Abstract
Purpose
To evaluate the feasibility of transrectal computed ultrasound tomography (TRCUT) in localization and targeted biopsy for men who have been shown to be negative in the first biopsy guided by two-dimensional transrectal ultrasound (2D TRUS) but continue to have figures suggestive of prostate cancer, so as to minimize with confidence the number of core needle biopsies, especially when the repeat or random required.
Materials and Methods
Sixty-three patients aged 53–78 years (mean, 64.9 years) with suspected prostate cancer were enrolled when a repeat or random biopsy was indicated after at least one prostatic biopsy with negative findings. The 2D TRUS random (sextant) and TRCUT guided shot-biopsies were obtained. Imaging-biopsy consequences were finally compared with the post-operative findings and the histopathologic staging.
Results
TRCUT enabled display of the prostate gland in planes usually not obtainable at conventional 2D TRUS. Transverse and oblique thin cuts of TRCUT were more significant in all patients than other planes. Diagnostic performance of TRCUT imaging on prostate cancer localization for targeted biopsy proved an accuracy of 97.4% (P value = 0.001). TRCUT for bilateral disease detection proved an accuracy of 78.6% compared to the final post-operative pathology. Accuracy for identification of positive tumor margins proved 82.4%. Gleason score on final post-operative pathology was upgraded to 7, compared with a median score of 6 on imaging biopsies.
Conclusion
TRCUT guided target-biopsy of the prostate gland represents a technique with a higher rate of accuracy and can minimize the number of biopsy shots than using 2D TRUS in men who have high PSA levels and negative in the first biopsy procedure.
Introduction
Imaging techniques, especially two-dimensional transrectal ultrasound (2D TRUS), introduced by Watanabe and associates 1 in 1971, is the prevalent leading imaging modality for the evaluation of prostatic disease. However, when evaluating a patient for prostate cancer (PCa) by 2D TRUS, the anatomical reference points are restricted by the limited viewing planes guiding the needle to target sites. 2 Besides; any motion of the physician's hand or the patient during the procedure may diverge targeting of focal lesions. 3 Many authors, as well, consider the poor-edge definition of 2D TRUS, is the main for why some focal lesions still missed.4,5 What is more, some prostate cancers (PCa) are invisible on 2D TRUS, 6 and these are as important as those visible. These restrictions make it difficult to establish a consistent reference frame by 2D TRUS, which increase number of false-positive or false-negative results. Nevertheless, the use of color and power Doppler techniques, use of microbubble contrast agents and elastography have shown promise in improving the ability to better detect and/or localize prostate cancer.6–10
Developments in magnetic resonance imaging (MRI) and magnetic resonance spectroscopy (MRS) have as well improved understanding of tumor biology and particularly enabled the detection and characterization of PCa and lymph node metastasis.11–17 So far, in some studies, patients who underwent radical prostatectomy, the accuracy of combined endorectal MRI/MRS for sextant localization of prostate cancer was equal to that of MRI alone.18,19 Together, this may be somewhere impractical considering the technical limitations, cost and the availability of these machines.
Cancer tissue can exist anywhere in the prostate and accurate diagnosis of PCa is of importance; hence, improving ultrasound real time display, as an affordable, easy and minimally invasive test, is essential. Reports of the use of endocavitary Three-Dimensional Ultrasound (3D US), were produced, showing improved and promising results in minimizing to some extent the invasiveness of random biopsies.20–24
Transrectal Three-Dimensional extended Imaging (3D XI) has been recently introduced into the diagnostic imaging field. It is an upgrade of 3D US, based upon the production of Computed Ultrasound Tomography (CUT) of the 3D volume with optical multiresolution sequence instead of grey scale shade. This modality is designed to provide thin (1.0–1.5 mm) contiguous images enlightening the entire prostatic gland in coronal, sagittal, transverse and oblique planes, allowing for extended readability and easy exploration of subtle lesions.
Hence, the purpose of our study was to evaluate the feasibility of transrectal computed ultrasound tomography (TRCUT) in precise localization-guidance for targeted biopsy in patients with high PSA levels and prior negative in the first 2D TRUS guided-biopsy.
Materials and Methods
Patient characteristics
This study was institutional review board approved. Between January 2007 and April 2009; 63 patients (age range, 53–78 years; mean, 64.9 years) were referred to our University Hospital for repeat biopsy. Each patient provided a written informed consent. Patients’ information from their medical records—received from their doctors—was used. The inclusion criteria were dependant mainly upon high serum PSA levels and negative first 2D TRUS guided-biopsy results. Patients enrolled had a mean PSA (11.6 ng/mL); range, 10.1–39.3 ng/mL. Suspicious abnormality on DRE was not essential, because DRE, which helps detect stiffness, is limited anatomically to the posterior gland and cannot help detect lesions confined anteriorly or in the transition zone, where as many as 28% of cancers occur. 25 Patients having a positive bone scintigram, or previously undergone androgen therapy or radiation therapy have been excluded from our entry.
Procedures
Imaging was performed using (MEDISON: SONO-ACE–X8) ultrasound machine provided with 3D XI optional software and 5–8 MHz endocavitary probe competent for conventional and volume ultrasound. The machine is networked to a workstation (Sonoview Pro: version 5.05.01). All patients received appropriate antibiotic coverage and had bowel preparation.
The prostate gland was divided into sextants: the apex, middle, and base of the gland, according to Wefer and associates. 26 Two biopsy protocols were performed using the 18-gauge needles at the same setting. In conventional 2D TRUS guided biopsy procedure, biopsy was obtained from each zone in a systematic but undirected fashion. After the 2D TRUS sextant biopsies, 3D US volume was acquired for every patient and the volume imaging data were displayed in thin (1.0–1.5 mm) coronal, transverse, sagittal, and oblique planes, exploring the entire prostate gland. Selecting the best plane of images demonstrating and localizing each focal lesion was signified. With the oblique semi-sagittal plane, because it enables the best view for biopsy, guided shot biopsy was taken under live TRCUT. All specimens were signified according to their site.
All patients had retropubic, nerve sparing (when appropriate) radical prostatectomy. The surgical specimens were processed in a standard manner. Imaging and post-operative data were scored prospectively. Evaluation focused for all patients on a) number of positive cores, b) laterality, c) positive margins and d) Gleason score.
Statistical analysis
Having in consideration our selection bias, statistical data were calculated manually to determine whether there is a significant difference between the 2D TRUS sextant and the TRCUT guided targeted biopsies in suspicious patients. The reference standard in the present study is the post-operative specimens. P value of less than 0.05 indicated a statistically significant difference.
Results
Upon taking the selection bias into account (patients were not initially sent to our department), we can affirm that the study was based upon selected cases that proved negative in the first biopsy while PSA levels were high (>10 ng/mL) and all patients were referred for the second biopsy.
In the present study, from a total of 378 random biopsies (sextant for each), 67 specimens (17.7%) proved positive for cancer. On the other hand 79 targeted-core biopsies has been collected by means of live TRCUT guidance from the same 63 patients, 74 specimens (93.7%) were positive for cancer. None of the patients experienced significant biopsy related complications; specifically, no cases of urine retention or urinary tract infection occurred.
TRCUT enabled display of the prostate gland in planes usually not obtainable at conventional 2D TRUS. The entire gland was seen in all patients (100%) on coronal, sagittal, transverse and the oblique set of images, allowed the reduction of time to target lesions. However, the oblique and transverse planes were judged superior in demonstration of PCa than the sagittal and coronal images (66.2%) in the studied patients.
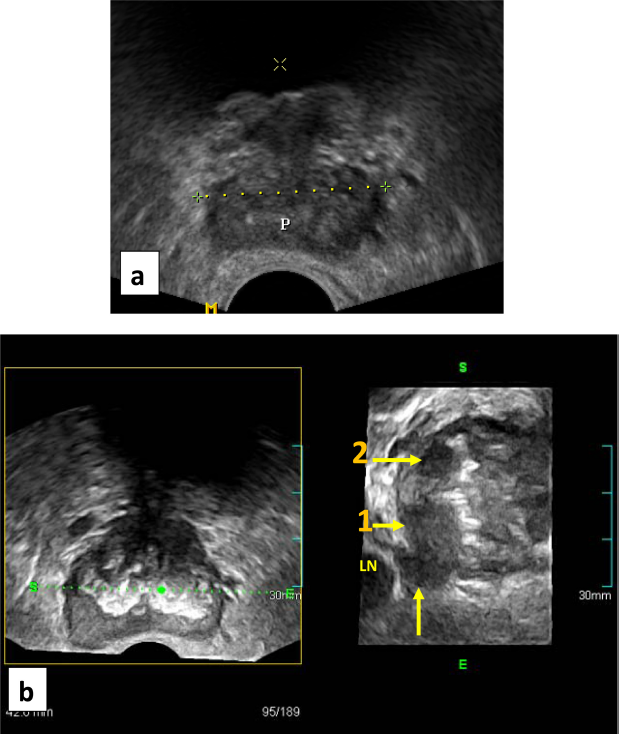
Of the 63 patients, 52 had unilateral disease and 11 had bilateral disease on TRCUT biopsy. Of the 52 patients with unilateral disease, 3 instances proved bilateral disease on final post-operative descriptions. Tumor behavior in terms of thinned out prostate capsule, jagged tumor margin and extending limb are the major observed criteria, proved compatible with tumor margin positivity in our series (Fig. 1). Of 63 patients, 9 (11.7%) had extracapsular extension, exact to the post-operative consequences, which improved our results in the evaluation of tumor behavior (Table 1).
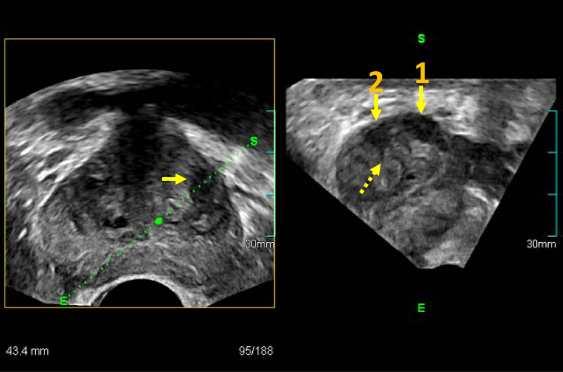
TRCUT oblique cut (right) along the focal lesion in the axial image (left) expressing two lesions; the larger is at the peripheral zone (long arrow) and the other one at the central gland (short arrow), with extended limb (dashed arrow), correlating with positive tumor margin.
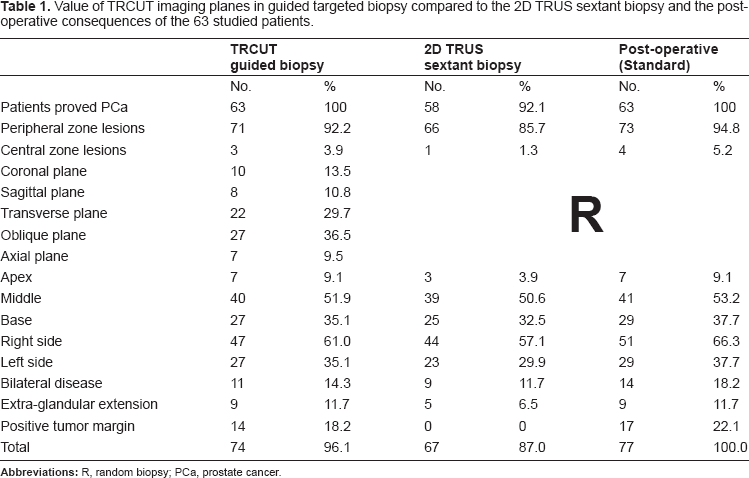
Value of TRCUT imaging planes in guided targeted biopsy compared to the 2D TRUS sextant biopsy and the post-operative consequences of the 63 studied patients.
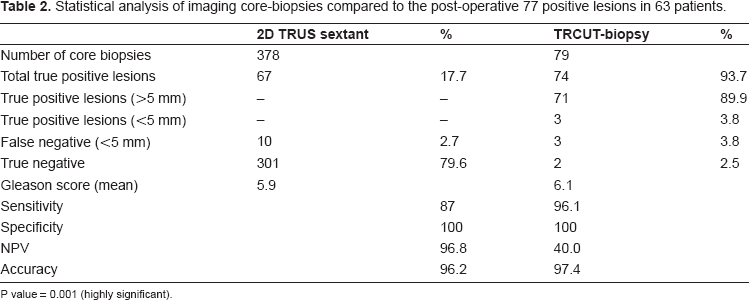
Successful targeting of focal lesions by TRCUT showed an accuracy of 97.4% and a sensitivity of 96.1% compared to the post-operative consequences (P value = 0.001). On the other hand, 2D TRUS sextant biopsies achieved a sensitivity of 87% (Table 2).
Statistical analysis of imaging core-biopsies compared to the post-operative 77 positive lesions in 63 patients.
P value = 0.001 (highly significant).
Despite all patients having proved cancers in their prostates by means of TRCUT, the missed focal lesions were of less than 5 mm diameter and all were observed in patients with bilateral tumors. On the other hand 5 patients out of the 63 showed negative for cancer with the sextant protocol.
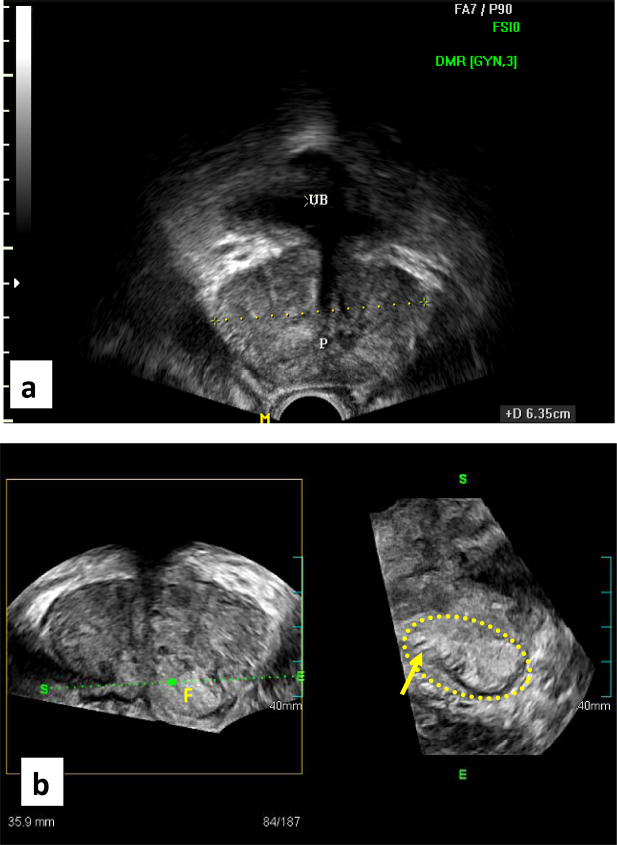
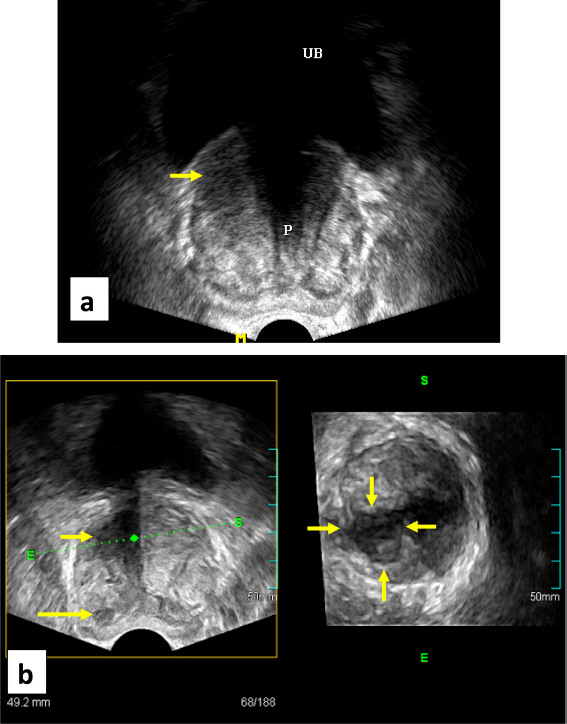
However, panoramic demonstration of PCa at the prostatic apex (Fig. 2) and in case of dense central gland calcifications (Fig. 3), have likely improved our results, mostly due to the free off-site planes. On the other hand, revealing small tumors around 5 mm by TRCUT was established in six instances that proved false negative in three (Fig. 4). Median Gleason score on final pathology was upgraded to 7, compared with a median score of 6 on imaging guided biopsy results.
Discussion
When a repeat biopsy is required for a man who has been shown to be negative in the first biopsy but continues to have figures suggestive of cancer, cancer is reportedly detected by repeat biopsy at a rate of about 20%.27,28 Then again, when the patient is a candidate for radical prostatectomy, diagnostic accuracy should be increased, and systematic biopsy up to saturation is recommended.26–30 The thought is that, prostate biopsy specimens collected in such a manner contain the posterior lateral side of the periphery, in which early prostate cancer is most frequently located. Other studies have shown that employing more biopsy cores may minimize misinterpretation in case of a lateral small volume of cancer that was not adequately sampled or missed by the needle.29,30 However, almost 32% of prostate cancers are missed when comparing results of random biopsies with prostatectomy specimens. 6 Therefore, no definitive criteria for repeat or systemic biopsy have been established despite the extensive literature on many aspects of PCa. In the interim, little has been written on minimizing the number of core needle biopsies.31,32
The analysis of our study results were performed by taking into account the selection bias. This bias is the result of the way patients were recruited into the present study, as mentioned earlier. Therefore, this study was designed to evaluate the impact of TRCUT on the diagnostic process of PCa, standardized by the prostatectomy specimens (n = 77 positive specimens) and including a few (n = 6) tumors around 5 mm each, from which the false negative specimens accounted. This could have resulted from inaccurate sampling of cancer tissue from those patients, because not all small lesions proved false results. Careful handling, as well, is of importance to consider during the histopathological assessment.
Nevertheless, our results demonstrate that, with TRCUT exploration of the entire gland, the sensitivity for PCa detection could be increased up to 96.1% compared to 87% with random biopsies, which are major improvements over the levels previously reported with conventional 2D TRUS.25–28 This is mostly due to the advantage of the high resolution real-time multi-planer display of thin (1.0–1.5 mm) contiguous sections offered by TRCUT imaging, facilitating accurate lesion targeting. In addition, the needle gauge used has played a significant role in the quantity of cancer within biopsy cores. All of which can be considered as the factors behind our score of diagnostic accuracy and the means to reduce the number of biopsy shots. Even with the presence of calcifications within the central gland, the ability to detect PCa is not mired, likely due to the flexibility and the unrestricted utilization of the 3D volume.
Taking into account the data from the literature and our findings, we suggest that TRCUT is a non-sophisticated imaging procedure, proficient in tumor mapping and can be an appropriate tool for confident targeted biopsy of PCa. Moreover, in TRCUT imaging, the volume data are standardized and can be stored, thus decreasing operator subjectivity, increasing certainty of findings and allows for later detection of lesions that may have been missed at the initial evaluation by less experienced radiologists.
Despite the bias of the way patients were recruited into the study, as mentioned earlier, our results demonstrate several novel findings. First, better expression of PCa can be achieved by using previously unattainable planes. Second, our procedure can significantly minimize the number of biopsy shots, and third, the tumor behavior can be estimated.
We can also postulate that the procedure in general can be affordable in the management protocol for prostatic cancer, especially when PSA elevations are in the “gray zone”.33,34
In terms of limitations, care should be taken when manipulating volume data because inappropriate settings may alter the diagnosis. We advise previous training before proceeding into interpretation of TRCUT image sequences. Nevertheless, this advanced ultrasound method can open a new promising prospect for further research to minimize the rate of the false negative result and to estimate how many patients would be avoided from the repeat or saturation biopsies.
Conclusions
TRCUT has the potential to become a valuable clinical tool, allowing prostatic gland exploration in previously unattainable planes, supporting better tumor detection and mapping for precise targeted biopsy, which can minimize the number of biopsy shots in men who have high PSA levels and are negative in the first biopsy procedure.
However, there are still a very few invisible cancers. Therefore, the policy of saturation biopsy for a minority of patients would be continued until this incidence can be eliminated.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest.
