Abstract
Pitavastatin is one of the most effective treatment agents for lowering serum low-density lipoprotein cholesterol (LDL-C) levels. Because pitavastatin is scarcely metabolized, the risk of drug-drug interactions during multidrug therapy is low, and the LDL-C target is achieved in the majority of patients. The incidence of adverse events associated with pitavastatin treatment has so far been comparable to or lower than that associated with other statins, and glucose metabolism is not affected by the drug. Pitavastatin promotes plaque regression and improves the plaque composition in patients with acute coronary syndrome. The serum level of high-density lipoprotein cholesterol (HDL-C) during treatment with pitavastatin was demonstrated to be inversely associated with the incidence of cardiovascular events. Because of its effective and sustained action to increase HDL-C, pitavastatin is expected to contribute to the prevention of cardiovascular events, in addition to its potent LDL-C lowering effect, especially in patients with low serum HDL-C levels.
Introduction
Statins play a major role in the prevention of cardiovascular events in patients with hypercholesterolemia. Combination of a standard-dose statin and a cholesterol absorption inhibitor or other dyslipidemia therapeutics, rather than a high-dose statin alone or statin dose escalation, is currently considered as the most suitable option for aggressively decreasing the serum level of low-density lipoprotein cholesterol (LDL-C). One of the reasons for such a trend is the withdrawal of cerivastatin from the market due to the frequent reports, after the launch of this drug, of life-threatening rhabdomyolysis associated with combined administration of the drug with gemfibrozil. The withdrawal of this drug drew attention to the possibility of drug-drug interactions associated with multidrug therapy. Hypercholesterolemia and combined dyslipidemia are often associated with multiple cardiovascular risk factors such as hypertension, diabetes mellitus and renal dysfunction. Most dyslipidemic patients who are at an elevated risk of cardiovascular events are treated with multiple drugs. According to a US database, more than 80 percent of dyslipidemic patients are treated with drugs metabolized by CYP3A4, an isoform of the drug-metabolizing enzyme cytochrome P450 (CYP), and one-fourth to one-third of them are concurrently treated with a CYP3A4 inhibitor. 1 Nine percent of the CYP3A4 inhibitors used by these patients are contraindicated for coadministration according to the package insert. Unnecessary dose adjustment and switch to other treatment may be avoided to a certain extent if lipid-lowering drugs that scarcely interact with each other are used.
It has been demonstrated that marked residual cardiovascular risk still remains even after aggressive lowering of the serum LDL-C. Hypertriglyceridemia and reduced serum high-density lipoprotein cholesterol (HDL-C) during statin treatment constitute cardiovascular risk factors. After lowering of the serum LDL-C, increasing the serum HDL-C and lowering serum triglycerides (TG) represent the subsequent treatment targets. 2 Previous clinical studies have failed to demonstrate the benefit of combined statin/fenofibrate treatment in patients with type 2 diabetes 3 or that of combined statin/niacin treatment in decreasing the risk of cardiovascular events in low-HDL-C/hypertriglyceridemic patients (AIM-HIGH study 4 ). However, a potential benefit of combined therapy is strongly suggested in low-HDL-C/hypertriglyceridemic patients treated with statins. 3 Ongoing studies of combination therapies and future studies of new drugs may further confirm these findings.
Clinical Studies
Safety
According to clinical data available to date, the safety of pitavastatin is similar or superior to that of the existing statins. Abnormal glucose metabolism, which is an increasingly recognized adverse effect of strong statins, has been reported to be infrequently associated with pitavastatin.
Pre-marketing clinical studies
In pre-marketing clinical studies of pitavastatin conducted in Japan, adverse drug reactions (ADRs) in the form of symptoms or signs were reported in 50 of 886 patients (5.6%). 5 Abnormal laboratory test results reported as ADRs in 167 of the 886 patients (18.8%) included elevated serum levels of gamma-glutamyl transferase (γ-GTP), creatine kinase (CK or CPK), alanine aminotransferase (ALT or GPT), aspartate aminotransferase (AST or GOT) and others. These ADRs were not unique to pitavastatin, and the frequencies and severities of the ADRs were comparable to those reported for the existing statins. No relationship was noted between the ADRs (symptoms, signs and abnormal laboratory test results) and the pitavastatin dose: the incidence was 20.3% (26/128) in patients treated with pitavastatin 1 mg, 23.3% (121/519) in those treated with pitavastatin 2 mg and 20.9% (50/239) in those treated with pitavastatin 4 mg (Table 1). 5 No increase in the incidence or severity of ADRs, or appearance of new ADRs was reported during long-term treatment with pitavastatin. A double-blind phase III study conducted in Japan, Korea and Europe showed that the safety of pitavastatin 2 to 4 mg was comparable to that of pravastatin 20 to 40 mg, simvastatin 20 to 40 mg and atorvastatin 10 to 20 mg.6–8
Incidence rate of adverse reactions in pre-marketing clinical studies of pitavastatin. 5
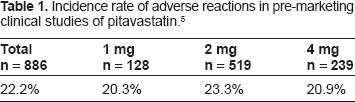
Post-marketing surveillance
The safety of pitavastatin was evaluated and established in a two-year large-scale prospective post-marketing surveillance of approximately 20,000 patients with hypercholesterolemia (LIVES Study 9 ). Analysis of the data reported during the initial three months revealed an incidence of ADRs of 6.1% (1,206/19,921). 10 As for other strong statins, the incidence of ADRs reported during the first three months of the post-marketing surveillance was 12.0% (576/4,805) for atorvastatin and 11.1% (978/8,795) for rosuvastatin. As compared with the results for these statins, the incidence was about 50% lower for pitavastatin.11,12 Among the patients treated with pitavastatin for the entire surveillance period of 2 years, ADRs occurred in 10.4%. The incidences of myopathy-related and hepatic dysfunction-related ADRs, the major ADRs associated with the use of statins, were 2.1% and 1.0%, respectively. Mild ADRs were reported in 1,045 patients, moderate ADRs in 155 patients and severe ADRs in six patients. Thus, most ADRs were mild in severity. The incidences of increased serum CK (CPK) and increased serum transaminase levels, other major ADRs, were lower as compared with those reported for other strong statins. No significant increase in the incidence of ADRs was reported in patients treated concurrently with azole antifungals, macrolide antibiotics, coumarin anticoagulants, antiplatelet agents, antihypertensive agents or antidiabetic agents. Thus, the safety of pitavastatin in routine clinical practice was demonstrated.
Effect on glucose metabolism
A number of recent studies reported an association between statin treatment and new onset of diabetes mellitus. According to a meta-analysis, statin use is associated with a 9% increase of newly diagnosed cases of diabetes mellitus. 13 While no large-scale study or meta-analysis to confirm such an association has been reported for pitavastatin until date, some comparator-controlled and uncontrolled studies have evaluated the effect of pitavastatin on glucose metabolism. In a two-year study conducted to evaluate the safety of pitavastatin in routine clinical practice in approximately 20,000 patients with hypercholesterolemia, the incidence of abnormal glucose metabolism reported as an ADR was quite low (LIVES Study 9 ). Subgroup analysis of patients with hypercholesterolemia and diabetes mellitus showed a significant decrease of the HbA1c after 104 weeks (LIVES study subgroup analysis 14 ). In a study performed to compare the efficacy and safety of atorvastatin and pitavastatin in 48 patients with hypercholesterolemia and type 2 diabetes mellitus, who were treated initially with atorvastatin and then switched to pitavastatin, significant decrease of the HbA1c was observed after six months treatment with pitavastatin, especially in obese patients. 15 A retrospective analysis of the effect of atorvastatin 10 mg, pravastatin 10 mg and pitavastatin 2 mg on the blood glucose and HbA1c in patients with type 2 diabetes mellitus showed that the glycemia status worsened only in the patients treated with atorvastatin. 16 While the safety of 16 weeks treatment with pitavastatin 2 mg, atorvastatin 10 mg and rosuvastatin 2.5 mg was comparable in a total of about 300 patients, significant increase of the HbA1c was seen only in those treated with atorvastatin or rosuvastatin, and not in those treated with pitavastatin (PATROL study 17 ). A subgroup analysis in a randomized controlled study conducted to compare the effect of pitavastatin and atorvastatin on the serum non-HDL-C showed a significant increase of the Glycoalbumin in patients treated with atorvastatin (CHIBA study subgroup analysis 18 ). In a controlled study of pitavastatin and atorvastatin in hyperlipidemia patients with type 2 diabetes, significant increase of the fasting blood glucose was observed after 12 weeks treatment in patients treated with atorvastatin. A significant inter-group difference was also noted. 19 While the increase was remarkable in female patients, no increase was noted in the male patients. A study to compare the five-year incidence of newly diagnosed diabetes mellitus between patients with glucose intolerance treated with pitavastatin and those without (n = 1,240) is ongoing (J-PREDICT Study 20 ), and the result is awaited.
Pharmacokinetic profile
Pitavastatin has an improved pharmacokinetic profile as compared with other statins. Most importantly, neither pitavastatin nor pitavastatin lactone is metabolized to any significant extent by CYP. 21 The lactone forms of most statins, including of rosuvastatin, are metabolized by CYP3A4. Increased blood concentrations of the lactone form are associated with myopathy. 22 Among the statins with a potent LDL-C-lowering effect at the starting dose, pitavastatin is the least associated with CYP-mediated drug-drug interactions.
Efficacy
Lipid modifying effect
In pre-marketing studies conducted in Japan, Korea and Europe, pitavastatin produced equivalent lowering of the serum LDL-C and serum total cholesterol (TC) in patients with hyperlipidemia or combined dyslipidemia. Pitavastatin 1 mg, 2 mg and 4 mg lowered the serum LDL-C by 31% to 34%, 36% to 42% and 43% to 47%, and lowered the serum TC by 22 to 23%, 26 to 29% and 31% to 33%, respectively.6–8,23,24 The serum LDL-C-and serum TC-lowering effect of pitavastatin 2 to 4 mg was shown to be comparable to that of simvastatin 20 to 40 mg and atorvastatin 10 to 20 mg in double-blind controlled studies.7,8 The effect was also comparable to that of rosuvastatin 5 to 10 mg. Only pitavastatin, atorvastatin and rosuvastatin lowered the serum LDL-C by 40% or more at the standard or recommended starting dose. 25 Thus, pitavastatin is one of the most potent statins available at present (Fig. 1).
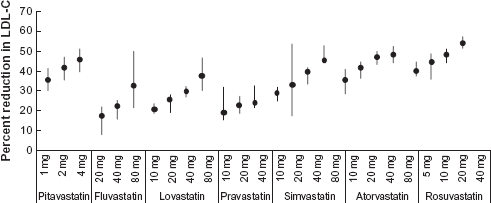
Percent reduction in LDL-C by dose by statin. Modified from reference 25.
The target serum LDL-C is achieved in a high percentage of patients with hypercholesterolemia or combined dyslipidemia treated with pitavastatin, regardless of age, sex or other characteristics. In a double-blind controlled study of pitavastatin and simvastatin conducted in patients with hypercholesterolemia or combined dyslipidemia, the target LDL-C recommended by NCEP ATP III was achieved in 70% of patients treated with pitavastatin 2 mg and 80% of those treated with pitavastatin 4 mg. 8 In a double-blind controlled study of pitavastatin and pravastatin in elderly patients with hypercholesterolemia or combined dyslipidemia, the target LDL-C recommended by NCEP ATP III was achieved in 83% of patients treated with pitavastatin 1 mg, 89% of patients treated with pitavastatin 2 mg, and 91% of patients treated with pitavastatin 4 mg. 6 In the LIVES study, in which about 40% of the patients were receiving treatment with 1 mg and about 60% were receiving treatment with 2 mg of the drug, the Japan Atherosclerosis Society (JAS)-recommended target LDL-C was achieved in 75% of the patients. 9 These studies suggest that pitavastatin allows the target LDL-C to be achieved in a majority of the patients, even at low doses (1 mg to 2 mg).
The effect of pitavastatin on familial hypercholesterolemia was evaluated in 30 heterozygous patients. The serum LDL-C decreased by 40% after 8 weeks of treatment with pitavastatin 2 mg and by 48% at 8 weeks after forced uptitration of the pitavastatin dose to 4 mg. 26 Of 20 patients treated with pitavastatin 2 mg for two years followed by combined treatment with pitavastatin 2 mg plus ezetimibe 10 mg for three months, the serum LDL-C decreased in 49% during treatment with pitavastatin alone and 63% during treatment with pitavastatin plus ezetimibe. 27 These data suggest that pitavastatin is one of the most potent LDL-C-lowering drugs available at present, when statin dose escalation and high-dose statin therapy need to be avoided.
Besides its effect of lowering the serum LDL-C, pitavastatin 2 to 4 mg also lowered the serum TG by as much or more than that by simvastatin, atorvastatin or pravastatin.6–8 Pitavastatin lowered the serum TG by 22% to 30% in patients with baseline serum TG levels of 150 mg/dL or more.23,24,28 Several studies have consistently demonstrated that pitavastatin produces strong and sustained elevation of the serum HDL-C.29–33 In a randomized, positive-controlled study of pitavastatin, the percent increase of the serum HDL-C at 4 to 12 weeks in patients with baseline serum HDL-C levels of 48.5 to 58.5 mg/dL was 3.2% to 8.9%.7,8,24,28,33,34 The LIVES study showed that pitavastatin produced a sustained increase of the serum HDL-C for two years (LIVES study subgroup analysis 31 ). The mean change of the serum HDL-C at the end of two years was 5.9%. In a subgroup of patients with baseline serum HDL-C levels of <40 mg/dL, significant increase of the serum HDL-C by 24.6% was observed at the end of two years. A study to compare pitavastatin 2 mg and atorvastatin 10 mg in patients with impaired glucose tolerance and elevated LDL-C based on the primary endpoint of change of the serum HDL-C at 52 weeks showed that pitavastatin was significantly superior to atorvastatin (Fig. 2; PIAT study 33 ). In view of the strong LDL-C-lowering effect at the starting dose, TG-lowering effect and the strong and sustained HDL-C-elevating effect, pitavastatin is expected as an effective approach to reduce the residual risk that persists even after aggressive LDL-C lowering. Further accumulation of evidence to support the efficacy of pitavastatin is awaited.
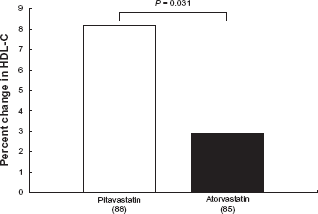
Percent change in HDL-C after 52-week treatment by pitavastatin 2 mg and atorvastatin 10 mg. 33
Residual risk
Despite the current standard treatment to achieve the target LDL-C, blood pressure and blood glucose, the cardiovascular risk remains high in patients with dyslipidemia, the so- called residual risk. Cardiovascular events cannot be prevented completely even when a serum LDL-C level of <70 mg/dL is achieved, which is quite low, by aggressive LDL-lowering therapy. Elevated serum TG and reduced serum HDL-C during statin treatment have been identified as independent cardiovascular risk factors (PROVE IT-TIMI 22 study, 35 TNT study 36 ). On the other hand, serum HDL-C may not be a reliable predictor of the residual risk in patients in whom marked lowering of the serum LDL-C level has been achieved with a strong statin, while the baseline serum HDL-C has been shown as a useful marker in the initial cardiovascular risk assessment (JUPITER study 37 ). In addition to the potent LDL-C-lowering effect, pitavastatin produces strong and sustainable elevation of the serum HDL-C, especially in patients with low baseline serum HDL-C. How the risk of cardiovascular events might be affected by the sustained effect of the drug on the serum HDL-C needs to be further evaluated. The post-marketing surveillance conducted to evaluate the safety and efficacy of pitavastatin in routine practice in about 20,000 patients with hypercholesterolemia was extended to three years (LIVES Extension 38 ). Pitavastatin produced sustained elevation of the serum HDL-C, which was associated with a reduced cardiovascular risk independently of the lowering of the serum LDL-C during pitavastatin therapy. In the retrospective study conducted to evaluate the major cardiovascular events in 743 consecutive patients who underwent percutaneous coronary intervention (PCI) and were treated or not treated with pitavastatin, atorvastatin or pravastatin, the serum HDL-C significantly increased only in patients treated with pitavastatin as compared with that in those not treated with statins. Pitavastatin was the most effective among the statins at reducing the risk of major cardiovascular events (CIRCLE study 39 ). These studies suggest that pitavastatin can exert the additional benefit of increasing the serum HDL-C, over its strong LDL-C-reducing effect, to reduce the risk of cardiovascular events. Whether the HDL-C-elevating effect of pitavastatin in addition to its LDL-C-lowering effect somehow provides long-term benefit in terms of reducing the cardiovascular risk needs to be investigated further. Analyses of the HDL particle function, as also of the HDL particle composition, are also required. Results of a currently ongoing prospective study to evaluate the reduction in the risk of cardiovascular events by pitavastatin (REAL-CAD study 40 ) are awaited.
Approach on atherosclerotic plaque
Statins have been shown to cause plaque regression, which is associated with the aggressive reduction of the serum LDL-C level (REVERSAL study, 41 ASTEROID study, 42 ESTABLISH study 43 ). Several studies have reported the effects of pitavastatin on coronary plaques. A study performed to evaluate the effect of pitavastatin 4 mg on intravascular ultrasound (IVUS)-measured plaque volume in patients with acute coronary syndrome successfully treated with PCI under IVUS, showed that pitavastatin induced plaque regression in the non-culprit lesions in the PCI-treated culprit arteries at 8 to 12 months (Fig. 3; JAPAN-ACS study 44 ). A direct angioscopic observational study revealed that pitavastatin 2 mg stabilized yellow plaques (TOGETHAR study 45 ). In addition to is strong LDL-C-lowering effect and its strong and sustained HDL-C-elevating effect, pitavastatin also lowers the serum TG and non-HDL-C, and improves the apo B/apo A1 ratio.7,8 Some studies have suggested other pleiotropic effects of pitavastatin, including antioxidant and anti-inflammatory actions, improvement of the endothelial function and plaque stabilization.46–55 An electron-microscopic study in a porcine coronary stent model showed that pitavastatin inhibited the adhesion of white blood cells to the stent, suggesting that the drug may inhibit post-stenting early inflammation. 56
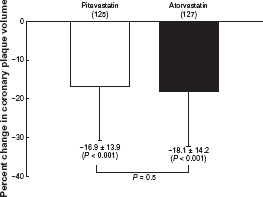
Percent change in coronary plaque volume from baseline to 8-to 12-month treatment with pitavastatin 4 mg and atorvastatin 20 mg. 44
Effects by Patient Phenotype
Studies conducted around the world have consistently demonstrated the efficacy and safety of pitavastatin, regardless of the age, sex and other clinical characteristics of the patients. The target serum LDL-C is achieved in most patients with hypercholesterolemia or combined dyslipidemia with pitavastatin 1 mg to 2 mg.6–9 Especially in patients with low serum HDL-C, pitavastatin produces a strong and sustained elevation of the serum HDL-C and may reduce the risk of cardiovascular events by lowering the serum LDL-C and increasing the serum HDL-C.31,38 Pitavastatin also lowers the serum TG.6–8,23,24,28 Serum LDL-C and TG could be reduced and serum HDL-C increased by pitavastatin in patients with diabetes mellitus or metabolic syndrome, who are likely to exhibit atherogenic dyslipidemia characterized by elevated serum TG and reduced serum HDL-C. Patients with dyslipidemia and diabetes mellitus or metabolic syndrome are usually treated with multiple drugs. Since pitavastatin is scarcely metabolized and is associated with a reduced likelihood of drug-drug interactions mediated by CYP, 21 its use is recommended in the aforementioned type of patients. So far, pitavastatin has not been shown to affect the glucose metabolism, which is an advantage.9,14–19 Like patients with dyslipidemia associated with diabetes mellitus or metabolic syndrome, high-risk patients with multiple cardiovascular risk factors also require multidrug therapies for the treatment of the risk factors. Also, elderly patients aged 65 years or over often have multiple cardiovascular risk factors. Pitavastatin can be a treatment of choice for these patients.
Place in Therapy
Several lipid modifying agents with different profiles are available for treatment of hypercholesterolemia and combined dyslipidemia as shown in Table 2. 62 Statins are the only treatment agents that improve the lipid profile with sufficient evidence of apparent improvement of the prognosis, and can therefore be considered as the first-line treatment for patients with hypercholesterolemia or combined dyslipidemia. Although statins with a potent LDL-C-lowering action at the starting dose are widely used, the target serum LDL-C is not achieved in many high-risk patients with multiple coronary risk factors. More aggressive lowering of the serum LDL-C is required in these patients. Ezetimibe and bile acid sequestrants are expected to play an important role, because high-dose statins and statin dose escalation often need to be avoided. At the standard dose, pitavastatin is one of the most potent LDL-C-lowering statins with effects comparable to those of atorvastatin and rosuvastatin. If more aggressive LDL-C reduction is required, reduction by over 60% can be achieved with concomitant use of a cholesterol absorption inhibitor.
Effects of lipid modifying agents.

Modified from reference 62.
In patients with combined dyslipidemia associated with high serum TG and/or low serum HDL-C, other lipid-lowering drugs are used together with statins, because adequate lipid control may not be expected to be achieved with statins alone. For multidrug therapies including statins, use of pitavastatin should be considered, because pitavastatin and its lactone form are not metabolized by CYP.
Fibrates are often used to lower elevated serum TG. However, whether this TG-lowering effect of fibrates is associated with cardiovascular prevention is still unknown. A meta-analysis of 18 studies in 45,058 patients showed that fibrate reduced the risk of development of coronary disorders by 13%. 57 The effect of fibrates in preventing coronary disorders, however, was only moderate as compared with that of the statins. No apparent additional effect of fibrates was demonstrated in patients treated with statins (ACCORD study 3 ). Therefore, statins are also the first-line treatment for dyslipidemia associated with elevated serum TG. However, some patients may benefit from the statin/fibrate combination, since fibrates prevent coronary events, especially in patients with elevated baseline serum TG, and cardiovascular event prevention by combined statin/fibrate treatment has also been suggested in patients with elevated baseline serum TG and low baseline serum HDL-C. Such potential benefit of this treatment combination should be evaluated in future studies. Pitavastatin has a milder TG-lowering effect as compared with fibrates. Combined use of pitavastatin and a fibrate could be considered when the serum TG cannot be lowered sufficiently, because the change of the blood pitavastatin concentration is small even when the drug is used concomitantly with fenofibrate or gemfibrozil.
Extended-release niacin elevates the serum HDL-C by 30% to 35% at the doses used in the US and Europe. While multidrug therapies including niacin have been shown to prevent coronary disorders and stroke, this prevention can hardly be attributed to niacin alone, since this drug is often used with the statins. 58 A subanalysis of PROVE IT-TIMI 22 reported that low serum HDL-C was associated with elevated cardiovascular risk. 35 Since the association between HDL-C elevation therapy and cardiovascular prevention is not clearly known yet, it should be evaluated in the course of development of various HDL-targeting drugs. Statins are the first-line treatment for dyslipidemia associated with low serum HDL-C, because they have been shown to prevent cardiovascular events in patients with low baseline serum HDL-C. Pitavastatin is an ideal treatment option, because it produces sustained elevation of the serum HDL-C over the long-term.
Although eicosapentaenoic acid (EPA) has a little lipid-lowering effect, EPA administration concomitantly with a statin prevented the major coronary events in hyperlipidemic patients (JELIS study 59 ). The EPA/arachidonic acid ratio has been shown to be associated with arteriosclerosis. EPA lowers the EPA/arachidonic acid ratio to prevent arteriosclerosis.60,61 EPA can be used in combination with a statin to potentially prevent cardiovascular disorders associated with the residual lipid abnormalities.
Conclusions
Pitavastatin is a well-tolerated, effective treatment agent for patients with hypercholesterolemia or combined dyslipidemia. It is especially useful in patients with low serum HDL-C. Pitavastatin is also associated with a low likelihood of drug-drug interactions in patients treated with multiple drugs and helps in achievement of the target serum LDL-C in the majority of patients. The ongoing and future studies will further identify the characteristics of pitavastatin and demonstrate its beneficial effect on the risk of development of cardiovascular events.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
