Abstract
Chronic obstructive pulmonary disease (COPD) is a leading respiratory disease in the world and third leading cause of death in United States. COPD is not recognized as an isolated respiratory disease anymore; in fact it is one of the dreaded diseases with significant systemic involvement and complications. Treatment of COPD requires team work and efforts among physicians, nurses, respiratory therapists and physical therapists. Treatment of patients with frequent exacerbations is a heavy burden on healthcare network. In the last few years, some improvements in the management of COPD have been made, owing to new drugs and management strategies, along with non-pharmacologic treatment of COPD with pulmonary rehabilitation and surgical interventions. Smoking cessation is still the best strategy to prevent COPD and prevent further progression of disease. In this review, we discuss pharmacotherapy of COPD including management of acute exacerbations, pharmacotherapy for smoking cessation and role of vaccination in COPD patients.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is characterized by airway inflammation and parenchy-mal destruction causing progressively irreversible airflow obstruction. COPD continues to be a major health problem worldwide with significant morbidity and mortality and has been projected to be the third leading cause of mortality and the fifth leading cause of disability by 2020.1,2 According to a recent multi-city study, prevalence of COPD varied from 11.4% to 26.1%. 3 COPD imposes an enormous economic burden on society. 4 As life expectancy rises across the world, and the burden of the disease increases, recognition of COPD and its optimal management has become essential. In the following text we will discuss pharmacological management of COPD including oxygen therapy. Non pharmacological management options like pulmonary rehabilitation, lung volume reduction surgery and lung transplantation will not be reviewed here.
Classification of Agents for Treatment of COPD
Classification of agents used in treatment of COPD based on their mechanisms is reviewed in Figure 1.
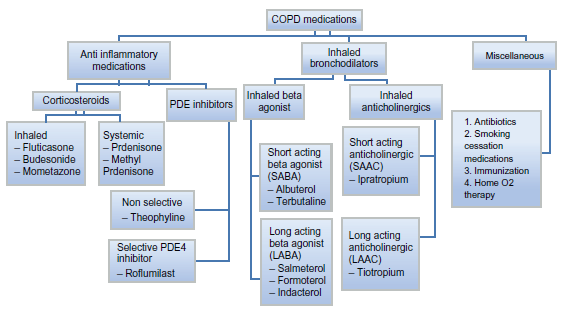
Classification of pharmacological agents used for treatment of COPD.
Properties of Pharmacological Agents
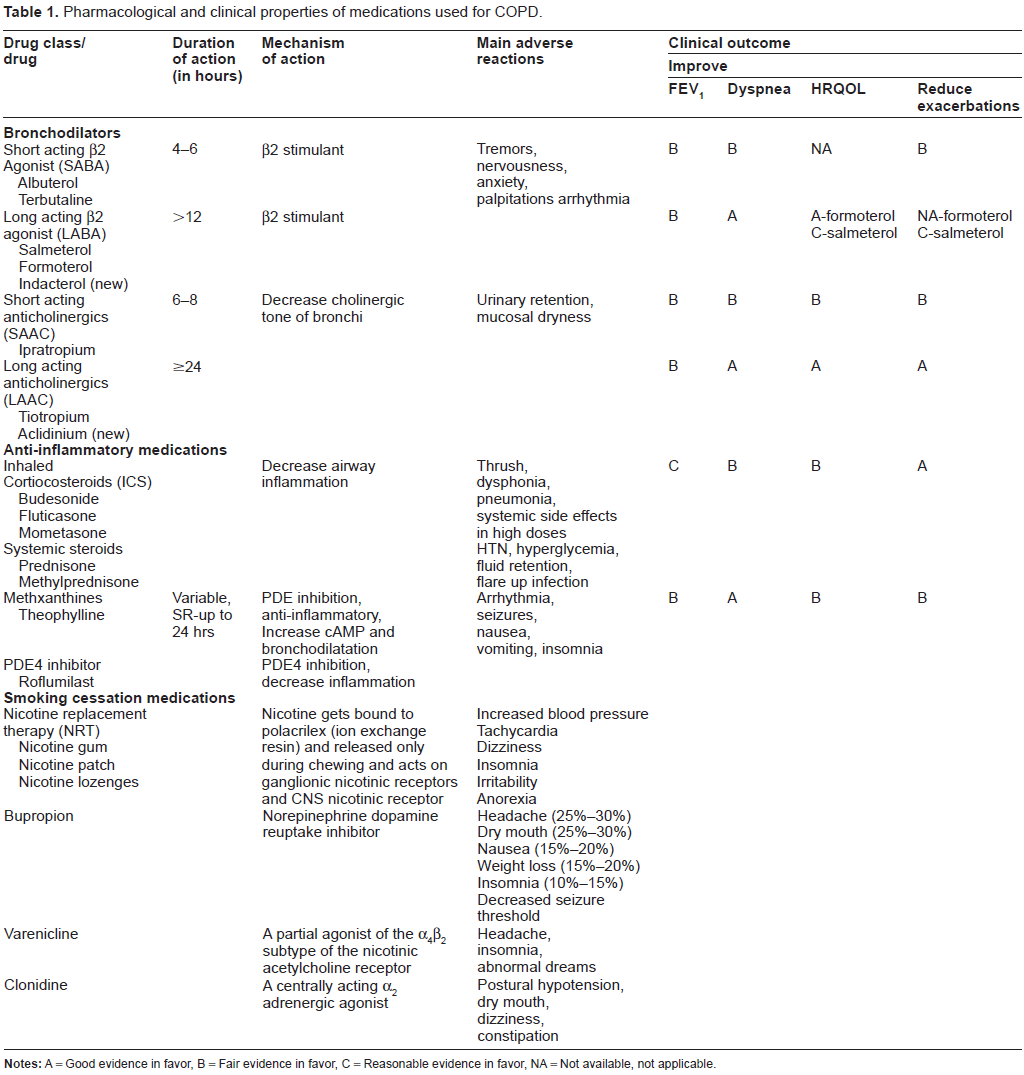
Pharmacological properties and clinical effects of commonly used agents in management of COPD are summarized in Table 1.
Pharmacological and clinical properties of medications used for COPD.
Pharmacological Management of Stable COPD
Pharmacological treatment in stable COPD improves quality of life and decreases the frequency of acute exacerbations. Asymptomatic patients with mild disease may not require any pharmacotherapy. As needed short acting bronchodilator therapy is recommended for mild symptomatic disease. As the disease becomes more severe, long acting bronchodilators become a mainstay of therapy. Inhaled corticoster-oids are also useful in patients with severe disease with frequent acute exacerbations. Long-term oxygen therapy has shown to improve survival and quality of life in patients with severe disease with hypoxia.
Short acting inhaled bronchodilators
This group consists of short acting anticholinergics and short acting β2 agonists (SABA). Anticholinergics cause bronchodilation by decreasing parasympathomimetic tone of bronchial wall smooth muscles and β2 agonists cause bronchodilation by sympathomimetic action. Both anti-cholinergics and β2 agonists improve expiratory flow rate and dyspnea in patients with COPD.5,6 Albuterol, a β2 agonist and ipratropium, an anticholinergic are commonly used medications in this group. Bronchodilation effects are dose-related; though higher doses, especially with β2 agonists, are associated with increasing frequency of side effects. 5 Anti-cholinergic therapy appears to be more efficacious in causing persistent bronchodilation than the short acting β2 agonists. 7 Combination therapy with both bronchodilators also causes significant airway dilation.8–10
Long acting inhaled bronchodilators
Long acting β2 agonists (LABA) and the long acting anti-cholinergic (LAAC) inhaler can be used alone or in combination for moderate or severe COPD and are the mainstay of maintenance therapy according to the Global initiative for Chronic Obstructive Lung Diseases guidelines (GOLD guidelines; available from: http://www.goldcopd.com). Long acting anticholinergics (LAAC) are also known as LAMA; long acting muscarinic antagonists. For discussion here we will use abbreviation of LAAC. Commonly used medications in this group are twice daily LABA; Salmeterol and Formoterol and once daily LAAC; Tiotropium. A new long acting once a day LABA, Indacaterol, has been recently approved by FDA for use in patients with COPD. Both groups of medications appear to have beneficial effects on dynamic hyperinflation as well as dyspnea indices, and are superior to short acting bronchodilators in terms of improvements in quality of life and dyspnea.11–14 The relative benefits of which agent to use first have not been systematically studied; however, Tiotropium is widely used and has generally been shown to provide better bronchodilation and clinical outcomes than the twice-daily LABAs.15–18 Tiotropium has been shown to decrease frequency of exacerbations, decrease hospitalizations and reduce length of stay in COPD patients.19,20 A recently conducted large scale double-blind placebo controlled trial (Understanding Potential Long-Term Impacts on Function with Tiotropium: UPLIFT) showed significant improvement in quality of life, lung function and frequency of exacerbations with Tiotropium use in patients with moderate to severe COPD regardless of other respiratory medications used. 21
Based on current evidence, initial treatment with an LAAC appears to be a rational approach, given its ability to reverse the heightened cholinergic tone that predominates in COPD patients. 22 However, Tiotropium should be used with caution, especially in patients with cardiovascular disease. LABAs initiate an alternative pathway of bronchodilation. As with short acting agents, combination therapy with both Tiotropium and long acting β2 agonists appears to have greater benefit (with improvement in spirometric indices, quality of life and symptoms) than monotherapy alone.17,23 Therefore when symptoms are not controlled with monotherapy, adding an LABA to LAAC or vice a versa may be more effective than either agent alone, without significant increase in side effects.24,25
Indacaterol improved lung function, dyspnea, quality of life, and rescue medication use compared with placebo in two different studies. Common side effect in both studies was COPD worsening though that also happened in placebo group.26,27 FDA approved Inda-caterol in US from July 2011 while it was approved for use in Europe since December 2009. Aclidinium is a twice daily LAAC. It is currently in an earlier stage of clinical development than Indacaterol. To date, clinical studies have demonstrated a broncho-dilatory effect and an improvement in quality of life versus placebo in patients with moderate-to-severe COPD, and an acceptable tolerability profile. 28
Inhaled corticosteroids (ICS)
Inhaled corticosteroids (ICS) are generally used in combination with long acting bronchodilators unlike the patients of asthma in which it is the mainstay of treatment. Combination therapy (ICS+LABA) is indicated in patients with severe COPD (FEV1 < 50%) who have frequent exacerbations. Criteria for frequent exacerbations are not well defined though in several studies they were defined as those with yearly exacerbation rates of greater than the median for the study, usually around three symptom-defined exacerbations per year or two per year if the exacerbation is defined by the requirement for therapy. 29 Treatment with ICS in this group of patients decreased the frequency of exacerbations and was associated with improved health status.30–32 Combined ICS and long acting β2 agonist therapy appears to be more beneficial than ICS alone. Patients on combination therapy had fewer exacerbations, improved lung function, symptoms and health status when compared to treatment with the individual component alone.33–36 A recent post-hoc analysis of the TORCH trial (Towards a Revolution in COPD Health study) suggests that combination therapy with ICS + LABA may decrease the rate of decline in lung function. 37 There is no significant change in mortality with treatment with ICS and ICS + LABA; however, use of ICS alone or in combination with LABA is associated with an increased likelihood of pneumonia.38,39
LAAC plus LABA plus ICS “triple therapy” may have additional clinical benefits and achieve better control in patients with severe COPD, reducing dyspnea and rescue medication use, 40 providing greater improvements in lung function,24,40,41 reducing day-and night-time symptoms, 24 improving quality of life (QoL) measures, 41 and reducing exacerbations. 24 Although triple therapy is often used in patients with severe and very severe COPD, cost constraints may limit its use in clinical practice.
Non steroid anti-inflammatory medications
Phosphodiasterase (PDEs) inhibitors and macrolide group of antibiotics are two groups of medications worth discussing as anti-inflammatory medications used for the treatment of COPD. Two types of PDE inhibitor currently used for treatment of COPD are methylxanthines (eg, Theophylline) and Roflumilast.
Methylxanthines
Methylxanthines, such as Theophylline, are reserved as third-line options due to their side effect burden and are only recommended for very severe disease according to GOLD guidelines. (available at http://www.goldcopd.com) A bronchodilator effect with Theo-phylline is seen only at high doses. However, low dose sustained released Theophylline, even in the absence of significant bronchodilation, was shown to decrease the frequency of exacerbations and improve quality of life in COPD in one small trial. 42 Metabolism and degradation of this medication is impaired significantly with age especially in the presence of co-morbid conditions and other drugs. Physicians must use caution when prescribing this medication due to its potential for significant side effects, even at therapeutic levels, particularly in the elderly. 43
Roflumilast
Eleven genetically distinct phosphodiesterase isoen-zymes have been identified. Amongst the isoenzymes, PDE4 is highly selective for cyclic AMP and represents the major cyclic AMP (c-AMP) metabolizing enzyme in all immunocompetent cells. Inactivation of c-AMP by PDE4 results in a pro-inflammatory state. So inhibition of PDE4 with agents like Roflumilast can help reducing inflammation. Roflumilast is once-daily, oral PDE4 inhibitor. In April 2010, Roflumilast received initial authorization in Europe as add-on therapy to bron-chodilator treatment for severe COPD (FEV1 < 50% predicted) in patients with a history of exacerbations. Roflumilast has been shown to improve lung function compared with placebo, although the improvements are moderate (pre-bronchodilator increase in FEV1 in the order of 48 mL) 44 compared with inhaled bronchodilators such as Tiotropium or Salmeterol (pre-bronchodilator increase in FEV1 in excess of 100 mL).45,46 Roflumilast was also shown to reduce exacerbations and the requirement for anti-inflammatory/anti-infective medications, and to improve QoL measures.44,47 Other studies have shown significant improvements in pre and post bron-chodilator FEV1 versus placebo when Roflumilast was added to Salmeterol or Tiotropium. 48 Nausea, diarrhea and weight loss are common side effects of PDE4 inhibitors, which may limit the use of Roflumilast in some COPD patients. Nevertheless, Roflumilast is potentially a valid alternative to concomitant ICS, and is an interesting new development as an add-on therapy to long-acting bronchodilators.
Macrolides
Macrolides are another newer generation of medications currently under study for COPD treatment. They belong to a family of agents that affect the 50 S ribosomes of both prokaryotic and eukaryotic organisms. Macrolides have been well studied in the treatment of bronchiolitis obliterans and have been shown to reduce airway inflammation. A recent trial evaluated the use of macrolides in COPD showed a significant reduction in COPD exacerbations with 12 month use of erythromycin 250 mg bid compared to placebo. 49 Azithromycin taken for 1 year has also been shown to decrease the frequency of exacerbations and to improve quality of life but was associated with increased hearing side effects. 50 Physicians should be cautious in frequently using azithromycin as azithromycin use was found to be associated with a small increase in cardiovascular deaths, especially in high risk patients for cardiovascular disease. 51
Drug delivery systems for inhaled medications and patient education
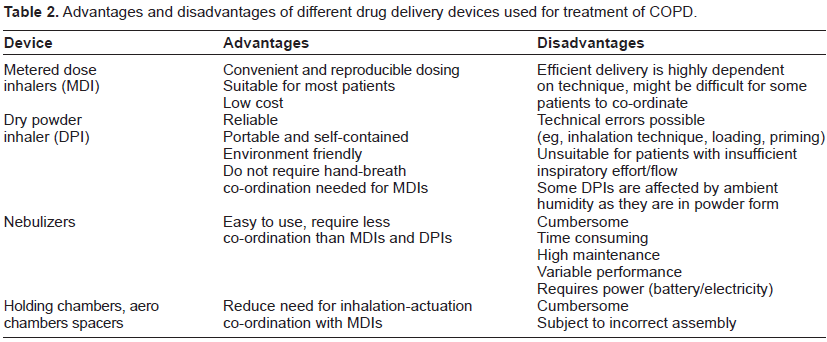
Most of the COPD medications are delivered via inhalation route. So it is very important to discuss different types of inhalation systems and proper inhaler technique. The inhaler devices used in management of COPD is described in Figure 2. Nebulizer converts liquid to aerosol through use of compressed air or ultrasonic power and is difficult to use as portable handy devices compared to MDIs and DPIs. MDIs can be used with spacers and aero chambers for convenience and better co-ordination. Advantages and disadvantages of these devices are narrated in Table 2.52–54
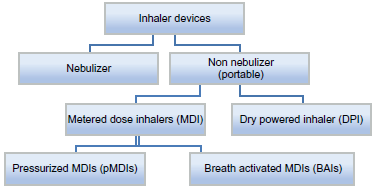
Types of Inhaler devices used in treatment of COPD.
Advantages and disadvantages of different drug delivery devices used for treatment of COPD.
Oxygen therapy
Long term oxygen therapy also requires prescription from physician; so we are covering this topic even though oxygen therapy can't be considered a trule pharmacological treatment. Supplemental oxygen, along with smoking cessation, is so far the only proven medical therapy to improve survival in selected COPD patients. Continuous oxygen therapy (15 hours/day, including at night) is recommended for patients with significant resting hypoxemia.55,56 The indications for supplemental oxygen in COPD are given in following Table 3. 57
Indications of continuous oxygen use in patient with COPD.

Algorithmic approach for treatment of stable COPD
Goals of pharmacological therapy include improvement of symptoms and quality of life along with reduction of mortality and morbidity (including exacerbations, emergency room visits and hospitalizations) and preservation of lung function while minimizing side effects of therapy. Complete management algorithm is abbreviated in Table 4.
Step care approach for treatment of stable COPD.
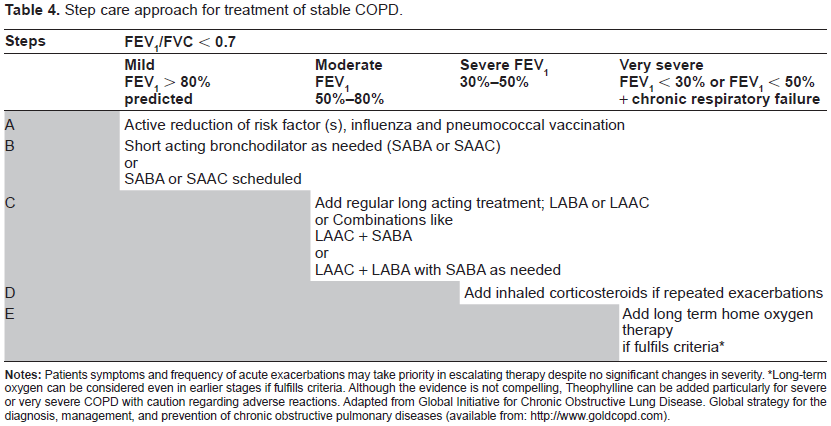
Long-term oxygen can be considered even in earlier stages if fulfills criteria. Although the evidence is not compelling, Theophylline can be added particularly for severe or very severe COPD with caution regarding adverse reactions. Adapted from Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary diseases (available from: http://www.goldcopd.com).
Pharmacological Management of Acute Exacerbation of COPD
Acute exacerbations are intermittent episodes of increased respiratory symptoms and worse pulmonary function that require a change in regular medications, which can happen anytime during course of COPD.58,59 Exacerbations may be accompanied by fever and other constitutional symptoms. These episodes contribute significantly to the morbidity associated with COPD, and in advanced disease, they are also the most frequent cause of death.38,60,61 Traditionally, the aims of treatment of an exacerbation are the recovery to baseline clinical status and the prevention of complications. Though these goals are undoubtedly important, several other important goals of treatment, both clinical and biological, should be considered. Among the clinical goals is rapid resolution of symptoms, prevention of early relapse, lengthening the ‘exacerbation free interval’ or ‘time to next exacerbation’, improvement in quality of life measures and improvement in exercise tolerance. Non-clinical goals of treatment include bacteriologic eradication, resolution of airway inflammation, resolution of systemic inflammation and prevention of FEV1 decline. The optimal setting of treatment is determined by the severity of the patient's symptoms, baseline lung function and co-morbid medical conditions. Patients who develop respiratory acidosis and require ventilator support have a significant associated mortality risk. Physicians should have a low threshold to hospitalize patients who have severe baseline pulmonary impairment and/or significant co-morbidities. Pharmacological management includes use of bron-chodilators, antibiotics and steroids, depending on the severity of exacerbation.
Bronchodilators during acute exacerbation
Bronchodilator therapy as covered in maintenance therapy is continued during exacerbations. Short acting bronchodilators, however, are used more frequently to provide symptomatic relief and improve airflow. 62 Inhaled short-acting β2-agonists and short acting anticholinergic have comparable effects and considered superior to parenterally administered bronchodilators. 61 A bronchodilator agent from a different class should be added to a patient's regimen who has persistent symptoms with a single bronchodilator. 62 The evidence base for the addition of a methylxanthines to inhaled bronchodilators is controversial. It is clear, however, that the high incidence of adverse reactions makes it difficult to recommend its routine use for acute exacerbation of COPD.63,64
Antibiotics during acute exacerbations
Antibiotics are recommended for an exacerbation if there is an increase in sputum purulence along with one or more cardinal symptom (increase in sputum volume, or increase in dyspnea) or if the exacerbation is severe enough to warrant ventilatory support (invasive or non-invasive). Antibiotics have been shown to enhance recovery from exacerbation and reduce airway inflammatory markers. Observational studies have identified advanced age, severity of airflow obstruction, recurrent exacerbations and co-morbid cardiac disease as predictive factors for poor clinical outcomes following an exacerbation. 65 A stratified approach to antibiotic choice based on these risk factors can be implicated.66,67 Though not prospectively validated, such a stratification approach addresses judicious use of antibiotics, as well as concerns of antibiotic resistance and optimizing clinical outcome. Our approach employing these principles is shown in this Figure 3.
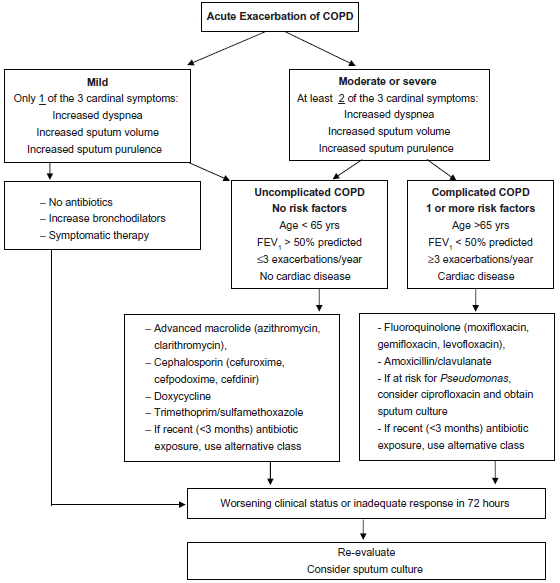
Algorithm for antibiotic treatment of acute exacerbations of COPD.
Corticosteroids during acute exacerbations
Systemic corticosteroid therapy improves lung function and treatment success, and reduces the length of hospital stay.68–70 However, they are associated with significant side effects. 69 They should be used for acute COPD exacerbations in all hospitalized patients, and should be considered for outpatient treatment of exacerbations if baseline FEV1 is <50% according to GOLD guidelines. The ideal dose and duration of steroid therapy are controversial. Generally, predni-solone of 30-40 mg per day or its equivalent is used for 7-10 days.71–73 Prolonged duration of treatment is not associated with greater efficacy and is associated with a higher risk of side effects. 69 The role of inhaled corticosteroids in COPD exacerbation has not been studied in randomized trials.
Prevention of COPD
Prevention of COPD not only includes the prevention of COPD development but also includes prevention of progression of disease and prevention of complications in established cases. Smoking cessation and vaccination are the two important interventions discussed here.
Smoking cessation
Smoking is the main risk factor for development of COPD and smoking cessation is the main stay for primary and secondary prevention which not only decreases the incidences of COPD but also decreases the rate of decline of lung function. 74 Combination of behavioral support and pharmacological treatment gives the best result for smoking cessation; we will discuss the pharmacological interventions only as per the scope of the article. Pharmacological treatments for smoking cessation can be divided in to two main groups for easy understanding: (1) Increase nicotine supply by nicotine replacement therapy (NRT) and (2) Decrease nicotine action by counteracting nicotine receptors.
Nicotine replacement therapy (NRT)
NRT works by alleviating nicotine withdrawal symptoms in smokers. There are several NRT products according to route of delivery; through skin (transdermal patch), through oral mucosa (lozenges, gums, inhalers) or through nasal mucosa (nasal spray). 75 The transdermal patch has a long half-life, slow onset of action and leads to generally stable nicotine levels over the course of 24 hours. Subjects generally have good compliance; however, the main drawback is its inability to counter acute cravings. Typically, treatment regimens include use of trans-dermal patch at its highest dose of 21 mg/day for those who smoke more than 10 cigarettes per day and 14 mg/day dose for those who smoke less than 10 cigarettes or whose weight is less than 45 Kg. Treatment is usually considered for 3-6 weeks at that dose and gradual taper over next 4-6 weeks. Supplementation with other short acting NRT products can be used as needed to alleviate nicotine cravings.
In contrast, the other nicotine replacement products that are absorbed by the mucosal surfaces have a rapid onset of action, short half-lives and variable nicotine blood concentrations, necessitating repetitive use during the day. However, they do offer flexibility for the subject's nicotine withdrawal symptoms. In general, NRT is safe and has minor side effects. Common side effects are gastrointestinal complaints, palpitations, insomnia, and skin irritation with the patch, especially if applied to the same spot daily. Compared to earlier myth no increase in the incidence of myocardial infarctions or death, or depression/anxiety was observed in a recent meta-analysis. 76 NRT should be used with caution in patients admitted to intensive care unit (ICU) as in one study NRT in a postoperative CABG surgery population resulted in a significant increase in mortality when adjusted for baseline characteristics. 77
Nicotine effect antagonism
Two main agents used for this purpose are Bupropion and Varenicline, which counteract nicotine at the nicotinic acetylcholine receptors in the central nervous system (CNS). Varenicline is a partial agonist, while Bupropion is an antagonist at these receptors. Bupropion also blocks the dopamine and norepinephrine reuptake pumps in the CNS, neurotransmitters that are intricately involved in the reward system associated with nicotine dependence. Multiple trials have demonstrated the efficacy of these medications in smoking cessation; comparisons between the two drugs have shown superior sustained abstinence rates with Varenicline. 78 Varenicline is generally used alone, whereas Bupropion can be used alone or in combination with NRT (combination therapy has been shown to have better success than Bupropion alone). The clinician needs to exercise caution when using these medications especially in patients with depression as there is an increased risk of suicidal ideations reported with both. As a result, the FDA placed a black box warning to this effect for both medications in 2009. Bupropion can lower the seizure threshold and is, contraindicated for use in patients with seizure disorders. 79 In 2011, the FDA issued an advisory that varenicline may increase the risk of adverse cardiovascular events in patients with known CVD based on a small increase in cardiovascular events (statistically not significant) reported in a study of 714 patients with stable cardiovascular disease. 80 The FDA also suggested that the known benefits of varenicline be weighed against potential harms in patients with CVD. In a recent single center study, Cytosine was found to be more effective than placebo for smoking cessation. 81 Cytosine is a partial agonist that binds with high affinity to the α4β2 subtype of the nicotinic acetylcholine receptor. One more medication in the same group is Clonidine though it's mechanism of action is different than former and it is not approved by FDA for this purpose and can be considered as second line agent. Clonidine, an antihypertensive agent, reduces central sympathetic activity by stimulating the α2-adrenergic receptors. It effectively suppresses the acute symptoms of nicotine withdrawal, such as tension, irritability, anxiety, cravings, and restlessness. 82 A Cochrane review of six clinical trials found Clonidine, oral or transdermal, more effective than placebo, with two fold higher abstinence rates. 83 Interestingly, Clonidine seems to be more effective in female smokers, although women generally respond less favorably to smoking cessation treatments. 84 The side effects of Clonidine, especially sedation, fatigue, orthostatic hypotension, dizziness, and dry mouth, limit its widespread use. 84
Vaccination
Vaccination is considered very important part of treatment of COPD as it prevents life threatening complications like pneumonia and possibly reduces exacerbations by preventing infections. Annual influenza vaccine and pneumococcal vaccine according to schedule are strongly recommended for patients with COPD. Viruses and bacteria, especially S. pneumoniae and influenza virus, are responsible for the majority of acute exacerbations in COPD patients and are associated with significant morbidity and mortality. 85 Influenza vaccine, in particular, has been shown to significantly decrease the rate of hospitalization and death in COPD patients. 86 Pneumococcal vaccine decreases the incidence of the possibly fatal invasive disease but has not been shown to reduce mortality in COPD patients.
Author Contributions
Wrote the first draft of the manuscript: HD, BD. Contributed to the writing of the manuscript: HD, BD. Jointly developed the structure and arguments for the paper: HD, BD. Made critical revisions and approved final version: HD, BD. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
Footnotes
Supplementary Data
A video abstract by the authors of this paper is available. video-abstract6563.mov
