Abstract
The newest antidepressant, desvenlafaxine (DVS) was approved by the FDA in early 2008 and since then, has been available in the market for general use. DVS is dual acting, a serotonin-norepinepherine reuptake inhibitor (SNRI). Like its parent compound, venlafaxine (VEN), DVS inhibits the neuronal re-uptake of both serotonin and norepinepherine while having minimal affinity for muscarinic cholinergic, H1-histaminergic, alpha1-adrenergic and opioid receptors. DVS is moderately effective for MDD at doses ranging from 100–400 milligrams per day. Its possible advantage is its lower risk of drug-drug interaction. There is no extant evidence that it is especially effective over and above the other existing antidepressants. Its niche will be defined by time but one speculation, given preliminary but un-replicated proof of its efficacy in vasomotor instability in peri-menopause, is that it may have a future utility for depressed women entering menopause.
Keywords
Introduction
The serotonin reuptake inhibitors have proven very useful in the treatment of various psychiatric disorders, notably major depression. As a class of psychotropic agents, they have been versatile in their indications and relatively safe not only in the incidence of side effects at therapeutic doses but also in cases of overdoses. The development of new and effective compounds to treat major depression remains an important arm of the pharmaceutical industry driven not only by the high incidence of major depression (MDD) and its attendant co-morbidities but also the fact that a certain percentage of the majorly depressed population will not respond adequately to monotherapy or to only one trial of a class of compounds. Complicating this picture is the emergence of more data suggesting that there are demonstrable structural changes in the brain on imaging studies of patients who suffer from long term depression.1,2
It is within this picture of a greater understanding of how major depression impacts its victims over their lifetimes, and other reasons of course not the least of which is the fact that the antidepressant market is a huge one for pharmaceutical companies, that new antidepressant compounds continue to be developed and put on clinical trials. The newest one, desvenlafaxine (DVS) was approved by the FDA in early 2008 and since then, has been available in the market for general use. This review considers basic pharmacokinetics of desvenlafaxine (DVS), the main clinical trials of its efficacy in major depression and the emergent experience so far as to its advantages as an antidepressant intervention with particular attention to its possible clinical niche in the treatment of MDD or possibly, other disorders.
DVS and its Action on Transporter Systems
DVS is a dual acting, a serotonin-norepinepherine reuptake inhibitor (SNRI). Like its parent compound, venlafaxine (VEN), DVS inhibits the neuronal re-uptake of both serotonin and norepinepherine while having minimal affinity for muscarinic cholinergic, H1-histaminergic, alpha1-adrenergic and opioid receptors. 3 The differences between desvenlafaxine and its parent compound venlafaxine can be seen in Table 1. 4
Difference between venlafaxine (VEN) and desvenlafaxine (DES).
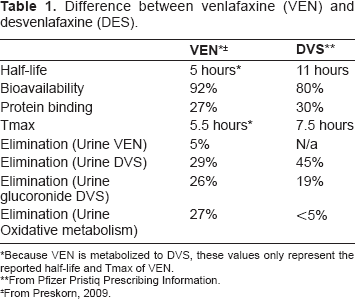
Because VEN is metabolized to DVS, these values only represent the reported half-life and Tmax of VEN.
From Pfizer Pristiq Prescribing Information.
From Preskorn, 2009.
Metabolism
DVS is metabolized through glucuronidation and to a much lesser extent (<5%) through cytochrome p450 (CYP3A4)-mediated oxidation. DVS has minimal interactions with the isoenzyme CYP2D6, and as such, it would likely have fewer drug-drug interactions with other pharmacologically active compounds metabolized through this isoenzyme. 5 Preskorn and his group 6 studied the effects of DVS on the CYP2D6 by comparing extensive versus poor metabolizers. Seven extensive metabolizers, determined through genotyping, and 7 poor metabolizers randomly received single doses of 75 milligram VEN or 100 milligrams DVS. Plasma concentration versus time (AUC) and peak plasma concentration (Cmax) of VEN was significantly greater for poor metabolizers. In contrast, there are no significant differences between poor and extensive metabolizers in these aforementioned drug concentration measures for those who received DVS.
In another review, Preskorn et al 7 surveyed 5 studies investigating the interaction between DVS and CYP450 2D6. They looked at the concomitant administration of DVS–-and comparatively, the concomitant administration of paroxetine and duloxetine–-as they separately interacted with the probe, the tricyclic desipramine, on the CYP450 2D6 enzyme system. Mean increases in the desipramine AUC was the measure of the level of activity for all the three aforementioned drugs studied as they competes with the desipramine in the enzyme system. The AUC for desipramine increased significantly when co-administered with paroxetine and duloxetine compared to DVS, suggesting that DVS by comparison is more inert at the CYP450 2D6 system.
DVS has a linear and dose-proportional linear pharmacokinetic profile when considered within the 100–600 milligrams dose range. Its absorption is not affected by food, has low protein binding in the plasma and has a half-life of approximately 11 hours. 5 There were differences in AUC, mean half-life and clearance in individuals with hepatic impairment as shown in Table 2. 4
DVS in hepatic impairment. ”
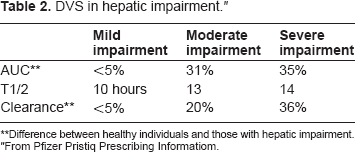
Difference between healthy individuals and those with hepatic impairment.
From Pfizer Pristiq Prescribing Informatiom.
The clinician must individualize DVS prescribing depending on the needs of the patient. Lastly, preclinical investigation of DVS showed good kinetic properties in plasma and brain tissues with rapid brain penetration. 8
Efficacy in Major Depressive Disorder
Patrice Boyer et al 9 studied the efficacy of DVS in outpatient subjects diagnosed with MDD (diagnosed using Mini-International Neuropsychitaric Interview) in several European countries in a multi-center study. In this trial, the primary investigator did not receive support but the clinical trial and analysis were supported by Wyeth Research, Pennsylvania. The outcome measures were the 17 item Hamilton Depression Rating Scale (HAM-D 17) and the Clinical Global Impression Scale (CGI). Secondary measures were the Montgomery Asberg Depression Rating Scale, Covi Anxiety Scale, Sheehan Disability Scale and the WHO 5-item well-being index. The design was an 8-week, randomized, double-blind, placebo controlled, parallel group design. The subjects were randomly assigned to 50 milligram dose of DVS, 100 milligrams or placebo. One hundred sixty-six subjects were randomly assigned to the 50 milligrams, 158 subjects to the 100 milligram DVS dose and 161 to placebo. Twenty seven patients (22.9%) in the DVS groups discontinued mostly because of adverse effects, reported as mild to moderate in severity, versus 13 (8.1%) who received placebo and discontinued mostly because of lack of efficacy. Eighteen patients had changes in blood pressure–-four in the placebo group, 8 in the 50-mg group, and 6 in the DVS 100-mg group. Statistically significant HAM-D 17 (Last observation carried forward or LOCF) compared to placebo became apparent at week 4 for DVS 100 milligrams and at week 6 for DVS 50 milligrams. The final evaluation LOCF CGI scores showed 73% of the 50 milligram and 100 milligram DVS groups responding at the 1 or 2 levels (very much improved or much improved) compared with 53% of the placebo group. Other studies that are similarly designed using MDD subjects with varying DVS dosages ranging from 100–400 milligrams showed efficacy for DVS when the HAMD 17 is used as efficacy measure.10–12 Daniel Lieberman et al 13 reviewed three studies of double blind, placebo controlled trial of DVS in the United States and abroad. He noted that there is a high placebo response, as is true in the Boyer study. To further analyze this phenomenon, a pooled analysis of two of these three studies was performed. The MDD criteria in these studies used DSM IV criteria with outcome measures using the HAM-D 17, CGI, MADRS and a Raskin Depression Scale score greater than the Covi Anxiety Score. In one of the studies, venlafaxine ER was used as an active control. The number of all pooled subjects totaled 713 patients (226 desvenlafaxine, 127 venlafaxine Extended Release 75–150 mg/day, 115 venlafaxine ER 150–225 mg/day, and 245 placebo). The analysis used a mixed effect model for repeated measures (MMRM). Using the latter statistic, DVS separated from placebo at week three with the significance maintained throughout the 8 week period of these studies. Venlafaxine groups showed significant difference from placebo at week 8 as well. They conclude that though individual studies may have high placebo response affecting the significance of the results when statistically analyzed, pooling the results may achieve a clearer picture of the effectiveness of a drug provided the individual studies were relatively similar. As a result, they concluded that DVS is a safe and efficacious treatment for MDD.
Post-Approval Experience and the Possible Niche(s) of DVS
In several cases, psychotropic agents would be approved by the FDA for one indication and only later in wide clinical post-marketing use would these drugs be found useful for other conditions. For example as a class, many of the serotonin reuptake inhibitors originally approved for MDD were later found to be efficacious for certain anxiety disorders. Several second-generation antipsychotic agents have now added indications for bipolar disorder. Regarding DVS, a medline search (April 2009) reveals no results for controlled double-blind studies commencing after DVS came out in the market in MDD–-though it is possible that these studies may be ongoing. Post-marketing standardized trials are important sources of knowledge especially if they draw subjects from a wide variety of clinical settings and not just from those usually drawn from research centers. Clinician feedback, specifically from those using post-FDA approval drugs “in the trenches” of actual patients as seen in individual practices often provide new data about previously un-highlighted beneficial and unwanted effects. Thus far, there is no evidence that DVS presents a significant advance beyond the already long list of available drugs for MDD. Sopko 5 notes that despite the availability of numerous anti-depressants, including those with both serotonin and norepinephrine reuptake inhibitory mechanisms such as venlafaxine, there have been no significant increases in the remission rates for major depression.
A recent study used DVS in the treatment of vasomotor instability during menopause. The trial was in part motivated by the need to find non-hormonal interventions for this symptom, a potentially significant and uncomfortable accompaniment of menopause. Serotonin and norepinephrine systems have been implicated in menopausal vasomotor instability. 14 Within these contexts, Archer et al 15 published the results of their 26-week study of more than 500 menopausal women with 50 or more hot flushes per week, randomly assigning them to DVS 100–150 milligrams or placebo. A statistically significant decline from baseline of the number of hot flushes occurred for the DVS group.
The occurrence of MDD within the peri-menopausal period has long been the subject of clinical discussion and recent literature. A study done among 460 pre-menopausal subjects in Boston, Massachusetts with no previous history of lifetime MDD showed that women were twice as likely to become significantly depressed, using the Center for Epidemiologic Studies Depression Scale as a measure, while entering peri-menopause compared to those who have not yet entered it. 16 Women with previous histories of MDD may be more likely to develop peri-menopausal depressive symptoms and may have an earlier decline in ovarian function as measured by follicle-stimulating and luteinizing hormone levels. 17 Mirtazapine, another SNRI, was tried in an open-label trial among menopausal women unresponsive to estrogen replacement therapy. Fourteen out of 16 completers responded well to mirtazapine with the HAM-D 17, the CGI and the Beck depression Inventory as outcome measures. 18
The question then begs asking: could the next trial for DVS be among peri-menopausal women with both depressive symptoms and vasomotor instability? And if efficacious in these co-morbid states, could this be a niche for DVS? We can only wait for future clinical trials specifically those that would compare it head to head with other SNRIs such as mirtazapine and venlafaxine and of course, placebo.
Kamath and Handrata 19 in their review sums up DVS, the advantages being simplicity in dosing, lower risk of drug-drug interaction and simple metabolism; the disadvantages are only moderate efficacy and possible discontinuation syndrome though they offer no controlled studies for the latter disadvantage. For DVS, nausea is a common adverse reaction as well as dizziness, hyperhidrosis, constipation and decreased appetite (Wyeth, op cit).
Considering the recent history of other previously approved SNRIs, duloxetine has found use in diabetic neuropathy20,21 and it could be argued that it is carving its niche within that indication especially when co-morbid MDD exists along with the neuropathy. Venlafaxine has efficacy for generalized anxiety disorder. 22 These multiple indications have extended these drugs’ competitiveness in an arena where many competing anti-depressants exist without any clear advantage of one over the others when considering their efficacies for MDD alone. It is also worth adding that clinical practice has attested to the complexity of MDD as it exists in individual patients. Often, for unclear mechanisms, one SSRI would be effective in a particular patient while another would not. The same could be true of SNRIs and perhaps merely having yet another choice may be beneficial in case there are patients who would idiosyncratically respond to it alone.
Conclusions
Desvenlafaxine, a serotonin and norepinephrine reuptake inhibitor metabolite of venlafaxine, is a moderately effective treatment for MDD. Thus far, its efficacy is comparable to with no demonstrable higher rate of efficacy over other available antidepressants with no demonstrable higher rate of efficacy over. Its possible advantage is its lower risk of drug-drug interaction. Though it has been tried successfully in subjects with vasomotor symptoms associated with menopause, these studies need replicating. Its specific niche in MDD and other psychiatric disorders remains to be defined.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
