Abstract
Type 2 diabetes mellitus (T2DM) has become an epidemic, with worldwide projections indicating that more than 336 million people will be afflicted with the disease by 2030. T2DM is characterized by inappropriately high blood glucose levels due to a deficiency in insulin secretion, action, or both. Despite the horrific complications that occur with chronic elevations of blood glucose levels, less than half of those with T2DM do not maintain proper glycemic control. Sitagliptin (Januvia, Merck and Co., Whitehouse Station, New Jersey) is a novel diabetes therapy approved for use in the U.S. and Europe. This small molecule inhibits the activity of DPP-4, a peptidase that degrades the glucoregulatory hormone GLP-1. Sitagliptin increases glucoregulation in individuals with T2DM both as a monotherapy and in combination with other antihyperglycemic drugs, with a low risk of adverse side effects.
Introduction
World Health Organization data projects that by 2030, 366 million people worldwide will have diabetes, twice the number of individuals estimated to have the disease in 2000. 1 Of those afflicted, approximately 90% will have type 2 diabetes mellitus (T2DM).2,3 Diabetes is characterized by inappropriately elevated circulating glucose levels due to defects in insulin secretion, insulin action, or both. 4 Specifically, T2DM occurs under conditions of insulin resistance, followed by β-cell failure. 5
Less than half of all individuals diagnosed with T2DM maintain long-term adequate glycemic control as defined by HbA1c of less than 7.0%. 6 While a decrease in HbA1c lowers the risk of microvascular complications, intensive blood glucose control (target HbA1c of ~6.5%) provides no additional benefit in reducing the risks of macrovascular complications in individuals diagnosed with T2DM for >8 years.7–9 Additionally, this intensive treatment increases the likelihood of weight gain, hypoglycemic events, and death due to any cause. 8 Some have suggested that increased macrovascular benefits of a HbA1c at ~6.5% may be masked by the detrimental cardiovascular effects of hypoglycemia, although at this point there no direct evidence. 10
Currently, standard care for T2DM typically results in HbA1c of ~7%–8.5%.7–9 This poses an increased risk for microvascular complications; 6 therefore, it is imperative that novel diabetes treatments are pursued. Ideally, these new diabetes therapies would decrease blood glucose levels in individuals with T2DM, have a minimal risk of hypoglycemia, be administered orally to increase the likelihood of compliance, and result in maintained or decreased patient weight.
The hormone glucagon-like peptide-1 (GLP-1) has been a prime target for novel diabetes therapies because the acute effects of the peptide decrease circulating glucose levels. GLP-1 is secreted from the L cells in the intestine after a meal. 11 In both healthy individuals and those with T2DM, GLP-1 administration increases postprandial insulin levels and slows the rate of gastric emptying.12,13 It also decreases inappropriately elevated glucagon levels often observed in individuals with T2DM. 14 These effects are only observed in the presence of hyperglycemia and euglycemia, thus decreasing the likelihood of hypoglycemic events often associated with diabetes therapies.15,16 Chronic administration of the peptide increases satiety, resulting in weight loss. 17
The active form of GLP-1 has a short half-life of 1–2 minutes due to its rapid degradation by dipeptidyl peptidase-4 (DPP-4). 18 A membrane bound form of DPP-4 is found throughout the vasculature, but predominantly in the brush-border epithelium and the gut capillary endothelium. 19 Approximately half of secreted GLP-1 is degraded by DPP-4 in the intestine before it reaches the hepatic portal vein. 20 Active GLP-1 that reaches the major vasculature is subject to degradation by a free form of DPP-4 in the plasma. 19
Currently, there are two different approaches to addressing the rapid degradation and deactivation of GLP-1. The first approach has been to develop compounds that act as GLP-1 receptor (GLP-1R) agonists, but are resistant to degradation by DPP-4. Examples include exenatide (Byetta, Amylin Pharmaceuticals, San Diego, CA and Eli Lilly and Co., Indianapolis, IN) and liraglutide (Victoza, Novo Nordisk, Bagsværd, Denmark), both of which are approved for use as T2DM therapy in the U.S. and Europe.21,22 The second approach has been to design molecules that selectively bind and inhibit the activity of DPP-4, thereby increasing the half-life endogenously released GLP-1. These include vildagliptin (Galvus, Novartis, Basel, Switzerland), which has been approved for use in Europe but not the U.S., saxagliptin (Onglyza, Bristol-Myers Squibb, New York, New York and AstraZeneca, London, UK), which has been approved in the U.S. but not in Europe, and sitagliptin (Januvia, Merck and Co., Whitehouse Station, New Jersey). Sitagliptin was the first DPP-4 to gain approval for use in individuals with T2DM in both the U.S. (2006) and Europe (2009).21,22 The remainder of this review will discuss the use of sitagliptin in treatment of T2DM.
Mechanism of Action, Metabolism, and Pharmacokinetic Profile
The degradation of GLP-1 occurs when DPP-4 cleaves a dipeptide from the
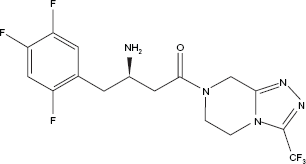
Chemical structure of sitagliptin.
In healthy individuals, across a wide dosage range (1.5–600 mg), approximately 80% of orally administered sitagliptin is excreted unchanged in urine. 26 The metabolism is not altered when the drug is administered with food. 27 When radioactively labeled [14C]sitagliptin is administered to healthy men, the radiolabel is recovered from the urine (85% of radiolabel) and feces (13%). 28 Approximately 74% of the radiolabel found in plasma, urine, and feces is in the original sitagliptin structure, while the remaining portion was accounted for in various metabolites (also found in plasma urine and feces). None of the metabolites account for more than 7% of the total radioactivity. 28
In healthy individuals, a single oral dose (ranging from 1.5–600 mg) of sitagliptin results in a dose proportional increase in AUC0-∞, a t½ of approximately 8–14 hours, and a median tmax of 1–6 hours (Table 1). 26 These parameters are not altered by the presence of food. 27 Mean DPP-4 inhibition is >80% for single doses of 50 mg or more after 12 hours and 100 mg or more after 24 hours. 26 Similar inhibition of DPP-4 occurs when sitagliptin is administered daily for more than 10 consecutive days. 29 Substantial inhibition of DPP-4 by sitagliptin increases active postprandial GLP-1 levels in normal and obese subjects by approximately 2-fold, but does not change total GLP-1 levels.26,29,30 This indicates that sitagliptin maintains the integrity of GLP-1, but does not change endogenous secretion.
Pharmacokinetic parameters with a single oral dose of sitagliptin. Comparisons are applicable for all directly preceding parameter data.
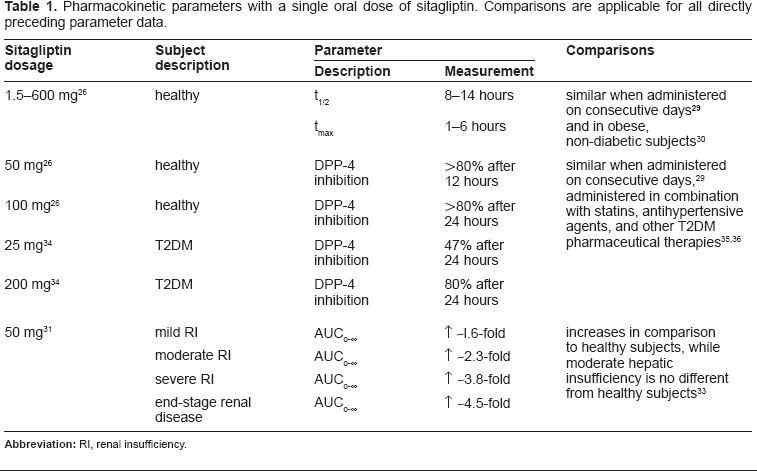
A single oral dose of 50 mg of sitagliptin administered to individuals with renal insufficiency increases AUC0-∞ values ~1.6-fold, ~2.3-fold, ~3.8-fold, and ~4.5-fold for mild, moderate, severe, and end-stage renal disease, respectively, compared to healthy individuals (Table 1). 31 Increases of >2-fold are considered clinically relevant, therefore justifying modifications in dosage. 31 Renal clearance of sitagliptin is decreased in individuals with renal insufficiency; however, drug clearance is proportional to creatinine clearance. This indicates that decreased renal clearance can be attributed to poor kidney function and is not drug specific. 31 Sitagliptin maintains its glycemic effects in individuals with T2DM and moderate renal insufficiently. 32 No changes in pharmacokinetics or clearance are observed in individuals with moderate hepatic insufficiency. 33
In patients with T2DM, a single oral dose of either 25 or 200 mg of sitagliptin inhibits DPP-4 activity (80% and 95%, 2 hours post-dose and 47% and 80%, 24 hours post-dose, respectively, all
Oral sitagliptin administration does not alter the pharmacokinetic profile or activity of statins, anti-hypertensive agents, or other T2DM pharmaceutical treatments. 36 When administered in combination, no dosage adjustments are required, with the exception of sulfonylureas. Although the pharmacokinetic profiles of sitagliptin and sulfonylureas are not altered when administered in combination, their combined insulinotropic effects may increase the risk for hypoglycemia and should be adjusted accordingly. 36
Safety
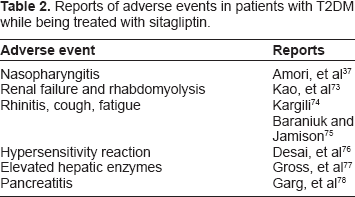
The majority of clinical studies report few or no adverse events when patients with T2DM are treated with sitagliptin. In comparison to other antihyperglyemic agents, incidence of hypoglycemia, nausea, vomiting, diarrhea, and abdominal pain are extremely low. 37 However, there have been several reports of adverse events, ranging from mild allergic reactions to pancreatitis and renal failure (Table 2). Meta-analysis of medical insurance transactions refute the reports of increased risk of pancreatitis in patients treated with sitagliptin.38,39 To further investigate these claims, a pooled analysis evaluating the treatment of 10, 246 patients with T2DM receiving either sitagliptin 100 mg q.d. (n = 5,429) or placebo/active control (n = 4,817) was conducted. 40 Results indicate that incidence rates for most specific adverse effects were similar between groups; however, increased likelihood of hypoglycemia with sulfonylurea, diarrhea with metformin, and constipation with sitagliptin treatment was reported. 40
Reports of adverse events in patients with T2DM while being treated with sitagliptin.
Clinical Studies and Efficacy
Sitagliptin vs. placebo
To determine the efficacy of sitagliptin, T2DM patients receiving oral antihyperglycemic agent (OHA) monotherapy or no prescribed therapy with HbA1c >7%, <10%, were given 100 mg q.d. sitagliptin as a monotherapy or placebo for 7 days.
41
Sitagliptin administration resulted in maximal DPP-4 inhibition (>95%).
41
Sitagliptin significantly decreased 24-hour weighted mean glucose (WMG) levels, compared to placebo (
During a 24-week, placebo-controlled trial, 100 mg q.d. sitagliptin as a monotherapy or placebo was administered to patients with T2DM with inadequate glucose control (baseline HbA1c 8%).
46
This trial indicated that continued sitagliptin treatment further augments the decrease in HbA1c from baseline (-0.61% vs. 0.18, sitagliptin vs. placebo, respectively,
Monotherapy: sitagliptin vs. metformin
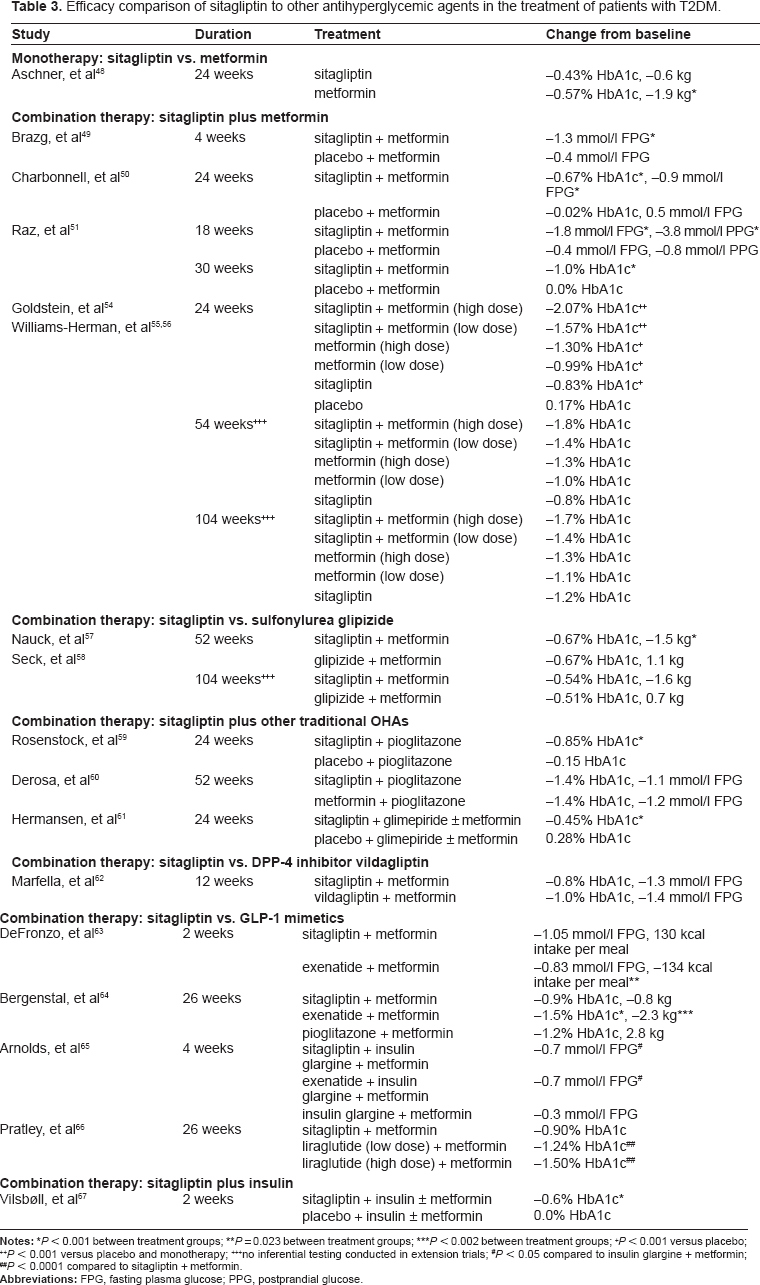
The efficacy of sitagliptin as a monotherapy has been directly compared to that of metformin (Table 3).
48
Metformin is the internationally recognized initial OHA for individuals newly diagnosed with T2DM.
16
A 24-week, double-blind, randomized, clinical trial compared sitagliptin 100 mg q.d. to metformin 1000 mg b.i.d. in 1,050 drug naïve patients with T2DM (HbA1c 6.5%–9%).
48
Results indicated that treatments with sitagliptin and metformin resulted in similar decreases in HbA1c (−0.43% vs. −0.57%, respectively). These decreases in HbA1c reached a stable maximum by 18 weeks of treatment.
48
Both treatments were generally well tolerated with low incidences of hypoglycemia in both groups. Incidences of diarrhea and nausea were significantly higher with metformin treatment,
48
a well-established side effect of the drug.
16
Body weight was reduced from baseline in both groups, but the decrease was significantly greater with metformin treatment (−0.6 vs. −1.9 kg,
Efficacy comparison of sitagliptin to other antihyperglycemic agents in the treatment of patients with T2DM.
no inferential testing conducted in extension trials;
Abbreviations: FPG, fasting plasma glucose; PPG, postprandial glucose.
Combination therapy: sitagliptin plus metformin
The efficacy of sitagliptin in combination with metformin has been extensively evaluated in patients unable to maintain adequate glycemic control with metformin alone (Table 3).49–53 Patients with T2DM with HbA1c of 6.5%–10% while on metformin, alone, received sitagliptin 50 mg b.i.d. in addition to their metformin therapy for four weeks.
49
This resulted in a significant decrease (
Long-term evaluation of the combination of sitagliptin plus metformin in individuals with T2DM has been conducted in a randomized, double-blind, clinical trial with reports at 24, 54, and 104 weeks.54–56 The initial cohort consisted of 1,091 patients with HbA1c ranging from 7.5%–11% while on a single OHA or no prescribed treatment. After a two-week run-in period, during which placebo was administered to all patients, baseline measurements were recorded. Patients were randomized into six treatment groups: sitagliptin 50 mg b.i.d. + metformin 1000 mg b.i.d. (S100 + M2000), sitagliptin 50 mg b.i.d. + metformin 500 mg b.i.d. (S100 + M1000), metformin 1000 mg b.i.d. (M2000), metformin 500 mg b.i.d. (M1000), sitagliptin 100 mg q.d. (S100), or placebo.
54
After 24 weeks of treatment, all active treatments reduced HbA1c from baseline by a significantly greater margin than placebo (
After the 24-week assessment, 885 patients participated in a 30-week extension trial (54 weeks of total treatment). 55 Subjects remained on the randomly assigned active regimen, while subjects previously receiving the placebo were placed on metformin 1000 mg b.i.d. (PBO/M2000). 55 An additional 50-week extension (104 weeks of treatment) was conducted with 685 patients, with subjects continuing with previously assigned therapy. 56 Glycemic improvements appear to be maintained at both the 54- and 104-week mark (Table 3); however, there was no statistical assessment between or among treatments.55,56 After 24, 53, and 104 weeks of treatment, the incidence of hypoglycemia was low.54–56 Gastrointestinal adverse effects in combination therapy were similar to respective metformin monotherapy, with a lower incidence rate with sitagliptin, alone.54–56
Combination therapy: sitagliptin vs. sulfonylurea glipizide
Sitagliptin administered in combination with metformin to patients with T2DM with inadequately controlled glucose levels (HbA1c ≥ 6.5% and ≤10% with metformin treatment, alone, 1500 mg per day) has been directly compared to the combination therapy of metformin and glipizide, an OHA sulfonylurea (Table 3).57,58 A double-blind, non-inferiority trial was conducted with 1,172 patients randomized into treatment groups receiving metformin plus either sitagliptin 100 mg q.d. or glipizide 5–20 mg/day (dependent on unspecified protocol-specified criteria). After 52 weeks of treatment, HbA1c was decreased by 0.67% in both groups, confirming non-inferiority.
57
Similar proportions of the treatment groups achieved the target HbA1c < 7% (63% vs. 59%, sitagliptin vs. glipizide).
57
In addition, a significantly larger proportion of subjects receiving glipizide experienced hypoglycemic events (5% vs. 32%, sitagliptin vs. glipizide,
Combination therapy: sitagliptin plus other traditional OHAs
To determine the efficacy of sitagliptin in combination with pioglitazone, an OHA thiazolidinedione (TZD), 353 individuals with T2DM with inadequate glycemic control (HbA1c ≥ 7% and ≤10%) with pioglitazone monotherapy were evaluated (Table 3).
59
Patients were randomly assigned to receive sitagliptin 100 mg q.d. or placebo treatment, in addition to their ongoing pioglitazone (30 or 45 mg).
59
After 24 weeks of treatment, sitagliptin decreased HbA1c (−0.85% vs. −0.15%, sitagliptin vs. placebo, respectively,
A non-inferiority trial compared the combination therapy of sitagliptin 100 mg q.d plus pioglitazone 30 mg q.d versus metformin 850 b.i.d. plus pioglitazone 15 mg b.i.d. in 151 patients with T2DM (HbA1c > 7.5% with pioglitazone 30 mg per day monotherapy).
60
After 9 and 12 months, both treatments significantly decreased HbA1c from baseline (−1.1% and −1.4%, sitagliptin at 9 and 12 months; −1.1% and −1.4%, metformin at 9 and 12 months, all
The efficacy of sitagliptin combined with a sulphonylurea was assessed when sitagliptin 100 mg q.d. or placebo was added to the treatment regimen of 441 individuals with inadequate glycemic control (HbA1c ≥ 7.5% and ≤ 10.5%) with glimepiride (≥4 mg/day) ± metformin (≥1500 mg/day).
61
After 24 weeks, HbA1c was significantly decrease from baseline compared to placebo (-0.45% vs. 0.28%, sitagliptin vs. placebo,
Combination therapy: sitagliptin vs. DPP-4 inhibitor vildagliptin
Thirty-eight patients with T2DM with HbA1c > 7.0% on maximal metformin treatment (3000 mg per day) received either sitagliptin 100 mg q.d. or vildagliptin 50 mg b.i.d. as add-on therapy to their continuing metformin treatment (Table 3).
62
After 3 months of treatment, both treatments significantly decreased HbA1c (−0.8% and −1.0%, sitagliptin and vildagliptin, respectively,
Combination therapy: sitagliptin vs. GLP-1 mimetics
A double-blind, randomized cross-over study in 95 patients with T2DM and inadequate glycemic control (HbA1c > 7% but <11%) on a stable regimen of metformin, compared the effect of adding either sitagliptin (100 mg q.d. for two weeks) or exenatide (5 μg q.d for one week, 10 μg b.i.d. the following week).
63
After 2 weeks of therapy, exenatide treatment resulted in significantly lower 2-hour postprandial glucose levels (11.6 mmol/l vs. 7.4 mmol/l, sitagliptin vs. exenatide, respectively,
A longer, double-blind, double-dummy, superiority comparison of sitagliptin, exenatide, and pioglitazone was conducted in 491 patients with T2DM and a HbA1c between 7.1%–11%.
64
In addition to a stable does of metformin, subjects in three treatment groups received sitagliptin 100 mg q.d. plus a weekly placebo injection, exenatide once weekly 2 mg plus daily oral placebo, or pioglitazone 45 mg q.d. plus weekly placebo injection for 26 weeks.
64
Treatment with exenatide reduced HbA1c by a significantly greater margin than sitagliptin or pioglitazone (Table 3,
Forty-eight individuals with T2DM and inadequately controlled HbA1c (7%–10%) with a stable regimen of metformin and insulin glargine, received either sitagliptin 100 mg q.d. or exenatide 5–10 μg q.d. as an add-on therapy.
65
After 4 weeks, both add-on therapies significantly decreased (
An open-label, randomized trial compared the use of sitagliptin 100 mg q.d. versus subcutaneous liraglutide 1.8 mg or 1.2 mg q.d. as add-on therapy in 665 individuals that maintained HbA1c 7.5%–10% while on a stable dose of metformin (≥1500 mg daily).
66
Both dosages of liraglutide resulted in significantly greater decreases in HbA1c from baseline, when compared to sitagliptin (−0.90% vs. −1.24% and −1.50%, sitagliptin vs. 1.2 mg liraglutide and 1.8 mg liraglutide,
Combination therapy: sitagliptin plus insulin
To determine if sitagliptin enhances glucose control in patients with T2DM that are unable to maintain adequate glycemic control with insulin ± metformin, 641 patients with HbA1c ranging from 6.6%–12.1% were given either sitagliptin 100 mg q.d or placebo, in addition to their previous therapy.
67
The addition of sitagliptin significantly decreased HbA1c from baseline, while there was no change with placebo (-0.6% vs. 0.0%,
Patient Preference
In addition to lifestyle changes, the first line of defense in the treatment of T2DM is often OHAs. Oral administration of these drugs, as opposed to injections of insulin or GLP-1 mimetics, increases compliance. 16 Oral administration of sitagliptin, in addition to low occurrence of gastrointestinal and hypoglycemic adverse events in comparison to traditional OHAs (ie, metformin and sulfonylureas), are appealing aspects of the drug. 37 The ability of sitagliptin to reduce or maintain patient weight, compared to the weight gain caused by both sulfonylureas and thiazolidinediones, 16 has the potential to enhance insulin sensitivity and cardiovascular parameters.68,69
The effects of sitagliptin are limited by the activity of endogenous GLP-1 secretion, as opposed to GLP-1 mimetics, which can activate GLP-1R at much higher levels. This is apparent when sitagliptin and exenatide are directly compared (Table 3), 64 however, once again, GLP-1 mimetics must be injected. It is likely that patients requiring more aggressive decreases in weight and HbA1c may select the injected mimetic, where patients close to their glucoregulatory goals may prefer the oral administration of sitagliptin.
For patients that no longer maintain adequate glycemic control with metformin, sitagliptin may prove to be ideal for combination therapy. Long-term clinical trials indicate that, independent of decreases in HbA1c, metformin decreases diabetes-related complications, cardiovascular events, and death from any cause. 70 Adding sitagliptin to a stable metformin regimen provides the long-term benefits of metformin, further decreases in HbA1c with the addition of sitagliptin (Table 3),49–51,–56 and maintains the ease of compliance accompanying oral administration. Patients may choose to administer each drug separately for enhanced control or use a combination tablet (Janumet, Merck and Co., Whitehouse Station, New Jersey) that has been approved recently in the U.S. and Europe for the use in individuals with T2DM unable to maintain glycemic control on sitagliptin or metformin alone.21,22
Place in Therapy
The American Diabetes Association and the European Association for the Study of Diabetes have developed an algorithm for the medical management of T2DM based on reviewing clinical trials and the clinical judgment based on the collective knowledge and clinical experience of the expert panel assembled for this task.16,71 The most recent iteration of the algorithm, indicates that upon diagnosis of T2DM, lifestyle changes in combination with metformin should be the initial treatment. 16 If glycemic outcomes are not achieved (HbA1c ≤ 7%), potential secondary measures include adding a GLP-1 mimetic, with the expectation of increase glycemic control, decreased body mass, and with low incidence of hypoglycemia, but possible side effects of nausea and vomiting. 16 At this point, DPP-4 inhibitors have not been included as part of the algorithm, because at the time of release (2009), long-term safety had not been established. 16 However, with the large number of recent peer-reviewed publications, it is possible that this decision will be revisited in the future.
The high efficacy of sitagliptin as a combination therapy with metformin (Table 3), makes it a likely candidate to serve as an add-on therapy for individuals with T2DM who are unable to maintain adequate glycemic control with lifestyle changes and metformin, alone. Sitagliptin may even be considered a viable option before the initiation of GLP-1 mimetics, as meta-analysis of clinical trials indicate that, although sitagliptin may be a more modest approach to achieving glycemic control (−0.7% vs. −1.0% change in HbA1c, sitagliptin vs. exenatide), it has fewer risks of side effects compared to both GLP-1 mimetics and other hypoglycemic agents. 37
The more modest approach to glucose control afforded by DPP-4 inhibitors may become desirable given the lack of cardiovascular protective effects7–9 and the potential increase in mortality 8 provided by intensive glucose control. It is possible that DPP-4 inhibitors, specifically sitagliptin, may be able to combat these potential hazards. It is possible that low incidence of hypoglycemia associated with sitagliptin, in comparison to other antihyperglyemic agents, 37 may reveal potential cardiovascular benefits of decreased HbA1c, that are currently masked with hypoglycemic events. 10 In addition, it has been suggested that GLP-1R activation, per se, has cardioprotective effects. 72
Although international guidelines have yet to clarify the exact role of sitagliptin in the treatment of T2DM, it is clear that sitagliptin is effective, and if deemed safe, it will likely be used in early treatment of T2DM.
Conclusions
Fewer than half of patients diagnosed with T2DM achieve glucose level low enough to decrease the risk of microvascular complications. 6 Sitagliptin is a novel therapy that has potential for enhancing glucoregulation in individuals that are unable to attain glucose control with their current regimen. Although its effects are limited by postprandial endogenous GLP-1 secreation, sitagliptin decreases HbA1c levels as a monotherapy or in combination with other antihyperglycemic agents. Sitagliptin provides an oral therapy for T2DM that has low risk of hypoglycemia and weight neutral/loss effects; all of which increase the likelihood of compliance. However, extended evaluation is still needed to address the long-term glycemic control, weight maintenance, and safety, as well as potential cardioprotective effects.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
