Abstract
Dear Editor
The etiology and pathomechanism of chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME) are unknown. This disabling illness is associated with poor memory and concentration; metabolic, cardiac, gastrointestinal, and immune dysfunction; and muscle fatigue often described as neuroimmune exhaustion and post-exertional malaise.1–18 We have previously identified single-nucleotide polymorphisms (SNPs) in genes for transient receptor potential (TRP) ion channels and acetylcholine receptors (AChRs), which have important roles in calcium (Ca2+) and acetylcholine (ACh) signaling.19–21 From this same cohort of patients, we now report additional data showing the prevalence of both TRP melastatin M3 (TRPM3) ion channel and muscarinic ACh M3 receptor (mAChM3R) SNP genotypes in patients with CFS/ME (Table 1).
Genotype frequencies of TRPM3 and mAChM3 gene polymorphisms in CFS/ME patients and nonfatigued controls.
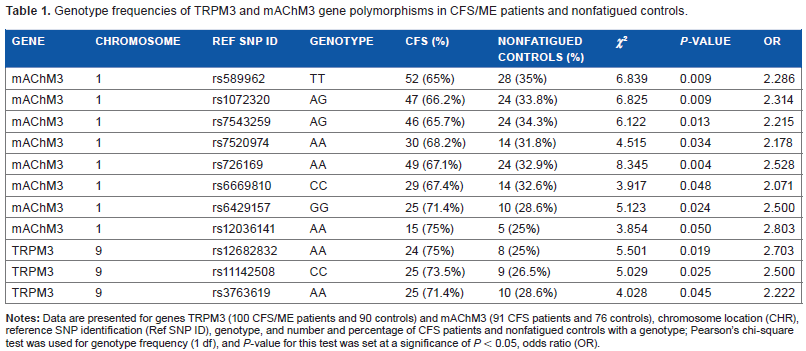
Genomic DNA extraction and SNP genotyping studies were performed as previously described.19,20 Existing, deidentified samples were used for the further study described here. The PLINK v1.0740 whole-genome analysis toolset and IBM® SPSS® Statistics (Version 21) were used to determine the genotype frequency between patients with CFS/ME and nonfatigued controls. A two-column χ 2 test was used, where the alpha level of significance was set at P < 0.05, and the results of genotype frequencies of TRPM3 and mAChM3R gene polymorphisms are shown in Table 1. Eleven genotypes were identified from SNPs that were significant in TRPM3 family (rs12682832; rs11142508; rs3763619) and mAChM3R (rs12036141; rs589962; rs1072320; rs7543259; rs7520974; rs726169; rs6669810; rs6429157) in patients with CFS/ME compared with nonfatigued controls (Table 1).
Muscarinic AChRs (mAChRs) are involved in autonomic function, particularly parasympathetic and exocrine function, such as in pancreas, exocrine glands, and inotropic and chronotropic cardiac regulation. Given that AChRs are distributed differentially around the body, it is axiomatic that tissues expressing a predominance of these receptors will be affected differentially by SNPs in muscarinic vs. nicotinic AChRs. Similarly, TRPs are distributed differentially around the body in all tissues. Adding to the complexity is the relative lack of knowledge about interactions between TRP and AChRs in humans. Interestingly, certain muscarinic ACh receptors are antagonists of TRPM3 via, for example, phospholipase C-coupled muscarinic ACh M1 receptor (mAChM1R).21,22 Given this developing research regarding the interdependence of mAChRs and TRP families, we question whether specific mAChM3R and TRPM3 SNP genotypes in CFS/ME contribute to the pathomechanism and phenotypes of this illness.
Even though the distribution of these receptors varies in peripheral blood mononuclear cells, SNP genotypes such as those we have identified in this patient cohort are likely to contribute to perturbations of TRP ion channel and AChR function mediated through altered Ca2+ and ACh signaling and manifest as physiological system compromise. The critical role of AChR and TRP ion channels in Ca2+ cell signaling suggests that further characterization of TRPM3 and mAChM3R may elucidate perturbations of second messenger signaling in CFS/ME.
Author Contributions
Conceived and designed the experiments: SM-G, AC, SJ, PS, BN, and DRS. Analyzed the data: SM-G, AC, SJ, PS, BN, and DRS. Contributed to the writing of the manuscript: SM-G, AC, SJ, PS, BN, and DRS. Made critical revisions and approved final version: SM-G, AC, SJ, PS, BN, and DRS. All authors reviewed and approved of the final manuscript.
Footnotes
Acknowledgments
The authors would like to thank Dr Lavinia Gordon, Australian Genome Research Facility, Melbourne, Australia, for completing the bioinformatics SNP analysis.
