Abstract
Urothelial carcinoma of the bladder represents a significant health burden globally. Patients who present with or develop metastatic disease experience poor overall survival even with salvage chemotherapy. Metastasectomy has shown promise in prolonging survival and even cure in patients with pulmonary and lymph node metastasis. We describe the second patient in the available literature who underwent liver resection for a solitary liver metastasis after cystectomy with disease-free survival at 64 months and present a review of the existing literature describing metastasectomy.
Introduction
Urothelial carcinoma of the bladder is the fourth most common cancer among men and the ninth most common cancer among women in the USA. 1 Seventy-five percent of all urothelial carcinoma is nonmuscle invasive at the time of diagnosis. The remaining 25% of patients will have muscle invasive disease, and of these, around 30% will have metastatic disease or local recurrence after cystectomy.2,3 Following recurrence, chemotherapy can achieve an overall survival with cisplatinbased chemotherapy between 8 and 12 months. 4 In renal cell carcinoma (RCC) and nonseminomatous germ cell tumor of the testicle (NSGCT), metastasectomy, including at multiple sites and for metachronous lesions, has been shown to improve survival compared to systemic therapy alone. 5 Given the overall poor prognosis of metastatic urothelial carcinoma and success of multimodal therapy involving metastasectomy in other solid malignancies, it stands to reason that patients with metastatic urothelial carcinoma might benefit from metastasectomy. We describe a patient who underwent liver resection for metastatic urothelial carcinoma after receiving adjuvant chemotherapy and remains alive and disease free over 8 years from initial cystectomy.
Case Presentation
The patient is a 73-year-old man with a history of sarcoidosis, hyperlipidemia, gastroesophageal reflux disease, sleep apnea, and obesity (body mass index 35) who initially presented with gross hematuria in February 2006. He had previously smoked tobacco; however, he quit in his early twenties and had been exposed to pesticides and other farming chemicals. Cystoscopy was performed that demonstrated a bladder lesion. Biopsy showed high-grade urothelial carcinoma invading into the muscularis propria. A computed tomography (CT) scan did not demonstrate metastatic disease but was concerning for lymphadenopathy. He then underwent cystectomy, lymph node dissection, and orthotopic neobladder formation in March 2006. Final pathologic evaluation revealed pT2bNlMx disease (Fig. 1) with one of the 18 lymph nodes positive (right pelvic lymph node).
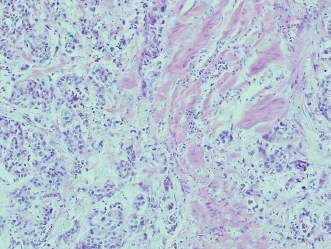
Muscle invasive nests of high-grade urothelial carcinoma in the cystectomy specimen.
The patient then completed five cycles of gemcitabine/cisplatin chemotherapy in June 2006. He was followed with clinic visits which included a history, physical, and serum chemistry every 3 months and CT and X-ray imaging of the chest at 6-month intervals. In October 2008 (31 months after cystectomy), a 1-cm liver lesion was identified on CT scan (Fig. 2). On a CT scan in April 2009 (37 months from cystectomy), the lesion had grown to 1.2 cm (Fig. 3). Fine-needle aspiration of the liver mass confirmed the diagnosis of metastatic urothelial carcinoma. He had no evidence of other metastatic site after positron emission tomography and CT, and thus underwent right partial hepatectomy in June 2009 for a 1.7-cm solitary lesion with surgical margins negative for disease (Fig. 4). Since this time, he has been followed for evidence of disease recurrence with CT scan and cytology. As of October 2014, there was no evidence of the disease, which represents a time period of 64 months from hepatic metastasectomy and 103 months from cystectomy.
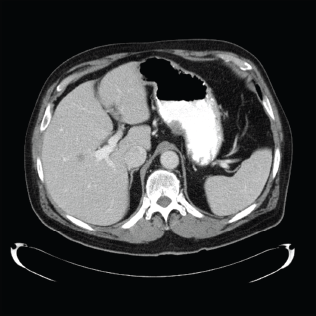
Hypodense, 1 cm lesion in the central right liver.
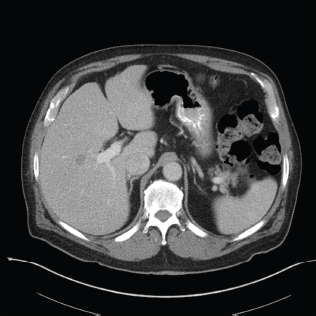
Hypodense, 1.2 cm lesion in the central right liver. Biopsy revealed this to be metastatic urothelial carcinoma.
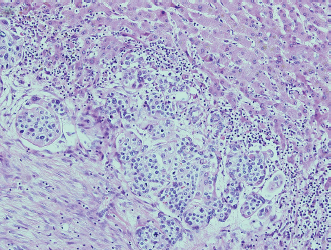
Metastatic urothelial carcinoma in the liver.
Methods for Literature Review
We performed a literature search using the PubMed and Embase databases using a combination of the search terms: bladder cancer, metastasectomy, urothelial carcinoma, pulmonary, hepatic, transitional cell carcinoma, and extended lymph node dissection. We identified articles published in English with the focus on large case series. Fourteen articles were identified and eight were selected for review. Two articles were not in English, one was not about bladder cancer, and one presented only two cases. The citations of all the articles were searched and additional articles meeting the inclusion criteria were reviewed and included as appropriate.
Discussion
Metastasectomy is common in malignancies such as colon cancer, NSGCT, and RCC and allows for improved overall and disease-free survival. 6 There is a growing amount of support in the literature for metastasectomy in urothelial carcinoma. A recent series published by Matsuguma et al reviewed 32 cases of pulmonary mastectomy for urothelial carcinoma. 7 Using the inclusion criteria of completely resectable disease, no areas of other detectable disease, pulmonary function compatible with resection, and absence of other effective therapies, they found that 9 of 32 patients (28%) were alive and without recurrence at 5 years. Significant variables in this study were tumor size <3 cm and complete resection.
The MD Anderson experience demonstrated a response rate to methotrexate, vinblastine, doxorubicin, and cisplatin (MVAC) for metastatic urothelial carcinoma of 60%–70% with median survival of ~13 months. 8 In these patients, they note that most have evidence of disease recurrence at prior sites responsive to chemotherapy. Therefore, 31 patients from 1985 to 2001 underwent metastasectomy at sites such as lung, lymph nodes, brain, and subcutaneous tissues. Twenty-two received some form of chemotherapy prior to surgery and 13 underwent more than one surgery. Thirty-three percent of the patients were alive at 5 years.
Abe et al 9 reviewed the Japanese experience of metastasectomy in 42 patients at four medical centers. Most of these patients had lymph node or lung resection; however, one patient underwent liver resection. In this series, 41/42 patients received either neoadjuvant (34 patients) or adjuvant therapy (7 patients) with MVAC, gemcitabine/cisplatin, or other salvage regimens. Inclusion criteria included single organ site with limited metastatic burden, good performance status, and a favorable response to chemotherapy. Following resection, patients were followed for recurrence at 3- to 6-month intervals. Median disease-free interval from surgical resection of the primary site to detection of metastatic disease was 16 months. Median overall survival was 26 months following metastasectomy from all sites, but on univariate analysis, patients with solitary lung or lymph node metastasis had median survival of 81 months versus 19 months for all other groups.
The German experience was reviewed by Lehmann et al 10 in 44 patients at 15 medical centers. They noted a 30% recurrence after cystectomy with 15 months median time to recurrence. Five-year overall survival in the metastasectomy cohort was 28%. They concluded that metastasectomy should only be performed in patients with single site, resectable lesions, who have received chemotherapy, de Vries et al and Sweeney et al studied chemotherapy and supraregional tumor-positive lymph node dissection.11,12 They demonstrated 24% 5-year and 27% 4-year survival, respectively. In 114 patients with metastasis after radical cystectomy from 1990 to 2010, the median survival was 11.2 months. Of the 13 patients who underwent metastasectomy, there was a 31.3-month versus 9.7-month overall survival difference. 13
In contrast to the above studies, Otto et al 14 performed metastasectomy in 70 patients who failed MVAC and found benefit only in the cohort who underwent resection for symptom relief and that asymptomatic patients had a worse sense of well-being postoperatively. Abe et al 15 performed multivariate analysis in 48 patients with metastatic disease, 12 of whom underwent metastasectomy. They showed that five or more chemotherapy cycles and metastasectomy as well as no bone, liver, or local recurrence predicted 42-month versus 10-month median survival with and without metastasectomy, respectively. Notably, however, no patients underwent liver metastasectomy as all with liver lesions had more than one site of metastasis and were ineligible for metastasectomy.
Conclusion
Multimodal therapy consisting of systemic therapy followed by surgical consolidation has been shown to improve survival in many solid malignancies. There is a growing body of literature to support metastasectomy in urothelial carcinoma after cystectomy and systemic therapy. Current data are retrospective in nature and reflect small cohorts treated with varied therapies but support metastasectomy especially in pulmonary and lymph node metastasis. Many studies have consistently shown that 20%–30% of the patients who undergo metastasectomy have significantly improved the duration of overall and disease-free survival when compared to treatment with chemotherapy alone. We present a patient with no evidence of disease recurrence 64 months after hepatic metastasectomy. This is only the second case in the available literature of hepatic metastasectomy. Given the success of liver resection in RCC, colorectal cancer, and NSGCT in prolonging survival and cure, consideration should be given to liver resection in properly selected patients with metastatic urothelial carcinoma of the bladder as multimodal therapy plays a larger role in the treatment of metastatic urothelial carcinoma. These patients should be carefully selected as a prolonged time to progression and recurrence, such as in our patient, as well as single metastatic site and slow tumor growth are likely to be important.
Author Contributions
Conceived the concepts: NAB, JAB. Analyzed the data: NAB. Wrote the first draft of the manuscript: NAB. Contributed to the writing of the manuscript: NAB, JAB, LD. Jointly developed the structure and arguments for the paper: NAB, JAB. Agreed with manuscript results and conclusions: NAB, JAB, LD. Made critical revisions and approved the final version: NAB, JAB, LD. All the authors reviewed and approved the final manuscript.
