Abstract
Angiotensin converting enzyme inhibitors (ACEIs) and angiotensin II receptor blockers (ARBs) are one of the renin angiotensin system (RAS) inhibitors widely used for the treatment of hypertension. Recently, Blood Pressure Lowering Treatment Trialists’ Collaboration (BPLTTC) meta-analysis showed that ACEIs had the blood pressure independent beneficial effects on coronary heart events. In this review, we summarized our current knowledge about ACEIs especially imidapril and re-evaluated ACEIs among other RAS inhibitors (RASIs) because ACEIs have wide ranges of beneficial effects in addition to the ACE inhibition which other RASIs do not have such as the up-regulation of bradykinin level, substance P level, the inhibition of matrix metalloproteinase (MMP) activity and stabilization of coronary plaque.
Keywords
Introduction
The renin angiotensin system (RAS) is a key regulator in maintenance of blood pressure. 1 In a broad sense, there are four major types of drugs to block the RAS, beta blockers, renin inhibitor, angiotensin II receptor blockers (ARBs) and angiotensin-converting enzyme inhibitors (ACEIs). Beta-blockers reduce renin release from juxtaglomerular apparatus, 2 renin inhibitor directly inhibits the activity of renin,3,4 ARBs competitively block the binding of angiotensin II to their receptors and ACEIs inhibit the enzymatic activity of angiotensin converting enzyme (ACE).5–8
In 1985, Taguma Y et al first reported that ACEIs reduced urinary protein excretion in diabetic patients with nephropathy. 9 After this report, many clinical studies have shown that ACEIs preserve renal function in diabetic nephropathy.7,10–12 Now, ACEIs are recognized as drugs which demonstrate organ protective actions mediated by not only ACE inhibition but also other various mechanisms.
This review summarizes our current knowledge about ACEIs, especially imidapril in respect to which it has been reported that the aspiration pneumonia and the cardiovascular disease protective effects are especially strong among ACEIs,13–16 how ACEIs provide beneficial effects for the management of non-diabetic and hypertensive type 2 diabetes.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Imidapril is one of the ACEI which inhibits the conversion of angiotensin I to angiotensin II. Furthermore, ACEIs including imidapril are thought to regulate many pathways such as kininogen/bradykinin/prostaglandin pathway and MMP2/9 pathway.17,18 These lead to marked peripheral vasodilation and the stability of coronary plaque (Fig. 1).
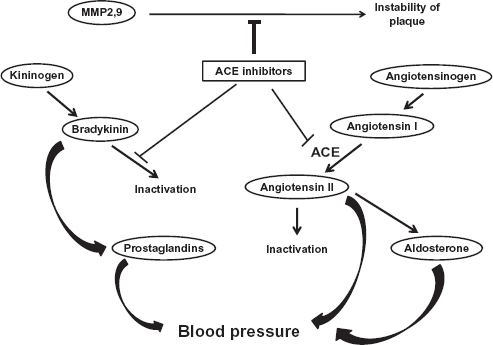
Schematic presentation of mode of action of ACEIs. ACEIs inhibit many molecules such as ACE, MMP2/9 and bradykinin inactivation pathway.
Furthermore, it is well known that ACE degrades substance P, a neurotransmitter of the swallowing reflex, and its action is potentiated by ACEIs.19,20 These lead to improvement of the swallowing reflex in older patients with aspiration pneumonia.
Imidapril is mainly hydrolysed to the active metabolite, imidaprilat and other inactive metabolites such as M2, M3 and M4 (Fig. 2). 21 It has been reported that imidaprilat up-regulated cellular retinol binding protein 1 (CRBP1) gene expression, transcriptional activity of peroxisome proliferator-activated receptor gamma (PPARγ)/retinoid X receptor alpha (RXRα) heterodimer partner and their many targets genes in adipocytes.22–24 ACEIs increased the serum bradykinin level and this increase accelerated the translocation of GLUT4 to the cell membrane in myocytes (Supplementary Fig. 1). 25 This lead to a suppressive effect of insulin resistance.
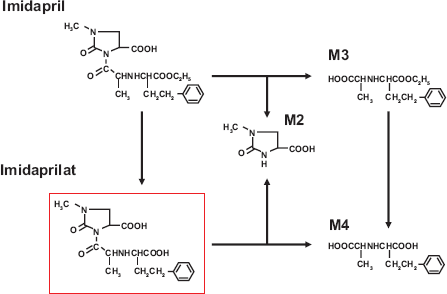
Postulated metabolic pathways of imidapril. Imidapril is converted into metabolically active metabolite, imidaprilat and inactive metabolites such as M2, M3 and M4.
About 70% of Imidapril is absorbed following oral administration and reaches maximum inhibition of ACE activity within 4 hours (Fig. 3). These results indicate that imidapril not only inhibits ACE activity but also influences a wide range of pathways.
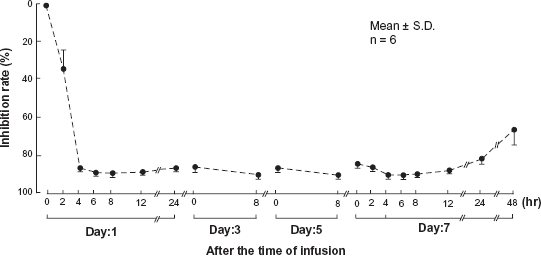
Schematic representation of the inhibition rate of ACE activity by imidapril (Phase I clinical study). Phase I clinical study of imidapril enrolled 6 healthy Japanese men. Men with renal disease, heart disease, liver disease, gastrointestinal disease and drug allergy were all excluded. Ten mg/day of imidapril were taken orally by 6 men, blood samples were collected at the indicated time points and ACE inhibition rate were determined by Kasahara method at SRL (Tokyo). 47
Clinical Studies
Many comparative clinical studies between ACEIs and ARBs were conducted on coronary heart disease, stroke and heart failure. In recent meta-analysis, Blood Pressure Lowering Treatment Trialists’ Collaboration (BPLTTC) analysis evaluated the blood pressure-dependent and independent effects between ACEIs and ARBs. This analysis showed that there are similar blood pressure (BP) dependent effects between ACEIs and ARBs about coronary heart disease, stroke and heart failure. Although BP- independent organ protective effects were recognized only in ACEIs treatment, the effects were not recognized in ARBs treatment (Fig. 4). 16 These results, therefore, suggest that the prevention of coronary heart diseases may depend on not only the blood pressure lowering effects but also the choice of antihypertensive agents.
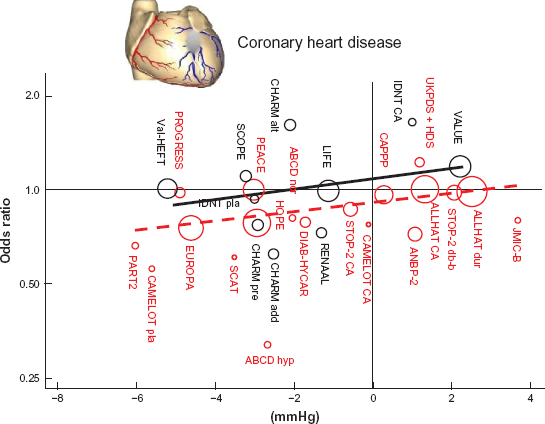
BPLTTC meta-analysis. Relationship between blood pressure lowering effects and the reduction of coronary heart disease in trials of ACEIs and ARBs. Red circles represent the trials of ACEIs and black circles represent the trials of ARBs with the area of each circle inversely proportional to the variance of the log odds ratio.
Shiga Plasminogen Activator in Coronary Circulation (SPAIC) study showed that imidapril increased the coronary release of tissue plasminogen activator (t-PA) in adult women but not in men (Fig. 5). 26 Coronary t-PA is considered to be an important defensive factor against coronary thrombosis, therefore cardiovascular protective effects of imidapril may be due in part to the increase of coronary t-PA release.
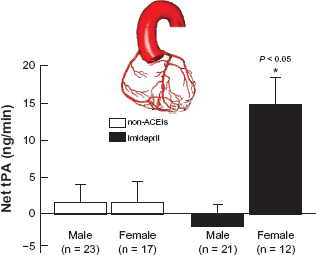
SPAIC study. SPAIC study enrolled a total of 140 patients with suspected coronary artery disease and hypertension (systolic blood pressure ≥ 140 mmHg or diastolic blood pressure ≥ 90 mmHg at the time of the recruitment into the study). Patients with the previous medication of anti-hypertensive agents, hormone replacement therapy, myocardial infarction, unstable angina pectoris, congestive heart failure, cardiomyopathy, severe valvular heart disease, premenopausal women, secondary hypertension, poor compliance, and significant coronary stenosis were all excluded. Eventually, 33 patients (21 men and 17 women) in the imidapril group (5 mg/day) and 40 patients (23 men and 17 women) in non-ACEIs group completed the study protocol. Net coronary t-PA release was determined by the coronary angiography. Net t-PA release in male and female imidapril and non-ACEIs groups (all patients). P < 0.05 vs. other groups. Note: Data are expressed as mean ± SEM.
Fibrinolysis and insulin sensitivity in imidapril and candesartan (FISIC study) showed that imidapril increased plasma t-PA activity. Furthermore, imidapril increased glucose infusion rate, which is considered to be an index of insulin sensitivity. 27
Another clinical study is Japan IDDM study (Japanese trial of ACE inhibitors on renal protection of nephropathy against IDDMs). 28 Japan IDDM study showed that imidapril prevented the increase of urinary protein excretion in micro- and macroalbuminuric type 1 diabetic patients (Fig. 6). Several lines of evidence showed that imidapril was one of the most effective anti-albuminuric and anti-atherogenic drugs among ACEIs in the treatment of hypertensive type 2 diabetic patients.
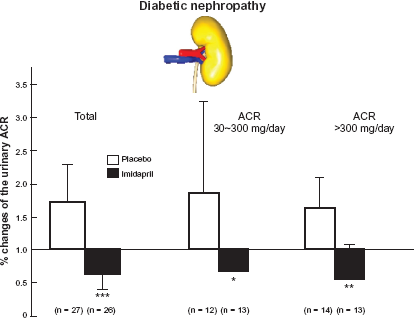
Changes in urinary albumin excretion from the baseline in the placebo group and the group treated with imidapril. JAPAN-IDDM study enrolled 83 patients who had developed type I diabetes mellitus before the age of 20, which was confirmed by lower serum levels and/or urinary excretion of C peptide. Patients with poor glycemic control with a HbA1c level greater than 10%, serum creatinine concentration greater than 2 mg/dl and other renal, endocrine, cardiac, liver, gastrointestinal, or connective tissue diseases were all excluded. Eventually 53 patients (27 placebo and 26 imidapril (5 mg/day)) completed the study.
Safety
Now ACEIs are widely used for the treatment of hypertension and heart failure around the world. However, some side effects to note have now been reported. For example, angioedema, dizziness, headache, general fatigue, hypotension, gastrointestinal (GI) symptoms, rash, cough, taste disorder and arrhythmia, etc (Supplementary Table 1).29–39
For imidapril there are some reports on its safety. Dews I and Vandenburg M reported that 46% of patients in the imidapril group and 53% of patients in hydrochlorothiazide group showed at least one adverse event in the multicenter, randomized, double-blind study compared oral administration of once-daily imidapril 5–20 mg with hydrochlorothiazide 12.5–50 mg in elderly patients for 24 weeks. The most frequent adverse events were cough (8%). Furthermore, three patients receiving imidapril died, two were possibly related with imidapril treatment, one was an unrelated case in this study. 40 Another group reported that 20.7% of patients in the imidapril group and 46.4% in captopril group showed some adverse events in 12-week, double blind, parallel-group, comparative trial. Again, the most frequent adverse events were cough (13.8%) in imidapril group, 35.7% in captopril group. 41
Efficacy
Patients with mild to moderate essential hypertension received placebo or imidapril 2.5 mg, 5 mg, 10 mg or 20 mg in a 2 week study (n = 91), or imidapril 5 mg, 10 mg, 20 mg or 40 mg in the 4 week study (n = 162). Compared with placebo, oral administration of 10 mg, 20 mg and 40 mg/ day of imidapril significantly reduced blood pressure without significant difference between these doses. The 2.5 mg of imidapril treatment showed no significant blood pressure lowering effect, and the 5 mg of imidapril treatment showed an intermediate blood pressure lowering effect. 42
In a long term extension study of 82 patients with mild to moderate hypertension treated with imidapril over one year, 49 patients (90.7%) on monotherapy (imidapril only) and 20 (76.9%) on combined therapy (imidapril plus thiazide diuretics) improved the reduction in blood pressure from 170/100 mmHg to 150/ 90 mmHg (Fig. 7 and Supplementary Fig. 2). 43
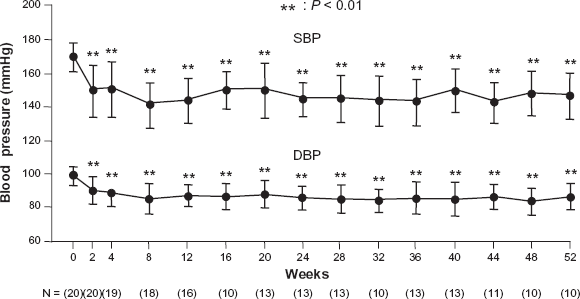
Efficacy of imidapril on essential hypertension in the long-term treatment (1 year). This study was designed as a multicenter, open-label clinical study. The patients subjected to this study were judged by physicians- in charged that showed favourable hypotensive effect and no problem in safety in the former phase II clinical study. Finally, 54 patients of mild to moderate essential hypertension were subjected to this study (31 men and 23 women). From 2.5 mg/day to 20 mg/day of imidapril were taken orally corresponding to blood pressure control and blood pressure was measured at the indicated time points.
In a multicenter, randomized, double blind and parallel group study, 161 patients with 95–115 mmHg of seated DBP were treated with placebo (n = 55) or imidapril 5–20 mg/day and were titrated at 2 week intervals for the first 4 weeks of the study if the DBP remained > 90 mmHg. After 8 weeks administration of placebo or imidapril, 33% of patients with placebo and 49% patients with imidapril attained a DBP of 90 mmHg. Thirty eight percent of patients with placebo and 61% patients with imidapril were classified as responders (P < 0.01 vs placebo). 44 Another multicenter, randomized, double blind and parallel group study, the Iberian Multicenter Imidapril Study on Hypertension (IMISH), reported that both 60 patients with the mean SBP and DBP, 155.7 mmHg and 96.7 mmHg respectively treated with imidapril up to 20 mg/day and 62 patients with the mean SBP and DBP, 158.4 mmHg and 98.3 mmHg respectively treated with candesartan up to 16 mg/ day achieved same blood pressure lowering effect and response rate after 12 weeks administration. 45
From the evidence of BPLTTC and major clinical trials, other physicians recommended that combination therapy such as ACEI and diuretics was effective in the treatment of Caucasians and Asians populations with hypertension. On the other hand, they preferred to avoid the treatment of ACEI and β-blocker combination for patients with uncomplicated hypertension and keeping as a second choice for the situation that other combinations were not effective or not well tolerated because the actions of 2 drugs in lowering BP are not fully independent nor fully additive. Furthermore, they also preferred to avoid the ACEI and ARB combination therapy because ONTARGET clearly indicated that this combination had no advantage in preventing hard outcome and disadvantages in the greater renal adverse events. 46
Patient preference
For many patients with hypertension, the uses of many antihypertensive agents have been decided by not only the efficacy but also patient's preference. Because some antihypertensive drugs have been associated with adverse events such as cough, which have been linked to no adherence, dissatisfaction with treatment, and side effect that ruins the quality of life, the clinical effects of these drugs should be further evaluated including the patient's preference to decide the likelihood of long-term acceptability. To continue a long-term treatment with ACEI successfully, the physician should fully explain the effects of their efficacy for the treatment of heart failure, diabetes, high coronary disease risk, post myocardial infarction, recurrent stroke prevention, chronic kidney disease, the necessity of the ACEI administration, and the side effect such as cough, and receive informed consent from the patients.
Conclusions
ACEIs, especially imidapril is as effective as other RAS inhibitors such as beta-blockers and ARBS in blood pressure reduction. Moreover, ACEI possesses more beneficial effects on the reduction of major coronary heart events than other RASIs and has superiority for these diseases.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
Footnotes
Acknowledgements
We thank Manami Shimizu and Mai Sasaki for preparing manuscript. We thank Drs. Kazuhiro Nako, Miho Senda and Takuya Sakamoto for helpful discussions. This work was supported by the Network Medicine Global-COE Program from the Ministry of Education, Culture, Sports, Science and Technology of Japan, Japan Heart Foundation & Astellas/Pfizer Grant For Research on Atherosclerosis Update and Kimura Memorial Heart Foundation Bayer Grant for Clinical Vascular Function (2010).
