Abstract
We conducted a systematic review and meta analysis of randomized controlled trials of galantamine hydrobromide in the treatment of mild to moderate dementia. Following a literature search and screening process, we included 15 trials and five companion papers in the review. Moderate-quality evidence suggested galantamine-treated persons generally had better outcomes than placebo-treated persons after a maximum 6-month follow-up. Outcome domains included cognitive function, global function, behaviour and mood, and activities of daily living. The evidence requires careful interpretation because ‘better outcomes’ can mean less deterioration, rather than improvement, relative to placebo. Galantamine has not been shown to halt dementia progression nor reverse disease course. The most frequently reported harms were nausea, diarrhea, and dizziness. Reported rates of these harms were highly variable (range, 0%–40%); reporting was at high risk of bias because authors rarely specified the frequency or timing of harms assessment, nor did they report active methods of collecting harms.
Introduction
Dementia is a major public health concern because of its chronic nature, rising prevalence, and high cost to society. 1 No cure exists for most dementias. Clinical management of common dementias (ie, Alzheimer's disease [AD], vascular dementia [VaD], and mixed dementia [MD]) often includes pharmacotherapy. The United States Food and Drug Administration (FDA) has approved five medications to treat the symptoms of cognitive decline in AD; clinicians also prescribe these medications to treat other dementias.
Four of the five approved medications are cholinesterase inhibitors (ie, galantamine, donepezil, rivastigmine, tacrine). These medications degrade acetylcholinesterase and increase acetylcholine, which is important for the neurons involved in cognition. The fifth approved medication, memantine, helps memory and learning through blockage of the N-methyl-d-aspartic acid receptor and prevention of excess stimulation of the glutamate system.
Galantamine (C17H21 NO3) has a molecular mass of 287.35 g/mol and is a tertiary alkaloid that can be made synthetically or extracted from plants such as the caucasian snowdrop or narcissus species. Only oral administration has been approved for clinical use and absorption is high with bioavailability ranging from 85% to 100%. Plasma elimination half-time is five to seven hours and 75% of total clearance is hepatic. 2
We conducted a systematic review and meta analysis of the evidence for the efficacy and safety of galantamine in dementia.
Methods
Literature search
We searched the Cochrane Central Register of Controlled Trials, MEDLINE, PREMEDLINE, EMBASE, Allied and Complementary Medicine Database, CINAHL, AgeLine, and PsycINFO for relevant articles published in English from January 1986 through November 2009. We also reviewed the bibliographies of retrieved articles.
We included randomized controlled trials comparing galantamine to placebo or another medication. The trials had to involve populations diagnosed with a major dementia such as AD or VaD. We excluded crossover trials because of possible bias due to period effects or period-by-treatment interactions. Table 1 shows the complete list of inclusion/exclusion criteria. Two reviewers independently applied the inclusion/exclusion criteria to each article retrieved in the literature search.
Inclusion and exclusion criteria.

Trial outcomes encompassed four domains: cognitive function, global function, behaviour and mood, and activities of daily living (ADLs).
Assessment of article quality
Two reviewers independently assessed article quality using the Jadad scale. 12 Reviewers awarded one point for each ‘yes’ answer to the following questions: authors described their study as randomized, authors described their study as double-blinded, and authors provided a description of withdrawals and drop-outs. Additional single points were awarded if the methods of randomization or double-blinding were described and methodologically appropriate (maximum score = 5). Single points were deducted if the methods of randomization or double-blinding were described and methodologically inappropriate.
The McMaster Quality Assessment Scale of Harms (McHarm) was used to assess the quality of reporting harms in the trials. McHarm has 15 items covering specification of harms, methods of collecting data on harms, and reporting of harms. 13
Meta analysis
We conducted a meta analysis to obtain summary estimates of the efficacy of galantamine versus placebo. We divided the meta analysis into four sub-analyses based on the outcome domains noted above and calculated a summary odds ratio (OR) for each sub-analysis. We chose the OR to report study results because of its ease of interpretation relative to measures of the standardized mean difference between groups (eg, Cohen's d or Hedges’ g).
To calculate ORs, we used binary event data from the published trial reports to develop 2 x 2 tables; we calculated 95% confidence intervals using the constant chi-square boundaries method. 14 When binary event data were unavailable, we used the formulas in Borenstein et al 15 to calculate standardized mean differences (SMDs [Cohen's d]) between treatment groups. SMDs were based on the mean differences in scale score from baseline to the end of follow-up in individual treatment groups. Since dementia is a syndrome, trial outcomes are typically measured using changes in score on scales such as the Alzheimer's Disease Assessment Scale-Cognitive Sub-scale (ADAS-cog). 16 We used formulas in Borenstein et al 15 to convert SMDs into log ORs and calculate 95% confidence intervals. We used Excel 2003's exponent function (Microsoft Corporation, Redmond, WA) to convert log ORs into ‘standard’ ORs with a null value of 1.00.
The OR computations were conducted such that ORs greater than 1.00 would indicate an increased odds of better outcomes (eg, improvement, less deterioration) in galantamine-treated versus placebo-treated persons. For example, we combined outcomes on the Clinician Interview-based Impression of Change with caregiver input (CIBIC-plus) scale 17 into two categories: improved/stabilized or deteriorated. Since persons with dementia deteriorate over time, we included stabilization with improvement. An OR above 1.00 would mean that persons treated with galantamine have greater odds of being improved or stabilized versus persons treated with placebo.
Data for OR calculations came from the results of intent-to-treat, observed case, or last observation carried forward analyses, in that order, depending on the type of analyses conducted in the trials.
To obtain summary ORs, trial-specific ORs were combined with a random effects model 18 that was weighted by the inverse variance of each effect estimate. Between-study statistical heterogeneity was measured using the I 2 statistic. 19 Review Manager v5.0 (The Nordic Cochrane Center, The Cochrane Collaboration, Copenhagen, Denmark) was used to perform the meta analysis.
Trials included in any of the four sub-analyses of the meta analysis had to contain a galantamine-placebo comparison, report data on the outcome domain in question, and include data that could be used to calculate an OR. In trials with more than one galantamine group, we selected the highest dose group as the treatment arm in the meta analysis.
Results
A total of 43,686 citations were retrieved in the literature search. Of these, 15 publications were randomized controlled trials of galantamine that met our inclusion criteria. We extracted data from these 15 trials, as well as from five companion papers containing supplementary results related to some of these trials. Figure 1 details the flow of articles through the retrieval and screening process.
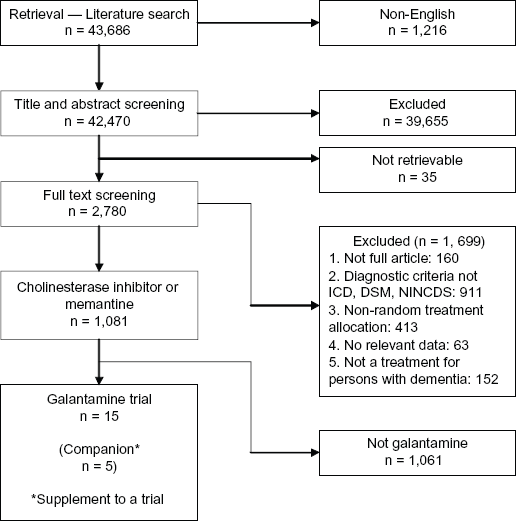
Flow diagram.
Twelve of the extracted trials compared galantamine to placebo.20–31 Additionally, the five companion papers32–36 all reported supplemental data for galantamine-placebo trials. A total of 4,393 patients completed the trials (range, 17–768 patients). Eight trials included persons with AD only. Four others included persons with AD and cerebrovascular disease, 22 AD and VaD, 24 VaD, 20 or frontotemporal dementia by primary progressive aphasia. 25 The trials titrated galantamine doses up to 24, 32, or 36 mg per day. One trial compared extended-release (once-daily) galantamine and the usual twice-daily formulation to placebo. 21 Double-blind follow-up periods ranged from eight 25 to 26 weeks. 21 Further trial data are shown in Table 2.
Study characteristics and effect sizes for outcomes measured within each study.
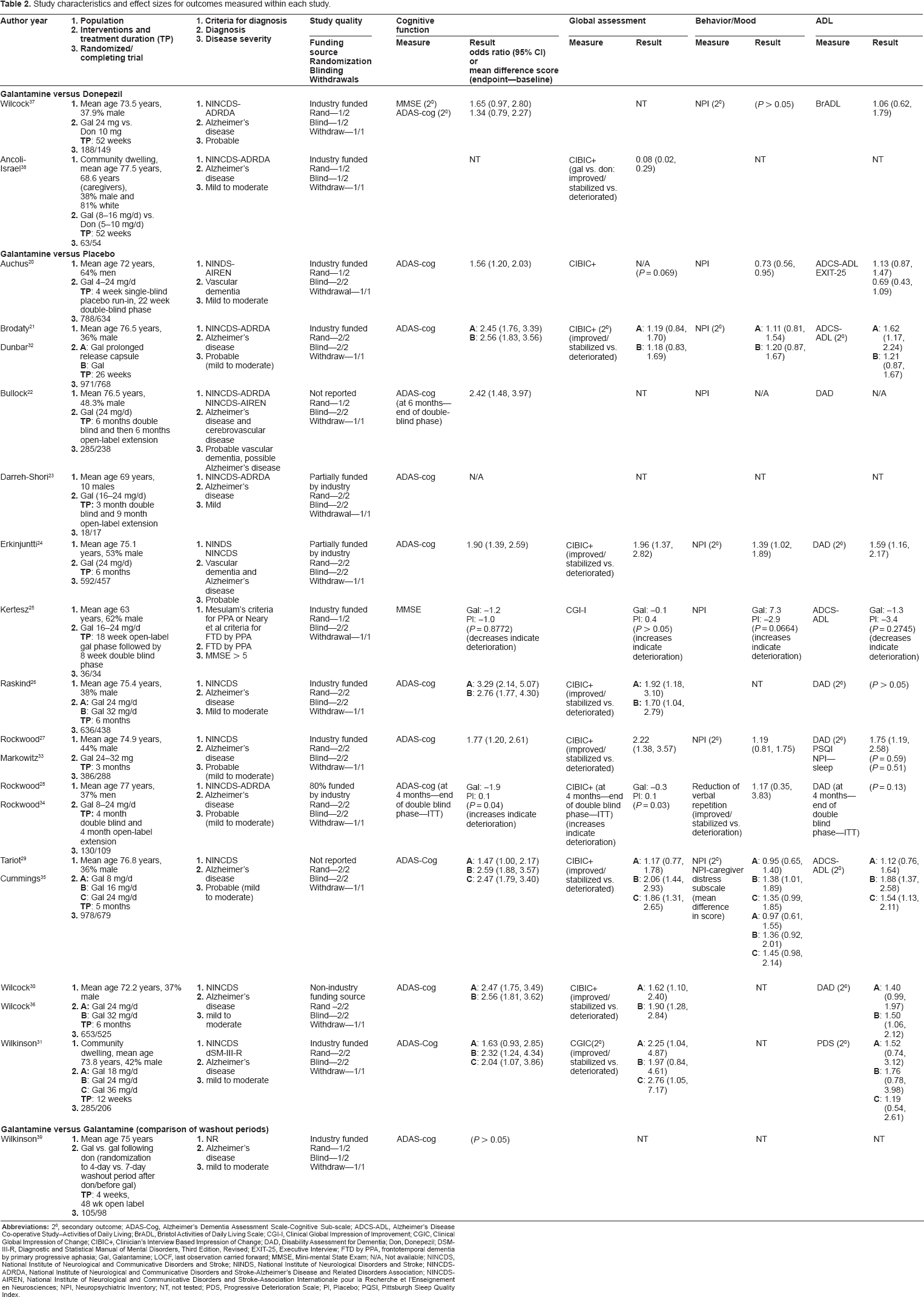
All 12 trials measured cognitive function: 11 trials used the ADAS-cog; one trial 25 used the Mini Mental State Examination (MMSE). 4 Global function, measured in 10 trials, was assessed using a group of related scales, ie, CIBIC-plus, Clinical Global Impression of Change (CGIC), 17 or Clinical Global Impression of Improvement (CGI-I). 40 Eight trials measured behaviour and mood; all except one 28 used the Neuropsychiatric Inventory (NPI). 41 The lone exception measured a specific behaviour, ie, reduction of verbal repetition, using goal attainment scaling (GAS). 42 Eleven trials measured ADLs, primarily using the Alzheimer's Disease Co-operative Study-Activities of Daily Living (ADCS-ADL) Scale 43 or the Disability Assessment for Dementia (DAD). 44 The details of these scales are described elsewhere.45,46
In general, galantamine-treated persons had better outcomes than placebo-treated persons in all four outcome domains. These results were consistent across trials. Most galantamine-placebo comparisons were statistically significantly different at the 5% level.
In the meta analysis (Fig. 2), the strongest treatment effect was in cognitive function (summary OR = 2.14; 95% confidence interval = 1.85, 2.48). Only one summary OR, for behaviour and mood, was not statistically significant at the 5% level. Heterogeneity was low (I 2 ≤ 30%) in three of four sub-analyses.
The I 2 was high (ie, 70%) for behaviour and mood. We felt a single trial contributed most of the heterogeneity to this domain. The trial was a study of 634 persons with VaD. 20 Despite the large sample size, the authors reported statistically non-significant results for global function and ADLs. We removed this study from the behaviour and mood sub-analysis and re-computed a summary OR of 1.29 (95% confidence interval = 1.09, 1.52) and an I 2 of 0% (not shown in Fig. 2).
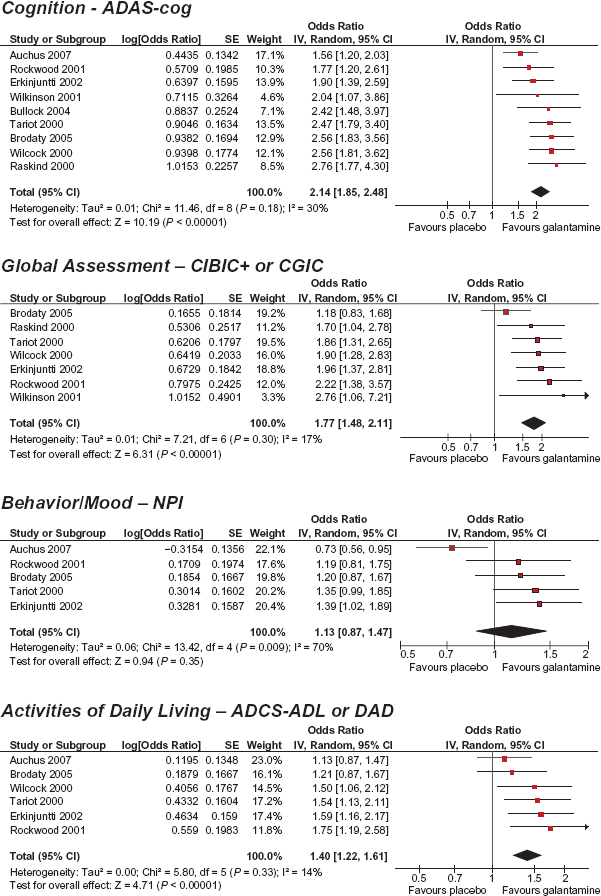
Meta analysis.
Three trials involved comparisons other than galantamine versus placebo.37–39 In one trial (Table 2), persons who received donepezil for at least 12 weeks were switched to galantamine following randomization to 4- or 7-day washout periods. After four weeks of treatment with galantamine, both ‘washout’ groups had similar mean score differences on the ADAS-cog (P > 0.05). 39
Two trials compared galantamine and donepezil (Table 2).37,38 In total, 203 persons completed these trials after 52 weeks of follow-up. In the first trial, galantamine (24 mg) was not statistically significantly better than donepezil (10 mg) on any outcome, although the galantamine group showed trends toward better cognitive and ADL outcomes. 37 In the second trial, global function was more likely to deteriorate in galantamine-treated versus donepezil-treated persons. 38 However, the galantamine dose in the second trial (ie, 8–16 mg) was lower than the standard recommended dose of 24 mg (donepezil patients received 5–10 mg in the second trial).
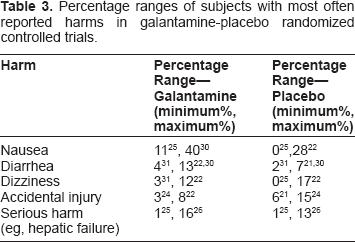
Specific harms were reported in all except two trials.23,39 The most often reported harms were nausea, diarrhea, and dizziness. Other reported harms included accidental injury, urinary disorder, and ‘serious’ harms such as hepatic failure. Percentages of subjects reporting these harms in galantamine-placebo trials are shown in Table 3.
Percentage ranges of subjects with most often reported harms in galantamine-placebo randomized controlled trials.
Numbers of subject withdrawals due to harms were reported in all except one trial. 39 In the galantamine-placebo trials, withdrawals ranged from 6 25 to 54% 31 in active treatment groups and 0 23 to 17% 22 in placebo groups.
Article quality
Article quality was generally good (Table 2). Nine published trial reports described appropriate methods of randomization and blinding;21,23,24,26–31 all 15 trials described withdrawals (though one trial did not report withdrawals due to harms). 39 Six trials mentioned subjects were randomized, although they omitted descriptions of appropriate methods of randomization;20,22,25,37–39 three trials reported blinding without describing appropriate types of blinding.37–39
All eight galantamine versus placebo trials in AD alone, plus the AD and VaD trial, scored the maximum 5 points on the Jadad scale. The galantamine versus donepezil37,38 and washout period 39 trials did not describe appropriate methods of randomization or blinding. The VaD, 20 AD and cerebrovascular disease, 22 and frontotemporal dementia by primary progressive aphasia 25 trials did not report appropriate methods of randomization.
Figure 3 shows the proportion of trials with potential bias in reporting harms. Trials were consistently poor at defining serious and severe harms a priori and most did not specify whether standardized checklists were used to record harms. Similarly, the majority of trials did not indicate who collected data on harms, nor the training of these persons. Approximately 30% of trials did not describe the frequency or timing of harms collection; approximately half of the trials did not explicitly state that subjects were being actively probed for the presence of harms.
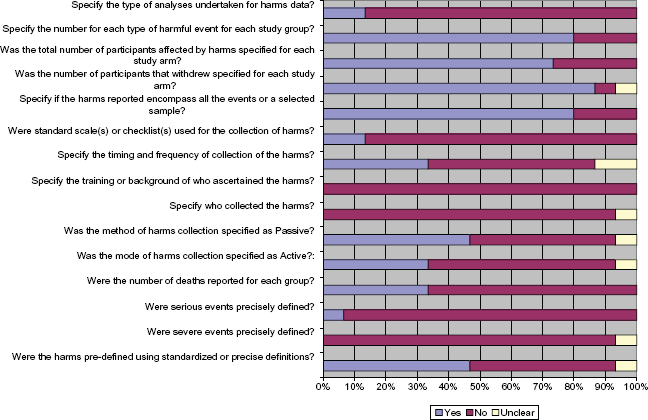
Percentage of randomized controlled galantamine trials with risk of bias in reporting harms.
The majority of trials reported the types and frequency of harms as a function of treatment group. Only two trials22,24 explicitly stated the method of analyzing harms (ie, frequency methods), although authors did not statistically compare between-group differences in harms.
Discussion
Extracted trial data (Table 2) and the meta analysis (Fig. 2) indicate that galantamine-treated persons on average have better outcomes than placebo-treated persons in cognitive function, global function, behaviour and mood, and ADLs. For comparison against donepezil, the evidence for galantamine is equivocal
The galantamine-placebo results should be interpreted cautiously. While results were generally better in galantamine groups, cognitive and functional decline is irreversible in most dementias and cholinesterase inhibitors do not halt or reverse disease progression. 47 Thus, better outcomes for galantamine often mean that the rate of deterioration in galantamine-treated persons is lower than in placebo-treated persons. Also, dementia is a chronic condition with median survival of 3.1 years for probable AD and 3.5 years for VaD. 48 However, the galantamine-placebo trials all had short follow-ups (≤26 weeks), so no comparative evidence exists to indicate whether the benefits of active treatment remain over the entire course of disease.
Another interpretation issue concerns the use of scales to measure efficacy in galantamine trials. Many galantamine-placebo comparisons, all based on changes in scale score, were statistically significant at the 5% level. However, statistically significant differences in scale score (and ORs > 1.00) do not necessarily translate into clinically meaningful differences between groups.
Researchers have established minimum clinically important differences (MCIDs) in scale score for some instruments used in dementia drug trials (ie, ±4-point change in ADAS-cog, 49 ±3.72-point change in MMSE). 50 Secondary analyses of double-blind galantamine data and open-label donepezil data at the group level found a 4-point change on the ADAS-cog to be associated with similar changes on the CIBIC+ or GAS.51,52 In the 15 extracted trials, none of the group differences in ADAS-cog score exceeded four points.
Researchers have conducted little work to establish MCIDs for other scales used in dementia drug trials, so the clinical value of statistically significant results based on these scales remains uncertain.
The reporting of harms within the 15 extracted trials was potentially biased, especially when defining serious and severe harms. Although authors generally reported the number of harms, they rarely specified the method (ie, frequency, timing) of reporting. This places the studies at risk of missing harms, particularly because active methods of collecting harms were rarely undertaken. Overall, we found a high risk of misclassification bias and underreporting of harms in the extracted trials.
Earlier systematic reviews of galantamine53,54 identified four areas for further research. These areas included studying persons with severe dementia, conducting follow-ups longer than six months, undertaking head-to-head comparisons with other cholinesterase inhibitors or memantine, and ascertaining drug effects on incidence and timing of institutionalization. Despite our inclusion of seven trials20,21,23,25,28,38,39 published after the earlier reviews, these four areas are still largely bereft of research.
Another important research gap is the lack of caregiver-related outcomes in galantamine trials. Caregivers are the ‘hidden victims’ of dementia 55 and they take on a greater role in treatment decision-making as their loved ones progress to more severe disease. Galantamine's impact on patient cognition, behaviour and mood, and ADLs can affect whether caregivers support and ‘promote’, or discourage, drug therapy. Also, dementia caregivers report lower quality-of-life and greater health challenges than non-caregivers.55,56 If galantamine can improve patient outcomes, then perhaps caregiver burden could decrease, thus improving caregiver health and delaying institutionalization. 57
While caregiver outcomes are germane to assessing the utility of galantamine, a consensus is needed to establish which outcomes are important and how to measure them. Trials for other cholinesterase inhibitors have examined caregiver stress and the amount of time devoted to caring for a loved one. 58 However, use of scales to measure constructs like stress requires determination of MCIDs and some evidence suggests caregivers cannot accurately estimate the time they devote to caregiving. 59
Conclusion
The moderate-quality evidence from 12 randomized controlled trials shows galantamine-treated persons have better outcomes than placebo-treated persons after a maximum 6-month follow-up. Outcomes included cognitive function, global function, behaviour and mood, and ADLs. Evidence from two galantamine versus donepezil trials does not favour one treatment over the other. A 4-day versus 7-day washout period was not shown to affect outcomes for galantamine-treated persons who received donepezil for at least 12 weeks prior to the washout period.
Evidence favouring galantamine must be interpreted carefully because ‘better outcomes’ sometimes means less deterioration than placebo and changes in scale score cannot easily be translated into clinically meaningful outcomes. Further research is required to assess galantamine's efficacy in more severely affected populations, over longer periods than six months, in head-to-head comparisons with other dementia medications, and on the incidence and timing of institutionalization. Caregiver outcomes are also important to assess in future research.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
Mark Oremus holds a Career Scientist Award from the Province of Ontario's Ministry of Health and Long-Term Care and the McLaughlin Professorship in Population and Public Health. Parminder Raina holds the Raymond and Margaret Labarge Chair in Research and Knowledge Application for Optimal Aging.
