Abstract
Background
We investigated the effects and safety of linagliptin as an add-on therapy in patients with advanced-stage diabetic nephropathy (DMN) taking renin–angiotensin–aldosterone system (RAAS) blockers.
Method
Twenty advanced-stage DMN patients (estimated glomerular filtration rate (eGFR): 24.5 ± 13.4 mL/min/1.73 m2) taking RAAS blockers were administered 5 mg/day linagliptin for 52 weeks. Changes in glucose and lipid metabolism and renal function were evaluated.
Results
Linagliptin decreased glycosylated hemoglobin levels (from 7.32 ± 0.77% to 6.85 ± 0.87%,
Conclusion
Linagliptin as an add-on therapy had beneficial effects on glucose and lipid metabolism without impairment of renal function, and did not have any adverse effects in this population of patients with advanced-stage DMN taking RAAS blockers.
Keywords
Introduction
The prevalence of diabetic nephropathy (DMN) is increasing worldwide. 1 DMN is the most common cause of end-stage renal disease.2,3 It is also a major risk factor for the development of cardiovascular disease. 4 A poorly controlled blood glucose level and hypertension are the main contributors to progression to end-stage renal disease and the development of cardiovascular disease in DMN.5,6 Appropriate management of blood glucose and blood pressure levels is important to improve the prognosis of patients with DMN.7–9
Renin–angiotensin–aldosterone system (RAAS) blockers are used as first-line agents for blood pressure control in DMN patients. They have been reported to decrease blood pressure and have beneficial nephroprotective and cardioprotective effects.10–12
For blood glucose control, although many kinds of hypoglycemic agents have been developed, most cannot be used in DMN patients with decreased renal function because they have diminished elimination by the kidneys, and may cause unfavorable side effects. Dipeptidyl peptidase-4 (DPP-4) inhibitors decrease blood glucose by inhibiting the degradation of glucagon-like peptide (GLP-1), which enhances insulin secretion from β-cells and decreases glucagon secretion from α-cells of the pancreas.13,14 Among DPP-4 inhibitors, linagliptin can be used for blood glucose control in patients with impaired renal function without any dose adjustment because it is mostly metabolized by the liver.15,16 Several clinical studies have reported that linagliptin improves glucose metabolism in patients with varying degrees of renal function, either as a monotherapy or in combination with other hypoglycemic agents.17–23 Linagliptin has also been reported to have beneficial effects on lipid metabolism and nephroprotective effects.22,23 This suggests that linagliptin as an add-on therapy in DMN patients taking RAAS blockers may show advantages in the management of DMN.
Only a few experimental and clinical studies have been reported on the effects of linagliptin in combination with RAAS blockers in DMN.22,24 Also, the effects and safety of linagliptin in advanced-stage DMN patients taking RAAS blockers have not been fully determined. In this study, we investigated the effects and safety of linagliptin as an add-on therapy in advanced-stage DMN patients taking RAAS blockers.
Participants and Methods
Ethical considerations
This study was performed in accordance with the ethical principles contained in the Declaration of Helsinki and was approved by the Ethics Committee of Saitama Medical Center, Jichi Medical University. Written informed consent was obtained from all patients.
Patients
Between March 2013 and July 2014, 30 patients were enrolled in the study. Inclusion criteria were as follows: >20 years of age, suffering from type 2 DMN, DMN with an estimated glomerular filtration rate (eGFR) ≤ 60 (mL/min/1.73 m2), urine protein/creatinine ratio (UACR) > 0.15 (g/g Cr), and taking angiotensin II-receptor blockers or angiotensin-converting enzyme inhibitors. Exclusion criteria were patients with type 1 diabetes mellitus or secondary diabetes mellitus, history of stroke or coronary heart disease, patients with malignancy, severe infection, urinary stones, steroid therapy, pregnant or lactating women, and patients with an allergy to linagliptin.
Study protocol
A diagram of the study design is shown in Figure 1. The study was a 52-week, single-center, prospective study. All eligible patients were administered linagliptin 5 mg orally in the morning once daily as an add-on to existing drugs including RAAS blockers, hypolipidemic drugs, and anti-hyperglycemic agents but not DPP-4 inhibitors. Six patients were changed from an existing DPP-4 inhibitor (vildagliptin) to 5 mg/day linagliptin. The dosage of the drugs, including RAAS blockers, hypolipidemic drugs, and anti-hyperglycemic agents, was not changed during the study period.
Diagram of study design.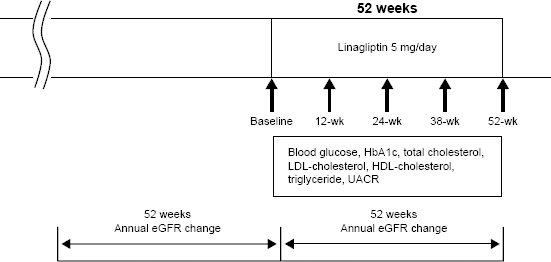
Changes in glucose metabolism [fasting blood glucose and glycosylated hemoglobin (HbA1c)] and lipid metabolism [total cholesterol, low-density lipoprotein (LDL)-cholesterol, high-density lipoprotein (HDL)-cholesterol, and triglycerides] were evaluated at baseline and at 12, 24, 38, and 52 weeks after administration of linagliptin. Changes in UACR were also measured at the same time points. The annual change in eGFR (mL/min/1.73 m2/year) was evaluated before and after administration of linagliptin.
Patients who underwent dialysis therapy because of progression to end-stage renal disease were removed from the study because changes in renal function could not be evaluated.
Laboratory methods
eGFR was calculated using a modified version of the Modification of Diet in Renal Disease formula of the Japanese Society of Nephrology: eGFR (mL/min/1.73 m2) = 194 × age-0.287 × serum creatinine-1.094 (multiplied by 0.739 for women). 25 Blood and urinary parameters were determined by the Department of Clinical Laboratory, Saitama Medical Center, Jichi Medical University.
Statistical analysis
Data are expressed as mean ± standard deviation. Repeated-measure analysis of variance was used to compare continuous data. Differences with a
Results
Thirty patients were enrolled in the study and administered linagliptin (Fig. 2). Four patients were discontinued because they progressed to end-stage renal disease and underwent hemodialysis. Another two patients were discontinued because of diagnoses of colon cancer and mediastinal tumor during the study period. One patient was lost to follow-up. Three patients were removed from the analysis because they were changed to different prescription drugs during the study period. Twenty patients completed the study (Fig. 2). The baseline characteristics of the analyzed patients who completed the study are listed in Table 1.
Patient flowchart. Patients’ baseline characteristic.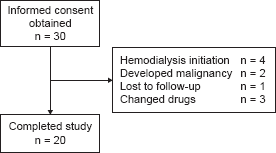
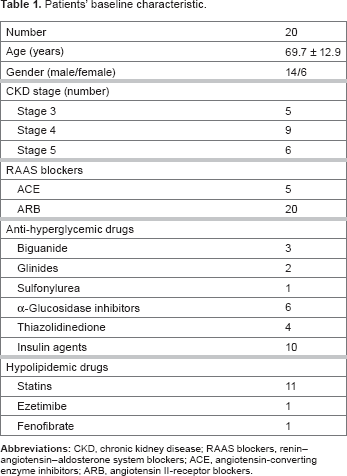
Effects of linagliptin on glucose metabolism
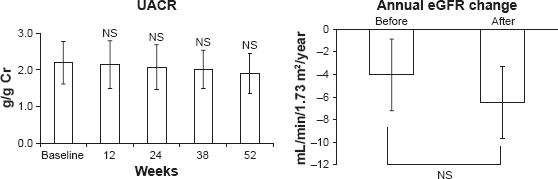
Linagliptin significantly decreased HbA1c levels, but did not change fasting blood glucose levels (Fig. 3 and Table 2).
Changes in HbA1c and fasting blood glucose (all patients, n = 20). Changes in parameters before and after linagliptin administration.
Effects of linagliptin on lipid metabolism and renal function
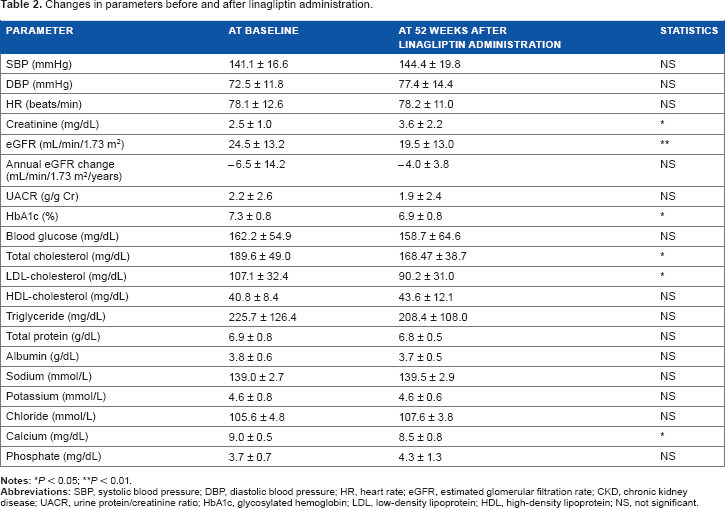
Linagliptin significantly decreased total cholesterol and LDL-cholesterol levels (Fig. 4 and Table 2), but did not change HDL-cholesterol and triglyceride levels (Fig. 4 and Table 2); nor did it change UACR and annual eGFR (Fig. 5 and Table 2).
Changes in total cholesterol, LDL-cholesterol, HDL-cholesterol, and triglyceride (all patients, n = 20). Changes in UACR and annual eGFR change.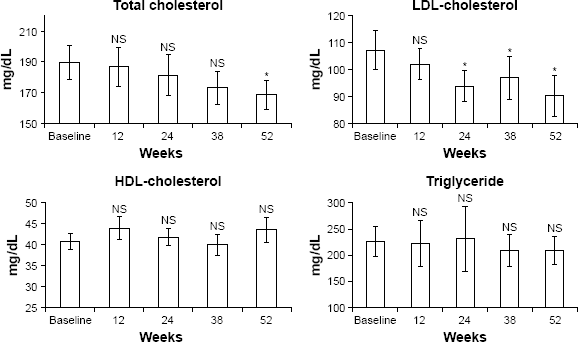
Changes in other clinical parameters and adverse effects
Other clinical and laboratory parameters were not changed by the administration of linagliptin (Table 2). No adverse effects, including joint pain, hypoglycemia, severe hyperglycemia, ketosis, or electrolyte abnormalities, were observed in patients administered linagliptin during the study period.
Discussion
In this study, linagliptin as an add-on therapy significantly decreased HbA1c and total cholesterol levels in advanced-stage DMN patients taking RAAS blockers. Linagliptin administration did not change UACR and annual eGFR, nor did it show any adverse effects in the patients. The results suggest that linagliptin has beneficial effects on glucose and lipid metabolism and can be used safely in such populations.
Linagliptin did not decrease fasting blood glucose levels. It has been reported that linagliptin decreases postprandial glucose levels rather than fasting blood glucose levels because GLP-1, increased by linagliptin, is secreted from the small intestine by the stimulation of food. 26 These blood glucose lowering mechanisms of linagliptin may explain the finding in the current study that linagliptin decreased HbA1c levels but did not decrease fasting blood glucose levels in DMN patients.
In addition to the beneficial effects of linagliptin on glucose metabolism, beneficial effects have also been reported on lipid metabolism as well as nephroprotective effects.22–24 Although the detailed mechanisms have not been fully determined, linagliptin may improve lipid metabolism and show nephroprotective effects by improving endothelial functions and reducing pro-oxidative and pro-inflammatory signals and inappropriate sympathetic nervous system activation through increasing levels of GLP-1 and other ligands. 26
Previous large-scale, double-blind clinical studies have reported that linagliptin improved glucose metabolism in DMN patients with renal impairment;21,22 however, the effects of linagliptin on lipid metabolism in this population were not studied. In the current study, linagliptin decreased total cholesterol and LDL-cholesterol levels in addition to improving glucose metabolism in advanced-stage DMN patients taking RAAS blockers. These results suggest that linagliptin has beneficial effects on both lipid metabolism and glucose metabolism in advanced-stage DMN patients. It should be noted that potential drug–drug interactions might have an effect on the results of the current study because the enrolled patients were on different types of drugs to control hyperglycemia and hyperlipidemia. Further studies are required to elucidate the mechanisms behind the effects of linagliptin on glucose and lipid metabolism and its interactions with other drugs.
It has been reported that linagliptin decreased UACR in the early to middle stages of DMN patients over the course of a 24-week study period. 22 Another study reported that linagliptin had little effect on renal function in DMN patients with severe renal impairment over a 1-year study period. 21 In the current study, the nephroprotective effects of linagliptin were not observed over 52 weeks. These results suggest that linagliptin does not have nephroprotective effects on advanced-stage DMN patients over the long term. Linagliptin may have nephroprotective effects at the early to middle stages of DMN, as previously reported. 22 Large-scale, long-term clinical studies investigating the nephroprotective effects of linagliptin at each stage of DMN are required.
Linagliptin did not induce any adverse effects, including joint pain, blood pressure, and electrolyte abnormalities, in the current study's population, which suggests that linagliptin can be used safely in advanced-stage DMN patients taking RAAS blockers.
This study had some limitations. It was a before–after study without a control group, and the number of patients was small. Large-scale, double-blind trials with an appropriate control group are required to investigate the effects of linagliptin on advanced-stage DMN patients taking RAAS blockers.
In conclusion, linagliptin as an add-on therapy significantly decreased HbA1c and total cholesterol levels in this population of advanced-stage DMN patients taking RAAS blockers without showing any adverse effects. Our results suggest that linagliptin has beneficial effects on glucose and lipid metabolism and can be used safely in such populations.
Author Contributions
Conceived and designed the experiments: HI, TK, MS, HM, KI, KH, YK, HM, TH, SO, MK, KT, and YM. Analyzed the data: YU, SO, and YM. Wrote the first draft of the manuscript: YU. Contributed to writing the manuscript: SO. Agreed with manuscript results and conclusions: HI, TK, MS, HM, KI, KH, YK, HM, TH, SO, MK, KT, and YM. Jointly developed the structure and arguments for the paper: HI, TK, MS, HM, KI, KH, YK, HM, TH, SO, MK, KT, and YM. Made critical revisions and approved the final version: YM. All authors reviewed and approved the final manuscript.
Footnotes
Acknowledgments
The authors wish to thank the members of the Division of Nephrology and the Division of Endocrinology and Metabolism, First Department of Integrated Medicine, Saitama Medical Center, Jichi Medical University. This study was supported by the Division of Nephrology, First Department of Integrated Medicine, Saitama Medical Center, Jichi Medical University.
