Abstract
Background
The transient receptor potential (TRP) superfamily in humans comprises 27 cation channels with permeability to monovalent and divalent cations. These channels are widely expressed within humans on cells and tissues and have significant sensory and regulatory roles on most physiological functions. Chronic fatigue syndrome (CFS) is an unexplained disorder with multiple physiological impairments.
OBJECTIVES
The purpose of this study was to determine the role of TRPs in CFS.
Methods
The study comprised 115 CFS patients (age = 48.68 ± 1.06 years) and 90 nonfatigued controls (age = 46.48 ± 1.22 years). CFS patients were defined according to the 1994 Center for Disease Prevention and Control criteria for CFS. A total of 240 single nucleotide polymorphisms (SNPs) for 21 mammalian TRP ion channel genes (TRPA1, TRPC1, TRPC2, TRPC3, TRPC4, TRPC6, TRPC7, TRPM1, TRPM2, TRPM3, TRPM4, TRPM5, TRPM6, TRPM7, TRPM8, TRPV1, TRPV2, TRPV3, TRPV4, TRPV5, and TRPV6) were examined via the Agena Biosciences iPLEX Gold assay. Statistical analysis was performed using the PLINK analysis software.
Results
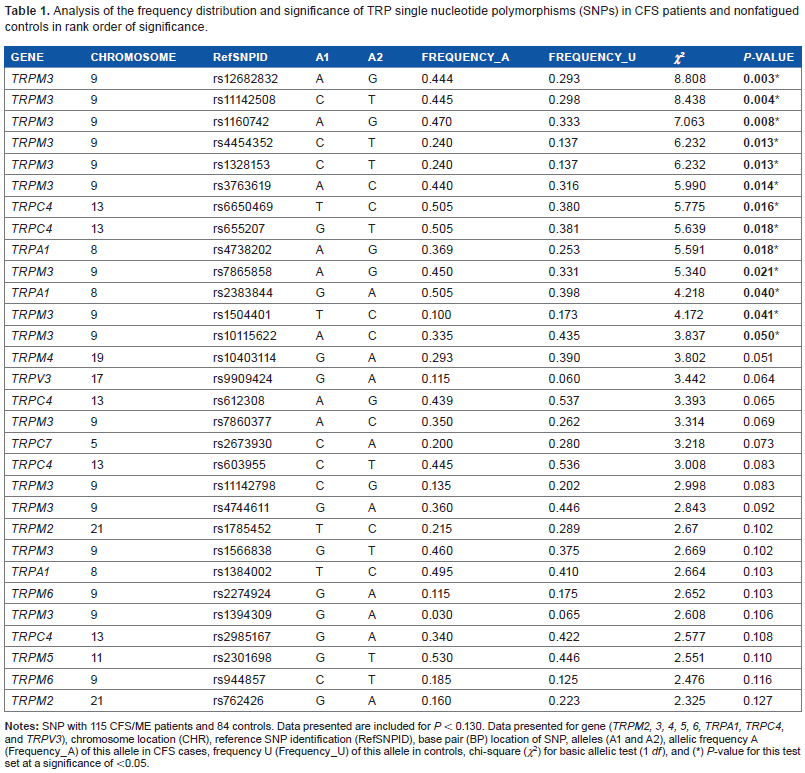
Thirteen SNPs were significantly associated with CFS patients compared with the controls. Nine of these SNPs were associated with TRPM3 (rs12682832; P < 0.003, rs11142508; P < 0.004, rs1160742; P < 0.08, rs4454352; P < 0.013, rs1328153; P < 0.013, rs3763619; P < 0.014, rs7865858; P ≤ 0.021, rs1504401; P ≤ 0041, rs10115622; P ≤ 0.050), while the remainder were associated with TRPA1 (rs2383844; P ≤ 0.040, rs4738202; P ≤ 0.018) and TRPC4 (rs6650469; P ≤ 0.016, rs655207; P ≤ 0.018).
Conclusion
The data from this pilot study suggest an association between TRP ion channels, predominantly TRPM3 and CFS. This and other TRPs identified may contribute to the etiology and pathomechanism of CFS.
Introduction
Transient receptor potential (TRP) ion channels are cation channels with putative roles in many physiological signaling pathways. Mammalian TRPs are comprised of six main groups: TRPA (ankyrin), TRPC (canonical), TRPM (melastatin), TRPML (mucolipin), TRPP (polycystin), and TRPV (vanilloid).1,2 Generally, the TRPC channels are nonselective cation channels; only two are highly permeable Ca2+ channels and two are impermeable for Ca2+. Importantly, several TRPs are permeable for Mg2+ and Zn2+. 3 TRPs are extensively expressed on almost all cells and therefore are likely to have significant effects on physiological functions. 3 Dysregulation in TRPs has been associated with pathological conditions and diseases including chronic pain, overactive bladder, diabetes, chronic obstructive pulmonary disease, cardiac hypertrophy, familial Alzheimer's disease, skin diseases, skeletal dysplasias, motor neuropathies, neurosensory neuropathies including Charcot–Marie–Tooth disease (type 2C), and cancer.4–8 TRP ion channels are activated following fluctuations or deviations in the cellular environment. Factors that may influence these changes are stressors including pathogens, temperature, pressure, chemicals, oxidation/reduction, toxins, osmolarity, and pH.9,10
Chronic fatigue syndrome (CFS) is an unexplained disorder with multiple physiological impairments. Research to date suggests significant immune impairment; however, the mechanism of this disorder remains to be determined. CFS patients may have reactions to a number of environmental and biological factors.11–13 Moreover, there is evidence to suggest that CFS may have an allergic component.14–16 Atypical TRP expression has been reported in CFS, particularly upregulation in the expression of TRPV1. 17 As TRPs regulate a plethora of physiological signaling pathways, they may have a role in CFS. A number of channelopathies have been associated with TRP genes and these have consequences for cellular function.4,18,19 Additionally, TRP channels may be targeted during inflammatory reactions, as they are easily activated in the presence of irritants, inflammatory products, and xenobiotic toxins. Incidentally, CFS patients report significant sensitivity to environmental toxins and irritants, but the causes of these sensitivities remain to be fully investigated. The purpose of this pilot study was to determine whether polymorphisms in SNPs associated with TRP ion channel genes are a contributory factor in the pathogenesis of CFS.
Methodology
Participants
One-hundred and fifteen CFS patients and 90 nonfatigued controls were recruited for this study. Of the 115 CFS patients (age = 48.68 ± 1.06 years), 84 (73.04%) were women and 31 (26.96%) were men. The 90 nonfatigued controls (age = 46.48 ± 1.22 years) comprised 59 (65.56%) women and 31 (34.44%) men. CFS patients were defined in accordance with the 1994 Center for Disease Prevention and Control (CDC) criteria for CFS. 20 All participants in the patient and nonfatigued control groups were of European decent, and all were residents of Australia at the time of blood collection. Approval for the study was granted by the Institutional Ethics Review Board at Griffith University (Ref No: MSC/18/13/HREC) and the research complied with the principles of the Declaration of Helsinki.
Ten milliliters of whole blood samples were collected from all participants in to ethylenediamine tetraacetic acid tubes. Written consent was obtained from all participants prior to sample collection.
DNA extraction
Genomic DNA was extracted from all whole blood samples using the Qiagen DNA blood mini-kit as per manufacturer's instructions (Qiagen). The Nanodrop (Nanodrop) was used to assess the quality and quantity of the DNA extracted. Approximately 2 µg of genomic DNA was used in the SNP assay.
SNP genotyping studies
SNP analysis was performed by Geneworks using the MassARRAY iPLEX Gold Assay (Sequenom Inc.) as previously defined. Customized assays were developed for 240 SNPs across the 21 TRP genes (TRPA1, TRPC1, TRPC2, TRPC3, TRPC4, TRPC6, TRPC7, TRPM1, TRPM2, TRPM3, TRPM4, TRPM5, TRPM6, TRPM7, TRPM8, TRPV1, TRPV2, TRPV3, TRPV4, TRPV5, and TRPV6). Primers and extension primers were created for each of the SNPs using the Assay Designer (Sequenom Inc.) according to the manufacturer's instructions. Briefly, DNA was amplified via polymerase chain reaction (PCR) under the following conditions: 94°C for 2 minutes, 94°C for 30 seconds, 56°C for 30 seconds, and 72°C for 1 minute. Amplification products were then treated with shrimp alkaline phosphatase at 37°C for 40 minutes, 85°C for 5 minutes reaction, and a final incubation at 4°C. Extension primers were optimized to control the signal-to-noise ratio where unextended primers (UEPs) were examined on the spectroCHIP and evaluated in Typer 4.0 to enable the division into low-mass UEP, medium-mass UEP, and high-mass UEP. To perform the iPLEX extension reaction, a mixture containing iPLEX Gold reaction was prepared using iPLEX Gold Buffer Plus, iPLEX termination mix, iPLEX enzyme, and primer mix. The iPLEX reaction was cycled at an initial denaturation of 94°C for 30 seconds, annealing at 52°C for 5 minutes, extension at 80°C for 5 minutes (five cycles of annealing and extension were performed, but the whole reaction was performed in 40 cycles) and extension again at 72°C for 3 minutes. Resin beads were used to rinse all iPLEX Gold reaction products. Following the iPLEX Gold reaction, MassARRAY was performed using the MassARRAY mass spectrometer, and the data generated were analyzed using the TyperAnalyzer software.
Statistical Analysis
The PLINK v1.07 20 whole genome analysis tool set was used to determine associations between the CFS patients and the nonfatigued control group. A two-column χ 2 test was used to determine significance, where a P-value of <0.05 was determined to be significant. Data analysis was performed by the Australian Genome Research Facility.
Results
SNP association studies
Of the 240 SNPs that were examined in the present study, 233 were successfully identified in both participants groups. Thirteen were observed to be significantly associated with CFS (Table 1). Nine of these SNPs were associated with TRPM3 (rs12682832; P ≤ 0.003, rs11142508; P < 0.004, rs1160742; P < 0.08, rs4454352; P ≤ 0.013, rs1328153; P ≤ 0.013, rs3763619; P ≤ 0.014, rs7865858; P ≤ 0.021, rs1504401; P ≤ 0041, rs10115622; P ≤ 0.050), while the remaining SNPs were associated with TRPA1 (rs2383844; P ≤ 0.040, rs4738202; P ≤ 0.018), and TRPC4 (rs6650469; P≤ 0.016, rs655207; P ≤ 0.018). A summary of the 233 SNPs that were reported in both participant groups can be found in Supplementary Table 1.
Analysis of the frequency distribution and significance of TRP single nucleotide polymorphisms (SNPs) in CFS patients and nonfatigued controls in rank order of significance.
Discussion
The purpose of this pilot study was to determine the presence of possible SNP variations in CFS patients with a specific focus on SNPs within the coding sequences of 21 TRP ion channel genes. Out of the 240 SNPs examined, 13 alleles were found to be significantly associated with CFS patients compared with the nonfatigued controls. As CFS is a heterogeneous condition, it is likely that further stratification of patients may elucidate further allelic associations. These alleles were located in the gene sequence of one of the canonical TRPs ion channels (TRPC4), one ankyrin (TRPA1), and one melastatin TRP ion channel (TRPM3).
SNPs located within a coding sequence may or may not necessarily change the amino acid sequence of the protein that is produced. As such, an SNP that does not alter the polypeptide sequence is termed synonymous (sometimes called a silent variant), while an SNP that results in a different polypeptide sequences is referred to as nonsynonymous, potentially resulting in altered gene transcripts and disease phenotypes. In this investigation, we report TRP SNP anomalies, suggesting they may mediate the potential onset or clinical presentation of CFS; however, this needs to be confirmed with larger cohorts. Nonetheless, this current study suggests the potential role for aberrant TRPs in CFS. Further rationale is provided below, whereby TRPs are demonstrated to play significant physiological roles. In particular, CFS patients report symptoms that may be associated with aberrant TRP function.
TRPC4 is activated via receptor-dependent activation of the Gq/11/PLC (phospholipase C)/γ pathway but also via Gαi proteins, PI(4,5)P2 proteins, and also intracellular Ca2+. 21 It is mainly involved in vasomotor function, aggregation of platelets, and smooth muscle function. Incidentally, Ca2+ is known to be required for the regulation of immune cells, as Ca2+ acts as a second messenger for most cells, particularly T cells and B cells. Intracellular Ca2+ increases when lymphocyte receptors are exposed to antigens. 22 In CFS patients, there are numerous reports on compromises to immune function, although there is limited information on the role of Ca2+ in these patients. However, dysregulation in TRPCs may affect intracellular calcium concentration and incidentally lymphocyte function. Lymphocytes such as natural killer (NK) cells and T cells have been shown to be compromised in CFS. In NK cells, Ca2+ enhances cytotoxic activity, and its depletion or excessive influx may have severe consequences on NK cells function. In CFS, reduced cytotoxic activity has been consistently reported,23–28 and this may be related to the dysregulation in Ca2+.
Dysregulation of TRPCs may affect neuronal responses, in particular those associated with the stimulation of muscarinic receptors. Following activation of TRPCs by PLCs, an influx of Ca2+ occurs, causing an induction in muscarinic receptors, and maintains incessant neuronal firing.29,30 Hence, secretion of Ca2+ and the availability of TRPCs in the neuronal environment are paramount to optimal muscarinic receptor function and overall function of the brain. Importantly, this process is essential for memory, attention, sensory acuity, emotion, pain, and motor control,31,32 and occurs in the amygdala, entorhinal cortex, hippocampus, and prefrontal cortex. 33 Neuronal deficits involving memory and attention have been identified in CFS.34–36 Deletion or compromises to TRPC4 may also affect intestinal function. TRPC4 and TRPC6 pair with muscarinic receptors in the intestine, activating smooth muscle depolarization, inflow of Ca2+, and smooth muscle contraction. 37 Intestinal dysfunction is a component of CFS; 38 however, the extent of damage to the intestinal wall or the exact role of ion channels in the intestine remains to be determined. TRPC4 may be simultaneously regulated by the G protein coupled receptors (GPCRs) Gαi and Gαq. 39
TRPA1 is a multiple chemical receptor that has been identified on nociceptive sensory neurons (C fibers) and has a role in the regulation of the release of neuropeptides, pain sensation, and inflammation. 40 It may be activated by both exogenous and endogenous inflammatory agents, resulting in inflammation and pain. 41 GPCRs also activate TRPA1 via PL C signaling, sensitizing the ion channel to various stimuli. 42 TRPA1 may be activated and subsequently inactivated in the presence of intracellular and extracellular calcium concentrations.43,44 TRPA1 gene has been proposed to affect sensitivity to nociceptive stimuli; 45 hence CFS patients expressing SNPs in the TRPA1 gene may increase their sensitivity to nociceptive stimuli. In the CNS, astrocytes express TRPA1 channels, and these channels are necessary for calcium uptake and neuronal regulation in the astrocytes. Changes in the level of calcium may therefore affect the function of astrocytes and interneuron communication.44,46 Activation of TRPA1 has been shown to induce acute headache, and this may occur through the calcitonin gene related peptide (CGRP), causing vasodilation in the meningeal artery.44,47 Importantly, headache is a prominent symptom of CFS. TRPA1 is also a key player in migraine and neuropathic joint and muscle pain, which is most often experienced by patients with fibromyalgia.48,49 TRPA1 forms functional heterotetramers with TRPV1; hence variations in the TRPA1 gene may suggest functional deficits to TRPV1 that may not be related to polymorphism in nucleotides. 50 Importantly, Carreno et al 51 reported that TRPV1 contributes to the genetic susceptibility to migraine in a large study population of 500 Spanish participants, suggesting TRPA1 and TRPV1 may play a role in symptom presentation of CFS patients. Interestingly, analgesics and antinociceptive drugs target TRPV1 and TRPA1, respectively, to alleviate pain sensation52–54 and these drugs are routinely prescribed to CFS patients. Perhaps in CFS, these drugs may not be effective due to impairments or variations in these ion channels.
TRPM channels are mostly permeable to magnesium and calcium. Only TRPM4 and TRPM5 are impermeable for divalent cations. TRPM3 is permeable for cations including Ca2+ and Zn2+. However, the permeation profile highly depends on the expressed spliced variant. 55 No hereditary TRPM3 channelopathy has been described to date. TRPM3 has been implicated in inflammatory pain syndromes, rheumatoid arthritis, and secretion of proinflammatory cytokines. As pancreatic β cells also have a high proportion of TRPM3 channels,56–58 there is the likelihood of perturbations in insulin/glucose regulation in CFS patients. Metabolic disturbance has also long been identified as a cardinal feature of CFS. The most characterized TRPM3 in humans is in the central nervous system (CNS) and eye 55 where missense mutation of the TRPM3 gene has also been found to underlie the development of cataract and glaucoma. 59 TRPM3 is involved in the detection of heat and in pain transmission. TRPM3-deficient mice exhibit clear deficits in their avoidance responses to noxious heat and in the development of inflammatory heat hyperalgesia. 55 Dysregulation in thermoregulatory responses has been reported in CFS patients. 60 Generalized pain is a characteristic of CFS and occurs in the absence of tissue damage, and this is suggestive of potential CNS impairments. 61 As TRPM3 has a role in nociception and thermoregulation, it may have a role in the pathomechansim of CFS. Additionally, TRPM3 is activated by pregnenolone sulfate, suggesting that it has neuroendocrine effects62,63 and might also be involved in the regulation of glutamatergic signaling in the brain. 64
These preliminary findings implicate TRP ion channels in the etiology and pathomechanism of CFS. Dysregulation of TRPs, including the TRPM3 family, is likely pertinent in predisposing CFS patients to calcium metabolism perturbations and aligns with symptom presentation. Potentially, dysregulated influx of calcium ions into cells will impact a number of vital components of cell regulatory machinery. These components include calcium-sensitive adenylate cyclases (ACs) and hence cAMP expression and function. For example, isolated cell types that have been shown previously to have calcium-sensitive cell regulatory mechanisms in CFS patients may enable further elucidation of TRP ion channels and the likely consequences in CFS. Furthermore, population analysis of TRP SNPs for CFS susceptibility, as well as the proposed various subtypes, needs to be considered. This undertaking will likely be of considerable importance to public health and public health practitioners, as well as to researchers to assess the role of TRP ion channels in CFS symptomatology, severity, and predisposition.
Author Contributions
Designed and developed all experiments as well as analysis, revisions and final preparation of this article: SMG, DRS, PS, BN. Involved in the sample preparation and drafting of the manuscript: SBR, EWB. All authors reviewed and approved of the final manuscript.
Supplementary Data
Supplementary table 1
Analysis of the frequency distribution and significance of TRP single nucleotide polymorphisms (SNPs) in CFS patients and nonfatigued controls in rank order of significance.
Footnotes
Acknowledgments
The authors would like to thank Dr Lavinia Gordon, Australian Genome Research Facility, Melbourne, Australia, for completing the bioinformatics SNP analysis.
