Abstract
Exercise-induced bronchoconstriction (EIB) can be a troublesome problem in asthmatic children and in some children without asthma. Self-reported exercise related symptoms should be verified formally. EIB is mediated by changes in temperature and humidity in the airway and is secondary to release of several mediators of the bronchoconstriction including leukotrienes. Montelukast as a leukotriene receptor antagonist offers protection against but does not completely ameliorate EIB. Regular use does not appear to lead to tolerance. Nightmares, abdominal pain, fever, nausea and aggressiveness are commonly described side effects; however, the overall safety profile of montelukast is good and does not change with long term use. There are individual differences in response to montelukast for protection against EIB. Bronchoconstriction triggered by exercise responds to cessation of exercise in its natural course. Due to its pharamacokinetic profile, montelukast may be more useful as a prophylaxis than to relieve symptoms.
Introduction
National 1 and international 2 guidelines stress the lack of a clear definition of asthma. A descriptive definition is offered by the Global Initiative for Asthma (GINA): 2
Asthma is a chronic inflammatory disorder of the airways in which many cells and cellular elements play a role. The chronic inflammation is associated with airway hyperresponsiveness (AHR) that leads to recurrent episodes of wheezing, breath-lessness, chest tightness and cough particularly at night or in the early morning. These symptoms are usually associated with widespread but variable airflow limitation within the lung that is at least partly reversible either spontaneously or with treatment. 2
This definition highlights the two underlying cardinal pathophysiological factors of asthma. These include airway hyperresposiveness leading to intermittent airflow limitation and airway inflammation. Breathlessness and wheezing secondary to airflow limitation is experienced by asthmatic subjects in response to various stimuli including exercise and is an indication of AHR. The Greek physician Aretaeus offers the first description: “If from running, and exercise, and labor of any kind a difficulty of breathing follows it is termed asthma”. 3
Exercise-induced asthma (EIA) and exercise-induced bronchoconstriction (EIB) are terms that are often used interchangeably in literature. The term exercise-induced asthma (EIA) is imperfect as it implies that exercise causes asthma, rather than being a trigger that exacerbates it. EIB is a more accurate description. 4 EIB is common in persons with clinical signs and symptoms of asthma, but it also occurs in persons who do not have clinical signs and symptoms. A 10%-15% drop in forced expiratory volume in 1 second (FEV1) from pre-exercise levels after 20-30 minutes of exercise is defined as EIB. 5 For the purposes of this review, EIA and EIB will be used interchangeably.
Diagnosis of EIB
Self-reported symptoms of dyspnoea, increased effort or work to breathe, chest tightness, shortness of breath, wheezing or cough with exercise are suggestive of EIB. 6 This needs to be confirmed by objective testing as not all exercise related breath-lessness is EIB and clinical signs and symptoms are not good predictors of bronchoconstriciton.6,7 Only 8 out of 52 (15.4%) children referred for poorly controlled EIA fulfilled criteria for EIA when formally tested in the respiratory laboratory, 8 and only 8 out of 39 (20.5%) patients evaluated by cardiopulmonary exercise testing in adults had EIB. 9 It is important to be aware that exercise-induced symptoms could mask a variety of other diseases or conditions, such as poor physical fitness, vocal cord dysfunction (VCD), exercise-induced paradoxical arytenoid motion (EPAM), exercise-induced laryngomalacia (EIL), exercise -induced hyperventilation and hyper-trophic cardiomyopathy or arrhythmias with long Q-wave to T-wave interval QT. 10 The timing of the appearance of breathlessness in relation to exercise can provide some clue about an alternative diagnosis as the nadir of EIB happens 5 to 10 minutes after vigorous and sustained exercise, 11 as opposed to the shortness of breath experienced due to a lack of fitness or pre - existing airflow limitation where shortness of breath develops during exercise but improves afterwards.
The diagnosis of EIB is confirmed by changes in pulmonary function after bronchial provocation. Pragmatically, the provocation test used most commonly is exercise. A fall in FEV1 of 10%–-15% or more after an exercise challenge is usually considered suggestive. Exercise protocols based on achievement of specific physiological parameters such as heart rate are usually used. Treadmills or bicycle ergometers can be used to achieve exercise endpoints. The exercise load needs to be high as assessed via heart rate, and the test should be standardized with respect to environment temperature and humidity.
This approach to the diagnosis of EIB may need to be modified in athletes. Subjects may be required to be tested in their usual environment (sport specific) at times. Surrogate provocation tests have been used and are accepted by sports bodies in diagnosis of exercise induced asthma. There may be advantages to using surrogate tests, especially when evaluating children and elderly or obese adults.
Hyperventilation with dry cold air (eucapnic hyperventilation) is also used instead of exercise as a stimulus for EIB. This is a physically demanding test and requires the athlete to perform hyperpnoea by inhaling air containing 5% carbon dioxide at ventilation equivalent to 30 times the baseline FEV1. Hyperosmolar aerosols like mannitol and hypertonic saline are other indirect stimuli for bronchial provocation tests. Eucapnic voluntary hyperpnea (EVH) is a sensitive test especially for elite athletes but there are some concerns about safety. In asthmatics and non athletes, hyperosmolar aerosols may offer practical advantages over EVH or exercise testing. Mannitol in particular is available as a simple standardized single use kit. 12
Mechanism of EIB
Hyperpnoea in response to exercise causes net water loss from the airway surface. This drying appears to trigger EIB by changes in osmolality of the perciliary layer of fluid, leading to bronchial epithelial cell shrinkage and release of inflammatory mediators. 13 Anderson 14 suggests that EIB appears to be an exaggerated response to airway dehydration in the presence of inflammatory cells and mediators in a person with a responsive bronchial smooth muscle.
Several mediators are likely to contribute to EIB including prostaglandin (PG) D2, leukotriene (LT) C4, adenosine and histamine, and in turn different cell types including mast cells, eosinophils, sensory nerves and epithelial cells are mediator sources. Drugs that inhibit the release of mast cell mediators or prevent the bronchoconstrictor effects, or reduce the production of mediators or mast cell number have been shown to have beneficial effects on EIB. 5 9α, 11β-PGF2, a metabolite of PGD2 can be detected with the help of sensitive assays in the urine and sputum of asthmatics and athletes with EIB. 5 A significant association between change in its levels from baseline and percentage changes in FEV1 indicate that PGD2 is likely to be the most important mediator of EIB. In healthy subjects, PGD2 and LTE4 are 100 and 1,000 times as potent as histamine or methacholine in provoking bronchoconstriction, respectively. A single class of receptors in human airway smooth muscle appear to mediate contractions induced by LTC4, LTD4 and LTE4. 15 The cysteinyl LT 1 (cysLT1) receptor is a G-protein-coupled receptor that is expressed in peripheral blood leukocytes and other tissues. The major intracellular signaling pathway for the cyLT1 receptor is via calcium release. CysLT receptor antagonists (CysLTRAs) block the actions of cysteinyl leukotrienes at this receptor on target cells. 16 Hence montelukast as a CysLTRA has found a place in the therapeutic armamentarium against EIB.
Metabolism and Pharmakokinetic Profile
The pharmacokinetic profile of montelukast has been extensively studied, and there is a multitude of data identifying the pathway of the drug during its activity in vivo. Such studies have been performed within paediatric populations in addition to adults to support its appropriate and safe use in both populations.
Montelukast is a potent, orally active selective leukotriene receptor antagonist (LTRA) that is rapidly absorbed following administration.17–21 The different formulations of tablet (chewable tablet and granules) have been shown to have differing bioavailabilities. Consequently, peak plasma levels are reached at different rates.18–21
A 10 mg dose in adults has been shown to have an oral bioavailability of 64% and achieves a peak plasma concentration (Cmax) within 3-4 hours (Tmax) with no effect on administration prior to or after a meal.18–21 The 5 mg chewable tablet achieved a Cmax in 2-2.5 hours and demonstrated a bioavailability of 73% (fasted) and 63% (post meal).18–21 The 4 mg chewable tablet and 4 mg oral granules are bioequivalent to the 5 mg chewable tablets, but a Cmax is achieved within 2 hours. However, a meal with administration reduced Cmax by 35%, and prolonged Tmax by 1 hour and 2.9 hours for the chewable tablet and granules, respectively.18–21
From the literature it is evident that montelukast is rapidly absorbed from the gastrointestinal tract and bioavailability is affected by formulation choice; however, plasma concentration differs in the youngest population of infant patients. The plasma concentration profile following oral administration to adolescents ° 15 years is similar to that seen in adults (10 mg dose).22–24 The plasma concentration profile following administration of the 4 mg or 5 mg chewable tablets, in children 2-5 years or 6-14 years, is also similar to that of the adults receiving 10 mg tablets. Variability of plasma concentrations has been shown to be greater for the oral granules in infants 12-23 months than in adults and even more so in infants of 6-11 months. 22
Peak plasma concentrations are achieved at different rates, but all formulations are appropriate for once daily dosing, as therapeutic effects persist for at least 24 hours in all demographics.22–24 Montelukast is more than 99% protein bound with minimal distribution across the blood brain barrier, 25 and is extensively hepatically metabolized,22–24 specifically by the cytochrome P450 (CYP) enzymes. Earlier studies identified that metabolism predominantly involved the 3A4, 2A6 and 2C9 microsomes; 25 however, more recent studies have shown more involvement by the 2C8 microsomes. 26 In addition, metabolism occurs via the acyl glucuronidation pathway within the gastrointestinal tract. 26 At a therapeutic dose, montelukast does not inhibit the CYP enzymes and therefore will not affect other drugs administered that are metabolized by the same hepatic pathway. However, when administering montelukast with known CYP 3A4 inducers, such as rifampicin and phenytoin, care should be taken. 22
Clinical Use
Role of montelukast specifically in EIB
As well as the above mentioned indications for the use of montelukast in wheezing disorders, there have been several studies over the last 15 years specifically looking at the effectiveness of montelukast in protecting against EIB in older children and adolescents. Leukotrienes are implicated in sustaining a bronchoconstrictive and inflammatory response in EIB. Consequently, leukotriene inhibition is an attractive option to consider for EIB treatment. This review will focus on studies that examined paediatric populations.
Montelukast may start acting within 2 hours of oral administration, unlike Beta-2 (β2) agonists, which have an instantaneous effect. This effect reaches its maximum by 12 hours after administration and persists for up to 24 hours.29,30 Bronchoconstriction triggered by exercise responds to cessation of exercise in its natural course. Due to its pharamacokinetic profile, Montelukast may not be the optimal medication for relief of symptoms, but may be more useful when given as a prophylaxis.
Several studies have looked at the long term use of montelukast.31–34 Montelukast improves the magnitude of bronchoconstriction and is significantly protective in EIB. Protection is measured as the magnitude of post-exercise fall in FEV1, as well as maximal decrease in FEV1 with exercise.31–34 It also shortens the time to recovery of FEV1 to pre-exercise level,31–34 and in some children it also improves the late phase response to exercise. 32 This protective response is evident even after a few days of prophylaxis with montelukast. 31 The response is sustained for the duration of montelukast prophylaxis uniformly in all studies, but the length of time for which this protection persists is variable. Leff 35 showed no lasting protection 2 weeks after stopping montelukast; however, in a paediatric study of 6-14 year olds, Kim 33 showed that improvements in asthma symptom scores were present even after 8 weeks of discontinuing montelukast.
The other beneficial property of montelukast is that no tolerance develops to its effect when given as a prophylaxis, as compared to β2 agonists for a prophylaxis.34,35
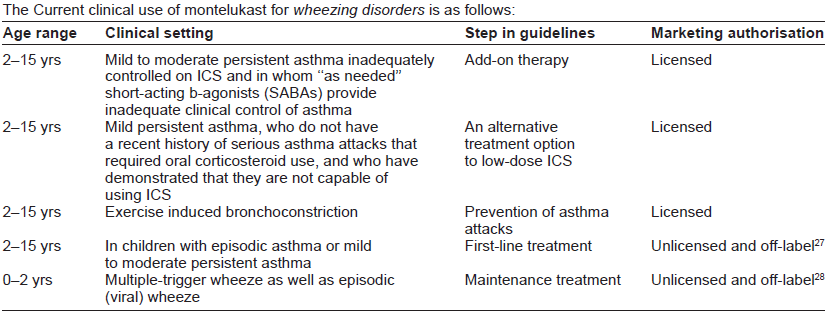
The Current clinical use of montelukast for wheezing disorders is as follows:
Timing of day dosing of montelukast in relation to its protective effect has also been studied. Pajaron-Fernandez 36 looked at EIB after 2 weeks of morning or evening dose of montelukast and found no difference whether morning or evening dose was used.
These protective effects appear to be independent of concomitant use of inhaled corticosteroids.32,34 However, unlike β2 agonists, protection from EIB is not complete (eg, 59% reduction in the study by Kemp and colleagues 31 ), and a proportion of patients may be non-responders to montelukast. 32
There is heterogeneity of response with montelukast; specifically, some children respond very well while others do not respond at all. This heterogeneity is thought to be due to variations in gene coding for components of the LT pathway. CysLTs are potent mediators of asthma inflammation. CysLTs are synthesized from arachidonic acid located in membrane phospholipids by cytosolic phospholipase A2 in response to stimulation. Arachidonic acid is converted to 5(S) HETE and LTA4 by membrane-bound 5-lipoxygenase (ALOX5). LTA4 is then in further steps converted to LTC4 and LTD4. Polymorhisms in the ALOX5 and the LTC4 synthase (LTC4S) genes contribute to variability in response to LT modifiers and LT selective antagonists. 16
Specifically in EIB, Kim 37 looked at the effect of genetic polymorphisms of the Thromboxane A2 receptor on the efficacy of montelukast in EIB in Korean children. They identified certain polymorphisms (TBXA2R + 924T > C homozygote, TBX2AR795T > C hetero - and homo - zygote) had a threefold poor response to 8-week montelukast treatment with respect to maximum percent fall in FEV1 after exercise, in comparison to patients with more common alleles.
Overall the evidence supports improvement in EIB with montelukast only, though this improvement is not complete. The protection seems to last as long as montelukast is taken regularly without appearance of tolerance,33,34 but this protection disappears after stopping montelukast in some studies. 34 The available evidence supports the use of regular montelukast treatment for children where EIB is the main manifestation of their asthma.
Montelukast Compared to other Therapies in EIB
Short acting B2 agonists
The improvement in the extent of EIB after inhaled β2 agonist use is almost complete, as compared to montelukast, with which the response is modest. 38 Additionally, a problem alluded to earlier involves the heterogeneity in response to Montelukast as compared to inhaled β2 agonists.
Long acting Beta2 agonists
In a three way crossover study with montelukast, salmetrol and placebo for EIB, investigators measured the percentage change in FEV1 at 2, 8.5 and 24 hours post-dose. Montelukast and salmetrol attenuated a decline in FEV1 at 2 and 8.5 hours post-dose, but the protective effect of salmetrol had subsided by 24 hours. 39
With long-term administration, lack of tolerance to montelukast supports its use over salmetrol. Viallarn 40 and Edelman 41 both found improvement with montelukast and salmetrol, however, both studies noted a waning of the bronchoprotective effect in the salmeterol group at 4 and 8 weeks whereas the montelukast effect was maintained. There were more discontinuations in the salmetrol group.
Storms and colleagues 42 investigated the response to short acting β2 agonist (SABA) rescue following 4 weeks of montelukast or salmetrol given to patients with EIB on fluticasone and persisting symptoms. Both the magnitude and rate of rescue bronchodilation were greater with montelukast compared with salmeterol. Patients treated with montelukast had significantly greater alleviation of EIB compared to placebo, but the salmeterol group did not.
Inhaled steroids
Vidal and colleagues 43 compared montelukast to budesonide in 20 patients with EIA. Patients received 10 mg of montelukast once daily for 3 days compared to 400 mcg of budesonide twice daily for 15 days, with a 15-day intervening washout period. Both therapies showed significant improvement as compared to baseline. However, there were considerable individual variations in response to both medications. 43 The study concluded with the recommendation that both medications be tested in each patient prior to the final decision.
Studies comparing LTRAs to some other medication or combinations are summarized in Table 1.
Montelukast compared to other therapies in EIB.

Montelukast is at least similar to long acting β2 agonists (LABAs) in the amelioration of EIB in the short term 39 and affords better protection than LABAs in the long term.40–42 Concerns over tolerance to LABAs would make montelukast a more suitable drug to be used in the ongoing management of asthmatic patients where EIB is a troublesome manifestation of their asthma. Even when LABAs are combined with inhaled corticosteroids (ICS), montelukast is superior for the protection against EIB.46,47 The protection afforded by montelukast is not as complete as with SABA, 38 though the latter works only for a short duration compared to montelukast.
Safety and Tolerability
Adverse drug reactions (ADR) are a common concern when prescribing treatments. Brunlof and colleagues 48 reviewed safety reports for numerous drugs administered to children and identified LTRAs having the greatest number of concerning reports. The ADRs reported (such as nightmares, abdominal pain, fever, nausea and aggressiveness) are all considered in the manufacturer's summary of product characteristics;17–19 in addition, this review identified the potential need for further exploration of psychiatric ADRs in those children taking montelukast.
Montelukast is currently used widely in paediatrics and it is important to discuss the appropriate safe use of the drug for therapy. A review was carried out by Bisgaard and colleagues 49 of numerous double blind, placebo controlled trials of montelukast in addition to the following open-label extension studies with data from 2751 patients aged 6 months to 14 years, diagnosed with allergic rhinitis, persistent asthma and intermittent asthma. The overall inference was that the safety profile of montelukast in these groups of patients was not significantly different from placebo, nor did it change with long term use.
Studies with montelukast have identified more specific tolerability and safety profiles within different demographics. In particular, appropriate dosing has been tested in various studies in hopes of reducing any risks that may arise from selecting montelukast as a therapy for children.
Use of montelukast in infants aged 6 to 24 months is documented to be well-tolerated at a dose of 4 mg. 50 Significantly comparable pharmacokinetics were observed in this group of infants when compared to adults receiving a 10 mg dose. 50 This is further reinforced by evidence from a study involving children aged 6 to 24 months with asthma, in which patients received the same 4 mg dose of treatment. 51 There was no significant difference in clinical adverse experiences found in the active treatment group compared to placebo and overall this group of patients tolerated montelukast 4 mg granules well for a period of six weeks.
Safe use was further evaluated in infants 1-3 months old 50 and 3-6 months old. 52 Interestingly, in a small study of 1-3 month-old infants (N = 12), the area under the curve concentration was found to be 3.6 times higher than the value for 6-24 month old infants. Despite this, at a tested dose of 4 mg and 8 mg there were no drug related adverse experiences and the drug was well-tolerated overall. 50
A study investigating a safe dose for children aged 2-5 years determined that a 4 mg dose of montelukast was preferable to a dose of 5 mg. 53 This finding was further confirmed by a similar study comparing the pharmacokinetics in this same age group, compared to adult pharmacokinetics. 54 In groups of older children from 6 years to 14 years, it was found that patients receiving montelukast did not have significantly higher adverse reactions (drug and non drug related) than placebo. Therefore, a tolerability and safety profile similar to placebo was determined for both short-term and long-term use of montelukast.55,56
Patient Preference
In a study comparing attitudes toward montelukast and inhaled cromolyn, parent and patient preference, parent and patient satisfaction, and patient adherence to therapy were all significantly better with oral montelukast compared with inhaled cromolyn. β2 agonist use was decreased when taking montelukast, which was safe and well-tolerated. 57 Similarly in an open-label study comparing montelukast and beclomethasone, 58 parents reported that montelukast was more convenient, less difficult to use and was used as instructed more often as compared to beclomethasone. However, in another study 59 comparing fluticasone to montelukast in asthmatic subjects, significantly more patients were satisfied with fluticasone compared to montelukast. Hence, it is difficult to draw any conclusive inference of patient preference for montelukast over other treatments. We were not able to identify any studies which specifically looked at patient preference for use of Montelukast in treatment of EIB.
Conclusions
In conclusion, exercise-induced bronchoconstriction is mediated by changes in temperature and/or humidity in the presence of inflammatory cells in the asthmatic airway, and is secondary to release of several mediators of bronchoconstriction. Leukotrienes are a major group of mediators causing bronchoconstriction, and regular or intermittent use of montelukast offers protection against EIB; however, this protection is not complete. Regular use does not appear to lead to tolerance. There are individual differences in response to montelukast for protection against EIB. Nightmares, abdominal pain, fever, nausea and aggressiveness are the commonly described side effects; however, the overall safety profile of montelukast is still very good and does not change with long-term use. There is some evidence to suggest that patients prefer this oral therapy to inhaled medications.
Author Contributions
Wrote the first draft of the manuscript: SK, JB. Contributed to the writing of the manuscript: AM Agree with manuscript results and conclusions: SK, AM, JB Jointly developed the structure and arguments for the paper: SK, AM, JB Made critical revisions and approved final version: SK, JB. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
