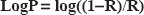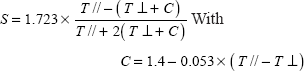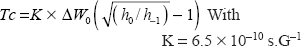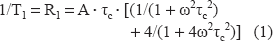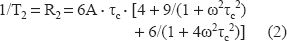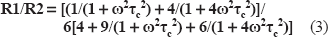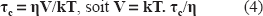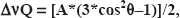Abstract
Shark liver oil (SLO), is used in natural medicine as immunity stimulant, cardiovascular protector and anti ageing reagent. These properties were related with the high amounts of alkylglycerols (22%) obtained from Greenland shark liver. After a control of the mean SLO composition by NMR and MS, surface and membrane interactions and antioxidant properties were investigated using NMR, ESR and ST measurements and the in vitro consequences on erythrocytes and cells were studied. An estimation of the composition of this extract was performed. Moreover, SLO was found not haemolytic (A concentration inducing 50% haemolysis, HC50 could not be reached) and superficial tension measurements revealed slight tension active properties. The 31P and 2H-NMR and ESR studies of phospholipid dispersions (dimyristoyl phosphatidyl cholin, DMPC) in the presence of SLO showed a significant increase in membrane fluidity at low temperature (below phase transition temperature) predominantly observed at the surface level. The anti oxidant activity was also confirmed, similar as that observed for vitamin E.
Introduction
Greenland sharks, and especially somnosius microcephalus are very robust species well adapted to hard environmental conditions such as deep and cold surroundings. In empiric traditional Scandinavian medicine, meat and oil from Greenland shark have been extensively used for healing of wounds, physical stress tolerance, and also immune stimulation and antitumor properties. These uses continuously faded out during the 19th century until early 20th when specific lipids (up to 50% in shark livers [1]) were identified as alkylglycerols [2] (1-O-alkyl-2,3-diacylglycerols and their metoxy derivatives [3]). This led to the first trials of A. Brohult [4] who evidenced increased production of granulocytes and thrombocytes and proposed the use of alkylglycerols to counterbalance the bone marrow depletion after radiotherapy in the therapy of carcinomas of the uterine cervix [5]. Later, alkyglycerols have been found to inhibit the growth and spread of transplanted or chemically induced tumors [6]. Immunological system stimulation was also identified, leading to bacteriostatic properties. It is noteworthy that the same substance, shown to be effective per os, exhibited both immunoreactivity stimulating and antitumor activity. Among the different mechanisms proposed, the ability of alkylglycerol to penetrate the cell membranes would stimulate the body’ s own defense system, mainly the macrophages. Besides, the results found when alkylglycerol was given before radiotherapy would also support the hypothesis of a direct interaction with radio induced free radical production. However, no precision of the mechanism involved was clearly proposed at the molecular level. This led us to investigate the biophysical properties of shark liver oil (SLO), the commercial form, by using both NMR and ESR spectroscopies, and assignment biophysics methods both in synthetic systems (phospholipidic membranes) and in red blood cells.
Experimental
Materials
ALKYROL®
ALKYROL® oil extract from Greenland shark liver was purchased by NUTRILYS® Company (Divonne les bains, France) and used without further purification. As this extract is a natural mixture, the amounts of SLO are better expressed in this paper in mg rather than in mM concentrations (even if the apparent density was estimated at 0.71).
This product was characterized by NMR (see Fig. 1 for peak assignment) and MS (ES+) analysis. As major MS peaks were found at m/z = 701; and also 633; 517; 351(m/2z), and from
1
H-NMR peak integration and
13
C direct spectra and DEPT datas, an very coarse estimation of the mean apparent molecular weight M#650 was proposed SLO. The corresponding molar ratios might be considered as only indicative and close to W/W ratios (molecular mass for Dimyristoylphosphatidylcholine, DMPC, is 678).
Top: proton nomenclature used for acyl chain labeling; middle:
1
H-NMR spectrum of SLO; 1 mg in CDCl3, 298 K: bottom: 1 mg in D2O, 298 K; Glycerol proton labeling: G1, G3, methylenic groups, G2, methinic group.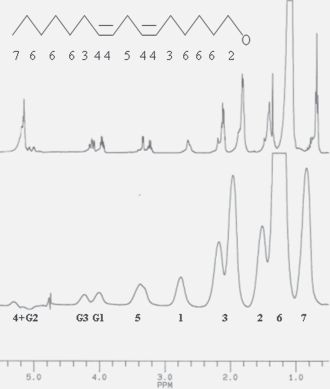
Chemicals
Dimyristoylphosphatidylcholine (DMPC), egg yolk phosphatidylcholine (EPC), and deuterated solvents were purchased from Sigma (La Verpillère, France) and were used as received. Chain perdeuterated DMPC-d54 was from Interchim, Montluçon, France.
Multibilayers (MLV)
DMPC liposomes for 31 P experiments were prepared in pure deuterated water by successive freezing and thawing cycles [7] until an homogenous milky sample was obtained. [8] The suspensions were degassed under nitrogen gas then introduced into NMR tubes and sealed. The final lipid concentration was 50 mM (in 500 μL samples), while SLO/DMPC in mixed systems was ranged from 1/50 to 1/25, mg/M. The same procedure was used for multilayers for 2 H-NMR experiments, except that 25% DMPC with perdeuterated chains were used (DMPC-d54) to build the liposomes.
Methods
Haemolytic activity
All procedures were in accordance with the standards for animal care established by our institute and were approved by our animal use ethic committee (decree 87–848 19 October 1987).
Blood from male Sprague-Dawley rats was collected in heparinated tubes and washed twice using isotonic NaCl solution; the hematocrit was then brought to 10%. 1 mL cuves were filled with the SLO solutions to test (0 to 32 μL) in 50 μL of DMSO and with 100 μL of the diluted blood in saline. The samples were stocked for 1 hour at 37 °C, then centrifuged at 2400 rpm, 4 °C for 10 minutes. Absorption measurements were finally performed on a Shimazu MCS-2000 absorption spectrometer at 540 nm, as described elsewhere. [1,9]
The haemolytic activities were expressed in terms of HC50, the concentration giving 50% haemolysis as referenced to i) the total haemolysis induced by triton X-100 addition or on sonicated samples ii) the absence of any haemolysis (0% haemolysis) evaluated on samples where only isotonic NaCl (0.9% W/W) solution was added.
NMR experiments
All NMR experiments were recorded on a Brüker AM-400 spectrometer. 1 H-NMR spectra in D2O were acquired at 298 K using a presaturation of the water resonance and a spectral width of 10 ppm. The chemical shifts were referenced by setting the water resonance at 4.75 ppm. 1 H-NMR control spectra were recorded using classical 1D and 2D (COSY, TOCSY (Sanders, 1989) experiments at 300 K, 2 mg, in perdeuterated di methyl sulfoxide (DMSO-d6).
In 1 H-NMR T1 and T2 measurements in water preparation (1 mg, D2O, 298 K) used the inversion recovery method [10] with a 10 ppm line width and a 5sec recycling delay to ensure relaxation.
Partition coefficient (LogP) calculation was realized by using an NMR method derived from the classical Shake-flask method [9]: A first
1
H-NMR spectrum was recorded acquired as previously in water, while using. 1 mg SLO in 1 mL D2O to ensure that the NMR observation area is fully included in the aqueous solution. Then an equal volume (1 mL) of perdeuterated octanol was added, and the sample vortexed and centrifugated to allow phase separation. The octanol phase was then removed. Another spectrum was the acquired and the intensity signal
31
P-NMR experiments were performed at 162 MHz. Phosphorus spectra were recorded using a dipolar echo sequence (π/2-t-π-t) [11] with a
2
H-NMR experiments were performed at 61 MHz. MLV were formed as for
31
P experiments whereas in deuterium depleted water. Deuterium spectra were recorded by using a quadrupolar echo sequence (π/2-t-π/2-t) with a
Surface tension measurements
Measurements were done on a Tensiometer CSC-Du Nouy (CSC N°70535) using the ring method of measurement in 20 ml of water. Pure water from MilliQ (18.2 Mω.cm) was used as reference (75.8 mN/m at 293 K).
Electron spin resonance (ESR): spin trapping investigation
Anti radical activity was assessed by in vitro spin trapping experiment. Reactive oxygen species were generated immediately before ESR experiment by a Fenton reaction (FeSO4, 0.1 mM and H2O2, 0.1 mM). The formation of short-lived radical species (OH) was evidenced by addition of Water Soluble α-(4-pyridyl-1-oxide)-N-t-butylnitrone (4-POBN) (Sigma, France) at 150 mM (in DMSO/H2O solution 5% V/V) spin trapping agent. Reaction was performed in an Eppendorf tube, 100 μL of FeSO4 were mixed with 100 μL of 4-POBN spin trap and with 2 μL of Alkyrol®. The trigger of reaction was performed by adding 100 μL of H2O2. Reference samples were prepared by replacing Alkyrol® by distilled water, Anti radical properties were also compared by replacing Alkyrol by 2 μL of Vit E.
The samples were transferred in 20 μL Pyrex capillary tube, an placed in 3 mm diameter quartz holder.
The spectra were acquired using the continuous wave mode with a ESP 380 (Brucker) ESR spectrometer, operating at a microwaves frequency of 9.71 GHz. The instrumental parameter were: microwave power of 10 mW, modulation frequency at 100 kHz with a modulation amplitude of 0.51 G, receiver gain was 6.30 × 104 and scan range was 70 G with magnetic field centred at 3430 G. Each sample was scanned 3 times at controlled temperature 295 K, with the following acquisition parameters: Time constant 20.48 ms, conversion time 20.48 ms and 5 repetitions. Figure 6 B shows typical ESR spectra of the control groups with the 4-POBN spin trap.
An estimation of free radical promotion was obtained by measuring the amplitude of the central doublet.
ESR spin label study
The fluidity of rat red cell membrane was investigated by ESR spin label experiments. Two spin labels (Sigma France) were used: 5 nitroxide stearate (5 NS) and 16 nitroxide stearate (16 NS). This fatty acids self incorporate the membrane and the nitroxide groups provide information of motional freedom of the label in biological membrane. So the former probes the superficial part of the membrane layer, the latter in its hydrophobic core. [12]
The experiments were performed on rat red cells. The erythrocytes were isolated from fresh blood by centrifugation at 4 °C (10 min, 1000 × g), then rinsed using saline, recentrifuged, this procedure being repeated until a clear supernatant was obtained, then brought to 30% packed cell volume. 2 μL of Alkyglycerol solution were added in each 1 mL sample and then labelled with 20 μL of spin label solution (5 ns 10– 3 M or 16 ns 10– 3 M). After 30 min incubation at room temperature, sample were transferred by capillarity in 20 μL Pyrex capillary tube. This tube was placed in a 3 mm diameter quartz holder, and insert into the cavity of the ESR spectrometer.
The ESR spectra were recorded at different controlled temperature (288, 293, 298, 303, 310 and 315 K) with the following conditions: microwave power 10.00 mW, modulation frequency 100 kHz, modulation amplitude 2.05 G, receiver gain 6.105conversion time 40.96 ms, time constant 20.48 ms. Sweep range was 160 G with a central field value of 3435 G.
The complete membrane incorporation of the spin labels was ascertained by the absence on the spectra of the extremely resolved ESR lines corresponding to free rotating markers.
5 NS experimentations: The value of outer and inner hyperfine splitting were measured (2T// and 2T⊥ respectively), on ESR specra (Fig. 5B), and order parameter S was calculated following the equation: [13]
The increase of the order parameter value means a decrease of local membrane fluidity.
16 NS experimentations: The changes in freedom motion of 16 NS were analyzed with the calculation of τc, the rotational correlation time. τc was calculated following the formula: [14]
In this formula, ΔW0 is the peak-to-peak line width of the central line; h0 and h-1 are the peak high of the central and highfield lines respectively (Fig. 5C).
The decrease of the rotational correlation time means a decrease of local membrane fluidity.
Mass spectroscopy
The ES-control spectra were acquired in CH2Cl2 as solvent with 1% formic acid, using a VG. Quatro II spectrometer from Micromass/Waters, and treated with the Masslink 4.00 V software. The capillary tension was 3.88 kV, and the cone tension and ion energy 88 V and 1.8 V, resolution values were set to 15.2, and the multipliers 1 and 2 set to 650 V.
Investigations and Results
SLO structure evaluation in solution and in water samples
Chloroformic solution
As expected a true solution of SLO was obtained in chloroform (see Fig. 1 top trace) and the control of SLO main composition could be easily obtained from standard 1D and 2D 1 H- 13 C- 31 P NMR and ES-MS experiments. Especially, no other hydrophobic components such as phospholipids, sterols an squalene were detected and the resonances of glycerol moiety (labeled G1,2,3) and those of the chain (see the nomenclature on the Fig. 1) were clearly identified. As relaxation times were quite homogenous (relaxation times T1 and T2 close to 1sec) [15] an estimation of the average length and insaturation of the chains was obtained by building indexes from 1 H-NMR peak integrals as follows:
As shown on Figure 1 the resonance labeled (4) at 5.2 ppm is representative of methynic group; however, since this resonance is completely overlapped by glycerol signal (G2), the unambiguous resonance of methylenic groups (3) nei ghbouring methynic (4) was used. The resonances (5) were representative of polyunsaturation, and terminal methyle peaks (7) of number of chains. For each group, the value of the integral was divided by the corresponding number of protons (3, for methyl, 2 for methylen) to allow a count of the number of groups. Finally, an estimation of the chain length reference,
Within the different samples controlled, no variation exceeded 10% from the following values:
Number of groups per chain;
MS spectra confirmed this homogeneity by giving exclusively a dominant line at m/z = 351.9, another of half the intensity at m/z = 517 and four minor components at m/z = 301–417–467–633.
Aqueous samples
Depending on chain length and insaturation found, partial apparent solubilization in the water was not excluded. [16] Hence,
1
H-NMR lines were detected on the NMR aqueous sample containing 1 mg SLO (see Fig. 1, bottom trace). However, the linewidthes measured (from 30 to 50 Hz) suggested the that supramolecular assemblies had been formed, such as micelles or droplets. This led to measure T1 and T2 relaxation times. These parameters are closely related to the correlation time τ
Similar relaxation values were found within the molecule (T2 = 30+/– 10 ms, T1 = 420+/– 20 ms) except for terminal methyl groups (resonance 7, T2 = 50 ms, T1 = 310 ms).
This allowed to calculate the range of correlation time by using the ratio T1/T2 as follows:
Relation in
Then, assuming a spherical approximation for the molecular assembly, the Stockes-Einstein relation allows an evaluation of the average apparent volume:
From these features, collective properties of SLO could be studied by using these aqueous dispersion in biological medium that organic solutions precluded. By considering the great importance of interfacial systems in biology, such as cell surfaces, complementary physico chemical tests were then performed.
Surface properties
The partition coefficient (LogP) was calculated as described in the method section. The value LogP = 1.3 well confirmed that, even if the solubility in organic solvent–-octanol- is more than tenfold that in water, the presence of SLO at the interfacial area is highly probable. As a consequence, possible tensioactive properties had to be tested. The result is shown on Figure 2: starting from 75.8 mN/m (pure water at 293 K) successive additions of SLO resulted in progressive diminution of surface tension down to ST = 53 mN/m around 5 mg/mL. Higher amounts of SLO induced no further decrease of ST. Such a limited evolution runs counter any detergent effect or soap-like interactions of SLO, as found for instance for SDS or amphiphilic [19] molecules like cyclodextrins. [20]
Superficial tension (dG-G°, mN/m), 298 K as a function of SLO concentration (mg/mL) (•), and percentage of haemolysis following the concentration of SLO (•□).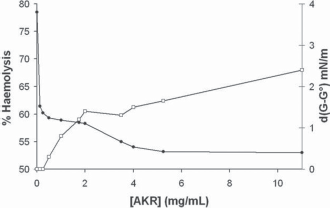
However, these negative tensioactive properties suggest interactions with membranes. This point is the topic of the next section.
Membrane structure and dynamics study by
2
H and
31
P-NMR
31 P and 2 H-NMR spectroscopies of phospholipid dispersions (MLV) were used to observe the structural and dynamics consequences of the presence of SLO at the polar head ( 31 P) and chain ( 2 H) levels of the membrane.
The polar head group level
As shown Figure 3A (bottom of column), the spectrum of pure DMPC dispersion (MLV) is typical of an axially symetric powder pattern, with a chemical shift anisotropy of 69 ppm, classical of DMPC bilayers in their liquid crystallin phase around (296 K) phase transition
7
The chemical shift difference between the lowfield and the highfield edges of the
31
P-NMR spectrum is called Chemical Shift Anisotropy (
31
P-NMR of DMPC: Column: A) typical spectrum of (top) DMPC bilayers (50 mM) close to transition temperature (296 K) (bottom) and in the presence of SLO (SLO/DMPC = 1/25 W/W); column B) spectra of ghosts prepared from rat erythrocytes (top), and in the presence of 3 mg SLO (bottom). Bottom traces: temperature dependence of the chemical shift anisotropy for pure DMPC (•), and SLO/DMPC systems, 1/25 W/W (□) and 2/25 W/W (Δ). The arrow indicates the point of the curve corresponding to the top traces.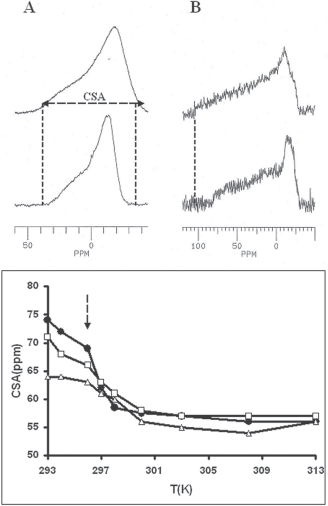
Such was also the case for the spectra recorded under the same conditions on SLO containing systems at various temperatures. Especially, no isotropic contribution typical of detergent effect was observed. However, a significant reduction in CSA value were measured at low temperature (under transition temperature, see Figure 3A, bottom trace); this increase in local fluidity was not detected at higher temperatures while transition temperature was found unaffected by the presence of SLO (297 K). The presence of structural rearrangements was also supported by this decrease in CSA at low temperature, with a normal transition temperature (297 K) and CSA values close to those of DMPC at higher temperatures.
The acyl chain level
2 H-NMR lineshape
Figure 4A (top) shows the spectrum of DMPC-d54 (dimyristoyl phosphatidyl choline with perdeuterated chains) dispersions. This spectrum is typical of phospholipid bilayers in the liquid crystal phase close to transition temperature (296 K) [21]. Such a spectrum appears as a super-imposition of symetrical doublets, each doublet corresponding to a methylenic
2
H-NMR spectrum of A) pure DMPC-d54 dispersions at 296 K (the spectrum is expanded in B to show the splitting of CD3 groups of pure DMPC -top- and in the presence of 1 mg SLO–-bottom-), Bottom traces temperature dependence of the half quadrupolar splitting (kHz) for pure DMPC (•), and SLO/DMPC systems, 1/25 W/W (□) and 2/25 W/W (Δ), for plateau resonances (C) and terminal CD3 group resonances (D).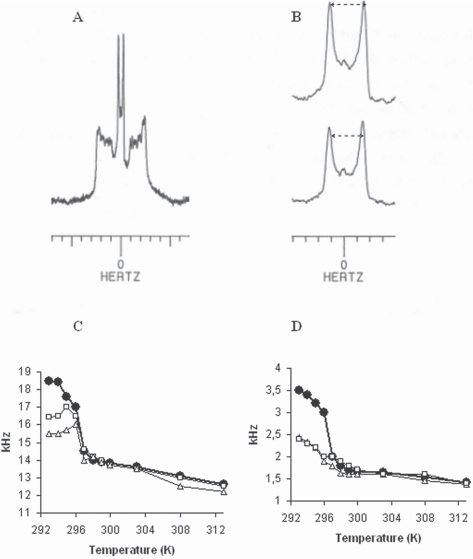
The main spectrum recorded under the same conditions (296 K) in the presence of SLO (R = 1/25 and 2/25 W/W) also shows a dramatic reduction in quadrupolar splittings both at the superficial level (the plateau region) and in the deep part of the membrane (right traces and curves Fig. 4). Besides, no other contribution indicative of isotropic rapid motion was found.
From this part, one can conclude that SLO induces an overall fluifization of synthetic membrane, exclusively present below transition temperature, without inducing any detergent effect and membrane structure and dynamics modification over phase transition. The following step was to test the relevance of these observations in biological systems, i.e. red blood cells, by using macroscopic haemolysis tests and ESR biophysical measurements.
Biological relevance of biophysical results
Haemolytic activity
The haemolysis curve is shown on the Figure 2. By comparison with well identified haemytic molecules (for instance natural β-cyclodextrin has a 50% haemolytic concentration of 13 mM,) [22] SLO haemolytic activity is found very low, since the maximum haemolysis obtained was 6.6% (21 mg/mL SLO) and 50% haemolysis could not be obtained.
31 P-NMR of erythrocyte ghosts
The Figure 3B (top) shows a typical spectrum of red blood cell membranes (100 mg ghosts in D2O for a total sample volume of 500 μL) recorded at 296 K with the same parameters as DMPC dispersions. Due to cellular organization (cytoskeleton, proteins) the overall membrane structure is significantly more rigid than synthetic systems, according with a CSA (chemical shift anisotropy) of 110 ppm. The addition of 3 mg SLO results in a significant reduction of this value (83 ppm), revealing an increase in collective fluidity without local membrane damages that should have been evidenced by the presence of isotropic line at 0 ppm. However, this effect required at least 2 mg SLO and was not observed for lower amounts.
ESR Spin labeling experiments
Spin label experiments were then realised to investigate the red cells membrane fluidity in different temperature conditions. Two probes were separately used, 5 NS gives information about superficial membrane fluidity, while 16 NS concerned the inner membrane region. The results are shown on Figure 5. An increase of the mobility of the two probes contribution could be observed in SLO groups, related to a global enhancement of the membrane fluidity.
ESR spin labeling experiment. (A) 5 NS and 16 NS results. Left Y axis: temperature dependance of the order parameter (5 NS) for control red cells (black diamond) and Red cell in presence of SLO (grey square). Right Y axis: temperature dependence of the rotational correlation time (16 NS) for control red cells (black triangle) and Red cell in presence of SLO (grey circle). (B) Typical 5 NS spectrum, parameter used for order parameter estimation are inner (2T⊥) and outer hyperfine (2T //) splitting. (C) Typical 16 NS spectrum, parameter used for rotational correlation time was central peak intensity H0, High field peak intensity and the with of the mid-field line W0.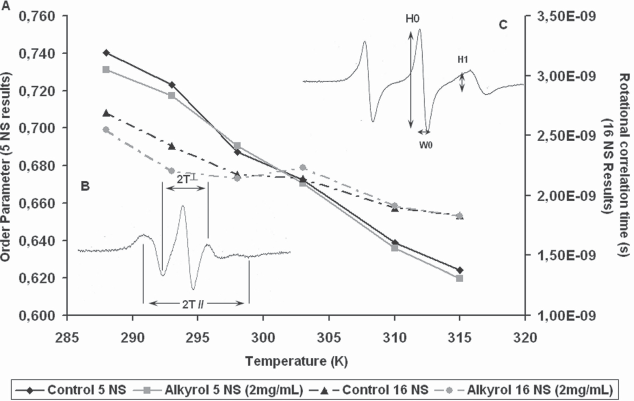
Furthermore, at low temperature (288 K and 293 K), a drop in the order parameter was measured in the SLO group compared to control, that disappeared at the physiological and at the above temperature (up to 315 K). The same observation was done for the rotational correlation time of the 16 NS probe with a decrease of τ in SLO group.
This feature indicated an overall increase of the membrane fluidity of the erythrocytes induced by SLO at the lowest temperature. This effect was not noticeable at physiological temperature.
ESR spin trapping experiments
The Fenton reaction in presence of 4-POBN yields characteristic six-line spectra (Fig. 6B showing 4-POBN results). The spin adduct hyperfine splitting constants were aN = 15.73 G and aH = 2.57 G. According to Finkelstein et al. [23] value: 4-POBN spin trap: aN = 15.60 G and aH = 2.55 G and to Augusto et al. [24] who found aN = 15.50 G and aH = 2.50 G; these hyperfine splitting constants correspond to a α-hydroxyethyl adduct. This is stable adduct results from a reaction between hydroxyl radicals initially generated and the spin trap.
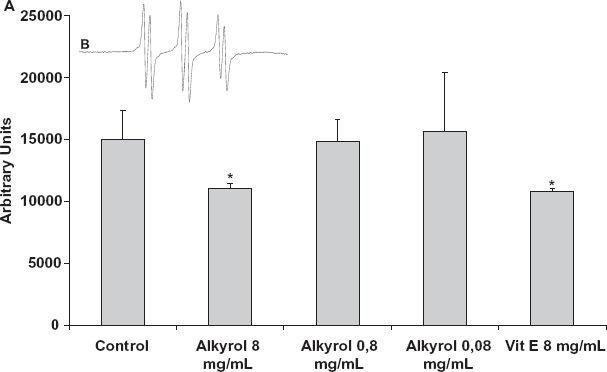
ESR spin trapping experiment. (A) Mean free radical production after exposure using spin trap N-tert-Butyl-α-(4-pyridyl)nitrone N'oxide (4-POBN). For each group the value was the average of 3 measurements ± SD. (B) Typical ESR of N-tert-Butyl-α-(4-pyridyl)nitrone N'oxide (4-POBN) radical adducts following Fenton reaction. (*) represents P < 0.05.
The histogram presented on Figure 6A, shows the free radicals promotion for 3 different concentrations of SLO (8 mg/mL, 0,8 mg/mL and 0,08 mg/mL) versus control and Vitamine E (8 mg/mL). Statistical comparisons were achieved using non-parametric tests (Kruskal-Wallis). Vitamine E is a reference antioxidant molecule able to recombine with free radical. In the presence of Vitamine E and Alkylglycerol in the same concentration, a significant strong decrease in the trace amplitude compared to control was observed (-26% SLO and -28% Vit E). For lower concentrations of SLO (0.8 and 0.08 mg/mL) no significant decrease in spin adduct detection could be shown.
Discussion
Beside the well established effects of alkyglycerols and polyunsaturated fatty acids on platelet aggregation and inflammatory reactions, the aim of the present work was to investigate physico chemical properties of SLO, especially membrane interactions. This work could be undertaken due to two initial conditions fulfilled:
the average composition was found homogenous between numerous samples tested; this was also in agreement with previous analysis showing a composition exclusively made of alkylglycerols and polyinsatured fatty acids [25] (PUFA); due to relatively amphiphilic properties, SLO exhibits a significant solubility in the water, by the way of self-organisation in supramolecular assemblies with an average diameter of 50å. This point allows the use of SLO in aqueous preparation without requiring to other organic cosolvents (DMSO…) or special preparations (encapsulation…).
The fundamental part of the study, performed on phospholipidic synthetic membranes allowed to identify the fluidifization of the membrane, as evoqued elsewhere. [26] An intercalation of the oil into the hydrophobic core of the membrane have been observed, affecting the order, packing and overall mobility of the lipid acyl chains. This effect was only observed when the chains are in the gel phase and ordered. This intercalation in the membrane, coupled with possible antioxidant effect should constitute the basis for a reasonably model for its action. However, our results clearly show that a dramatic fluidifization is obtained at low temperature, while this effect completely vanishes at temperature (over 296 K). This feature observed both in synthetic systems and in erythrocytes would be of interest in cold environmental conditions if related with the clinical effects expected (a better resistance to intense training…). However, it is worth to note that temperature regulation in sharks is very limited, even at very low temperature. Under this point of view, increased fluidity at low temperature would contribute to maintain cell and tissue functions in extreme environments where sharks live in. By the way of contrast, homeotherms such as humans generally maintain their internal temperature around 37 °C by active metabolic mechanisms such as increasing blood pressure, vasoconstriction and active shivering (from 37 to 34 °C) [27]. Lower temperatures lead to collapses and severe hypothermia to death by ventricular fibrillation. Here the observed effect of SLO at low temperature could appear of limited practical interest. However, in cold environments (e.g. high mountain training, outside work in winter…) cutaneous and subcutaneous temperature are often dramatically lower and SLO properties would play here a physiological role. This is particularly true in some pathologies such as Raynaud's syndrom or microcirculation abnormalities (Malan's syndrome) where both blood cell viscosity and capillary membrane fluidity are involved [28]. Also, cardiac surgery frequently uses extra corporal circulation systems: during operating time, central temperature is set down to 15 °C to protect the brain from the consequences of long lasting hypoxia (20–30min or more). Possible benefits in these circumstances remain to study.
SLO antioxidant properties are also of interest: hence, vitamin E is routinely used as adjuvant agent in radiotherapy, used for its antiradical properties in the prevention of radio induced fibrosis [29]. Another advantage is the apparent extremely low toxicity as tested by insignificant hemolytic activity and complete absence of detergent effect.
Another point of interest is that the amphiphilic properties of SLO are very favorable to overcome biological barrier (cellular membranes, intestinal wall…) by allowing both surface binding and spontaneous cell integration as shown by paramagnetic broadening experiments in cells (not shown). [30] Also, the anti oxidant properties were found similar as those of vitamin E and were related mainly to the general properties of polyinsatured fatty acids. [31]
Finally, the presence of non specific membrane properties of SLO, associated with its low toxicity is consistent with the great diversity of biological effects evoqued in the past, [32] such as anticancer, antioxidant and anti-inflammatory properties. A promising way for future research would be to evaluate the specific applications for work or physical effort in cold environments.
The extreme environmental conditions (pressure and cold) met in all day life of Greenland sharks would also been related with an adaptative Evolutional process leading to optimize biochemical composition of this species. From this point of view, the properties of SLO under high pressure conditions (diving) should also be evaluated.
Abbreviations
α-(4-pyridyl-1-oxide)-N-t-butylnitrone;
5 nitroxide stearate;
16 nitroxide stearate;
Chemical Shift Anisotropy;
egg yolk phosphatidylcholine;
Electron Spin Resonance;
dimyristoyl phosphatidyl cholin;
Hemolytic constant 50%;
Multibilayer Vesicle;
Electron Spray-Mass spectroscopy;
Nuclear Magnetic Resonance;
Platelet activating factor;
Shark liver oil;
Superficial Tension;
Vitamin E.
Footnotes
Aknowledgments
Thanks to J. Morin and T. Lerond for stimulating discussions, and Prof. J. Hàn-peuh-Pluu for manuscript relecture.