Abstract
Alzheimer's disease (AD) affects the majority of the 35 million people with dementia worldwide. Four pharmacological treatment options are available for this patient group, of which memantine is licensed for treatment of people with moderate to severe stages of the condition. Memantine acts through its function as an NMDA-glutamate receptor blocker and has an established safety profile. The evidence supporting its efficacy in people with AD includes a number of large randomized clinical trials showing benefit to cognition, function, and overall clinical outcome. Additional favorable health economics analyses have confirmed the clinical and cost-effectiveness of this drug. More recently an extended-release formulation has been developed. This review outlines the key evidence base supporting memantine as a treatment for moderate to severe AD, in addition to discussing the conditions under which it may provide additional value in combination with other drugs. The review also discusses the use of memantine to address behavioral and psychological symptoms of dementia (BPSD) arising in people with AD and the limited evidence around its use in AD in people with Down's syndrome. Finally the review considers the potential value of the extended release formulation in AD.
Introduction
There are 35 million people with dementia worldwide, and the most common cause is Alzheimer's disease (AD). AD follows a pathway of progressive cognitive decline associated with significant pathology, the most prominent of which is the accumulation of beta-amyloid and hyper-phosphorylated tau in the brain. The resulting symptoms of gradual cognitive loss and impairment are devastating, leading to eventual functional impairment and loss of independence. The condition affects 5% of people over 65, 20% over 80, and more than a third of those over 90. 1 The number of people with AD is expected to rise with the current ageing population with higher life expectancies. It is estimated that there will be over 115 million people with dementia worldwide by 2050, of which the majority will have AD. 1 AD incurs extremely high costs, both on a personal level to those affected, including their families and carers, and on a financial level, with an estimated $604 billion spent worldwide on treatment and care for people with dementia worldwide. 2 AD has become a major public health issue which will only continue to worsen. While research continues to develop more effective treatments to treat the underlying pathology and delay emergence of AD, it is critical that existing pharmacological therapies are positioned to maximize benefits to people with the condition today.
Two drug classes are currently available for treatment of AD. Acetylcholinesterase inhibitors (AChEI), including donepezil, rivastigmine, and galanthamine, are available for people with mild to moderate AD, and NMDA-glutamate receptor blockers, of which the only licensed drug is memantine, are licensed for moderate to severe AD cases. The efficacy of AChEI in improving symptoms of AD has been established through more than 30 randomized controlled trials (RCT). The RCT literature demonstrates significant improvement in cognition, function, and global clinical outcomes in people with mild to moderate AD treated with AChEI over six months compared to a placebo group, with standardized effect sizes of approximately 0.4.3–5 Three key RCTs of memantine reported significant benefit in moderate to severe AD.6–8 Both licensed drug classes have been shown to be cost-effective based on health economic analyses. 9
This review focuses on memantine, providing an overview of the structure and mechanisms of action of the compound, reviewing the safety and tolerability, and summarizing the established benefits in the treatment of AD. The review will also focus on possible further therapeutic uses in AD such as the treatment and prophylaxis of behavioral and psychological symptoms of dementia (BPSD) and treatment of AD in people with Down's Syndrome, in addition to considering the potential benefits of combining memantine and AChEI to determine whether there are opportunities for greater clinical benefits beyond the current license indications. Finally, the review considers the evidence to support the recently developed extended-release formulation of memantine and any implications for practice.
Memantine
Two-thirds of cortical neurons are pyramidal cells and use glutamate as a neurotransmitter, hence the glutamatergic system is understood to be a vital element in all regions of the cortex and hippocampus. The role of glutamate in learning and memory is long-established and it is also likely that any cortical function will be underpinned by glutamate neurotransmission. The vital role of glutamate in learning and memory is exemplified by the NMDA receptor, which acts as a co-incidence detector in long-term potentiation (LTP) formation. A key feature of the function of this receptor is the transient high peak of glutamate in the synaptic cleft during depolarization, detected against a low resting background level, and resulting in a high signal-to-noise ratio.
Memantine hydrochloride acts in mimicry with physiological magnesium as a voltage-dependent, moderate-affinity uncompetitive NMDA-receptor antagonist. 10 Reduced efficiency of this voltage-dependent Mg2+ block alters the ability of the NMDA subtype of glutamate receptor to generate LTP, an important underpinning of learning and memory. Evidence for dysfunction in glutamate neurotransmission in AD is extensive and such changes are considered to contribute to cognitive impairment in AD.10,11 For example, reduced glial glutamate uptake is reported in AD 12 perhaps reflecting oxidative damage to the transporters. Additionally, there is loss of the vesicular glutamate transporter (VGluT) in several cortical regions. 13 Since memantine has a moderate affinity for the NMDA receptor and blockade is voltage dependent, it is hypothesized to allow the molecule to prevent this receptor from opening during the constant background ‘noise’ of pathological glutamatergic activation at rest, while still being displaced upon arrival of a physiological signal.10,11 In vivo studies in a number of animal models of both neurodegenerative and vascular pathology have shown memantine to be an effective neuroprotectant.14–17 Finally, emerging evidence suggests that memantine may influence the status of tau phosphorylation, an important correlate of tangle formation, acting either to prevent addition of phosphate to tau or increasing its removal. 18
Memantine was first used to treat AD in Germany in 1991 and has shown some benefit in other conditions, including Parkinson's disease and vascular dementia.19–23 When taken orally, memantine is completely absorbed after six hours, undergoes limited metabolic breakdown, and has a terminal elimination half-life between 60 and 100 hours. 24 The licensed form of memantine is provided as a 10 mg bd oral dose. More recently, an extended-release formulation has been developed, as a once-daily 28 mg dose. The extended release formulation of memantine is licensed but not marketed in the USA and remains unlicensed in Europe.
Safety of Memantine
Safety and efficacy of memantine for treatment of dementia, and particularly AD, at a dose of 20 mg/day has been established in RCTs. 25 The current literature, which includes 1784 people receiving memantine and 1595 people receiving placebo, shows no difference in incidence of adverse effects between the two groups. 26 Any reactions following treatment with memantine were mild to moderate in severity, with the most common being dizziness (6.3% vs. 5.6%), headache (5.2% vs. 3.9%), constipation (4.6% vs. 2.6%), somnolence (3.4% vs. 2.2%), and hypertension (4.1% vs. 2.8%). Venous thromboembolism was also significantly more common following treatment with memantine, albeit in very few cases. To reduce this risk, monitoring of blood pressure and general good clinical practice is recommended. Caution should be taken when considering memantine for patients with epilepsy, a history of convulsions, or those with predisposing factors for epilepsy, and a lower dose is recommended for people with renal failure. 26 Overall, memantine has a good safety profile and is well tolerated. This safety evidence is summarized in Table 1. Despite this safety and tolerability record, theoretical advantages have been postulated for a once daily extended-release formulation. Certainly this may have advantages for people with AD who are reluctant to take medications and for those where medication administration needs to be supervised by a family member or professional carer. There is only a limited evidence base available for the extended-release form of memantine. However, to date, studies indicate that it has a similar adverse event profile to the 10 mg bd memantine formulation. This is also consistent with the findings of an RCT which compared once-daily doses of 20 mg memantine with twice-daily doses of 10 mg and showed no difference in safety and tolerability. 27
Adverse effects associated with memantine.
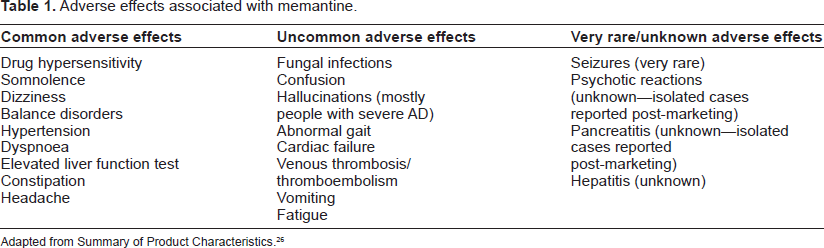
Adapted from Summary of Product Characteristics. 26
Memantine in the Treatment of AD
Although 12 RCTs comparing memantine to placebo have been completed, a number have not been published in full. A Cochrane review includes data from all 12 trials, but only three trials contribute data with respect to outcomes in people with moderate to severe AD at six months. 28 A further three trials contribute data regarding the impact of memantine in people with mild to moderate AD. Pooled data from all three trials included in the Cochrane evaluation of memantine for the treatment of moderate to severe AD indicate benefit to cognition (2.97 points on the 100 point SIB, 95% CI 1.68 to 4.26, P < 0.00001), activities of daily living (1.27 points on the 54 point ADCS-ADLsev, 95% CI 0.44 to 2.09, P = 0.003), and clinical global impression of change (0.28 points on the 7 point CIBIC+, 95% CI 0.15 to 0.41, P < 0.0001).2,6,28,29 The results indicate a reduction in the rate of decline compared to placebo rather than improvement in cognition and function following treatment. Nonetheless, this effect is important in this patient group where extending the period of time when individuals retain a level of independence and function is of great value, as well as having a direct impact on quality of life. This is emphasized by retrospective reports that show that specific elements of function, such as mobility, remain more stable in people treated with memantine than those in the placebo group. A more recent RCT provided further evidence to support the benefit of memantine in moderate to severe AD, showing consistent benefits to cognition (MMSE 1.2 [95% CI 0.6 to 1.8; P < 0.001]) and function (BADLS 1.5 [95% CI 0.3 to 2.8; P = 0.016] compared to placebo memantine in over 12 months of treatment. 30 The potential impact of memantine in people with mild to moderate AD are less substantial. RCTs in this group of people show only limited benefits compared to placebo in both cognition (0.99 points on the 70 point ADAS-Cog, 95% CI 0.21 to 1.78, P = 0.01) and overall benefit (0.13 CIBIC+ points, 95% CI 0.01 to 0.25, P = 0.03).28,31 As a result, memantine is established as both clinically and cost-effective for the treatment of moderate to severe AD, but is not currently licensed for treatment of mild to moderate stages of the condition.
The evidence base for the new extended-release formulation of memantine is extremely limited. All of the published RCTs of memantine in AD have been based upon an oral prescription of 10 mg bd. Whilst several clinical trials have been undertaken using the 28 mg/day extended release formulation, which is orally administered once a day, none of these studies have been fully published in peer review journals. The only RCT is a six month comparison of memantine combined with a cholinesterase inhibitor compared to a cholinesterase inhibitor alone in 661 patients with moderate to severe AD. Some of the data from this study have recently been published as part of a meta-analysis 32 indicating significant advantages with the combination of extended-release memantine and a cholinesterase inhibitor alone with respect to cognition (standardized mean difference SMD -0.21 95% CI -0.37 to -0.05), global outcome (SMD -0.25 95% CI -0.40 to -0.10), and behavior (SMD -0.20 95% CI -0.35 to -0.04), but not activities of daily living. Summary data from this study, one open label study and two open-label extension studies are available on the Forest pharmaceutical company website, 33 and indicate good tolerability. However, there are no head to head comparisons with the currently licensed version of memantine. It is difficult to interpret this evidence base in the absence of any placebo-controlled trials or head-to-head comparisons of the two memantine formulations. In addition, it is difficult to comment on the quality of these studies in the absence of any peer review publications. From the available information, there is a much more substantive evidence base to support the use of the 10 mg bd formulation and it is difficult to see any advantages of the extended release formulation without a further clinical trial program.
Combined Treatment of Memantine and Donepezil
Three RCTs have evaluated the potential benefit of combined therapy with memantine and the AChEI donepezil for treatment of AD. The outcomes are difficult to interpret, but point towards possible beneficial effect.29,30,34 The first of these trials recruited 403 people with moderate to severe AD, with a MMSE score of four to 14. People randomized to a combination of memantine and donepezil showed significantly better performance in cognition with a 2.5 point advantage on the Severe Impairment Battery (SIB) compared to placebo (SIB Standardized Mean Difference 0.42, 0.20–0.63) compared to people receiving donepezil alone. 29 The subsequent study evaluated combined treatment in 202 people with mild to moderate AD (MMSE of ten to 22) over 24 weeks. The treatment group received memantine in addition to their existing AChEI prescription (donepezil, rivastigmine, galanthamine), but showed no significant improvement compared to the control group. The most recent study to investigate this approach was the DOMINO study which included 295 people with moderate to severe AD (MMSE five to 13), all of whom were already prescribed donepezil but were under consideration for withdrawal. The cohort was randomized to receive either continuation of donepezil alone, a switch of prescription to memantine or combined treatment. A fourth group received placebo only. As expected, the study reported significant benefit with both donepezil and memantine compared to placebo over 12 months. 30 The combination treatment resulted in numerical improvements on all measures, but none reached statistical significance. It should be noted that interpretation of these findings are difficult since the study was powered for comparison with the placebo arm, and not for comparison between active treatments. A recent meta-analysis of the Tariot and Porsteinsson studies moves forward our understanding of the evidence, suggesting that combination treatment with memantine and donepezil does lead to significant benefit compared to donepezil alone in people with moderate-severe AD, but not for individuals with mild AD. Across both studies all outcomes were improved in the combined treatment group in people with moderate to severe AD (MMSE 5–19 SIB SMD 0.36, 0.19–0.54, ADCS ADL SMD 0.21, 0.04–0.39; Global Clinical Outcome SMD 0.23, 0.06–0.41). 35 Taken together, although the evidence is not straightforward, this appears to indicate that combined treatment does confer overall benefit in comparison to maintaining treatment with donepezil alone. Additional support for this interpretation is provided by a cohort study that reported improved long term outcomes in people receiving combination treatment. 36 Importantly, both of these compounds are now off-patent and the cost of widespread prescription continues to decrease as generics are manufactured. This raises the urgent need for a cost-effectiveness analysis to identify whether combination treatment could be a viable therapy for people with moderate to severe AD.
Memantine for Behavioral and Psychological Symptoms in Dementia (BPSD)
Behavioral and psychological symptoms of dementia (BPSD), including delusions, hallucinations, agitation, and aggression, are very common in people with AD and affect up to 90% of people with dementia. 37 BPSD causes significant distress to the individual and their carers. They present a challenge for clinicians making decisions about treatment frequently resulting in placement in long term care and prescriptions of sedatives, most commonly atypical antipsychotic medications.38,39 The short term (less than 12 weeks) use of antipsychotics in these patients is known to be useful in certain severe cases where symptoms are causing unacceptable risk or distress. This is based on evidence from 18 RCTs showing improvements in BPSD including aggression and psychosis following short-term treatment. The best established evidence of efficacy is for risperidone, with five RCTs showing benefit.40–46 However, antipsychotics are associated with significant safety issues, including increased risk in mortality by up to 80%, and increased likelihood of severe cerebrovascular events such as stroke.47–49 Although strict monitoring protocols go some way to help control for these risks, it is clear that the modest benefit conferred by antipsychotics are overshadowed by the risks when prescriptions extend beyond 12 weeks.47–49
There is therefore imperative to introduce alternative pharmacological approaches to treat BPSD which have a more acceptable safety profile. A key question in the field currently is whether memantine may hold value in addressing these symptoms. Interrogation of secondary outcomes from existing RCTs of memantine highlight some promising findings to date. A meta-analysis of data from a number of trials have reported a significant, if modest, improvement in BPSD measured by the Neuropsychiatric Inventory (NPI) in people treated with memantine compared to placebo (2.76 points on the 144 point NPI, 95% CI 0.88 to 4.63, P = 0.004).50–52 A subsequent post-hoc analysis of additional RCTs, which included at least one measure of BPSD, included data from 983 people with moderate to severe AD, of whom 593 were experiencing these symptoms. The analysis of NPI data revealed significant improvement in these individuals when treated with memantine after both 12 (-0.8 points vs. 0.5 points; P = 0.0014) and 24/28 weeks (-0.7 points vs. 0.7 points; P = 0.0004). 53 This effect was particularly prominent in reducing agitation and aggression at both time points (54.8% vs. 42.1%; P = 0.008 and 61.0% vs. 45.0%; P < 0.001, respectively). Interestingly, these individuals also showed better response in cognitive and functional outcomes compared to the overall cohort, potentially indicating an overall treatment response. 53 It is important to note however that this study was a post-hoc analysis, and must be interpreted with caution. The overall baseline level of BPSD was also reported to be mild (mean total NPI score of 20) and so only provides a limited view of the potential value of memantine in treating clinically significant BPSD.
Three more recent studies add useful data to this issue. One RCT compared memantine and placebo in 149 people with AD and BPSD. The study reported no improvement in agitation or global clinical outcome over 6 or 12 weeks (CMAI: 6 weeks -3.0; -8.3 to 2.2, P = 0.26, 12 weeks: [-3.8 (95% CI -9.1 to 1.5 P = 0.16], Clinical Global Impression of Change: 6 weeks P = 0.45, week 12 P = 0.11). 54 Two further studies reported more promising data. The DOMINO study described above showed a benefit of 4.0 [99% CI 0.6 to 7.4; P = 0.002] NPI points in the memantine treatment group compared to those receiving placebo memantine over 12 months. Adding memantine to donepezil continuation resulted in an even larger benefit on the NPI of 5.1 [99% CI 0.3 to 9.8; P = 0.006] points compared to placebo. 30 The MAIN-AD study, currently only published in abstract, 55 randomized 200 patients with AD with an antipsychotic prescription to memantine or to continue with the antipsychotic. So far the study has reported similar outcomes over 26 weeks with respect to agitation. In a similar way to the post-hoc analysis performed by Wilcock et al, 53 the level of baseline BPSD was modest. Again this is a conflicting evidence base which is not straightforward to interpret. Overall, the evidence indicates that memantine is not efficacious in the acute (6 to 12 weeks) treatment of clinically significant agitation and aggression, but does have value and similar efficacy to antipsychotics in the longer term treatment and prophylaxis of mild BPSD, with safety advantages.
There is only one study which was included in a recent systematic review which examined the effectiveness of the extended-release formulation of memantine on behavior, in combination with a cholinesterase inhibitor. The study reported a significant improvement in behavior compared with cholinesterase inhibitor alone. 32 However, there is no direct comparison with the licensed form of memantine therefore this outcome provides limited information regarding the potential benefit of the newly developed formulation on these symptoms.
Memantine for AD in Down's Syndrome
Development of AD in people with Down's syndrome is very common, with many people with Down's syndrome over the age of 40 showing AD pathology. There is limited evidence upon which to base treatment decisions. In vivo studies have shown promising findings with memantine treatment in transgenic mice.56,57 However a recent prospective RCT of 173 people with Down's syndrome with and without dementia showed that despite being well tolerated, memantine did not confer benefit incognition, function, or global outcome either in the overall group or the subgroup of individuals with dementia. 58 Despite promising indications from animal studies this study strongly indicated that memantine is not an effective treatment in older people with Down's syndrome, and highlighted potential issues with assuming that treatments that are effective in people with AD will also be effective in people with Down's syndrome.
Author Contributions
Analyzed the data: AC, PF, CB. Wrote the first draft of the manuscript: AC. Contributed to the writing of the manuscript: AC, PF, CB. Agree with manuscript results and conclusions: AC, PF, CB. Jointly developed the structure and arguments for the paper: AC, PF, CB. Made critical revisions and approved final version: AC, PF, CB. All authors reviewed and approved of the final manuscript.
Funding
All authors thank the National Institute for Health Research (NIHR) Mental Health Biomedical Research Centre and Dementia Unit at South London and Maudsley NHS Foundation Trust and Institute of Psychiatry, King's College London for supporting the work for this manuscript. This article presents independent research funded by the National Institute for Health Research (NIHR). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
Competing Interests
AC has received consultancy fees from Acadia pharmaceuticals, and speaking honoraria from Lundbeck, Novartis and Bial pharmaceutical companies. None of this was directly related to the content of this manuscript. PF has received funding and speaker fees from Lundbeck pharmaceuticals and provided expert testimony on behalf of Janssen Alzheimer Immunotherapy and Novartis. CB has received speaking and consultancy honoraria from Lundbeck, Novartis, Bristol Myer Squibb, Janssen and Acadia pharmaceuticals, research funding from Lundbeck pharmaceuticals and royalties from Oxford University Press and Informa.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests. Provenance: the authors were invited to submit this paper.
