Abstract
Overactive bladder (OAB) is a common problem whose prevalence increases with advancing age and compromises health-related quality of life. Tolterodine was the first antimuscarinic drug specifically designed to treat OAB.
Through MEDLINE, we reviewed articles, standardization reports and meeting abstracts, published between 2001 and 2011, on the efficacy and tolerability of Tolterodine extended release (ER). Our search terms included Tolterodine, extended release, overactive bladder, therapeutic use, efficacy, tolerance, and adverse events.
Tolterodine ER, via steady drug release during the daytime, gives the benefit of constant serum concentration and thus better efficacy and tolerability in comparison to Tolterodine immediate release, even in elderly patients with co-morbid conditions.
Introduction
Overactive bladder (OAB) syndrome is defined as “symptoms of urgency, with or without urge incontinence, often with frequency and nocturia”. 1 OAB affects 6% to 17% of adults, and its prevalence increases with advancing age,2–4 compromising health-related quality of life (HRQoL). Also, co-morbidities (diseases of the central nervous system (CNS), cardiovascular system, etc.) are common among OAB patients, 5 and their occurrence as well as medications must be taken into account when making decisions on the most appropriate treatment.
Antimuscarinic drugs are keystone therapeutics of OAB symptoms, 6 with Tolterodine being the first drug specifically designed to control OAB. However, they are limited by their side-effects, due to the lack of urinary tract selectivity and possible drug interactions, especially in elderly patients. Extended release (ER) drugs may help reduce the side-effects, ensuring patient compliance and improving HRQoL.
Evidence Acquisition
We conducted a Medline search to identify the articles published between 2001 and 2011 on Tolterodine ER in OAB. Our keywords were Tolterodine, extended release, overactive bladder, therapeutic use, efficacy, adverse events, elderly, tolerance, Oxybutinin, Propiverine, Trospium, Darifenacin, Solifenacin, Fesoterodine, trial. We included all original research articles, review articles, standardization reports, meeting abstracts and case reports.
Pharmacology
The main mechanism of bladder contraction, for normal voiding as well as involuntary bladder contraction, is the synaptic release of acetylcholine and its binding to muscarinic receptors in the detrusor smooth muscle. There are 5 different muscarinic receptor subtypes. M2 receptors predominate over the M3 subtype in the bladder, and both are responsible for cholinergic bladder contraction. 7 The M3 subtype, liable for salivary secretion and papillary constriction, is present in the brain—with the M2 and M1 subtypes. 8
Tolterodine, a competitive antagonist at muscarinic receptors, is absorbed and undergoes hepatic metabolism by the cytochrome P450-2D6 system (CYP2D6) in extensive metabolizers, resulting in 5-hydroxylation derivate that has a similar pharmacological profile and makes a significative contribution to Tolterodine's therapeutic effect. 8 However, in poor metabolizers (10% of the Caucasian population), Tolterodine undergoes a slow CYP3A4-mediated N-dealkylation, and its metabolite has virtually no metabolic activity. Thus, higher serum Tolterodine concentrations are achieved in these patients because of its slow metabolism.10,11 Overall, a major portion of Tolterodine (77%) is excreted in the urine, with a minor amount in the feces within 7 days of its administration.8–10
Even though studies have demonstrated no specificity of either Tolterodine or its metabolite(s) for any muscarinic subtype, it has displayed functional specificity in vivo for muscarinic receptor subtypes in the bladder over the salivary glands. 9
Tolterodine ER was designed and became available in 2001. 8 The ER formulation consists of numerous little soluble microspheres contained within a gelatin capsule. The drug's slow release delivers constant serum concentration for 24 h. The maximum concentration is 25% lower and minimal serum concentration is approximately 150% higher than with the immediate release (IR) formulation. 10 Also, Tolterodine has low lipophilicity, which would limit its passage through the blood-brain barrier with consequent effects on the CNS. 9 Theoretically, this means fewer adverse events (AEs) and better tolerance.
No clinically significant interactions have been reported between Tolterodine and other commonly-prescribed drugs, including those metabolized by major CYP enzymes (eg, Fluoxetine, Warfarin, and oral contraceptive). Nevertheless, potent CYP3A4 inhibitors (azole antifungals, macrolide antibiotics, cyclosporin) may increase Tolterodine's plasma concentration. Also, medications affecting gastric pH must be considered with caution acknowledging the possible effect of pH changes on ER technology. 10
Efficacy of Tolterodine ER
Tolterodine ER (4 mg once daily) was designed to improve efficacy, tolerability and compliance over the IR formulation. Its efficacy and tolerability have been evaluated in multiple studies in both treatment-naïve patients and those who previously received medications against OAB symptoms. Overall, Tolterodine ER-treated patients experience significant improvements in the frequency of micturition, urgency episodes and urge urinary incontinence (UUI) episodes. These results are maintained over time.7,12–15
In males with lower urinary tract symptoms (LUTS) and non-obstructive benign prostate hyperplasia (BPH), urinary frequency, urgency episodes and UUI are reported to be significantly reduced, and median volume voided per micturition increased with Tolterodine ER, 16 while nocturnal micturitions, both severe and non-severe, have been found by Rackley et al 17 to be diminished in comparison to placebo (P = 0.0086). In association with Tamsulosin, significant improvement in the International Prostate Symptom Score (IPSS) storage subscale (–2.6 vs. –2.1 with tamsulosin and placebo; P = 0.0370) and OAB symptom bother questionnaire (OABq) scores (–17.9 vs. –14.4; P = 0.0086) has been observed, along with a low incidence of acute urinary retention (less than 1%).18,19 Kaplan et al 20 reported significant improvement of post void residual (PVR) in these patients. Men with small prostates and moderate-to-severe LUTS, including OAB symptoms, seem to benefit from Tolterodine ER monotherapy, while Tolterodine ER plus Tamsulosin combination appears to be effective regardless of prostate size.21,22
In patients with mixed urinary incontinence with predominant urge incontinence, Khullar et al observed that those treated with Tolterodine ER experienced less urinary frequency, urgency episodes and UUI episodes (–12.3 versus –8.0) and increased median volume voided per micturition compared to placebo. 23
In OAB patients without incontinence, Tolterodine ER is well-tolerated and reduces both diary and nocturnal severe OAB micturitions (–3.2 and –1.1 episodes respectively) as well as mean urgency ratings and frequency-urgency sum (treatment difference vs. placebo: –19.5; P = 0.003).24,25
Reinberg et al 26 reported significant improvement in daytime incontinence episodes in 47% of children enrolled in their study, but, in the same investigation, Oxybutynin ER was significantly more effective than Tolterodine ER in controlling daytime incontinence and frequency.
Two studies described the evolution of results after 12 months of treatment.27,28 Although the patient populations were dissimilar, there was no significant difference in the results. In both studies, more than 70% of patients completed their 12-month protocol, and among them, more than 85% were >75% compliant. No development of tolerance was noted for patient perception variables. Both studies reported significant improvement in the number of UUI episodes per week (median change –83% in Kreder's report), in the number of micturitions per day (–21% in Takei's report) and in volume voided per micturition (+19% to 25%) at 12 months compared to baseline. Most of all, both studies encountered no increment in AE frequency with long-term compared to short-term treatment.27,28
Effects of Tolterodine ER on Patient-Reported Outcomes (PROs)
Because patient satisfaction with treatment, although affected by patient perceptions of OAB symptoms, influences the long-term use of prescription treatments, assessing PROs has become an increasingly common means of evaluating treatment efficacy.
The IMPACT (IMprovement in Patients: Assessing symptomatic Control with Tolterodine) trial enrolled 896 patients in primary care and obstetrics/gynecology offices in the United States in an open-label, single-arm study. 29 It showed significant improvement in diary-evaluated symptoms, in perception of bladder condition, symptom bother and HRQoL.
The STARGATE study 30 enrolled 56 female patients with OAB to assess symptom-specific, patient-reported goal achievements. Most OAB patients manifested improvement of their OAB symptoms with 12-week Tolterodine ER 4 mg treatment and significant attainment of symptom-specific goals.
Rogers et al 31 conveyed the results of a multicenter, double-blind, placebo-controlled trial that included 411 sexually-active women with OAB symptoms. 78% of patients reported treatment benefit and satisfaction. Tolterodine ER led to improvement in Patient Perception of Bladder Condition (PPBC) responses, OABq symptom bother and HRQoL compared to placebo. Similar findings were obtained by Van Kerrebroeck et al in a double-blind, placebo-controlled trial conducted at 167 centers and enrolling 987 patients. 32
Höfner et al 19 documented 741 male patients with OAB and LUTS suggestive of BPH not sufficiently improved after alpha-blocker and treated with Tolterodine ER in a non-interventional, observational study. OABq and IPSS indicated marked recovery of HRQoL with Tolterodine ER.
Age, concomitant diabetes mellitus, concomitant alpha-blocker use, duration of symptoms, baseline maximum flow rate (Qmax) and PVR do not appear to have a relevant effect on the symptomatic response to treatment. 19
Adverse Effects
All antimuscarinic agents prescribed to control OAB have high withdrawal rates because of AEs. Although Tolterodine ER does not have pharmacological selectivity for any muscarinic receptor subtypes, it has functional selectivity for muscarinic receptor subtypes in the bladder over the salivary glands.
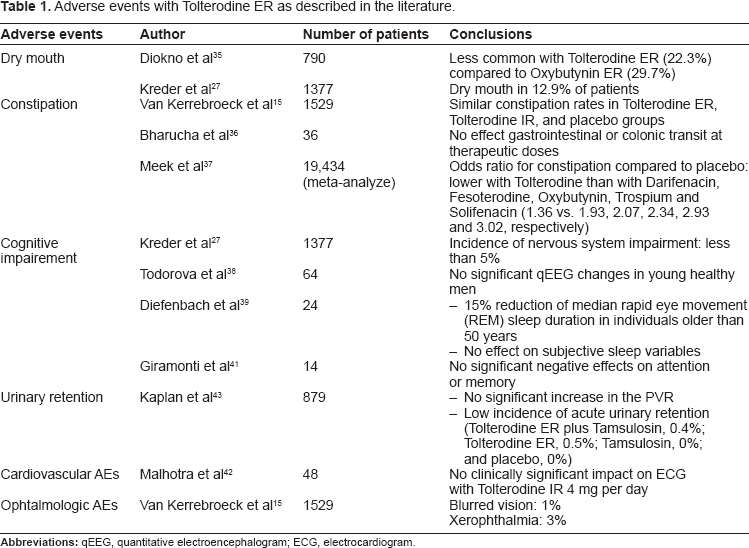
Adverse events described with Tolterodine ER are summarized in Table 1. Chapple et al 33 noted that Tolterodine ER was associated with a statistically significant lower risk of AEs than placebo (RR = 0.71; 95% CI 0.53–0.95; P = 0.02). Gopal et al accessed a large administrative database on 29,369 women on anticholinergics to ascertain the discontinuation rate. 34 Tolterodine ER had the lowest adjusted cumulative incidence of discontinuation at 6 months compared to Oxybutynin, Tolterodine IR and Oxybutynin ER (54% vs. 71%, 61% and 57%, respectively). Takei et al presented no evidence of increased AEs frequency with long-term exposure. 28 Dry mouth is generally mild and its incidence seems to diminish with treatment duration.27,28 At therapeutic doses, Tolterodine ER have minor effects on bowel habits 37 and no effect on cognitive function. The incidence of urinary retention in patients on Tolterodine ER is low. 43
Adverse events with Tolterodine ER as described in the literature.
Efficacy and Safety of Tolterodine ER in the Elderly
The older generation is growing in both total number and proportionally across most of the world. It is estimated that, in 2030, 1 in 5 Americans will be aged more than 65 years. 44 The number of elderly patients with OAB will continue to rise as the older population expands. 3
OAB symptoms are often coupled with co-morbid conditions. Bowel symptoms and falls related to night-time urgency are major concerns among the elderly. Also, the physical changes associated with age involve altered bladder function as well as drug dosing, solubility, metabolism and clearance. Because of multiple co-morbidities, poly-pharmacy is common among older adults and heightens the risk of AEs and drug-drug interactions. All these conditions affect OAB diagnosis, treatment efficacy and tolerance. 45
Therapeutic effects of Tolterodine ER can be seen within a week, but maximum outcome is evident after 5–8 weeks of administration. 46
Numerous trials have assessed Tolterodine ER efficacy in older patients. Although much of the data are from post hoc studies of clinical trials that were not powered to review Tolterodine ER in old patients, overall, the drug is associated with improvement of OAB symptoms and HRQoL. In an open-label study, Michel et al reported that increased age was linked with a slight but statistically significant decline of Tolterodine treatment efficacy. 47 However, in a 12-week, double-blind, placebo-controlled clinical trial conducted at 167 medical centers, comparing old (over 65 years) to young patients on Tolterodine ER, Zinner et al 48 discerned that it improved symptoms, HRQoL and self-reported measures in older patients with OAB with no significant difference between age groups. Ulahannan and Wagg 49 reported significant and comparable reduction of micturition frequency, incontinence episodes per week with increased volume voided per micturition among old and young patients on Tolterodine ER.
No differences in AEs were found in studies directly comparing older and younger patients. Griebling et al reported similar discontinuation rates (less than 5%) because of AEs in both the Tolterodine ER and placebo groups. 50 Dry mouth and constipation were common in most trials. Mild to moderate dry mouth occurred in 15% of elderly patients (as in patients under 65 years of age). No significant difference in constipation rates (5% vs. 3% in the placebo arm) was noticed between age groups—a real concern among older patients—and none of the other AEs was higher in older subjects treated with Tolterodine ER. Zinner et al 48 reported a similar frequency of constipation in younger (aged <65 years) and older (aged >65 years) patients receiving Tolterodine.
Another important concern in older patients is the cognitive impact of Tolterodine. OAB medications can worsen cognitive performance measures in some individuals, particularly those with existing dementia. 51 There have been isolated case reports of cognitive impairment in old patients on Tolterodine.52,53 Todorova et al 38 published the results of a randomized, single-blind, parallel-group, topographical qEEG clinical phase I study investigating potential CNS AEs of antimuscarinic drugs. Tolterodine showed limited effects on qEEG activity. 38 However, this outcome has not translated into differences in cognitive function. Kay et al 54 detected no CNS AE with Tolterodine when they performed cognitive testing (recent memory on the Name-Face Association Test) in older patients (mean age 63 years) randomly assigned to either 3 weeks of Tolterodine ER (4 mg) or Oxybutynin ER (20 mg).
Comparison with other Antimuscarinic Agents
Pharmacological treatment of OAB comprises oral antimuscarinic agents, which are divided into older-generation medications, including Oxybutynin, and new-generation drugs, such as Solifenacin, Tolterodine, Darifenacin and Fesoterodine. The latter have reduced CNS penetration and better selectivity for the M3 subclass of acetylcholine receptors, resulting in improved tolerability.
Oxybutynin
Oxybutynin IR was the gold standard for pharmacological treatment of OAB for nearly 30 years. Intolerable systemic side-effects led to poor patient compliance and thus limited its clinical utility. Several other formulations have since been developed to ensure tolerable AEs and compliance.
Overall, Oxybutynin is slightly more effective than Tolterodine in improving baseline urinary symptoms, but patients taking Tolterodine show significantly lower AE and dose reduction rates compared to those on Oxybutynin.
Two major, randomized, double-blind, active-controlled studies compared the tolerability and efficacy of Tolterodine ER and Oxybutynin ER. Sussman and Garely 55 reported the results of the Antimuscarinic Clinical Effectiveness Trial (ACET) which enrolled a total of 1,289 patients. After 8 weeks, more patients on Tolterodine ER perceived an improved bladder condition compared to Oxybutinin ER (70% vs. 59%). The therapeutic response was greater in a subgroup of patients whose perception of bladder condition was moderate to severe at baseline.
The OPERA study 35 found no statistical significance in average weekly UUI episodes between groups, but Oxybutynin was significantly more effective in reducing micturition frequency and urinary incontinence episodes than Tolterodine (23% vs. 16.8%, P = 0.03).
In post hoc analysis of the OPERA trial, Anderson et al 56 compared the efficacy of Tolterodine ER and Oxybutynin ER in women with and without previous anticholinergic treatment of OAB. In the group of previously-treated patients, Oxybutynin ER was more effective in reducing micturition frequency and UUI episodes. Fewer differences between Oxybutynin and Tolterodine were seen in the antimuscarinic-naïve group.
Dry mouth is the most common AE reported. Armstrong et al 57 combined the results of 2 large clinical trials: OBJECT and OPERA. They reported a lower percentage of dry mouth in patients on Tolterodine ER compared to Oxybutinin ER (22% vs. 29%). Other AEs included constipation, diarrhea, headache and urinary tract infection, with no apparent difference across treatment groups, and most of them (>90%) were mild or moderate in intensity in all treatment groups. Sussman and Garely 55 observed that patients treated with Tolterodine ER reported significantly lower severity of dry mouth compared to those on Oxybutynin ER. Chu et al 58 published a subanalysis of the OPERA trial, comparing the CNS tolerability profiles of Oxybutynin ER and Tolterodine ER formulations. The incidence of CNS events was low and similar between groups (9% for Oxybutynin, 8% for Tolterodine), despite the fact that Tolterodine is 30 times less lipid-soluble than Oxybutynin.
Propiverine
No randomized control trials have compared Propiverine and Tolterodine ER. A randomized, double-blind, multi-center study compared Propiverine to Tolterodine IR in patients with detrusor overactivity over a period 4 weeks. 59 Tolterodine and Propiverine showed comparable efficacy, tolerability and improved HRQoL.
Trospium
Tolerability and efficacy data on Tolterodine ER vs. Trospium chloride are not available.
A comparative trial on the safety and efficacy of Trospium chloride, Tolterodine IR and placebo was conducted in subjects with urge syndrome in Russia, Bulgaria and Poland. 60 Participants (n = 234) were randomized to 1 of 3 treatment arms for 3 weeks: Trospium chloride 20 mg twice a day, Tolterodine IR or placebo. 24-h micturition frequency from baseline was –3.4 in the Trospium chloride group, –2.6 in the Tolterodine group, and –1.9 in the placebo group. AEs were similar, mostly consisting of gastrointestinal disorders, such as dry mouth, which was generally mild (6.3%, 27.2% and 28.9% in the placebo, Trospium chloride and Tolterodine groups, respectively). In their topographical qEEG evaluation study, Todorova et al 38 reported that Tolterodine showed limited qEEG disturbances comparable to Trospium chloride and milder than those caused by Oxybutynin.
Darifenacin
No controlled trial comparing Darifenacin and Tolterodine ER is available.
Zinner et al investigated patient-reported outcomes and clinical parameters during Darifenacin treatment in 497 OAB patients who expressed dissatisfaction with prior Oxybutynin ER or Tolterodine ER therapy. 61 Darifenacin administration resulted in statistically significant improvements in Patient Perception of Bladder Condition scores, micturition frequency, urgency and UUI episodes at 12 weeks from baseline. Improvements were similar in patients previously treated with Oxybutynin ER or Tolterodine ER.
Solifenacin
The STAR (Solifenacin and Tolterodine as an Active Comparator in a Randomized) trial compared the efficacy and tolerability of Solifenacin (5 mg and 10 mg) and Tolterodine ER (4 mg) in a prospective, double blind, double-dummy, parallel group study. 62 Of 1355 patients screened, 30% were over 65 years of age. Solifenacin treatment (5 mg and 10 mg) was associated with statistically significant improvements in urgency (P = 0.035), UUI (P = 0.001) and overall incontinence (P = 0.006) compared to patients receiving Tolterodine ER. Solifenacin-treated patients also reported a larger reduction of nocturia episodes (–0.71 versus –0.63). Moreover, 59% of Solifenacin-treated patients who were incontinent at baseline became continent, compared to 49% of patients treated with Tolterodine ER. Dry mouth and constipation were seen more in the Solifenacin group, but most of these AEs were mild to moderate. Withdrawal from both groups because of AEs was comparable.
Chancellor et al 63 conducted a multi-center, open-label, flexible-dose study on the efficacy, tolerability, and effects of Solifenacin on HRQoL in 606 patients previously treated with Tolterodine ER and who wished to switch therapy because of insufficient subjective improvement of urgency. Among these patients with residual urgency after treatment with Tolterodine ER 4 mg, Solifenacin was associated with significant amelioration of urgency and other diary-documented OAB symptoms as well as enhancement of HRQoL and perceived OAB bother.
Fesoterodine
The US Food and Drug Administration approved Fesoterodine 8 mg in 2008. Its efficacy is superior to that of Tolterodine ER 4 mg in reducing UUI episodes and in improving most patient-reported outcome measures but with a higher risk of AEs.
Kaplan et al 64 randomized 2,417 patients in a 12-week, prospective, double-blind, placebo-controlled trial to fesoterodine (4 mg for 1 week, 8 mg for 11 weeks), tolterodine ER 4 mg or placebo. Fosoterodine showed superior efficacy over Tolterodine ER and placebo on UUI episodes, micturitions and urgency episodes as well as on self-reported patient assessments of bladder-related problems, urgency, symptom bother and HRQoL. However significantly more patients reported dry mouth (28% vs. 13%) and constipation with Fesoterodine compared to Tolterodine ER.
Herschorn et al 65 compared Fesoterodine and Tolterodine ER in a 12-week double-blind, double-dummy, placebo-controlled, randomized clinical trial that included 1,712 patients. Fesoterodine showed superior efficacy over Tolterodine ER 4 mg in reducing UUI episodes (primary endpoint), total and nocturnal voids, urgency episodes, severe urgency episodes, and frequency-urgency sum per 24 h, mean voided volume per void, except for nocturnal voids, and in improving most patient-reported outcome measures, except for the OABq sleep domain.
Actual Indication of Tolterodine ER in OAB Management
Tolterodine was the first antimuscarinic drug designed specifically for OAB treatment. The ER formulation has been evaluated extensively and has shown high efficacy with good safety and tolerability profiles.
However, providing clear indications for clinical practice is complex. Meta-analyses and evidence-based guidelines concerning antimuscarinic medications against OAB provide no consensus about appropriate first-line or second-line therapy.
We believe that Tolterodine ER represents a good first choice in naïve patients. Solifenacin or Fesoterodine could be cost-effective alternatives.66,67
As for the choice of second-line drug, no randomized controlled trials have aimed to identify the most efficient medication in case no success is met with some of the other first line choices. Thus, this decision cannot really be based on evidence. From available data, Fesoterodine and Solifenacin are possible options.
Conclusion
OAB patients suffer decreased QoL and productivity. Antimuscarinic agents are the mainstay of OAB therapy. Although Tolterodine ER does not have pharmacological muscarinic receptor subtype selectivity, it has been shown to possess functional selectivity for muscarinic receptor subtypes in the bladder over the salivary glands. It has demonstrated high efficacy and good safety and tolerability profiles.
Author Contributions
Conceived and designed the experiments: OEA, JC. Analysed the data: OEA, JC. Wrote the first draft of the manuscript: OEA, JC. Contributed to the writing of the manuscript: OEA, JC. Agree with manuscript results and conclusions: OEA, JC. Jointly developed the structure and arguments for the paper: OEA, JC. Made critical revisions and approved final version: JC. All authors reviewed and approved of the final manuscript.
Competing Interests
JC is a board member and consultant to AstellasPharma Canada and both him and his institution have received payments for lectures and development of educational presentations from AstellasPharma Canada. JC also has received or has pending grants from AtellasPharma Canada and Paladin. OEYA reports no actual or potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
