Abstract
No abstract supplied.
Introduction
The incidence of most cancers is increasing in India. With respect to bladder cancer, there has been a steady increase in the number of bladder cancers diagnosed per year, according to the ICMR Cancer Atlas. 1 Our recent interest in bladder cancers, as diabetologists, stems from the fact that use of the anti-diabetic drug pioglitazone has been linked to bladder cancer. 2 This has led to the discontinuation of pioglitazone in France and Germany 3 and the US-FDA has also issued warnings regarding the use of pioglitazone. 4 A large study from the UK showed that there was an 83% increase in bladder cancer among those who used pioglitazone. 5 Another recent study from the United States also confirmed that pioglitazone was associated with bladder cancer. 6 In order to study whether urologists are aware of this association, we conducted a study using a questionnaire on bladder cancer, which was sent to 200 urologists in South India using an address book of their association. Seventy-one urologists responded (35.5%) and sent back the questionnaires duly filled in.
Results
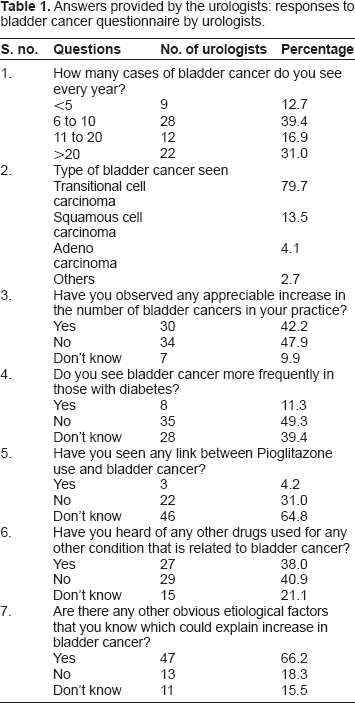
From Table 1, it can be seen that 22 urologists (31%) reported that they were seeing more than 20 cases of bladder cancer per year. The most common bladder cancer in practice was transitional cell carcinoma, which was seen in 79.7% of patients, followed by squamous cell carcinoma (13.5%) and adenocarcinoma (4.1%). Over 40% of urologists felt that there was an increase in bladder cancer cases. About 12% of urologists felt that bladder cancers were more common among people with diabetes, but only 3 (4.2%) were aware that there was a link between pioglitazone use and bladder cancer. Two of these three urologists worked in major teaching institutions. 38% felt that there were other drugs which were related to bladder cancer. Almost 60% of urologists reported that the use of certain dyes was related to bladder cancer.
Answers provided by the urologists: responses to bladder cancer questionnaire by urologists.
Conclusion
This short survey shows that the need for increasing awareness of the possible link between pioglitazone and bladder cancer in India among urologists and other doctors such as physicians, diabetologists, oncologists. It also calls for increased pharmacovigilance in India on this subject. 7 We propose this could be best done by performing retrospective studies using the medical records of biopsy-proven bladder cancer patients. We also propose that treating doctors should tell their patients on pioglitazone therapy about the possible risk of bladder cancers, if pioglitazone treatment cannot be discontinued for any reason.
Footnotes
Author Contributions
Conceived and designed the experiments: SC, VM. Analysed the data: SC, VM. Wrote the first draft of the manuscript: SC, VM. Contributed to the writing of the manuscript: SC, VVJ, VM. Agree with manuscript results and conclusions: SC, VVJ, VM. Jointly developed the structure and arguments for the paper: SC, VVJ, VM. Made critical revisions and approved final version: SC, VVJ, VM. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
VVJ has received payment for speaking from Eli Lilly and Boehringer Ingelheim. Other authors disclose no competing interests.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
