case were considered dead.19,29
Duration and survivorship of immature stages
The duration of the immature stages and stage-specific survivorship were computed according to the life table analysis suggested by Olayemi and Ande. 14 Survival rates during the life stages were determined as the proportion of individuals at the beginning of a life stage that successfully entered the next stage. 19
Sex ratio and adult longevity
The sex of each emergent mosquito was noted, and all adult mosquitoes were subsequently monitored for daily survival rates and life span. During this period, the mosquitoes were fed with only sugar solution (10%) soaked in cotton wool. 30
Adult fitness attributes (adult wing length and fluctuating asymmetry)
The adult fitness attribute, which is basically a measure of how well formed a mosquito is, to fly, locate mate, locate host, bite, and transmit pathogen, was estimated from data on body size and symmetry of the wings. The body size of adult mosquitoes is reflected by the wing size, which was determined as described by Gafur 16 and Ukubuiwe et al. 17 The wings of emergent mosquitoes were carefully detached with the aid of entomological pins. The left and right wings were preserved in separate envelopes for further analysis. Wing length was measured according to the techniques of Gafur, 16 and fluctuating asymmetry (FA) was determined as the difference between the left and right wings.
Data analyses
Differences in the mean values of duration and survival rates of immature life stage, as well as adult wing lengths, among the egg rafts were compared for statistical significance using analysis of variance at
Results
Egg raft productivity and duration of immature life stages
Egg raft fertility and developmental rates among the families of
Hatchability of egg rafts and mean ± SD of duration (days) of immature stages (progeny) of egg rafts of
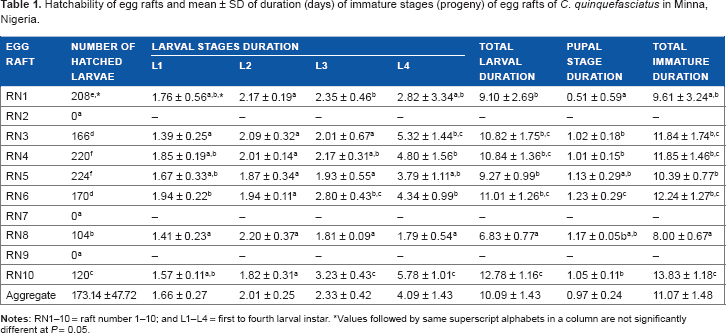
Values followed by same superscript alphabets in a column are not significantly different at
Like larval development, the duration of pupal stage (PSD) and total immature duration (TID) differed significantly among the mosquito families, averaging 0.97 ± 0.24 days and 11.07 ± 1.48 days, respectively. Significant (
Survivorship of immature life stage
Survivorship of the immature life stage is highlighted in Table 2. Mean aggregate survivorship of the immature mosquitoes was 89.91 ± 5.08% and was higher in the pupal stage (95.88 ± 3.16%) than that in the larval stage (88.42 ± 6.07%). Unlike the duration of development, survivorship of larval instars decreased steadily from L1 (98.29 ± 1.64%) to L3 (89.23 ± 8.78%) and, then, drastically in L4 (72.52 ± 17.02%). Differences within families of the species were significant (
Mean ± SD of stage-specific survivorship (%) of immature stages (progeny) of egg rafts of
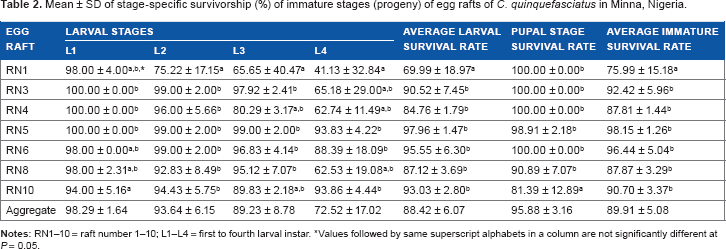
Values followed by same superscript alphabets in a column are not significantly different at
Adult emergence rate and survivorship
Table 3 shows the number of emergent imagines from the egg rafts, their daily survivorship, average postemergence longevity (APL) of adult mosquitoes, and their average life expectancy. Analyses showed a significant variation (
Adult mosquito emergence rate and longevity of
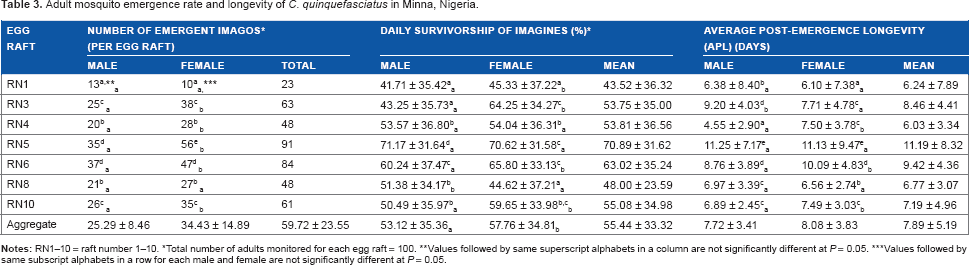
Total number of adults monitored for each egg raft = 100.
Values followed by same superscript alphabets in a column are not significantly different at
Values followed by same subscript alphabets in a row for each male and female are not significantly different at
The influence of family source on daily survivorship of the adult mosquitoes was also significant (P < 0.05) and ranged from 41.71 ± 35.42% (in RN1) to 71.17 ± 31.64% (in RN5) and 44.62 ± 37.21% (in RN8) to 70.62 ± 31.58% (in RN5) for the male and female mosquitoes, respectively. On the whole, daily survivorship of the female mosquitoes (57.76 ± 34.81%) was significantly higher than that of the male mosquitoes (53.12 ± 35.36%), thus revealing the variation among families; apart from RN5, other rafts had significant difference (P < 0.05) in the survivorship of male and female mosquitoes. Generally, RN5 imagines were the most daily survived for the sexes, and RN1 adults being the least survived (Table 3).
Life expectancy (ie, longevity) of the male and female mosquitoes followed the same pattern as daily survival rate, with longevity of 7.72 ± 3.41 and 8.08 ± 3.83 days, respectively. Analyses of APL revealed variation between and among sexes of the egg rafts, with RN5 producing adults with the longest APL for males (11.25 ± 7.17 days) and females (11.13 ± 9.47 days). These variations translated into mean APL for the species as 7.89 ± 5.19 days, ranging from 6.24 ± 7.89 days in RN1 to 11.19 ± 8.32 days in RN5 (Table 3).
Wing length and FA
Wing length (proxy for adult body size) was longer in the female mosquitoes (3.15 ± 0.18 mm) than male mosquitoes (2.97 ± 0.12 mm) (Table 4). However, the left and right wing lengths in both sexes varied within narrow limits. Noticeably, family source had significant (P < 0.05) influence on wing lengths of the mosquitoes, as the biggest male mosquitoes were from families/egg rafts RN1 and RN10 (mean, 3.20 ± 0.03 mm and 3.23 ± 0.10 mm, respectively), while RN8 produced the smallest male mosquitoes (mean, 2.61 ± 0.14 mm). In addition, RN1 and RN10 produced the biggest female mosquitoes (mean, 3.27 ± 0.09 mm and 3.24 ± 0.18 mm, respectively), but unlike for the male mosquitoes, RN5 produced the smallest female mosquitoes (mean, 2.92 ± 0.15 mm). This result is further buttressed by the analyses of aggregate adult wing lengths (male and female), which revealed that RN1 and RN10 produced the largest mosquitoes, while RN5 produced the smallest mosquitoes. FA of the wings was very low (aggregate population mean = 0.02 mm) and similar for both sexes.
Wing length and FA of
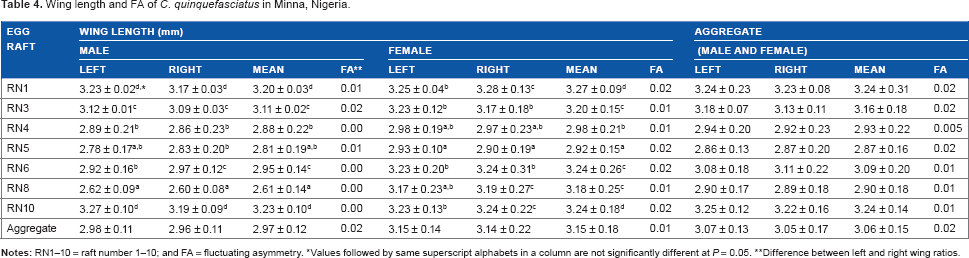
Values followed by same superscript alphabets in a column are not significantly different at
Difference between left and right wing ratios.
Discussion
Knowledge of genetic diversity present within families of a species is important in understanding the evolutionary adaptability of the organism to function well in its ecological niche. The life cycle of mosquitoes starts from the laid egg (singly or in rafts), and life attributes of the progeny depend largely on the quality of the eggs. In the present study, which is a preliminary investigation, the egg rafts were not all productive, as 30% of the randomly collected eggs were not viable; this confirms earlier assumptions that not all egg rafts are viable. However, egg raft fertility was relatively high (70%) for the species (
Duration and survivorship of immature stages of the mosquito species averaged about 11.07 ± 1.48 days and 90%, respectively. These results are similar to those of Olayemi et al, 22 who reported values of 11.57 ± 0.00 days and 90.38 ± 6.52%, respectively. However, their results contradicted those reported by Ukubuiwe et al, 19 who reported a duration of development of 8.67 ± 2.03 to 10.10 ± 0.94 days and survivorship range of 88.87 ± 7.58% to 95.08 ± 1.68% for species collected from different areas of the city. In addition, they differed from those reported by Olayemi et al, 23 who reported survivorship of 95.40 ± 2.87% and duration of immature development of 9.97 ± 0.74 days for the same species. These similarities/dissimilarities may be due to genetic and/ or environmental concurrence/differences between the populations of mosquito species, as they were raised in the same laboratory conditions.
In the present study, familial variation in the immature duration showed that mosquitoes from RN8 are fast developers, while those from RN10 are very slow developers (eliminating the influence of habitat source by rearing in the same breeding conditions), which is of great importance epidemiologically and entomologically, as it can affect the prediction of population explosion from existing data. Thus, it points to a differential endowment of the species genetically; this could have been the reason for the conflicting results from previous studies on the species.
Increased duration of instar stage with larval age as reported in this study and supported by earlier studies 19 ,22–24 may be occasioned by the increasing need to accumulate teneral reserve for egg development in female mosquitoes during the adult stage. In addition, Briegel et al 33 and Briegel 34 reported the climax teneral reserve accumulation in mosquitoes during the L4 instar stage, which in the present study varied significantly between families/egg rafts and could signify differential accumulation tendencies.
While the duration of larval instar stage increased from L1 to L4, the reverse was the case with survival rates. The decreasing survivorship of the larval instars (of all the families/ egg rafts) with age (ie, L1–L4) seems to disfavor the ecological adaptability of
Significantly, higher densities of and better survivorship of female mosquitoes per egg raft that varied among egg rafts/ families were not unexpected, as nature, through evolutionary selection, tends to invest more in the female sex of many species, as they are regarded as better assets. For example, female mosquito needs to produce eggs (a costly physiological and metabolic process), accumulate more teneral reserve during the larval stage, 35 and take blood meals (a highly proteinous food source), which stand them in good stead for better adult life performance than the males.34,36,37
Wing length of mosquitoes gives an estimate of body weight
38
and body size
39
and affects longevity, fecundity, and blood meal volume, which may influence the fitness of the vector for disease/parasite transmission. In addition, FA measures deviation from perfect bilateral symmetry caused by environmental and/or genetic stress experienced during ontogeny and usually is an interaction between the environment and the genotype or wholly genetic expression.40,41 In the present study, differential wing lengths were noted across the families and the earlier productive and perceived genetically efficient egg raft (RN5; with the greatest immature survivorship, highest adult emergence, and longest surviving mosquitoes) produced the smallest mosquitoes with greater FA, with the female mosquitoes having longer wing lengths (ie, adult body size) than the male mosquitoes. This is supported by earlier studies by Agnew et al
42
and Kaufmann and Briegel,
37
who reported greater value of wing length for female species for
Although familial variation exists, the FA of the wings of
The influence of family variations on the reproductive and adult fitness of
Conclusion
This preliminary study shows that
Author Contributions
Conceived and designed the experiment: ACU and IKO. Analyzed the data: ACU. Wrote the first draft of the manuscript: ACU. Contributed to the writing of the manuscript: IKO and AIJ. Agreed with manuscript results and conclusion: ACU, IKO, and AIJ. Jointly developed the structure and arguments for the paper: ACU, IKO, and AIJ. Made critical revisions and approved the final version: all authors. All the authors reviewed and approved the final manuscript.
Footnotes
Acknowledgment
We wish to acknowledge the management and staff of the Department of Biological Sciences, Federal University of Technology, Minna, for the unrestricted use of the equipment and space in the laboratory.
