Abstract
Recent advances in virus detection strategies and deep sequencing technologies have enabled the identification of a multitude of new viruses that persistently infect mosquitoes but do not infect vertebrates. These are usually referred to as insect-specific viruses (ISVs). These novel viruses have generated considerable interest in their modes of transmission, persistence in mosquito populations, the mechanisms that restrict their host range to mosquitoes, and their interactions with pathogens transmissible by the same mosquito. In this article, we discuss studies in our laboratory and others that demonstrate that many ISVs are efficiently transmitted directly from the female mosquito to their progeny via infected eggs, and, moreover, that persistent infection of mosquito cell cultures or whole mosquitoes with ISVs can restrict subsequent infection, replication, and transmission of some mosquito-borne viral pathogens. This suggests that some ISVs may act as natural regulators of arboviral transmission. We also discuss viral and host factors that may be responsible for their host restriction.
Keywords
Introduction
Mosquito-borne viruses are objects of intense research due to their complex biology, ecology, and evolution and their potential to produce large and unpredictable outbreaks of disease. 1 Indeed, explosive outbreaks of disease in many regions of the world have been caused by mosquito-borne viruses such as dengue virus (DENV), Zika virus (ZIKV), chikungunya virus (CHIKV), yellow fever virus (YFV), Japanese encephalitis virus (JEV), Ross River virus (RRV), and Rift Valley fever virus. In the absence of safe and effective vaccines and antivirals against many of these viruses, new approaches are being explored to control these diseases and their transmissions.
Currently, there is a strong research focus on a novel group of “insect-specific” viruses (ISVs) that persistently infect mosquitoes, but do not infect vertebrates.2,3 Importantly, persistent infection of mosquitoes with some ISVs appears to interfere with the replication and transmission of medically significant viruses, such as West Nile virus (WNV).4–7 These findings suggest that ISVs may act as natural regulators of transmission of some arboviruses and may provide a new avenue for developing vector control strategies. Furthermore, the potential to genetically manipulate ISVs to develop new platforms for the production of safe diagnostic antigens and vaccines for mosquito-borne pathogens is now recognized as an innovative and a viable strategy. 8
In this article, we review the key research conducted on mosquito-borne ISVs with a focus on the recent isolation and characterization of several new ISVs that group within a range of virus families including flaviviruses, alphaviruses, bunyaviruses, mesoniviruses, negeviruses, and reoviruses. We also discuss mechanistic and evolutionary features that are likely to be associated with the adaption of ISVs to a mosquito-only transmission cycle and the potential applications of these viruses as novel model systems for research, recombinant technology platforms, and agents of biological control.
Flaviviruses
The genus
The first recognized ISF, cell-fusing agent virus (CFAV), was identified as an endogenous virus in a cell line derived from
In 2010, our laboratory initiated a project to assess the biodiversity of ISFs in Australian mosquitoes. This was initially performed by the detection of flavivirus RNA directly in archival samples of homogenized mosquito pools or by inoculation of the samples onto C6/36 cells prior to the detection by reverse transcription polymerase chain reaction and/or the presence of cytopathic effect (CPE) in the cells.
5
Subsequently, we enhanced the speed and sensitivity of the ISF isolation protocol by the detection of viral replicative dsRNA in inoculated mosquito cell culture by the use of anti-dsRNA mAbs in enzyme-linked immunosorbent assay – referred to as “MAVRIC” (monoclonal antibodies against viral
Several ISFs were subsequently isolated from Australian mosquitoes including Palm Creek virus (PCV) from

Map of Australia showing general locations of insect-specific flavivirus isolations.

Bayesian phylogenies of flaviviruses over the whole open reading frame nucleotide sequence. The tree was constructed in Geneious using MrBayes v3.2.2 under the Bayesian Marko chain Monte Carlo (MOMC) model with a general time reversible substitution model, gamma distribution (five discrete gamma categories), and invariant rates among sites. Horizontal branch lengths represent posterior probabilities. The tree has been rooted using the outgroup Modoc virus (MODV), a flavivirus with no known vector. The colored nodes represent insect-specific flaviviruses (ISFs), with Lineage I in blue and Lineage II in red.
ISF-like viruses
Most of the ISFs reported to date belong to a group that is phylogenetically separate from the vertebrate-infecting flaviviruses and are described as “classical” ISFs.
2
For clarity, these viruses are referred to, in this article, as Lineage I ISFs (Fig. 2). However, a smaller subset of ISFs, termed dual-host affiliated ISFs, display an insect-specific phenotype but group phylogenetically with the mosquito-borne pathogenic flaviviruses.18–20 These viruses are referred to, in this article, as Linage II ISFs (Fig. 2). While Lineage II ISFs have been assessed
Another subset of ISF-like viruses shows some replication in vertebrate cells, but only in a limited range of cell types or under specific growth conditions. Rabensburg virus, considered to be a strain of WNV, showed little to no replication in vertebrate cell lines or live birds.
21
However, further studies revealed that this virus replicates and causes CPE in vertebrate cells if they are incubated at temperatures below 35 °C.
22
A recent report from our laboratory also showed that a new Australian flavivirus named Bamaga virus (BgV), which groups phylogenetically with vertebrate-infecting flaviviruses in the YFV group, displays restricted replication in vertebrates, both
The unusual phenotype described earlier for the two viruses may be indicative of their adaption to cryptic vertebrate hosts with low optimal body temperature. Alternatively, the data may indicate that they are in transition between a vertebrate-infecting and an insect-specific transmission cycle.22,23
Interference by ISFs with the replication and transmission of flaviviral pathogens
Studies by Bolling et al.
6
, on a population of
The effect on DENV and ZIKV transmission by
The mechanism(s) involved in both
The evolutionary origin of ISFs is an enigma. Did they evolve from vertebrate-infecting flaviviruses by adapting to replication in mosquitoes or do they represent the ancestral lineage of the dual-host flaviviruses? While it has been proposed that Lineage I ISFs represent the precursors to dual-host flaviviruses, previous attempts to address this by bioinformatics analyses were inconclusive, due to the limited number of fully sequenced ISF genomes available at the time. 24 However, the discovery and full genome sequencing of several additional ISFs may now allow more meaningful bioinformatics to be undertaken to elucidate the evolutionary origins of the flavivirus genus.
The molecular basis for the restricted host range of ISFs is another poorly understood phenomenon. Alignment of the deduced amino acid sequences of Lineage I ISFs with vertebrate-infecting flaviviruses reveals significant changes in several genes including large conserved deletions in domain III of the envelope protein (EDIII).
17
These deletions are of particular interest since EDIII contains the putative flavivirus receptor-binding site.
25
Furthermore, four additional cysteines in the EDIII of ISFs suggest the formation of additional S-S bonds in this domain, which likely change the tertiary structure and may alter the ability of ISFs to recognize specific cell receptors or interfere with membrane fusion.17,26,27 The identification of conserved deletions in the
The use of novel methods to manipulate infectious genomes of ISFs will allow the identification of viral and host factors that restrict the replication of ISFs to mosquitoes and help us to understand the complex dynamics of the transmission of mosquito-borne viruses30,31 (Piyasena et al, unpublished data). These investigations will also provide valuable insights into the evolution of mosquito-borne viruses and underpin the future development of new strategies to genetically manipulate ISFs as new technological platforms to prevent mosquito-borne viral diseases and control their transmission.
Alphaviruses
In contrast to the ever-expanding group of ISFs discussed earlier, only a single member of the
The construction of chimeric viruses between EILV and CHIKV, with the viral structural protein derived from the latter and the replicase components from the former, has also yielded a viable virus that is antigenically similar to the CHIKV parental virus, but unable to replicate in vertebrate cells like EILV. 8 This represents a novel platform for the production of diagnostic antigens or vaccines for pathogenic alphaviruses without the need for inactivation.
Similar to findings with ISFs,4,6,7 prior infection of mosquito cells with EILV also delayed replication and reduced viral titers of other alphaviruses, including SINV, WEEV, and CHIKV.
34
Furthermore, infection of
Bunyaviruses
Recently, four divergent groups of bunyaviruses, which do not appear to replicate in vertebrate cells, have been identified in mosquitoes.35–37 The first of these viruses to be isolated and characterized from mosquitoes collected in Cote d'Ivoire was Gouleako virus (GOLV).
35
The low genetic homology and lack of antigenic relatedness with other members of the

(
Although the three goukoviruses most studied to date (GOLV, CUMV, and BADUV) show restricted host range
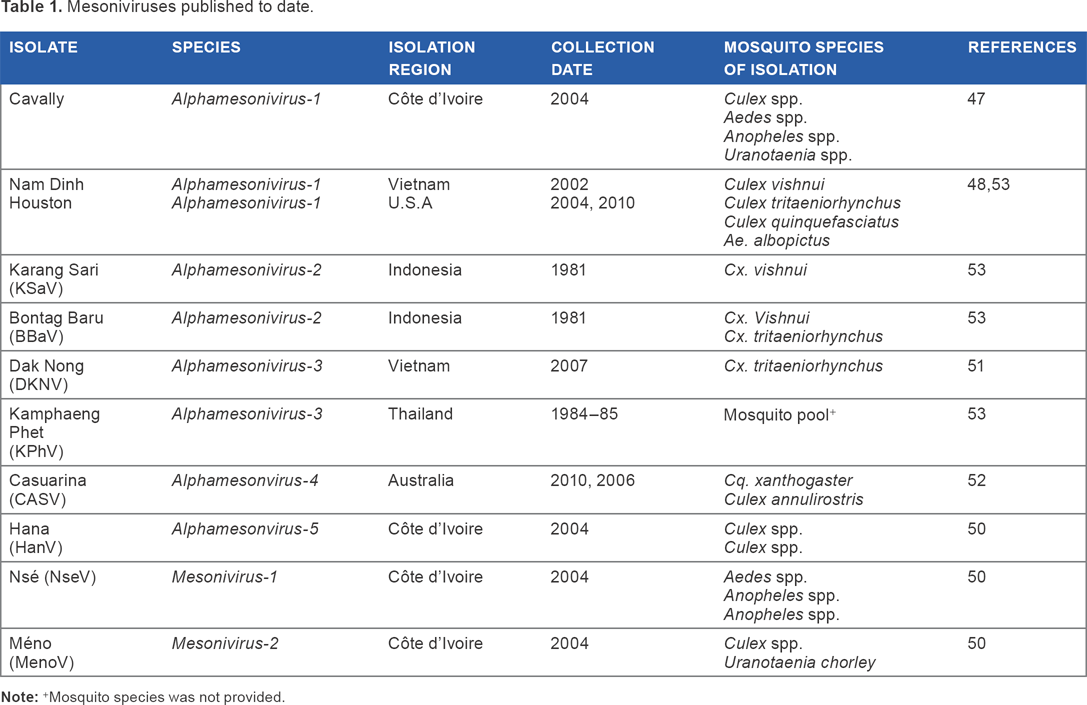
Mesoniviruses
The order
Mesoniviruses published to date.
Australian mesoniviruses
In 2014, our laboratory reported the first mesonivirus discovered in Australia. Casuarina virus (CASV) was isolated from

Phylogenetic tree showing the genetic relationship between the proposed species of the
Following the discovery of CASV, the first Australian isolates of
Reoviruses
The family
However, PLV shows a remarkably different phenotype in cell culture, failing to replicate in several mammalian and avian cell lines that supported efficient growth of CORV.
55
In contrast, PLV grew well in mosquito cells, suggesting that it has developed a restricted host range, indicative of a mosquito-only life cycle. Several other reoviruses also show a restricted host range (Table 2).
Putative insect-specific reoviruses published to date.

Currently unclassified. The authors concluded that Cimodo virus putatively defines a novel genus within the subfamily of
Another newly classified group of viruses in the
Negeviruses
The taxon negevirus was originally described by Vasilakis et al. 63 , who reported the isolation and characterization of six viruses that formed an orphan group with no strong similarity to previously described families. Since this first report, at least six new species of negeviruses have been discovered along with a number of reisolations of previously described negeviruses often from different countries and host species (Table 3).
Negeviruses published to date.

Bioinformatic analyses have indicated that the taxon negevirus is distantly related to the mite-transmitted, plant-infecting cileviruses and is likely to form two clades (
Research into these viruses to date has been mostly
Conclusions
With the application of deep sequencing approaches to virus discovery, many novel viral genomes have been detected in arthropod samples.42,61,66 While the outcomes of these studies have redefined the taxonomy and phylogenetics of mosquito-borne viruses at the genus and family level and provided a wealth of data for the analysis of their evolution, additional efforts to isolate these new viruses are essential to determine their phenotypic properties. These properties include host range, mode of transmission, potential for pathogenesis, as well as interaction with the mosquito host and viral pathogens vectored by the host.
Our studies and those of several other groups demonstrate that many ISVs are carried by mosquitoes and occur at high prevalence in some populations, apparently transmitted vertically. Furthermore, some of these viruses can regulate the transmission of pathogenic arboviruses in coinfected mosquitoes. Future studies should be directed at determining the mechanisms by which ISFs interfere with arbovirus transmission and the viral and host factors associated with their restriction to mosquito hosts and efficient vertical transmission. These studies will also require the development of essential ISV-specific research tools, including antibodies, molecular detection reagents, and reverse genetics systems.
Author Contributions
Conceived and designed the experiments: RAH, HBO, NAP, JH-P. Analyzed the data: RAH, HBO, BJM, CAO, AMGC, JJH, NDN, NAP, JMD, MGM, JH-P. Wrote the first draft of the manuscript: RAH. Contributed to the writing of the manuscript: RAH, HBO, CAO, AMGC, TBHP, JJH, NDN, RTB, NAP, JH-P. Agreed with the manuscript results and conclusions: RAH, HBO, BJM, CAO, AMGC, TBHP, JJH, NDN, RTB, NAP, JMD, MGM, JH-P. Jointly developed the structure and arguments for the article: RAH, HBO, CAO, AMGC, JJH, NDN, NAP, JH-P. Made critical revisions and approved the final version: RAH, HBO, BJM, CAO, AMGC, TBHP, JJH, NDN, RTB, NAP, JMD, MGM, JH-P. All the authors reviewed and approved the final manuscript.
Footnotes
Acknowledgment
The authors would like to thank Dr Sonja Hall-Mendelin for proof reading the manuscript.
