Abstract
Chronic kidney disease (CKD) introduces a unique set of nutritional challenges for the growing and developing child. This article addresses initial evaluation and ongoing assessment of a child with CKD. It aims to provide an overview of nutritional challenges unique to a pediatric patient with CKD and practical management guidelines. Caloric assessment in children with CKD is critical as many factors contribute to poor caloric intake. Tube feeding is a practical option to provide the required calories and fluid in children who have difficulty with adequate oral intake. Protein intake should not be limited and should be further adjusted for protein loss with dialysis. Supplementation or restriction of sodium is patient specific. Urine output, fluid status, and modality of dialysis are factors that influence sodium balance. Hyperkalemia poses a significant cardiac risk, and potassium is closely monitored. In addition to a low potassium diet, potassium binders may be prescribed to reduce potassium load from oral intake. Phosphorus and calcium play a significant role in cardiovascular and bone health. Phosphorus binders have helped children and families manage phosphorus levels in conjunction with a phosphorus-restricted diet. Nutritional management of children with CKD is a challenge that requires continuous reassessment and readjustment as the child ages, CKD progresses, and urine output decreases.
The “Renal” Diet
In children with chronic kidney disease (CKD), there is no
Initial Evaluation
The initial evaluation of a child with renal disease includes height, weight, head circumference (up to 36 months of age), and body mass index. 1 These parameters should be plotted on the appropriate percentile charts. In children, serial measurements are required to properly assess growth. When plotting values, age should be adjusted for prematurity. In the United States, Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines recommend the growth charts of World Health Organization to monitor children at the age of 0 and 2 years and the growth charts of Centers for Disease Control and Prevention to monitor children at the age of 2 years and older. Disease-specific growth charts (eg, Trisomy 21, Williams syndrome) can also be used when appropriate.
Calories, Formula, and Supplements
Per KDOQI 2008 guidelines, the energy needs of a child with renal disease are expected to be the same as those of a healthy child of the same age. 2 Estimated needs can be adjusted depending on growth and weight gain trends. Nausea, vomiting, gastroesophageal reflux, oral aversion, delayed gastric emptying, renal tubular acidosis, elevation of cytokine levels, and changes in leptin and ghrelin hormones all affect appetite and contribute to inadequate caloric intake.
In infants, breast milk is the feeding of choice. If breast milk is not available, standard infant formula may be appropriate. When a low potassium and/or low phosphorus intake is required, Similac PM 60/40 can be offered. The choice of formula takes into account the need for a low renal solute load. Formulas such as Similac PM 60/40® (Abbott Laboratories) contain lower potassium and phosphorus than standard infant formulas. Breast milk or formula may be supplemented with glucose polymers, fats, and carbohydrates to increase caloric density. Concentrating the formulas by increasing the ratio of formula powder to water is not recommended as it also increases electrolytes, which may be dangerous in the face of a sodium-, potassium-, and phosphorus-restricted diet.
For older children, commercially prepared formula nutritional supplements can be considered. There are currently no commercially available pediatric renal formulas for children older than one year. Potential options for supplementation that are currently used include pediatric nonrenal supplements such as Nestle Nutrition's Compleat Pediatric®, Nutren Junior®, Boost Kid Essentials®, and Abbott Nutrition's PediaSure®, and adult renal formulas such as Abbott Nutrition's Nepro® and Suplena®, Nestle Nutrition's Nova-Source Renal®, and Renalcal®. Serum electrolytes should be closely monitored when any supplementation is used. 3
Tube Feeding
Children born with CKD may develop oral aversion and may not be able to take in sufficient calories for growth. Tube feeding should be considered in these children as nutrition is extremely important for physical and neurodevelopmental growth. Some children will start with nasogastric (NG) feeding to supplement oral intake. However, dependence on NG feeds to achieve adequate fluid, caloric, and protein intake should prompt the medical team to consider gastrostomy tube (G-tube) placement.4,5 Gastrostomy placement approaches include percutaneous endoscopic gastrostomy versus laparoscopic versus open gastrostomy with or without Nissen fundoplication. Some patients may have difficulty in tolerating gastric feeding volumes and may require gastrojejunostomy or jejunostomy tube placement. Special consideration is given to patients requiring peritoneal dialysis (PD) and G-tube placement. Complications including tube blockage, leakage around exit site, exit site infection, gastrointestinal bleed, peritonitis, the need for PD catheter replacement secondary to infection, and the need for G-tube replacement have been reported.6,7 It is recommended that tube placement occurs before or concomitant with tunneled PD catheter placement to minimize the risk of peritonitis. If G-tube placement occurs after PD catheter placement, avoid using the PD catheter for two to three days and administer prophylactic antibiotics and antifungals. 8
Depending on the appetite and oral feeding skills of the child, G-tube feeds can be used to supplement oral intake or they may provide the soul source of nutrition. Tube feeds can either be delivered by bolus or as continuous feeds. Higher volumes required with bolus feeds may be poorly tolerated, resulting in retching and vomiting. Potential aspiration risk with gastroesophageal reflux is a concern with continuous overnight feeds in infants in supine positions. Continuous nighttime feeds for older children may be beneficial to promote daytime hunger and encourage oral intake.
Protein
The current KDOQI guidelines recommend providing 100%–140% of dietary reference intake (DRI) of protein for ideal body weight for children with CKD stage 3. Children with CKD stages 4 and 5 require 100%–120% of DRI for ideal body weight. Children on dialysis should receive 100% of DRI of protein for ideal body weight in addition to losses from hemodialysis or PD. For patients on hemodialysis, an estimated additional 0.1 g/kg/day of protein is required. For patients on PD, an estimated additional 0.2–0.3 g/kg/day may be required.
Figure 1 is from “KDOQI Clinical Practice Guideline for Nutrition in Children with CKD: 2008 Update,” which provides guidelines on recommended dietary protein intake in children with CKD. 2
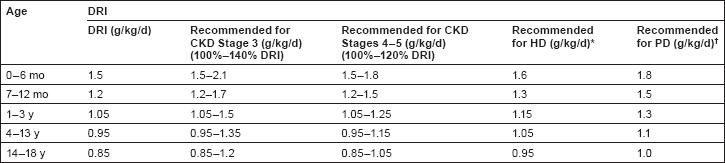
Recommended dietary protein intake in children with CKD stages 3 to 5 and 5D. Reprinted with permission from: National Kidney Foundation Kidney Disease Outcomes Quality Initiative. KDOQI clinical practice guideline for nutrition in children with CKD: 2008.
Sodium
Children with advanced CKD who have poor urine output typically require a sodium-restricted diet. A no-added-salt diet that encourages cooking without salt is a starting point. Teaching families and patients how to read nutrition labels helps them to better assess their salt intake. KDOQI guidelines recommend <1500–2400 mg/day for children who require sodium restriction. Table 1 provides some practical suggestions for patients requiring a sodium-restricted diet. Children with salt wasting may require supplemental sodium. Children on PD with increased sodium losses in their ultrafiltrate may also require sodium supplementation.
Discussion points to help children lower their sodium intake.

Potassium
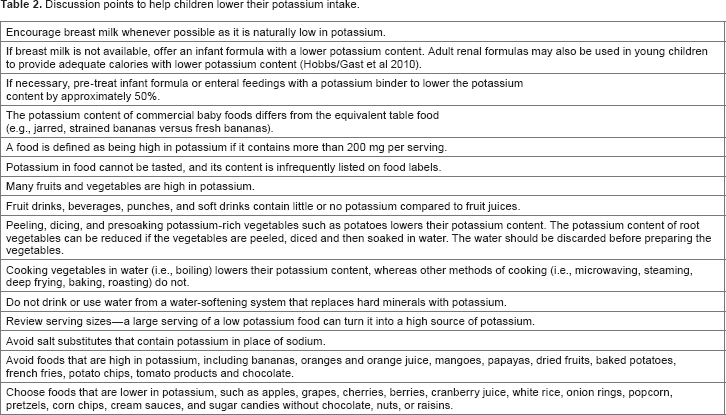
Hyperkalemia poses a more significant risk than hypokalemia in children with CKD, increasing risks of arrhythmias and cardiac arrest. KDOQI recommends a restriction of 40–120 mg/kg/day for infants and younger children and 30–40 mg/kg/day for older children. 2 Breast milk has low potassium content and should be encouraged for infants. Families should be educated about high potassium foods to avoid. Salt substitutes that contain potassium should be avoided as well. The Food and Drug Administration recently proposed changes to add potassium content to nutrition labels, which often do not list potassium. Table 2 provides some practical suggestions for patients requiring a potassium-restricted diet.
Discussion points to help children lower their potassium intake.
Potassium binders, such as sodium polystyrene sulfonate, can be used to pretreat formulas to lower the potassium content. The medication is mixed with the formula and allowed to form a precipitate. The supernatant is then decanted and used to feed the child. For older patients, oral sodium polystyrene sulfonate may be prescribed to lower serum potassium levels if diet restrictions alone are not effective. Sodium polystyrene sulfonate is a cation-exchange resin that can be administered orally or rectally as an enema. Approximately 1 mEq of potassium in the gastrointestinal tract is exchanged per gram sodium polystyrene sulfonate. In exchange for potassium, 4.1 mEq (~100 mg) of sodium per gram of the drug is delivered, which may need to be added to total daily sodium intake calculations. Sodium polystyrene sulfonate is not a fast-acting medication and should not be used as a sole therapy in the management of acute moderate-to-severe hyperkalemia. Sodium polystyrene sulfonate causes significant diarrhea at the time of use. Fluid loss from diarrhea should be included in total daily fluid losses with potential fluid replacement if required. Small amounts of magnesium and calcium may also be removed. Patients should be monitored for potential risk of colonic necrosis even with sorbitol-free sodium polystyrene sulfonate. 9
Phosphorus
Hyperphosphatemia or hyperparathyroidism may necessitate a dietary phosphorus restriction, which is often challenging for children. Hyperphosphatemia is an independent risk factor that increases cardiovascular morbidity and mortality such as vascular calcifications in patients with CKD and end stage renal disease (ESRD) on dialysis.10,11 Poor bone health affects linear growth as well. 12 Management of hyperphosphatemia is therefore key in improving these outcomes. Limiting phosphorus intake can help improve metabolic bone disease and cardiovascular morbidity and mortality associated with CKD. In the setting of elevated parathyroid hormone (PTH), KDOQI guidelines recommend limiting phosphorus intake to 100% of DRI for age. If serum phosphorus and serum PTH levels are both elevated, it is recommended that phosphorus intake be limited to 80% of the DRI. Phosphorus binders may be prescribed in conjunction with a dietary phosphorus restriction to help control serum phosphate levels. In children, calcium carbonate, calcium acetate, and sevelamer carbonate are the typical choices. These should be administered at the time of food consumption and will bind with dietary phosphorus to prevent absorption. When choosing which phosphate binder to prescribe, one must consider dietary calcium intake as well as the calcium content of the binder. Calcium-containing binders should be carefully considered as they are associated with the development of calcification. More calcium is absorbed from calcium carbonate when compared with calcium acetate. 13 Table 3 provides some practical suggestions for patients requiring a phosphorus-restricted diet.
Strategies for lowering dietary phosphorus intake and/or absorption in infants and children.

Calcium
Calcium balance is important to promote bone health and mineralization. The current KDOQI recommendations suggest that total calcium intake should be between 100% and 200% of the DRI for age with a maximum of 2500 mg of elemental calcium per day in older children. The total calcium intake calculated should include the calcium-based phosphate binders. Calcium supplementation may be necessary if dietary intake does not meet DRI. Calcium gluconate, lactate, acetate, and carbonate are the most effective forms of supplementation. Calcium chloride may cause or worsen metabolic acidosis. 2 Calcium citrate can increase intestinal absorption of aluminum, leading to aluminum toxicity in CKD patients. 14 The addition of vitamin D supplementation increases intestinal absorption of calcium.
Patients should be monitored for hypercalcemia as this could increase the risk for calcification and cardiovascular diseases. Hemodialysis and PD can be modified with lower dialysate calcium concentrations. The most recent meta-analysis does not show significant differences between paricalcitol or calcitriol used in the treatment of secondary hyperparathyroidism in causing hypercalcemia. 15 Calcimimetics such as cinacalcet hydrochloride are considered to treat secondary hyperparathyroidism in patients with hypercalcemia.
Iron
Anemia can be found in the early stages of CKD (as early as CKD stage 2) and is secondary to loss of erythropoietin production. In addition, many patients are found to be iron deficient as well. Ongoing research on the role of hepcidin in iron metabolism and subclinical inflammation as part of the pathomechanism of anemia in CKD.16,17 Individual needs such as age- and sex-specific hemoglobin distribution, neurocognitive development, and exercise capacity are considered when targeting hemoglobin in children with CKD between 11 and 12 g/dL. 18
The treatment of anemia in CKD begins with the full evaluation of a child's current iron status with the measurement of iron level, ferritin, and iron saturation. Supplemental oral or intravenous iron is usually required by children of all ages on erythropoietin-stimulating agents to avoid storage iron depletion, prevent iron-deficient erythropoiesis, and achieve and maintain target hemoglobin concentrations. Iron supplementation can be provided in liquid formulation as well for GT-dependent children. The starting dose recommendation is 3–4 mg/kg/day of elemental iron. 18 KDIGO 2012 recommends the evaluation of iron status at least every three months.
Vitamin B12 and Folate
Children with CKD and ESRD on dialysis can develop vitamin and mineral deficiencies due to anorexia, poor intake, dietary restrictions, abnormal renal metabolism, drugnutrient interactions, poor gastrointestinal absorption, and potential losses due to dialysis.19,20 In addition to iron supplementation, some children require vitamin B12 and folic acid supplementation as part of the management of their anemia of CKD. Canepa et al estimated the prevalence of hyperhomocysteinemia in up to 40% in children with chronic renal failure. 21 A small percentage of children with hyperhomocysteinemia have been found to have low folic acid levels and a smaller percentage have been found to have low vitamin B12 levels. 21 Elias et al reported an association between hyperhomocysteinemia and arterial stiffness in those with CKD compared to those without CKD. 22 However, a recent meta-analysis did not find that folic acid supplementation reduced cardiovascular events in adults with kidney diseases. 23 Due to lack of supportive evidence, KDIGO 2012 does not support folic acid supplementation as an adjuvant for ESA treatment. 18 However, current practice provides folic acid and vitamin B12 to pediatric dialysis patients as part of a standard water-soluble vitamin supplement.
Post-renal Transplantation
With a well functioning graft, the nutrition goals post-kidney transplant shift. While the primary goal remains the optimization of nutrition status with adequate calories and protein, there is a new focus on maximizing hydration while minimizing nutrition-related side effects of transplant medications. The continued involvement of a renal dietitian is critical.
The target goal for caloric intake is 100% of energy efficiency ratio (EER) for age, adjusted for activity, and body size. Protein needs increase after surgery to permit wound healing but return back to 100% of the DRI once healing is complete. Significant weight gain is often seen with prednisone immunosuppression, 24 and excessive caloric consumption becomes a prominent focus of discussion with encouragement of heart-healthy eating habits and increased physical activity. New-onset diabetes after transplantation requires further adjustment of the nutrition care plan with blood glucose monitoring and diabetic diet education. GT-dependent children, particularly those with normal cognition, may show a quick improvement of oral food intake after transplantation with the goal of transition off of G-tube feeds as soon as they are able. 25 Electrolyte imbalances such as hyperkalemia, hypomagnesemia, and hypophosphatemia are often seen and can be managed through dietary changes and medication adjustments. These are usually secondary to medication effects as well as the presence of secondary hyperparathyroidism prior to transplant.
Summary
Nutritional management of children with CKD is a challenge that requires continuous assessment and adjustment of the nutrition care plan as the child ages and CKD progresses. A renal dietitian plays a vital role in the multidisciplinary approach to CKD management. Achieving a balance between calorie, protein, and electrolyte needs can be challenging in the face of multiple dietary restrictions, but with education and support, it is possible to promote growth and weight gain in children with CKD.
Author Contributions
Conceived the concepts: LN, RHM. Analyzed the data: LN, RL, RHM. Wrote the first draft of the manuscript: LN. Contributed to the writing of the manuscript: LN, RL, RHM. Agree with manuscript results and conclusions: LN, RL, RHM. Jointly developed the structure and arguments for the paper: LN, RHM. Made critical revisions and approved final version: LN, RL, RHM. All authors reviewed and approved of the final manuscript.
