Abstract
Introduction
This multicenter, randomized, double-blind, parallel-group, Phase III, pivotal trial investigated the efficacy and safety of solifenacin succinate 10 mg, a once-daily (OD) oral antimuscarinic agent, in overactive bladder syndrome (OAB).
Materials and methods
A total of 634 adult patients with OAB symptoms were randomized to either solifenacin 10 mg (n = 318) or placebo (n = 316) OD over 12 weeks, to examine changes from baseline in micturition-, incontinence-, urgency- and nocturia-episodes/24 hours, measured using a 3-day diary.
Results
Solifenacin significantly reduced the mean number of micturition-, urgency- and incontinence-episodes/24 hours at end of treatment (
Conclusions
Solifenacin 10 mg OD was well tolerated and effective in treating major OAB symptoms, including urinary incontinence, frequency and urgency.
Introduction
Overactive bladder (OAB) is a symptom syndrome suffered by many individuals (approximately 17% or 33 million individuals in the United States [US]),1,2 and is defined by the International Continence Society as ‘urgency, with or without urgency incontinence, usually with frequency and nocturia, in the absence of pathologic or metabolic conditions that might explain these symptoms’. 3 The symptoms of OAB are inconvenient and burdensome, and can result in significant impairment of quality of life.4–6
The established first-line drug therapies for OAB are antimuscarinic agents, which act to reduce the severity of detrusor contraction by inhibiting the parasympathetic pathway in the bladder. 7 However, a lack of selectivity among such agents for muscarinic receptors located in the bladder can lead to a high incidence of anticholinergic side effects, such as dry mouth, dry eyes, blurred vision and constipation, due to blockade of muscarinic receptors elsewhere.8,9 Solifenacin succinate is an antimuscarinic agent that has demonstrated in a number of preclinical models to be more selective for the muscarinic receptors within the bladder, relative to those located in the salivary glands, compared with other antimuscarinic agents, such as oxybutynin and tolterodine.10,11
Such preclinical evidence led to the clinical development of solifenacin in patients with OAB, which included four 12-week, randomized, controlled, double-blind, parallel group, Phase III studies. 12 Collectively, these studies showed that 5 or 10 mg/day doses of solifenacin significantly improved urinary frequency, as measured by the number of micturitions per 24 hours, relative to placebo. 12 Solifenacin was also associated with significant improvements in the number of incontinence episodes per 24 hours and the volume voided per micturition. 12 On the basis of the efficacy and safety demonstrated in these four Phase III studies, the US Food and Drug Administration granted approval for the use of solifenacin in the treatment of OAB with symptoms of urge urinary incontinence, urgency and urinary frequency. 13 Data from three of the pivotal trials have been published previously, outlining the efficacy of solifenacin against all OAB symptoms including the key symptom of urgency.14–16 Here we present the results from the fourth pivotal Phase III study.
Methods
Study design
This multicenter, Phase III, randomized, double-blind, placebo-controlled, parallel-group, pivotal trial was designed to evaluate the efficacy, safety and tolerability of solifenacin succinate 10 mg in reducing symptoms of OAB, based on a 3-day patient voiding diary. The study comprised of 2-week screening/washout period, a 12-week treatment period and a 2-week post-treatment follow-up period. Men and women aged ≥18 years with OAB symptoms including urinary frequency, urgency or urgency incontinence, were eligible for entry into the 2-week screening phase. Patients were required to keep micturition diaries for 3 days during the screening period; those who had recorded a mean of eight or more micturitions per 24 hours plus a mean of one or more incontinence episode per 24 hours and/or a mean of one or more urgency episode per 24 hours were eligible for inclusion into the randomization phase. Exclusion criteria included stress incontinence or mixed incontinence where stress was predominant (mixed incontinence was allowed otherwise); patients with a neurological cause of detrusor overactivity; urinary retention; and Grade III/IV prolapse with cystocele and recurrent or active urinary tract infection. Women of childbearing potential were required to have a negative serum pregnancy test at screening and to use a medically acceptable form of contraception during study participation.
The study was conducted in 33 centers across the US in 2001. Six hundred and thirty-four patients were randomized and received at least one dose of either solifenacin (10 mg OD, n = 318) or placebo (n = 316); the first dose of study medication was taken at the baseline visit for the treatment period.
Patients were instructed to complete a micturition diary for the 3 days preceding each scheduled visit (Weeks 4, 8 and 12). The patient recorded the date and time for micturitions, and incontinence and urgency episodes. The volume voided per micturition was recorded during any 2 of the 3 days. Nocturnal voids and episodes of nocturia (defined as an urge to urinate that woke the patient from sleep) were captured as all micturitions that occurred between the time the patient reported going to bed and the reported time of waking. The patient also recorded the time they went to bed and the time that they woke up.
The study was performed in accordance with the International Conference on Harmonization Good Clinical Practice guidelines, and the principles of the Declaration of Helsinki. The study protocol was approved by the responsible ethical committee at each study site. All patients were informed of the nature and purpose of the study, and written informed consent was obtained before screening.
Efficacy assessments
The primary efficacy variable in this trial was the change from baseline to endpoint (defined as the last available on-treatment visit on or before Week 12) in the mean number of micturitions per 24 hours. Secondary efficacy variables included the change from baseline to endpoint for the mean number of incontinence-, urgency-, nocturnal voiding- and nocturia-episodes per 24 hours, and the mean volume voided per micturition.
Safety assessments
Safety was evaluated throughout the trial by monitoring treatment-emergent adverse events (AEs), which were categorized by severity and likelihood of causal relationship to study medication. AEs were recorded during the scheduled visits (Weeks 4, 8 and 12) and safety assessments were performed, including vital signs and electrocardiograms. A final follow-up visit (Visit 6) was scheduled in order to record AEs and perform physical examinations. Laboratory assessments (hematology, clinical chemistry and urinalysis) were also carried out at these time points. Physical examinations were performed at screening, baseline, Week 12 and at post-treatment follow-up. Post-void residual volume was measured by bladder scan at screening and at Week 12.
Statistical methods
Based on the detection of an active–placebo difference of one micturition per 24 hours, using a standard deviation (SD) of three, a significance level of α = 0.05 (two-sided) and a power of 90%, it was estimated that a sample size of 250 patients per treatment arm would be required to complete the study. Assuming a dropout rate of 20%, approximately 630 patients needed to be randomized.
All statistical comparisons were made using two-sided tests at a significance level of α = 0.05. Continuous variables were summarized using descriptive statistics, and frequencies and percentages were used to report categorical data. Efficacy analysis was performed on the mean change from baseline to endpoint. Patients with a baseline and at least one on-treatment efficacy assessment were included in the analysis. Treatment groups were compared using analysis of variance with terms for center and treatment. Last observation carried forward was used for endpoint values; endpoint was the last available on- treatment visit on or before Week 12 (Visit 5).
Treatment group comparisons of the percentage of patients with incontinence at baseline who were restored to continence (i.e. did not report incontinence in the 3-day diary at endpoint) were based on the Cochran–Mantel–Haenszel test. This test was also used to evaluate the percentage of patients meeting a set of criteria with respect to the number of micturitions, incontinence, nocturnal voids and nocturia episodes, comparing the incidence estimates between treatment groups at each visit and endpoint. For the safety analyses, Cochran–Mantel–Haenszel tests were used to compare incidence estimates of AEs between treatment groups.
Results
Patient characteristics
Six hundred and thirty-four patients were randomized and received at least one dose of study medication (n = 318 solifenacin; n = 316 placebo). The safety population comprised all randomized patients. The primary efficacy analysis population included all randomized patients who received at least one dose of double-blind study medication and had diary data for the screening phase and at least one on-treatment period.
There were no notable differences between the treatment groups in terms of baseline patient characteristics (Table 1). The majority of the study population was female (82%) and predominantly Caucasian (90%), with a mean age of 60 years; less than 40% of the study population was ≥65 years old. Overall, the mean ± SD duration of OAB symptoms was 10 ± 10.5 years. Fifty-one percent of patients had a history of urgency-only incontinence.
Baseline patient characteristics.
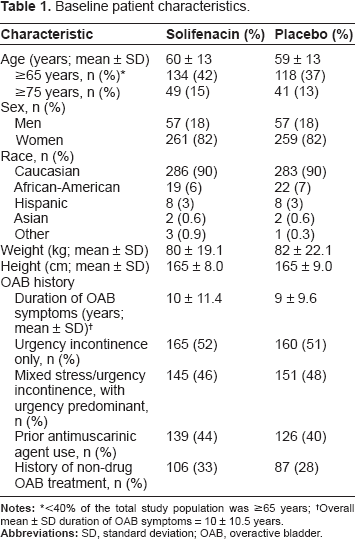
< 40% of the total study population was ≥65 years;
Overall mean ± SD duration of OAB symptoms = 10 ± 10.5 years.
The proportion of patients completing the study was similar for the two treatments (85% for solifenacin vs. 86% for placebo), and the most common primary reason for study discontinuation was an AE (28/318 [9%] and 15/316 [5%] for solifenacin and placebo, respectively; Fig. 1).
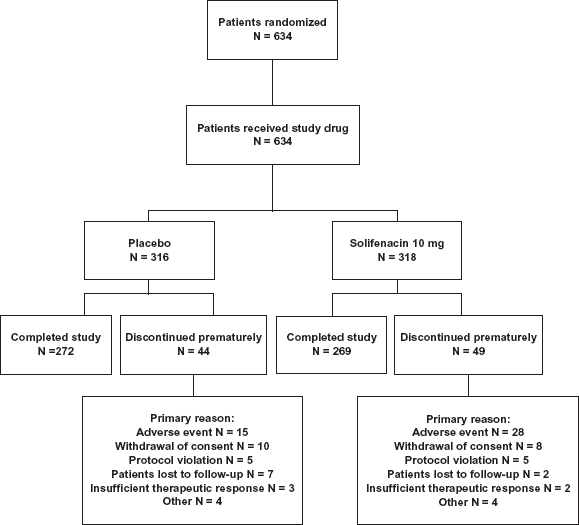
Patient disposition throughout the study.
Efficacy
Solifenacin was significantly more effective than placebo in reducing the mean ± standard error (SE) number of micturitions per 24 hours from baseline to endpoint (solifenacin –2.4 ± 0.15 vs. placebo –1.3 ± 0.16,
Change from baseline to endpoint * in efficacy variables.

Endpoint defined as last available on-treatment visit on or before Visit 5 (Week 12);
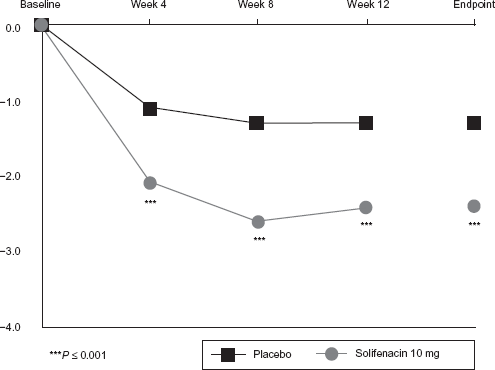
Mean change from baseline in number of micturitions per 24 hours.
At endpoint and at all study visits, solifenacin was also significantly superior to placebo in reducing the mean (± SE) number of urgency episodes per 24 hours (solifenacin 10 mg at endpoint: –3.3 ± 0.23; placebo at endpoint: –1.8 ± 0.22;
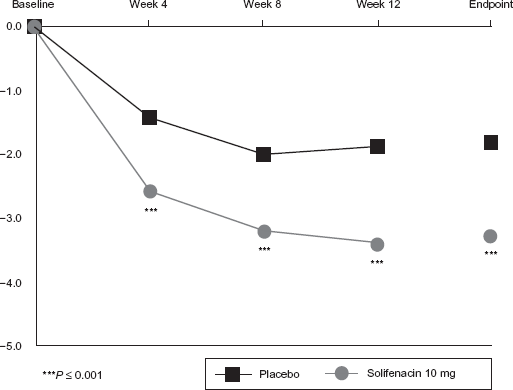
Mean change from baseline in urgency episodes per 24 hours.
Solifenacin was significantly more effective than placebo in reducing the mean number of incontinence episodes per 24 hours from Week 4 onwards (change from baseline to endpoint: -2.0 ± 0.15 vs. –1.2 ± 0.15;
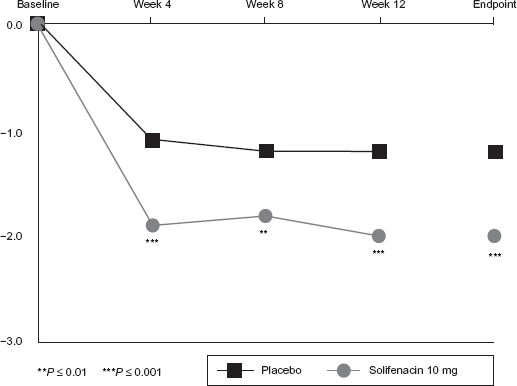
Mean change from baseline in number of incontinence episodes per 24 hours.
Among the patients who had at least one nocturia episode during the baseline period, 49 patients receiving solifenacin (18%) and 30 patients receiving placebo (11%) had no nocturia episodes at endpoint (
Safety
More than half of the patients in both treatment groups experienced treatment-emergent AEs (79% for solifenacin and 63% for placebo). The majority of these were mild-to-moderate in severity and the discontinuation rate due to AEs was low in both groups (28/318 [9%] and 15/316 [5%] for solifenacin and placebo, respectively).
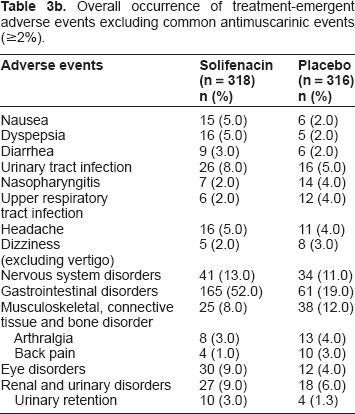
The occurrence of common antimuscarinic AEs is shown in Table 3a, and the occurrence rates of other AEs are shown in Table 3b. The most common treatment-emergent AEs were those associated with the anticholinergic nature of solifenacin, namely dry mouth and constipation, the majority being mild or moderate in severity. Mild-to-moderate and severe dry mouth occurred in 36 and 3% of solifenacin-treated patients and 6 and 0% of placebo recipients, respectively. Similarly, mild-to-moderate and severe constipation occurred in 17 and 2% of solifenacin-treated patients and 4 and 0% of placebo recipients, respectively. Urinary retention was reported as a treatment-emergent AE for 3% of patients in the solifenacin arm and 1.3% in the placebo arm. Six (1.9%) solifenacin recipients discontinued due to dry mouth, versus 0% of placebo recipients; five (1.6%) solifenacin recipients discontinued due to constipation, versus one (0.3%) patient receiving placebo. Discontinuations due to any other AEs occurred in less than 1% of patients per AE.
Most common treatment-emergent antimuscarinic adverse events.

Overall occurrence of treatment-emergent adverse events excluding common antimuscarinic events (≥2%).
Two patients in the placebo group and 10 patients in the solifenacin group experienced a serious AE (SAE) during the treatment and follow-up period. Of the SAEs reported for solifenacin, three were considered by the investigator to be related to solifenacin. Two of these SAEs (hyponatremia secondary to polydypsia and hypotension secondary to cardiac disease) resolved following treatment discontinuation. A third patient with fecal impaction was treated with an enema and NuLYTELY and recovered with no change to the solifenacin dose.
There were no clinically relevant changes in vital signs, physical examination findings, hematology, urinalysis or chemistry. Similarly, there were no clinically relevant changes in post-void residual volume (five patients [1.6%] in the placebo group and 10 patients [3.1%] in the solifenacin group experienced shifts in post-void residual volume from ≤150 mL at baseline to >150 mL at Week 12). There was a small increase in QTc interval (3.3 msec relative to placebo) that was statistically significant, but not clinically meaningful.
Discussion
Solifenacin gained regulatory approval for the treatment of OAB on the basis of the significant efficacy and safety demonstrated in four Phase III pivotal trials of more than 3000 patients with OAB. 13 The results presented here are the outcomes of one of those pivotal studies which demonstrate that solifenacin 10 mg is significantly superior to placebo in reducing the symptoms that define OAB: urgency, with or without incontinence, usually with frequency and nocturia. 3
Of the OAB symptoms, urinary incontinence represents a particularly embarrassing and bothersome problem to patients; 6 thus, the achievement/restoration of continence is an important outcome in clinical trials of OAB treatments. Here we demonstrated that solifenacin was superior to placebo in reducing the number of incontinence episodes. Moreover, treatment with solifenacin allowed over 50% of the patients who were initially incontinent at baseline to achieve continence; less than 30% of placebo-treated patients achieved the same goal. These results are consistent with the outcomes of three additional pivotal Phase III studies, which have demonstrated that solifenacin reduces the incidence of incontinence episodes at doses of 5 and 10 mg over 12 weeks of treatment.14–16
Our study also showed that solifenacin was significantly superior to placebo in reducing urgency, which is considered to be the defining symptom of OAB. 3 These findings are again consistent with those observed in previously reported clinical studies. 17 In addition, micturition frequency was normalized (fewer than eight per day) in 40% of patients receiving solifenacin 10 mg in this study.
The beneficial effects of solifenacin on incontinence, frequency, urgency and volume voided in this current study were obtained as early as Week 4 of treatment and maintained throughout the remainder of the 12-week study duration. These results indicate that solifenacin provides a rapid onset of symptom improvement that is sustained. An extension study has also subsequently been conducted to determine whether the improvements in symptoms are maintained over a longer treatment period than 12 weeks. Haab et al 17 have demonstrated the long-term (1-year) efficacy and safety of solifenacin as part of an open-label follow-up of the two European Phase III clinical trials.14,15
Although expected anticholinergic AEs, including dry mouth, constipation and blurred vision, were seen with solifenacin in this study, these were generally mild-to-moderate in severity. Furthermore, the overall completion rates were 85 and 86% for solifenacin 10 mg and placebo, respectively, suggesting that the solifenacin-associated AEs were not of a sufficiently severe nature to result in frequent elective discontinuation of therapy. The rate of AEs and completion rate of 85% with solifenacin 10 mg in this study is consistent with those in a second Phase III trial of solifenacin 10 mg. 16
It should be noted that although subjects in the current study were randomized to receive solifenacin 10 mg or placebo, the recommended dose of solifenacin is 5 mg, which can be increased to 10 mg if the 5 mg dose is well tolerated. Thus, treatment with solifenacin enables flexible dosing to provide an appropriate balance between efficacy and safety. The two additional Phase III trials of solifenacin used a flexible dosing regimen of 5–10 mg/day, which was associated with somewhat lower AE rates and higher completion rates (92.2 14 and 92.9%) 15 suggesting that the option of a lower solifenacin dose may have improved patient retention in these studies.
Evidence of the benefits of flexible dosing are also provided in a randomized, double-blind trial that compared solifenacin 5–10 mg/day versus 4 mg/day tolterodine extended release. 18 Over the duration of the study, low rates of anticholinergic AEs were reported for both treatments with comparable rates between the treatments. However, patients treated with solifenacin achieved significantly greater improvements in urgency, incontinence and mean volume voided compared with tolterodine-treated patients. 18 Solifenacin was also non-inferior to tolterodine with respect to reducing frequency of micturitions, the primary efficacy variable. 18 The demonstrated benefits of solifenacin over tolterodine may reflect the fact that 48% of patients in solifenacin arm requested and received a dose increase from 5 to 10 mg/day while the 51% of patients in the tolterodine arm who requested a dose increase received 4 mg/day tolterodine plus placebo, reflecting the maximum recommended doses of these two agents. 18
Conclusions
The results of this study confirm the safety of solifenacin 10 mg OD and the efficacy in treating the key OAB symptoms of urinary frequency, incontinence and urgency episodes per 24 hours.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgments
The authors would like to thank the following investigators and statisticians for their help:
This study was funded and sponsored by Astellas Pharma Inc., Tokyo, Japan, including editorial support and statistical analysis. Editorial assistance was provided by Medicus International.
