Abstract
Objective
To compare the efficacy and tolerability of paroxetine and mirtazapine in the treatment of major depression.
Data sources
Searches were conducted to identify studies through Medline (1980-2011), PsycInfo (1980-2011) and PubMed databases up to June 2011. The searches were not restricted to publication type or clinical trial design.
Study Selection
A clinical trial was included if it described a trial of paroxetine versus mirtazapine in patients with major depression, based on the research evidence of reviews.
Data Abstraction
Three assessors analyzed the quality of the trials and extracted study design data, trial features, efficacy and toler-ability assessment tools, discontinuation reasons for both antidepressants and remitter and responder rates.
Results
We included six randomized controlled trials, one open-label, randomized controlled trial and four systematic reviews and metaanalysis. Rates of remission and response between mirtazapine and paroxetine were compared: at the beginning (1-2 weeks) there were statistically significant differences in mirtazapine treated patients, but these were not found at the end of assessment period (6-8 weeks). Discontinuation rates between the two drugs showed no differences, with an adverse event profile characteristic of each drug.
Conclusions
Mirtazapine and paroxetine were equally effective and well-tolerated in major depressive disorder. Differences in effectiveness were only observed in the first or second week of treatment when mirtazapine showed earlier onset of action.
Introduction
Major depressive disorder is the leading cause of psychiatric morbidity in developed countries with a prevalence of about 20%. 1
The most indicated treatment of major depression disorder is antidepressants and their choice depends particularly on the mechanism of action involved and the severity of symptoms. 2 Multiple action mechanisms of have been considered beneficial in terms of efficacy and tolerability. 3
Mirtazapine and paroxetine are two antidepressants with an efficacy and tolerability profile. The former shows a dual effect; noradrenergic and serotonergic, while the latter belongs to the selective serotonin reuptake inhibitor group.
Specifically, mirtazapine is an antagonist of alpha-2 adrenergic auto and heteroreceptors, and also shows 5-TH2 and 5-TTH3 receptor antagonism.4–6 As such, it increases noradrenergic and serotonergic neurotransmission and demonstrates excellent antidepressant action. 6
On the other hand, paroxetine is a selective and potent reuptake inhibitor of 5-hydroxytryptamine and its antidepressant action is related to specific inhibition of 5-HT reuptake in brain neurons. 7
The results of earlier clinical trials show equivalent efficacy for both drugs with mirtazapine showing faster onset of action than paroxetine. 7
We aimed to determine differences in therapeutic efficacy and tolerability between paroxetine and mirtazapine for patients with major depression based on the earlier and newer scientific literature.
Method
Search strategy
Searches were conducted to identify studies through Medline (1980-2011), Psyclnfo (1980-2011), and PubMed databases up to June 2011. The following search topics were used: mirtazapine, paroxetine, depressive disorder, affective disorder and major depression. The searches were not restricted to publication type or clinical trial design.
In addition, relevant abstracts related to the search terms were obtained to identify full-text articles.
Eligibility criteria
A clinical trial was included if it described a comparison of paroxetine and mirtazapine in major depression patients. Patients of all ages were selected.
Data abstraction
Two independent assessors extracted the data which, once completed, was reviewed by another senior researcher. The abstracted data included study design, trial features, patients’ characteristics at trial commencement, efficacy and tolerability assessment tools and reasons for discontinuation, such as adverse events.
Results
We included six randomized controlled trials, one open-label, randomized controlled trial and four systematic reviews and meta-analysis, which included results from clinical trials.
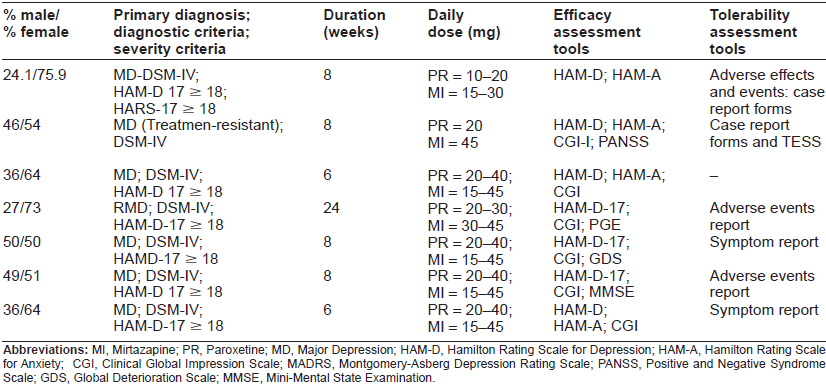
At the initial assessment, samples of the clinical trials were required to fulfill DSM-IV-TR criteria for a single or recurrent major depressive episode and have a score of at least 18 on the 17-item Hamilton Rating Scale for Depression (HAM-D-17). With regard to demographic data such as educational level and ethnicity, we highlight a lack of information in the included studies. Other sociodemographic and clinical data, such as nationality of the patients are specified in a table on description of the sample (Table 1).
Study and patients features for all trials (n = 7).
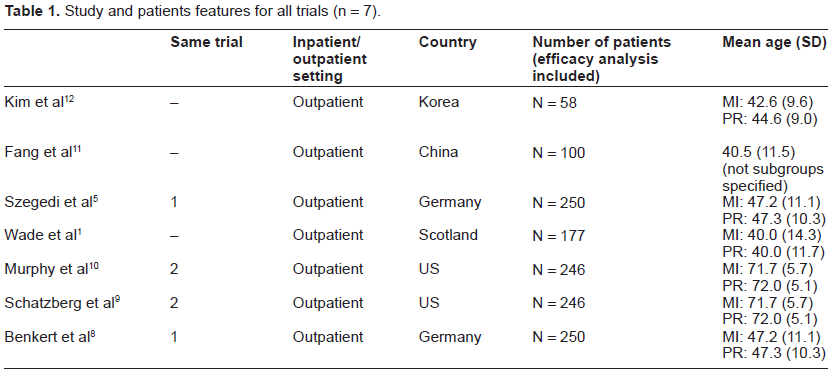
Patients in all selected trials were classified as responders (50% reduction in HAM-D-17 score from baseline), remitters (score of 7 or less on HAM-D-17) and patients on symptom reduction (change score from baseline). The mean time to response was also an outcome tool used for comparison of data. As an indirect assessment of tolerability, adverse events report and total dropouts due to adverse events were compared for the two drugs.
Rates of mirtazapine and paroxetine remission and response in the selected clinical trials were compared at the beginning of the drug administration (1-2 weeks) and at the end of assessment period (6-8 weeks).
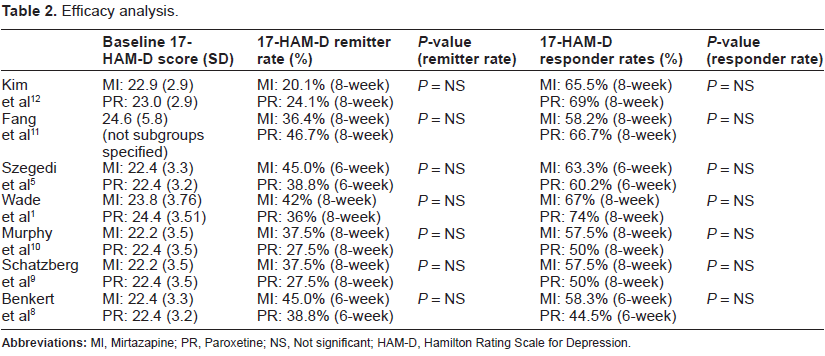
With our initial search strategy we obtained 7 trials comparing paroxetine and mirtazapine in major depression. Study and Patients Features for all Trials (n = 7) are shown in Table 1, efficacy analysis in Table 2 and discontinuation reasons in Table 3.
Efficacy analysis.
Discontinuation reasons.
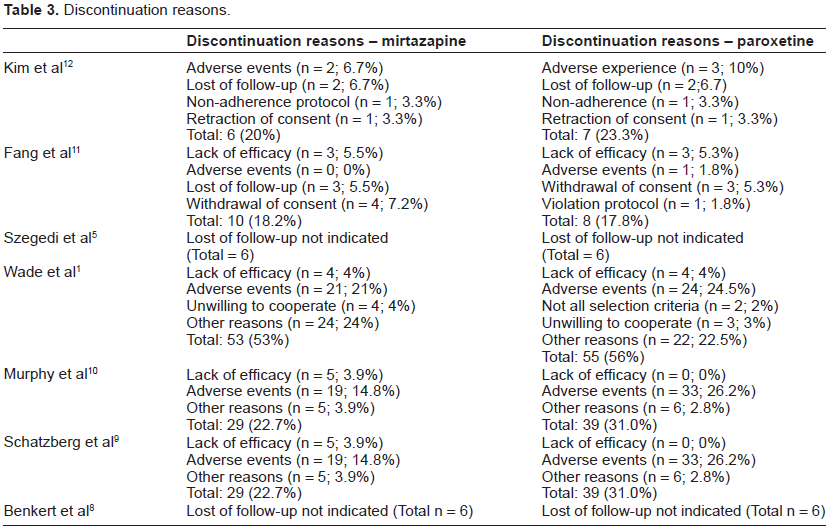
In two multicenter, double-blind, randomized controlled trials, Benkert et al 2000 and Szegedi et al 20035,8 included 250 German outpatients between 18 and 70 years old, treated with mirtazapine and paroxetine between 20-40 mg and 15-45 mg respectively for 6 weeks.5,8 Patients who fulfilled criteria for alcohol abuse or dependence, as well as seizures, were excluded. The average age of the sample was 47.2 years and most were women (63%-65% vs. 37%-35%) depending on the drug received. The mean dose was 32.7 mg for the mirtazapine group and 22.9 mg for paroxetine. Remission rates at 6 weeks of treatment were similar for mirtazapine and paroxetine (45% vs. 38.8%). Despite the similar reduction in HAM-D-17 score at the end of the period, there was greater reduction in the mirtazapine group compared with paroxetine at week 1 (16.5 vs. 18.8, P = 0.0032).
These results were confirmed in two 8-week, double-blind, randomized controlled trials by Schatzberg et al 2002 and Murphy et al 20039,10 which included patients from US aged at least 65 years old. They were treated in the outpatient setting. Patients with psychotic features, suicide attempts or previous symptoms compatible with cognitive impairment were excluded. A total of 246 patients (126 mirtazapine, 120 paroxetine) were included in the efficacy analysis with a mean age of 71.7 and 72 years respectively. 29 patients in the mirtazapine group (22.7%) and 39 (21.0%) in the paroxetine group discontinued treatment. When analyzing reasons for discontinuation, it was observed that patients receiving mirtazap-ine discontinued treatment due to adverse effects less frequently than those receiving paroxetine (14.8% vs. 26.2%). With respect to efficacy outcomes, the mirtazapine group had a higher rate of responders and remission compared to paroxetine at 14, 21, 28, 42 and 56 days after starting treatment. For the responders, the differences were statistically significant in the evaluation on day 14, when the mirtazapine group showed a 27.8% response rate versus 13.3% for the paroxetine group (P = 0.005).
Wade et al in 2003, 1 in a 24-week, double-blind, randomized controlled trial 1 compared the efficacy and tolerability of mirtazapine and paroxetine in depressed patients in primary care. The study included 177 Scottish outpatients between 17 and 74 years of age. The average age of the sample was 40 years and both groups included more women than men (75% mirtazapine group vs. 71% paroxetine group). Patients with a history of substance abuse or dependence, or suicidality were excluded. The mean doses were mirtazapine, 34.6 mg and paroxetine, 23.9 mg. The number of patients who discontinued due to adverse events was similar in both groups, with 21% for mirtazapine and 24.5% for paroxetine. The most frequently adverse event in patients treated with mirtazapine was fatigue (20%) and in the paroxetine group it was nausea (31%) and headache (29%), which reached statistical significance. Regarding efficacy, statistically significant differences were found in the assessments at weeks 1, 2 and 4, patients treated with mirtazapine showing a significant improvement in depressive symptoms.
In a double-blind, randomized pilot study in a Chinese population, Fang et al 11 compared one hundred fifty patients with a history of resistant depressive disorder 11 who were randomly assigned to treatment with venlafaxine, mirtazapine and paroxetine. Fifty-five patients received mirtazapine 45 mg/day and 45 patients received paroxetine 20 mg/day. Remission rates based on HAM-D-17 were 36.4% for mirtazapine and 46.7% for paroxetine. However, no statistically significant differences in mirtazapine and paroxetine efficacy were found. With regard to the tolerability of these drugs, it was observed that only one patient discontinued paroxetine treatment due to adverse effects. This clinical trial determined that patients treated with mirtazapine had more adverse effects than those treated with paroxetine.
On the other hand, in an 8-week, open-label, randomized paroxetine vs. mirtazapine-controlled trial, Kim et al, 2011 12 included 58 patients diagnosed with major depressive disorder with prominent anxiety symptoms. 12 Patients were assigned to fixed dose treatment with mirtazapine (15-30 mg/day) and paroxetine (10-20 mg/day). Efficacy was assessed with the HAM-D-17, HAM-A-17 for anxiety symptoms and tolerability of adverse events.
Week-by-week analysis of the two treatment groups showed that mirtazapine-treated patients improved earlier in depression and anxiety symptoms. There was no difference in the overall frequency of adverse events. Despite this, the most common adverse event in the mirtazapine group was somnolence (n = 8) and in the paroxetine group, it was gastrointestinal discomfort (n = 9).
Discussion
The Diagnostic and Statistical Manual of Mental Disorders-Fourth Edition, Text Revision (American Psychiatric Association, 2000) is the current reference used by mental health professionals and physicians to diagnose mental disorders.
On the other hand, the Hamilton Depression Rating Scale (HAMD) is one of the most commonly-used rating scales that assess symptoms that are most frequently observed in patients with major depression. It is not a diagnostic instrument, but is considered to be a measure of illness severity. Moreover, this scale was primarily developed as measurement tool to assess symptom change in studies of psychopharmacologic agents, and it has been more commonly used in antide-pressant trials. The original scale created by Hamilton contained 17 items that were considered to be most consistent in detecting mood change. Other HAMD items were developed later and added by Hamilton and other researchers. However, the most commonly used form of the HAMD is the original 17-item version.
This review includes results from clinical trials comparing mirtazapine and paroxetine efficacy and tolerability, which fulfilled DSM-IV-TR criteria for major depressive disorder, assessed by HAM-D-17 scale.
When we consider the improvement in anxiety symptoms, mirtazapine presents a better efficacy profile (Kim et al, 2011 12 ) which leads to statistically significant differences in the first week of administration. In the 8-week assessment no statistical differences were found, so it was concluded that both drugs are equally effective when adherence is good.
Wade et al, 1 and Schatzberg et al, 9 excluded patients with a history of suicidality from their clinical trials. On the other hand, Benkert et al, 8 excluded patients with major depressive disorder who verbalized suicidal ideation. Therefore, we conclude that evidence on the efficacy and tolerability of mirtazapine and paroxetine in major depressive disorder with suicidal ideation or behavior is sparse. In addition, systematic reviews and meta-analyses do not collect data on anti-depressant response to mirtazapine and paroxetine in patients with suicidal ideation or behavior. Further studies involving these patients are needed.
The efficacy and tolerability of these drugs in major depressive disorder resistant to antidepressants is not well described in the clinical trials evaluated. Benkert et al, 8 and Szegedi et al, 5 excluded patients if a lack of response to at least 2 adequate antidepressant therapy treatments was observed. On the other hand, Schatzberg et al, 9 and Murphy et al, 10 excluded patients who failed to respond to more than one adequate antidepressant treatment for the current depressive episode. The only clinical trial in patients with resistance to antidepres-sant treatment was performed by Fang et al, 11 in a Chinese population and no statistically significant differences were found between mirtazapine and paroxetine efficacy.
With regard to treatment in elderly depressed patients, the only clinical trial involving this population was performed by Schatzberg et al, 9 who excluded patients with psychotic symptoms. It was concluded that during the first weeks of mirtazapine treatment, antidepressant efficacy was higher when compared to paroxetine and that mirtazapine showed a better tolerability profile in elderly patients. 13 No analysis was performed according to age groups in other studies.
In children or young population no clinical trials have been conducted which evaluate the efficacy of mirtazapine and paroxetine. Moreover, the pharmacological responses to these drugs with regard to gender differences were not analyzed.
We concluded from the evaluated clinical trials that mirtazapine probably has a faster onset of action with respect to paroxetine, which could begin between days 7-14 of drug administration. Moreover, other drugs, such as escitalopram, have shown earlier anti-depressant efficacy in the treatment of early-onset major depressive disorder. 14 This would be advantageous in patients with severe depressive symptoms with a decline in its functionality.
Nevertheless, we conclude that in 6-8 weeks of treatment, mirtazapine would be as effective as paroxetine in the treatment of major depressive disorder.
As an indirect measure of tolerability assessment, we analyzed the discontinuation reasons for both drugs, with particular interest in lost to follow-up due to adverse effects.
When consider this measure, loss rate due to adverse effects does not differ between mirtazapine and paroxetine. Mirtazapine-treated patients more frequently reported somnolence, dizziness and weight gain, while those treated with paroxetine complained of gastrointestinal discomfort and headache.
Limitations
This is a review of published evidence regarding the efficacy and tolerability of mirtazapine versus paroxetine in the treatment of major depressive disorder. The reviewed clinical trials, reviews and meta-analysis included heterogeneous populations with respect to demographic data and other clinical characteristics. Due to the paucity of information, we could not describe differences in antidepressant efficacy of both drugs according to age, sex, socioeconomic and educational level of the patients. Also due to a lack of information has not been able to compare the antidepressant efficacy between paroxetine and mirtazapine in relation to the improvement of psychotic symptoms or suicidal behavior, which can sometimes accompany the depressive symptomatology.
Author Contributions
All authors have worked in all phases of the manuscripts and all authors reviewed and approved of the final manuscript (AGR, CGF, VNO).
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper. All authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. We have no conflicts of interest.
