Abstract
The induction of glycosuria using phlorizin, a nonselective inhibitor of renal and intestinal transport was well recognised to lower glucose levels and induce calorie loss in animal models of diabetes. Phlorizin and other similar molecules however were not suitable for clinical use due to adverse effects of non selective inhibition of extra-renal glucose transport system. More recent understanding of the physiology of renal glucose transport system and increased knowledge of rare genetic syndromes of renal glycosuria has resulted in the development of drugs that selectively inhibit the Sodium Glucose Transporter-2 (SGLT2). Among the various agents currently being developed within this drug class, dapagliflozin is the most advanced in clinical development. This article discusses the basic physiology of the SGLT2 transporter system, pharmacokinetics and pharmacodynamic information of dapagliflozin, its efficacy in lowering HbA1c and weight as well as its safety and adverse effects profile. This is discussed based on evidence derived from clinical trials involving a spectrum of patients with diabetes, from drug naïve to individuals already on insulin therapy.
Introduction
Kidney plays an important role in glucose homeostasis, mainly by two mechanisms: gluconeogenesis and reabsorption of filtered glucose in the proximal convoluted tubule. 1 About 180 g of glucose is filtered by the kidneys each day and in healthy individuals, virtually all of this is reabsorbed as a function of renal regulation of glucose homeostasis. 2 Glucose reabsorption in kidneys occurs via sodium glucose co-transporter (SGLT), of which there are 6 types known to date. 3
The two most important glucose co-transporters are SGLT1 and SGLT2. SGLT1 is a high affinity low capacity transporter, responsible mainly for the absorption of glucose in the gastrointestinal tract. It is also expressed in the liver, lungs and kidneys. SGLT2 on the other hand is a low affinity high capacity transporter, found primarily in the S1 segment of proximal convoluted renal tubule, where >90% of renal glucose reabsorption occurs. 2 Transport of each glucose molecule is coupled to co transport of one sodium (Na+) ion in the kidneys and once inside the cell, glucose diffuses into blood via facilitated transport. Reabsorption of glucose in proximal tubules of kidneys is an active process requiring energy, which comes from electrochemical gradient generated by reabsorption of Na+ across the brush border and maintained by continuous transport of Na+ across the basolateral membrane into blood via Na+/K+ ATPase 4 (Fig. 1).
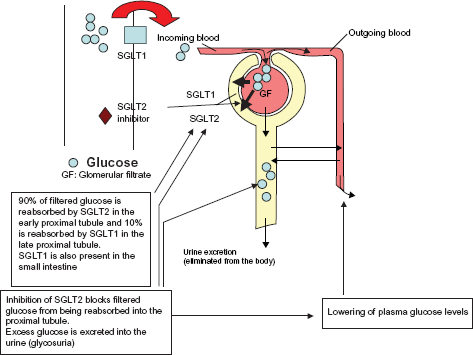
Antidiabetic mechanism of SGLT2 inhibitors.
Blocking the reabsorption of glucose in the kidneys as a strategy to treat hyperglycaemia has been the focus of recent ongoing research, though compounds causing renal glycosuria have been known for a long time. Phlorizin, a naturally occurring phenol glycoside, first isolated from the bark of apple tree in 1835, was first discovered to induce glycosuria and diuresis in 1855. In 1933, Chasis, Jolliffe and Smith showed that administering phlorizin intravenously to man increased glucose clearance via kidneys to that of simultaneous xylose and sucrose clearances. William Goldring subsequently gave oral phlorizin to healthy volunteers but found that it was much less efficient in inducing urinary glucose excretion compared with intravenous injection. 5 This relative lack of efficacy of oral phlorizin, a competitive inhibitor of SGLT1 and SGLT2, in inducing complete glycosuria is due to its poor intestinal absorption. When given orally, phlorizin causes galactosemalabsorption (via SGLT1 inhibition) and diarrhoea. Phlorizin has also been used in animals to study the mechanism of glucose transport. Lausser et al showed that phlorizin, by correcting glycaemia, significantly increased glucose clearance in alloxan-diabetic dogs. 6 Rossetti et al, by using partially pancreatectomized rats, in which fasting insulin levels remained normal but insulin response to hyperglycaemia was markedly impaired, demonstrated that hyperglycaemia per se could lead to the development of insulin resistance and that phlorizin normalized insulin sensitivity, without any change in insulin level, by correcting hyperglycaemia. 7
Despite this, the development of Phlorizin as a potential drug was not pursued due to its poor absorption from the GI tract and its non-selective inhibition of both SGLT1 and SGLT2. Therefore recent emphasis has been on developing a selective SGLT2 inhibitor, of which there are at least seven compounds currently being tested and at various stages of development. Of these, dapagliflozin is the most advanced in its clinical development. Other compounds are shown in Table 1.
List of SGLT2 inhibitor in clinical development.

Currently available treatments for diabetes mellitus
Apart from insulin, there are at least five different classes of oral and parenteral drugs currently available for the treatment of diabetes. These are:
Insulin secretagogues
sulphonylureas (SU)
Non SU secretagogues (Meglitinides)
Insulin sensitizers (thiazolidinediones)
Metformin
α Glucosidase inhibitors
GLP1 analogues/incretinmimetics.
Except for α Glucosidase inhibitors, all other drugs appear to target the pathophysiological defects seen in type 2 diabetes ie, beta cell dysfunction and insulin resistance. The side effect profile of these drugs however, may limit their use. Weight gain for example, is associated with insulin, SU and thiozolidinediones whilst hypoglycaemia is well associated with the use of insulin and sulphonylureas. Other drugs such as metformin and GLP-1 analogues may cause gastrointestinal upset and drug specific adverse effects such as oedema, cardiovascular or fracture risk may preclude the use of thiazolidinediones use in some.8,9 By virtue of their mechanism of action, SGLT2 inhibitors present a novel class of drugs, which has the potential of being used in various stages of the natural history of type 2 diabetes.
Dapagliflozin
Dapagliflozin is a C-aryl glucoside-derived SGLT2 inhibitor (Fig. 2), resistant to gastrointestinal β-glucosidase enzymes and can therefore be administered orally in an unmodified form. It is a potent (50% effective concentration [EC50] of 1.12 ± 0.065 nM against human SGLT2) and highly selective (1200-fold greater selectivity for SGLT2 versus SGLT1) SGLT2 inhibitor, and first of its class that has been tested extensively in animals and humans and has been shown to improve glycaemic profile. In-vivo studies in normal and Zucker diabetic rats 10 have shown that dapagliflozin produced dose dependent glycosuria, reduced hyperglycaemia and also improved insulin sensitivity as assessed by hyperinsulinaemiceuglycaemic clamp technique. Administration of dapagliflozin to streptozotocininduced diabetic rat, an animal model of type 1 diabetes, showed a dose dependent reduction in fasting glucose. Another study, using non-diabetic Sprague-Dawley rats, has shown that dapagliflozin induced weight loss was related to the degree of glycosuria and that weight-loss was associated with a significant reduction in body fat. 11
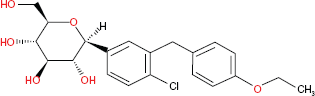
Chemical structures of dapagliflozin.
Clinical data
Studies in healthy adults–-pharmacokinetic and pharmacodynamics
Pharmacokinetic and pharmacodynamic studies have shown that dapagliflozin is adequately absorbed after oral administration and has a low overall clearance. Obermeier and colleagues enrolled 6 healthy adult males, in an open label, non randomized, single dose study. 12 Subjects were given single oral dose of [14C] dapagliflozin solution containing approximately 125 μCi of total radioactivity. Dapagliflozin was rapidly absorbed with maximum plasma concentration (Cmax) attained within one hour. Based on radioactivity recovered in urine, it was estimated that systemic absorption of orally administered dapagliflozin was about 75% within 24 hours of dosing. Maximum terminal half-life (t12) was 13.8 hours. Urinary excretion of parent dapagliflozin was very low, and therefore, contribution of renal clearance to total clearance of dapagliflozin was minimal.
Allometry was used to predict human clearance, and the predicted value (3.3 ml/min/kg) was close to observed oral plasma clearance (4.9 ml/min · kg). Dapagliflozin is therefore a low clearance compound in humans.
Studies in patients with type 2 diabetes
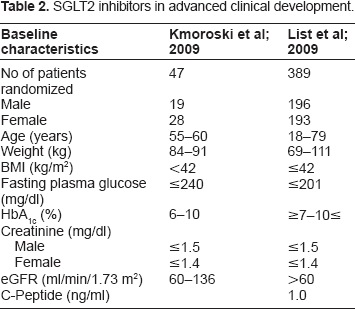
As far as efficacy and safety of dapagliflozin is concerned, studies in patients with type 2 diabetes have shown promising results. Two phase II clinical trials have looked at patients with early onset type diabetes. Baseline patient characteristics of patients in these two trials are summarised in Table 2.
SGLT2 inhibitors in advanced clinical development.
In the first study, 13 47 patients were randomised to receive dapagliflozin 5, 25 or 100 mg daily or placebo for 14 days. 18 patients were already on metformin and continued the treatment. Dapagliflozin significantly reduced fasting serum glucose and serum glucose following OGTT and produced a dose dependent increase in glycosuria. It was estimated that dapagliflozin inhibited up to 40% of filtered glucose from being reabsorbed. Hence there was absence of complete inhibition of SGLT2, which according to authors was either due to the effect of increase glucose in renal tubules competing with dapagliflozin for SGLT2 or due to the action of additional SGLT2 or SGLT1. Changes in body weight, however, were not measured as study endpoint. Two out of twenty four female participants, taking 25 and 100 mg dapagliflozin, were diagnosed with mild vulvo-vaginal candidiasis requiring 4 days treatment with miconazole. Overall, Dapagliflozin was found to be safe and well tolerated, with no reported serious adverse events.
In the second Phase II clinical trial, 14 389 treatment naive patients were randomly assigned to one of five dapagliflozin doses (2.5, 5, 10, 20 and 50 mg), Metformin XR (750 mg, up-titrated at 2 weeks to 1500 mg; therapeutic benchmark) or placebo for 12 weeks with a 2 week diet/exercise placebo lead-in and 4 week follow up period. Patients with fasting plasma glucose >13.3 mmol/L at weeks 4 to 6, >12.2 mmol/L at week 8 or >11.1 mmol/L at week 10 were discontinued. Primary outcome measure was mean HbA1c change from baseline of dapagliflozin compared to placebo at 12 weeks. Significant reductions in HbA1c and FPG were apparent with dapagliflozin compared to placebo. As expected urinary glucose excretion increased in all dapagliflozin groups. There were no deaths and no serious drug related adverse events. Also no symptomatic hypoglycaemic events were reported.
Data from phase III trials have also emerged, but most are currently in abstract form. Key inclusion and exclusion criteria are comparable across all available phase III studies, ie, type 2 diabetic patients with HbA1c between 7% and 10% and BMI ≤ 45. Included patients have evidence of residual beta cell function and exclusions are those with history of type 1 diabetes, symptoms of uncontrolled diabetes, or significant renal, liver or cardiac disease. Primary endpoint for most studies is change in HbA1c and secondary end points include reductions in fasting and post prandial plasma glucose and improvements in weight. Some studies have also highlighted reductions in systolic and diastolic B P.
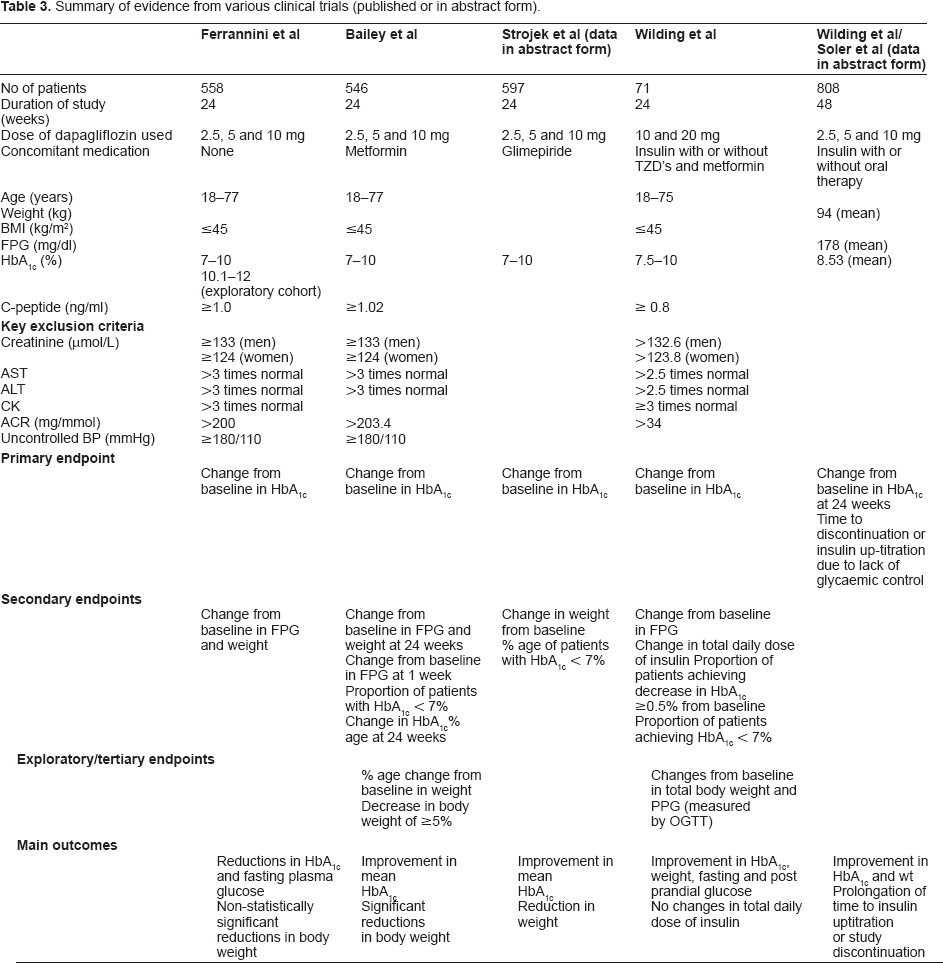
Patient population included in these trials presents a spectrum of disease, ranging from treatment naïve patients, to those inadequately controlled on single oral agent, to those with suboptimal HbA1c on a combination of insulin and insulin sensitizers. Doses of dapagliflozin used range from 2.5 mg once daily to 20 mg once daily, given in the morning, with only one study looking at the efficacy and side effect profile of evening dose. 15 Patient demographics, key inclusion/ exclusion criteria, primary and secondary endpoints and main outcomes of these studies are summarised in the Table 3.
Summary of evidence from various clinical trials (published or in abstract form).
In a another study, 15 Ferannini and colleagues investigated the efficacy and safety of dapagliflozinmonotherapy in treatment naïve patients with type 2 diabetes inadequately controlled by diet and exercise. Patients (HbA1c 7%–10%) were randomised to one of seven arms to receive placebo or 2.5, 5, or 10 mg dapagliflozin once daily in the morning (main cohort) or evening (exploratory cohort). A high HbA1c (10.1%–12.0%) cohort (n = 73), were randomly assigned 1:1 to receive 5 or 10 mg dapagliflozin once daily in the morning. There was a 2 week diet/exercise placebo lead-in period (1 week for high HbA1c cohort). Mean HbA1c reductions were apparent by 4th week (−0.58% to −0.89% for dapagliflozin, 0.23% for placebo) and maintained thereafter and statistically significant for dapagliflozin 5 and 10 mg only. Reductions in fasting plasma glucose were apparent by 1st week, more marked with 5 and 10 mg and statistically significant at 24 weeks.
In the exploratory evening cohort, changes were similar to those seen in the morning cohort. In the exploratory high HbA1c cohort, numerically greater reductions in mean HbA1c and fasting plasma glucose from baseline than in other cohorts were seen. Subgroup analysis showed that dapagliflozin produced higher HbA1c reduction in patients with high baseline HbA1c. Proportion of patients achieving therapeutic response ie, HbA1c < 7% was higher with dapagliflozin than with placebo at the end of 24 weeks. Non-statistically significant mean body weight reductions were seen in all dapagliflozin groups. Interestingly changes from baseline in fractional renal glucose excretion at 24 week were significantly related with corresponding changes in body weight. Across all treatment arms, greater renal glucose losses were associated with greater body weight loss. Increased glucose excretion persisted for full 24 week study period and was consistent with urinary loss of ~200–300 calories/day. There were no clinically meaningful changes in electrolytes and no clinically relevant changes in renal parameters including creatinine, blood urea nitrogen, cystatin C and no hypoglycaemic events were reported. There was however an increased incidence of genitourinary tract infections with dapagliflozin. Six patients in the evening exploratory cohort reported nocturia. The observed small reduction in mean systolic and diastolic BP and small rise in haematocrit (up to 2.4%) was in keeping with the diuretic effect of dapagliflozin.
In another large phase III clinical study, Bailey and colleagues 16 studied the effects of dapagliflozin in patients with type 2 diabetes who had suboptimal HbA1c level despite metformin therapy. They reported a dose dependent improvement in mean HbA1c at 24 weeks (−0.3% with placebo, vs. −0.67%, −0.70% and −0.84% with 2.5, 5 and 10 mg dapagliflozin respectively). At week 24, significant reductions in body weight were noted in all dapagliflozin groups (−2.2, −3.0 and −2.9 kg with 2.5, 5 and 10 mg respectively) compared with placebo (-0.9 kg). Patients taking dapagliflozin also showed a decrease in mean seated systolic and diastolic blood pressure. No major hypoglycaemic events requiring third party assistance were reported. No clinically meaningful changes in serum electrolytes were noted although there was a small dose dependent rise in blood urea nitrogen and haematocrit in dapagliflozin groups, consistent with its diuretic effect. There were 17 serious adverse events, 4 each in dapagliflozin groups and 5 in placebo, with no association with any treatment group.
Another phase III clinical trial (in abstract form), looking at the efficacy and safety of dapagliflozin in patients with type 2 diabetes mellitus and inadequate control on glimepiride monotherapy, 17 showed that dapagliflozin added to glimepiride, significantly improves HbA1c, reduces weight and is well tolerated. In this study, patients on at least half the maximum recommended dose of sulfonylurea alone were assigned to dapagliflozin 2.5, 5 and 10 mg or placebo, added to open label glimepiride 4 mg/day. Absolute change in HbA1c from baseline at 24 weeks (adjusted mean) was significant with dapagliflozin (-0.13% with placebo vs. −0.58% to −0.82% with dapagliflozin). Dapagliflozin also showed greater absolute change in weight compared to placebo at 24 weeks (adjusted mean), statistically significant for dapagliflozin 5 and 10 mg (-0.72 kg with placebo vs. −1.18 to −2.26 kg with dapagliflozin). Also more patients on dapagliflozin than placebo achieved HbA1c ≤ 7% at 24 weeks. Adverse events were similar across all treatment groups, while events suggestive of genital tract infection were higher in dapagliflozin groups than placebo (3.9%–9.6% vs. 0.7% with placebo). No cases of genitourinary tract infection led to discontinuation of treatment. Hypoglycaemic events were 4.8%, 7.1%, 6.9% and 7.9% with placebo, 2.5, 5 and 10 mg dapagliflozin respectively.
Two phase III clinical trials have shown the effectiveness of dapagliflozin in patients with poorly controlled type 2 diabetes, using a combination of insulin with insulin sensitizers or insulin alone. In the first study, 18 patients with type 2 diabetes mellitus, receiving high doses of insulin plus insulin sensitizers, were randomly assigned in a 1:1:1 ratio to placebo, dapagliflozin 10 mg or dapagliflozin 20 mg plus open labelled treatment with 50% of their usual insulin daily dose and oral anti diabetic treatment. No dose modification of the blinded treatment or oral anti diabetic drugs was allowed during the study; however insulin dose could be titrated up wards or down wards. Severe hypoglycaemia or lack of glycaemic control despite an increase in insulin dose greater than subject's base line dose lead to discontinuation. Patients should have been on metformin ≥1000 mg and/or pioglitazone ≥30 mg or rosiglitazone 4mg for ≥6 weeks and insulin ≥50IU of U100 for ≥12 weeks with a stable dose for ≥6 weeks. Patients with a history of type 1 diabetes, severe hypoglycaemia or symptoms of uncontrolled diabetes were excluded from the study as were patients with significant hepatic, renal or cardiac disease.
At 12 weeks, more patients in both dapagliflozin groups achieved ≥0.5% reduction from baseline HbA1c (65.2% vs. 15.8%). Mean changes in weight were significant in dapagliflozin groups (−4.3 to −4.5 kg vs. −1.9 kg with placebo). Effect of dapagliflozin on fasting plasma glucose and post prandial glucose showed a dose dependent response. There were no appreciable changes from base line in total daily dose of insulin. 4 patients in placebo group while 1 in 10 mg and 3 in 20 mg dapagliflozin groups required insulin up-titration. This trial also showed improvements in mean standing systolic and diastolic BP in both dapagliflozin groups (−6.1 to −7.2 systolic/-1.2 to −3.9 diastolic mmHg). In 20 mg group, supine BP also decreased (mean change −5.5 mmHg systolic/-5.8 mmHg diastolic).
Second study (in abstract form) shows sustained effectiveness of dapagliflozin over 48 weeks in patients with type 2 diabetes poorly controlled with insulin (mean dose 77 IU/d). 19 Patients were randomized to dapagliflozin or placebo added to their unchanged background treatment that included insulin with or without concomitant oral treatment. Adjusted mean change in HbA1c from base line at 24 weeks was sustained at 48 weeks (-0.43% with placebo vs. −0.74% to −0.94% with dapagliflozin). Body weight reductions after 24 weeks were maintained at 48 weeks even when including data for insulin up-titration (-1.5 kg with dapagliflozin vs. +0.9 kg with placebo). Total daily dose of insulin was escalated in patients taking placebo while it remained stable in dapagliflozin groups over 48 weeks (+10.54 IU/d with placebo vs. −0.70 to +0.30 IU/d with dapagliflozin). Similar reductions in fasting plasma glucose were reported with dapagliflozin with no clinically relevant increase in hypoglycaemia. There were increased reports of signs and symptoms suggestive of urinary (7.9%–10.8% vs. 5.1% with placebo) and genital (6.4%–10.7% vs. 2.5% with placebo) tract infections with dapagliflozin.
After the first 24 weeks, investigators looked at the rate of insulin up-titration/study discontinuation from lack of glycaemic control. 20 Insulin was uptitrated if HbA1c was >8% or FPG was >9.9 mmol/L from 24–48 weeks. Weight gain, peripheral oedema and discontinuation due to hypoglycaemia and other causes was also assessed. The authors concluded that the time to reach discontinuation/up titration was substantially prolonged in all dapagliflozin + insulin groups vs. placebo + insulin group. At 48 weeks proportion of patients with discontinuation/up-titration was 42.8% in placebo + insulin group vs. 15.3%–21.7% in dapagliflozin + insulin groups. Frequency of peripheral oedema was 7.6% in placebo + insulin group vs. 2.4%–4.6% in dapagliflozin + insulin groups. Frequency of discontinuation due to adverse events was 4.6% in placebo + insulin groups vs. 3.5–5.1 in dapagliflozin + insulin groups. There was no discontinuation due to hypoglycaemia.
Clinical use of dapagliflozin
Based on currently available clinical data, dapagliflozin seems to be an attractive new addition to the available treatments for type 2 diabetes. An important issue relates to its favourable effect on weight. Almost all the available studies indicate that dapagliflozin produced some degree of weight loss, albeit some studies reported statistically non-significant change due to the studies not adequately powered to assess weight loss. One phase III study 15 showed that weight loss is likely to occur as a direct effect of dapagliflozin induced glycosuria. Given that caloric loss may be counter-balanced by overeating, it will be interesting to see if weight loss persists in the long term and whether it is associated with caloric loss and reduction in adiposity. In animal studies dapagliflozin has been shown to reduce adiposity, 11 while veterinary literature shows that chronic administration of phlorizin induces lipolysis in lactating cows. 21
Another attraction of dapagliflozin as a pharmacological option for patients with type 2 diabetes is the fact that its use is not associated with an increased risk of hypoglycaemia. This observation is not only based on data from clinical trials thus far, but also from experiences gained with the genetic disorder of familial renal glycosuria. Furthermore, individuals with familial renal glycosuria showed no increased risks of dehydration, electrolyte imbalance or urinary tract infection, despite reporting significantly greater amount of glycosuria than that reported with SGLT2 therapies. 22 The dose of dapagliflozin used in clinical trials ranges from 2.5 to 20 mg once daily. Majority of trials have used 10 mg as their maximum dose, though one phase II study 13 used 100 mg as maximum dose and reported no serious adverse events.
Adverse effects
Dapagliflozin appears to be well tolerated when used alone or in combination with other anti diabetic treatments. Although none of the trials have reported any serious adverse events or clinically meaningful changes in serum electrolytes and biochemical markers of renal function, these reports has to be taken with a note of caution. All the available data is from short term trials in relatively fit subjects. It will be important to see what happens to all these parameters with the long term use of dapagliflozin, especially when used in combination with other drugs. Another important consideration is the elderly people with type 2 diabetes, as current data is derived primarily from middle aged population.
As seen above, symptoms and signs suggestive of genital and urinary tract infections have been reported more frequently with dapagliflozin. Although these have not lead to treatment discontinuation in any trial, yet it will be interesting to see the impact of long term use of dapagliflozin on the incidence of genito-urinary infections in a population. One clinical trial 14 reports a slight increase in mean 24 hr urinary output, but not enough to have any clinical impact. Furthermore, with the exception of one study, 15 where dapagliflozin was administered in the evening, none of the other trials report nocturia. In addition, none of the clinical trials reported incidence of severe hypoglycaemia irrespective of drug combinations.
Summary
Dapagliflozin is a promising new drug for patients with type 2 diabetes. We believe that the drug is likely to be most effective when given early in the course of the disease (by virtue of reducing hyperglycaemia induced beta cell toxicity), but also, in view of the fact that SGLT2 inhibitor works independently of insulin, it is likely to be effective in the later stages of diabetes, including as a combination therapy with insulin (Fig. 3). Given the rising prevalence of obesity, it's beneficial effect in inducing weight loss is particularly attractive, but further adequately powered study with analysis on the relationship between weight loss and the degree of glycosuria is required to justify this preliminary observation and to explain the possible mechanism of weight loss. The lack of hypoglycaemia associated with its use is a major advantage particularly due to recent concerns about the risks of adverse cardiovascular outcome as a result of hypoglycaemia when striving to achieve low HbA1c targets. 23 Since SGLT2 inhibition is associated with relevant diuretic effects, the use of this drug may be advantageous in patients with hypertension or used in conjunction with thiazolidinediones. Whist evidence from individuals with familial renal glycosuria syndrome is reassuring, it is important to note that these individuals do not have diabetes. Thus, further short and long-term data remains essential to assess the safety of this drug in patients with type 2 diabetes with regards to the increased risks of genital and urinary tract infections as well as renal function.
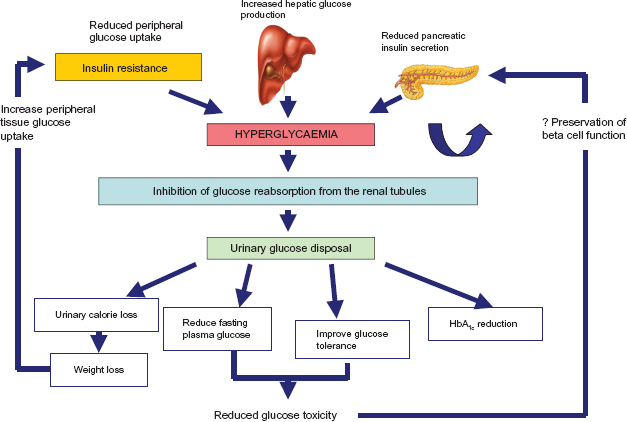
Schematic representation of the clinical effects of SGLT2 inhibitor. Irrespective of the underlying causes of hyperglycaemia in patients with diabetes, inhibition of tubular urinary glucose reabsorption leads to urinary glucose excretion and concurrent plasma glucose lowering. Urinary glucose ‘calorie’ loss leads to weight loss and reduction in insulin resistance while blood glucose lowering reduces the effects of glucose toxicity on the pancreatic beta cell–thus potentially preserving beta cell function.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
