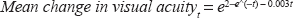Abstract
Objective
To evaluate the cost-effectiveness of monthly and as-needed dosing protocols using ranibizumab or bevacizumab for the treatment of neovascular age-related macular degeneration (AMD), when the treatment costs of severe ocular and systemic adverse events are considered.
Methods
A Markov model was developed to assess the cost effectiveness of each of the following protocols: monthly ranibizumab, monthly bevacizumab, as-needed ranibizumab and as-needed bevacizumab. Direct costs and utilities were assessed from the perspective of a third-party payer or an insurance company. Cost effectiveness was evaluated in 2011 US dollars per quality-adjusted life year (QALY).
Results
Considering the treatment costs of severe medical and ocular adverse events, the cost effectiveness of each protocol is as follows: monthly ranibizumab $63,333/QALY, ranibizumab as needed $18,571/QALY, bevacizumab monthly $2,676/QALY and bevacizumab as needed $3,333/QALY Sensitivity analysis of the treatment costs of medical and ocular adverse events demonstrated minimal impact on relative cost-effectiveness.
Conclusion
At current prices, monthly bevacizumab is the most cost-effective anti-VEGF AMD treatment protocol. Ranibizumab is as cost effective as bevacizumab at a maximum price of $158 per dose.
Introduction
The introduction of anti-vascular endothelial growth factor (anti-VEGF) monoclonal antibodies such as ranibizumab (Lucentis, Genentech, San Francisco, CA) and bevacizumab (Avastin, Genentech, San Francisco, CA) represented a significant milestone in the treatment of neovascular (wet) age-related macular degeneration (AMD). Ranibizumab is the Fab fragment of a humanized recombinant monoclonal antibody that targets vascular endothelial growth factor A (VEGF-A), and bevacizumab is a humanized recombinant monoclonal antibody that also targets VEGF-A. Prior to ranibizumab and bevacizumab, AMD was treated with either pegaptanib (Macugen, Eyetech Pharmaceuticals, New York NY, an anti-VEGF aptamer) or photodynamic therapy (PDT), both of which resulted in the reduction of the rate of vision loss.1–4 Improvement of the vision of a substantial portion of wet AMD patients was demonstrated for the first time in the 2006 ANCHOR and MARINA trials. The MARINA trial enrolled 716 patients, of which 96.6% were white and 64.8% were female. The MARINA trial demonstrated that, after two years of treatment with ranibizumab, one fourth to one third of patients had gained at least 15 letters and at least 90% of patients lost fewer than 15 letters. 5 The ANCHOR trial commenced with 423 patients, of which 97.6% were white and 49.9% were female. The ANCHOR trial similarly showed that 35.7%-40.3% of patients treated with bevacizumab gained 15 or more letters, while at least 94% of patients lost fewer than 15 letters. The fact that AMD is the leading cause of irreversible vision loss in the United States among patients over 50 years of age6–11 and that more than 7 million Americans have AMD, 12 made ranibizumab immediately clinically relevant. But the benefits of ranibizumab have been associated with significant cost. 13 One cross-sectional study showed that in 2006, the year that the United States Food and Drug Administration (FDA) approved ranibizumab, Medicare payments for eye care doubled relative to 1994, with most of the increase attributable to ranibizumab. 14
Concerns about the cost of ranibizumab led to many attempts to seek a lower-cost alternative. In 2006, the same year that he was lead author of the MARINA trial, Dr. Rosenfeld authored a study in which systemic bevacizumab was investigated as an alternative to ranibizumab. 16 That year, he also published an editorial entitled: “Intravitreal Avastin: the low cost alternative to Lucentis?”. This focus on the cost of ranibizumab has resulted in several studies evaluating its cost-effectiveness, both within and outside the United States. Hurley et al reported that if the care-giver costs (approximately $50,000 annually for legally blind patients) were factored into the cost-benefit analysis, ranibizumab was cost-saving relative to no treatment because the reduced rate of poor vision associated with ranibizumab would reduce the costs of caring for low-vision patients. However, ignoring care-giver costs, ranibizumab was cost-effective if it was less than $1,000 a dose. 15 Brown et al, by assuming disease in both eyes demonstrated that ranibizumab was cost effective relative to no treatment. 16 Comparisons of ranibizumab to PDT and pegaptanib sodium show that ranibizumab is relatively more cost effective, mostly due to its improved efficacy. 17
Pressure to migrate to cheaper alternatives than ranibizumab arises from the fact that the segment of society at greatest risk for AMD–-the elderly–-is rapidly increasing, one study projected that the elderly population would increase by 50% in 2020. 9 This increase would result in 1.82 million neovascular AMD cases and would require annual expenditure of $25-$42 billion (depending on monthly or as-needed dosing schedules) on ranibizumab. In other words 1.0%-1.7% of the United States 2009 total healthcare expenditures would have to be spent on a single drug. 18 These expenditures would likely be borne by Medicare (given the demographics of the typical AMD patient). Considering the weak state of the United States economy, the large and politically charged national debts, and the fact that Medicare and Medicaid consume approximately 20% of the federal budget, the government has a definite incentive to control health care expenditure. 19 These economic realities have heightened the desire to understand the economics of ranibizumab, which costs approximately 40 times more than bevacizumab. 20
Previous cost-effectiveness studies that attempted to compare bevacizumab to ranibizumab were limited by the absence of large clinical trials comparing both medications. Raftery et al reported that, from a cost-effectiveness perspective, ranibizumab would have to be 2.5 times more clinically effective than bevacizumab to justify the forty-fold disparity in price. 21 Using patient data from the San Diego Veterans Affairs Hospital, Patel et al concluded that bevacizumab was more cost effective than ranibizumab and suggested that it would be equivalent at a price of $44 a dose. 22 Subsequently the National Eye Institute sponsored the Comparison of Age-Related Macular Degeneration Treatments Trial (CATT) to directly compare ranibizumab and bevacizumab. The CATT trial started with 1,185 patients, of which 98.6% were white and 61.8% were female. The CATT trial concluded that bevacizumab and ranibizumab were equivalently effective in improving vision. However, the CATT study also reported a statistically significant increase in the number of serious systemic adverse events (mostly hospitalizations) in patients treated with bevacizumab (24.5% vs. 19.0%, P = 0.04), with excess adverse events broadly distributed in areas that were not identified in previous studies as areas of concern. 20 It is imperative to note that Dr. Philip Rosenfeld published an editorial, in the same issue of the New England Journal of Medicine that published the CATT study, stating that the CATT trial was insufficiently powered to detect differences in drug-related adverse events. 23 Thus it is unclear whether or not the differences in severe systemic adverse events between both drugs are real or not. It has been reported that patients receiving intravitreal injections of bevacizumab demonstrated a statistically significant decrease in their plasma VEGF levels that was not seen in patients treated with intravitreal ranibizumab. 24 Systemic suppression of VEGF by intravitreal bevacizumab was also noted by Qian et al. 25 Reasons for the increased systemic absorption of bevacizumab may include the persistence of the Fc fragment on bevacizumab, which provides a means for immune cells to deliver the drug to the systemic circulation 26 and the fact that bevacizumab has a 21-day half-life in plasma in contrast to ranibizumab which has a 0.5 day half-life. 27
In this paper, the authors attempt a cost-effectiveness analysis comparing bevacizumab to ranibizumab that incorporates the costs of both systemic and ocular side effects as presented by the CATT trial. 20
Materials and Methods
A Markov model was developed, using decision analysis software TreeAge Pro 2011 (TreeAge Software Inc. Williamstown, MA) to simulate the progression and treatment of neovascular AMD in a cohort of 65-year-old patients in the United States according to the treatment protocols reported in the CATT study over a ten-year period. 20 Four treatment groups were compared to each other: monthly treatments with bevacizumab, monthly treatments with ranibizumab, treatment with ranibizumab on an as-needed basis, and treatment with bevacizumab on an as-needed basis. The frequency of the treatments on an as-needed basis was assumed to be identical to that reported in the CATT trial. The four treatment protocols were compared in terms of their relative cost-effectiveness. In the context of this study, cost-effectiveness is defined as the ratio of discounted direct costs from the perspective of a third party payer or insurance company, denominated in 2011 US dollars, to the discounted treatment-derived utilities, denominated in quality-adjusted life-years (QALYs). Both quantities are discounted at a 3 percent per annum.
For each of the treatment protocols five vision-related health states were defined: better or equal to 20/20; 20/25 to 20/40; 20/50 to 20/80; 20/100 to 20/200; and worse than 20/200. Age-specific mortality rates were applied to the hypothetical cohort. 28 The initial distribution of patients among the vision-related health states was identical to the distribution of patients in the CATT study. 20 The vision-related health states were assumed to be associated with the patient's better-seeing eye. This assumption enabled utilities associated with the corresponding levels of vision to be immediately assigned to the patients.15,29–31 The transitions to the end of life were made using age-specific mortality rates derived from the 2007 United States Life Table of the National Vital Statistics System of the Center for Disease Control's (CDC). 28 Similar to Hurley et al, transition between the various vision-related health states were modeled as 15 letter gains, no change in vision, 15 letter losses or 30 letter losses. 15 Letters refer to the number of letters read on the Early Treatment Diabetic Retinopathy Study (EDTRS) visual chart. The proportion of letters lost or gained depended on the projected mean visual acuity of the cohort at a particular point in time.
Because the CATT study has only released the results of one year of follow-up, the two-years of follow-up data from the MARINA trial were used to model the gains in vision for the monthly bevacizumab treatment groups. 5 For the purposes of the 10 year simulation the two year visual acuity results of the MARINA trial were modeled with the following exponential equation (Fig. 1):
Graph of the mean change in visual acuity, measured in letters on the ETDRS chart, over a two-year period for patients treated with ranibizumab during the MARINA study. 5
The variable t refers to time measured in three-month intervals and e refers to the mathematical constant 2.7183. The exponential model appears to fit the MARINA data relatively well. Regression of the modeled data against the MARINA data showed close correlation with P value 4.87 x 10-5 and an adjusted R square of 90.6%. This model also predicts reduced mean gains in vision with the progression of time. These losses may be attributable to factors such as new neovascularization, persistent neovascularization, tachyphylaxis, treatment related complication such as endophthalmitis, and atrophic scar formation. 32 To model the gains in vision for the other treatment groups, it was assumed that the relative ratios of gains in vision reported by the CATT study would remain stable over time. The projections for monthly bevacizumab were therefore scaled by the relative ratios calculated from the CATT study. 20
Drug cost data was derived from the CATT study. The cost of ranibizumab is approximately $2,000 a dose and that of bevacizumab approximately $50 a dose. 20 To determine the costs of complications, all complications–-both ocular and systemic–-that were described as severe, life-threatening or disabling in the supplementary index of the CATT study were compiled and mapped onto corresponding DRG codes. The cost per hospitalization of each of these DRG codes was derived from the Healthcare Cost and Utilization Project (HCUP) of the Agency for Healthcare Research and Quality (AHRQ) of the US Department of Health and Human Services. 33 The annual weighted average costs of the various complications were calculated. The weighted average costs of the systemic and ocular complications that were reported for each arm of the CATT study are as follows: monthly ranibizumab administration required an average of $934 per patient per year to treat severe systemic and ocular adverse events; monthly bevacizumab administration required an average of $1,657 per patient per year to treat severe systemic and ocular adverse events; ranibizumab administered on an as-needed basis required $1,672 per patient per year to treat severe systemic and ocular adverse events; and bevacizumab administered on an as-needed basis required $2,350 per patient per year to treat severe systemic and ocular adverse events. It is important to emphasize that this analysis only captures the direct costs associated with the treatment of the adverse events from the perspective of a third party payer. This study does not account for costs such as pain and suffering of the patient and his/her family, disruption of the lives of the patient and his/her family as well as other financial costs such as lawsuits, lost productivity, and the foregone wages of the patient's care-takers. These other costs are not included in the analysis, despite being paramount to physicians and their patients, because they are extremely difficult to compute or model with any degree of accuracy. All costs and utilities used in this analysis were discounted at a rate of 3% per annum.
Results
Impact of age and treatment horizon on cost-effectiveness
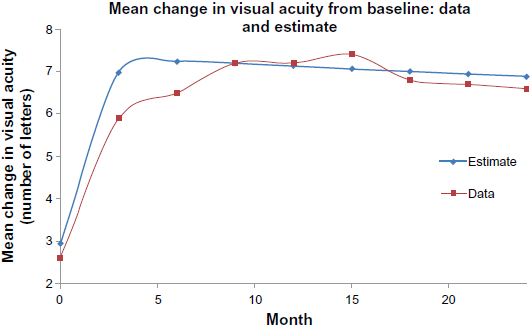
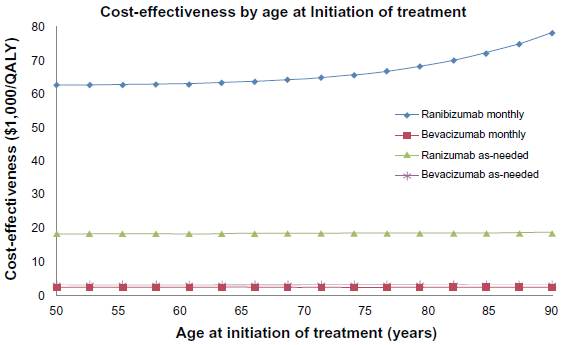
Accounting for the cost of treating severe medical and ophthalmic complications, the cost effectiveness ratios of associated with four sixty-five year-old cohorts undergoing a decade of treatment are: monthly ranibizumab $63,333/QALY, ranibizumab as-needed $18,571/QALY, bevacizumab monthly $2,676/QALY and bevacizumab as-needed $3,333/QALY. The generally accepted threshold of cost-effectiveness is $50,000 per QALY.34–36 As the average age of the starting cohort increased, the cost per QALY increases for all treatment protocols, implying reduced cost-effectiveness as the age of the treatment protocol increased. (Fig. 2). Thus, the cost-effectiveness of bevacizumab improved as the average age of the starting cohort decreased (Fig. 2). Younger cohorts have, on average, lower mortality and thus accrue more years of good vision, but AMD is typically not a disease of younger patients. Nevertheless, a hypothetical cohort as young as 50 years-old was insufficient to improve the cost-effectiveness of monthly ranibizumab to the $50,000/QALY threshold. Similarly the cost per QALY decreases as the time horizon of the treatment is increased, implying improved cost effectiveness as the treatment horizon increases (Fig. 3). Longer treatment horizons means more years of good vision, but even a twenty-five year long life expectancy is not enough to make monthly ranibizumab's cost-effectiveness less than $50,000/QALY. Generally, assuming reasonable starting ages and treatment time horizons, monthly ranibizumab, priced at $2,000 a dose, does not meet the $50,000/QALY benchmark.
Graph of Cost effectiveness ratios ($1,000/QALY) of the following treatment strategies: monthly ranibizumab, monthly bevacizumab, ranibizumab as-needed and bevacizumab as-needed, as the average age of the initial cohort is varied from 50 to 90 years of age.
Graph of Cost effectiveness ratios ($1,000/QALY) of the following treatment strategies: monthly ranibizumab, monthly bevacizumab, ranibizumab as-needed and bevacizumab as-needed, as the duration of treatment is varied from 5 to 25 years.
Impact of the rate of adverse events on cost effectiveness
The relatively lower rate of severe medical adverse events associated with ranibizumab improves the cost-effectiveness of the monthly ranibizumab protocol as the costs of treating adverse events increases (Fig. 4). The cost-effectiveness of monthly ranibizumab matches that of as-needed ranibizumab when the cost of treating severe adverse events increases by a multiple of 50.1, an unlikely and hypothetical scenario. Ranibizumab as-needed was as cost effective as bevacizumab as-needed when the cost of treating adverse events was increased by a factor of 19.1 and as cost-effective as monthly ranibizumab when the adverse event treatment costs were increased by a multiple of 35.2. These scenarios are similarly hypothetical and highly unlikely. Monthly bevacizumab is as cost-effective as monthly ranibizumab when the cost of complications increases by a factor of 71.1.
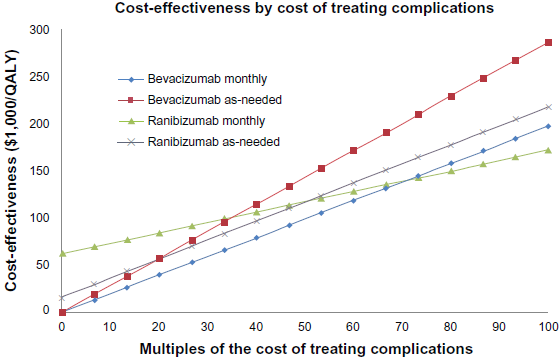
Graph of Cost effectiveness ratios ($1,000/QALY) of the following treatment strategies: monthly ranibizumab, monthly bevacizumab, ranibi-zumab as-needed and bevacizumab as-needed, as the cost of treatment-related severe systemic and ocular adverse events increases.
Impact of drug efficacy on cost-effectiveness
By varying the efficacy of ranibizumab, its cost effectiveness can be improved. A 27.4% increase in the efficacy of ranibizumab relative to bevacizumab would improve the cost-effectiveness of monthly ranibizumab treatments to $50,000 (Fig. 5). To make as-needed ranibizumab as cost-effective as monthly bevacizumab a 553.3% increase in efficacy would be required. For as-needed ranibizumab to match the cost-effectiveness of as-needed bevacizumab, a 692.3% increase in efficacy would be required. Monthly ranibizumab would require even larger gains in efficacy to attain the same cost-effectiveness as the bevacizumab regimens. A 1,905.8% gain in efficacy would make it as cost-effective as as-needed bevacizumab and a 2,376.3% gain would make it as cost-effective as monthly bevacizumab.
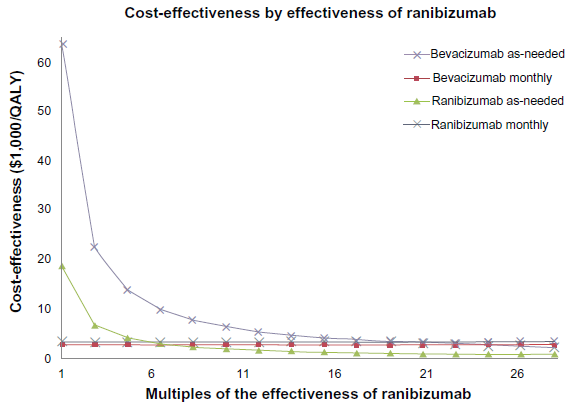
Graph of Cost effectiveness ratios ($/QALY) of the following treatment strategies: monthly ranibizumab, monthly bevacizumab, ranibizumab as-needed and bevacizumab as needed, as the relative efficacy of ranibizumab is increased.
Impact of medication costs on cost-effectiveness
The cost-effectiveness of ranibizumab can also be improved by reducing its price (Fig. 6). For the monthly ranibizumab protocol to meet the $50,000 cost-effectiveness threshold, ranibizumab would have to be priced at $1,560 a dose (78.0% of current price). For the monthly ranibizumab treatments to match the cost effectiveness of monthly bevacizumab, ranibizumab would have to be priced at $50.42 a dose (a 0.8% premium over ranibizumab). For monthly ranibizumab to rival the cost effectiveness of bevacizumab as-needed, ranibizumab would have to be priced at $71 a dose (a 42.7% premium over bevacizumab). The ranibizumab as-needed protocol is as cost-effective as the monthly bevacizumab protocol when ranibizumab is priced at $80 (a 59.1% premium over bevacizumab). But the cost-effectiveness of the ranibizumab as-needed protocol matches that of the bevacizumab as-needed protocol when ranibizumab is priced at $158 a dose (a 216.7% premium over bevacizumab).
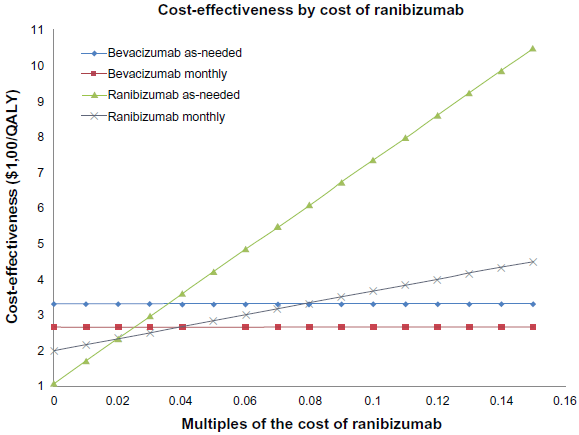
Graph of Cost effectiveness ratios ($/QALY) of the following treatment strategies: monthly ranibizumab, monthly bevacizumab, ranibizumab as-needed and bevacizumab as needed, as the costs of associated medical and ophthalmic complications are varied.
Discussion
The results of this study demonstrate that at its current price of $2,000 ranibizumab is not cost effective. Hurley et al similarly reported that above $1,000, ranibizumab was not cost effective. 15 When the costs of systemic and ophthalmic complications are factored into the analysis, the possibly lower rate of severe systemic complications associated with ranibizumab results in greater cost-effectiveness relative to bevacizumab only when the costs of the complications for both drugs are at least nineteen-fold higher than the rates reported in the CATT study. Assuming limited cost inflation, ranibizumab is less cost effective than bevacizumab unless the hypothetical situation that the rate of serious complications increases nineteen-fold. Stated differently, in spite of a forty-fold difference in price, ranibizumab and bevacizumab will be equivalently cost-effective if a serious medical adverse event occurred, on average, after every 2.9 injections of ranibizumab and every 2.1 injections of bevacizumab. In this hypothetical scenario, ranibizumab would still not meet the generally accepted threshold for cost-effectiveness (≤$50,000 per QALY),34–36 but the nineteen-fold increase in medical complications, would change the cost-benefit ratio of the medication to such an extent as to make it clinically unusable. Stated plainly if any monthly medication caused a heart attack (for example) after 2-3 doses, no one would use it.
By increasing treatment time-horizons or treating younger patients, the cost-effectiveness of ranibizumab can be improved, but not to the level of $50,000 per QALY. Greater efficacy may improve the cost-effectiveness of ranibizumab. To meet the $50,000/QALY benchmark, a conceivable 27.4% increased efficacy realized over 10 years would be required. In order to match the cost-effectiveness of bevacizumab, a minimum of a 553.3% increase in the efficacy of ranibizumab would have to be realized. Thus under any realistic scenario, when the costs of complications are considered, ranibizumab–-while priced at $2,000–-is unlikely to be as cost-effective as bevacizumab. In reality, however, considerations other than cost-effectiveness factor into medical decision making. For example the Veterans Affairs Hospitals (VA) suspended the use of intraocular bevacizumab throughout the United States after five of its patients in Florida developed endophthalmitis from contaminated bevacizumab from a single pharmacy. One of these patients has filed a $4 million lawsuit against the VA. This cluster of infections, also prompted a national FDA advisory regarding intraocular bevacizumab.37,38
This analysis suggests that the relative increase in systemic complications associated with bevacizumab means that ranibizumab can be priced at most 216.7% higher than bevacizumab ($156 vs. $50) and still be as cost-effective as bevacizumab. Furthermore, from a national perspective, the use of bevacizumab instead of ranibizumab would reduce the expected outlay of $25-$42 billion to a more manageable $0.7-$1.1 billion to treat the anticipated number of wet AMD patients in 2020.
An important limitation of this study, as well as most cost-effectiveness studies, is its dependence on the variables included in the analysis. For example, if adverse event-related lawsuits become the major factor deciding whether or not it is cost-effective to use bevacizumab, this study would not have captured that information. Another important limitation of this study is the uncertainty about the data used in the analysis. The relative gains in vision after one year of treatment reported in the CATT study were assumed to be stable over time, and were extended to the ten-year analysis period of the study. It is conceivable that the visual gains of the treatment groups of the CATT study may either converge or diverge with time. Only future studies can determine what the long-term changes in the visual function of treated patients will be. Furthermore, the causal relationship between the severe medical complications reported in the CATT study and ranibizumab and bevacizumab has not been proven. The costs associated with treating medical complications in both “as-needed” protocols were greater than the costs associated with treating complications arising in the corresponding monthly treatment protocols. With a causal relationship one would expect more complications with increased exposure to a causative drug and thus higher complication-related costs associated with the monthly treatment protocols, not the as-needed protocols. Nevertheless, where possible, the uncertainties were mitigated with sensitivity analysis.
In conclusion, the most cost-effective anti-VEGF treatment protocol at this time appears to be monthly bevacizumab treatments with a cost-effectiveness ratio of approximately $2,600/QALY. Ranibizumab, at the $2,000 per dose price point, does not cross the $50,000/QALY threshold for cost-effectiveness. If ranibizumab, at $2,000 a dose, is found to be at least 27.4% more efficacious than bevacizumab, then it will meet the $50,000/QALY cost-effectiveness benchmark, but will not be as cost-effective as bevacizumab. Ranibizumab is only as cost-effective as bevacizumab if it costs at most $158, when the costs of treating systemic and ocular complications arising during treatments are considered.
This work was funded in part by grant T15LM007092 from the National Library of Medicine, National institutes of Health, the Leir Foundation and Newman's Own Foundation.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributor-ship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.