Abstract
Objective
Oxybutynin chloride topical gel (OTG) significantly improved overactive bladder (OAB)–related urinary symptoms and was well tolerated in a phase III double-blind, placebo-controlled trial (ClinicalTrials.gov identifier NCT00350636). This study further evaluated the effect of OTG on health-related quality of life (HRQoL) in adults with OAB.
Methods
Adults aged 18 years or older with urge-predominant urinary incontinence were assigned randomly to OTG 1 g/day or matching placebo for 12 weeks. HRQoL was measured by the Incontinence Impact Questionnaire (IIQ) and the King's Health Questionnaire (KHQ) at baseline and at weeks 1, 4, 8, and 12. Between-group differences in IIQ or KHQ score changes from baseline to week 12 or last observation were compared by analysis of covariance.
Results
Of 789 study participants (704 women, 85 men), 389 received OTG and 400 received placebo. Mean age was 59 ± 12 years. IIQ total score improved significantly more in participants treated with OTG (P = 0.0005) than in those treated with placebo, as did scores on all four IIQ subscales: Emotional Health (P = 0.0002), Social Relationships (P = 0.0019), Travel (P = 0.0068), and Physical Activity (P = 0.0078). KHQ scores improved significantly more with OTG than with placebo in the more incontinence-specific domains: Incontinence Impact (P = 0.0023), Symptom Severity (P = 0.0024), Severity (Coping) Measures (P = 0.0058), Sleep/Energy (P = 0.0061), Role Limitations (P = 0.0133), and Personal Relationships (P = 0.0489).
Conclusion
Treatment with OTG resulted in significant HRQoL improvements in adults with OAB.
Introduction
Overactive bladder (OAB) is a prevalent condition characterized by the symptoms of urinary “urgency, with or without urge incontinence, usually with frequency and nocturia”. 1 OAB affects about one-sixth of US adults 2 ; its prevalence is similar among men and women and increases with age. 2 Treatment options for OAB-related symptoms include behavioral therapy, pharmacotherapy, or a combination.3,4 Oxybutynin and other antimuscarinic agents are the mainstay of pharmacotherapy. 5 Oral oxybutynin formulations are effective but associated with relatively high incidences of anticholinergic adverse effects, particularly dry mouth, which affected between 17% and 87% of patients in clinical studies, depending on dose and type of oral formulation. 5 One-year medication adherence rates for immediate-release and extended-release oxybutynin formulations in clinical practice were reported to be 14.8% and 23.5%, respectively. 6 The higher adherence rate for the extended- release formulation likely is attributable to its greater anticholinergic tolerability. 6 Comparative clinical and pharmacokinetic studies provided compelling evidence that transdermal compared with oral administration of oxybutynin is associated with improved anticholinergic tolerability because of the avoidance of presystemic metabolism.7,8
Oxybutynin chloride topical gel (OTG) is a newly approved transdermal formulation of oxybutynin9,10 that contains 10% oxybutynin chloride by weight. OTG has a small dosing volume (about 1 mL/dose), is colorless and fast drying, and leaves no residue. In a recent 12-week phase III study in adults with OAB with urge-predominant urinary incontinence, OTG significantly improved OAB-related symptoms compared with placebo, was safe, and generally was well tolerated. 11 Dry mouth, the most common anticholinergic adverse event (AE), occurred in 2.8% of participants receiving placebo and in 6.9% of those treated with OTG. Incidences of other treatment-related anticholinergic AEs were low and statistically similar for OTG and placebo. 11
OAB is a debilitating chronic condition that has a significant negative effect on health-related quality of life (HRQoL), including physical and mental well-being. 12 Urinary symptoms of OAB, particularly OAB with incontinence, reduce mobility and negatively affect social interactions, work productivity, and sexual activity.2,13–15 As a result, OAB with incontinence significantly increases the incidence and severity of depression.2,14 Therefore, it has been suggested that patient-reported outcomes, such as HRQoL, should be included in the design of studies evaluating treatments for OAB. 16 Disease-specific questionnaires are more reliable than generic HRQoL questionnaires in evaluating the impact of a specific medical condition on HRQoL. With proven validity and reliability, the Incontinence Impact Questionnaire (IIQ) and the King's Health Questionnaire (KHQ) are two urinary incontinence–specific questionnaires highly recommended by the International Consultation on Incontinence. 17 As disease-specific questionnaires, they also are sensitive in detecting patient-perceived changes in OAB-related symptoms caused by study treatment.
HRQoL was a secondary end point of the OTG phase III study. Here, we present a prospective evaluation of the treatment effects of OTG compared with placebo on HRQoL assessed by the IIQ and the KHQ.
Methods
Study design and treatments
Study design and treatments have been described previously. 3 In brief, this was a multicenter (76 centers), parallel-group, phase III study (OG05009; ClinicalTrials.gov identifier NCT00350636) conducted in accordance with Good Clinical Practice, health privacy standards, and the Helsinki Declaration of the World Medical Assembly. After an initial screening period of 3 to 4 weeks including a 2-week washout period for prior OAB pharmacotherapy, eligible patients were randomized 1:1 to receive OTG or placebo gel for 12 weeks. The gel (1 g) was applied daily to rotating sites on the abdomen, upper arms or shoulders, and thighs. At the first study visit (beginning of screening; Fig. 1), a component of behavioral interventions, including bladder and fluid management techniques, was taught to all participants. HRQoL baseline evaluation with the IIQ and KHQ instruments was performed at the end of screening. Participants also completed HRQoL questionnaires when returning to the clinic on an outpatient basis 1, 4, 8, and 12 weeks after initiation of treatment (Fig. 1).
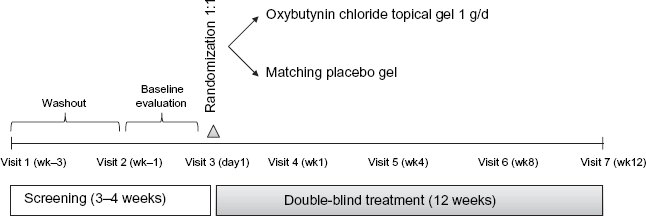
Study schema.
Study participants
Eligibility criteria have been described. 11 Briefly, study participants were men and women aged 18 years or older with OAB symptoms of urinary urgency, urge urinary incontinence, and urinary frequency, with or without neurologic disease. Eligible adults had a history of pure urge or mixed urinary incontinence with a predominance of urge incontinence episodes (on baseline diary), recorded average of at least eight urinary voids per day and at least four urge urinary incontinence episodes during a 3-day baseline diary period, mean recorded urine void volume of 350 mL/void or less during the 2-day urine collection of the 3-day baseline urinary diary, and postvoid residual urine volume of 250 mL or less.
HRQoL assessment
Main HRQoL outcome variables were changes in IIQ and KHQ scores from baseline to study end. The IIQ 18 includes 30 response items regarding the extent to which urinary incontinence affects varying activities and feelings. Possible responses to each item are “not at all”, “slightly”, “moderately”, and “greatly”. Mean scores were calculated for four subscales: Travel, Physical Activity, Social Relationships, and Emotional Health. Subscale scores range from 0 to 100; the total score is the sum of all subscale scores, ranging from 0 to 400.
The KHQ 19 consists of 32 response items that address 10 domains of HRQoL: General Health Perception, Incontinence Impact, Symptom Severity, Role Limitations, Physical Limitations, Social Limitations, Personal Relationships, Emotions, Sleep/Energy, and Severity (Coping) Measures. The General Health Perception domain asks individuals to rate their overall health as “very good”, “good”, “fair”, “poor”, or “very poor”. The Incontinence Impact domain asks individuals how much (“not at all”, “slightly”, “moderately”, or “a lot”) their bladder problems have affected their lives. The multi-item Symptom Severity domain measures the severity of urinary symptoms; possible item responses include “a little”, “moderately”, “a lot”, and “omitted or N/A”. Other domains ask individuals to what degree (Role Limitations, Physical Limitations, Social Limitations, and Emotions domains: “not at all”, “slightly”, “moderately”, or “a lot”; Personal Relationships domain: “not applicable”, “not at all”, “slightly”, “moderately”, or “a lot”; Sleep/Energy and Severity [Coping] Measures domains: “never”, “sometimes”, “often”, or “all the time”) their bladder control symptoms have affected different aspects of their lives. Each domain is scored on a scale from 0 (least impairment) to 100 (greatest impairment). For both instruments, a decrease in mean score represents improvement in HRQoL.
Statistical analysis
HRQoL evaluation included all randomized study participants who received at least one dose of OTG and provided baseline data for efficacy assessment. The last observation carried forward method was used to impute missing data. The statistical significance of intergroup differences in IIQ or KHQ score changes from baseline was tested with an analysis of covariance model using baseline as the covariate. The statistical significance of intergroup differences in baseline scores in the OTG was assessed with a two-sample t test. No adjustments were made for multiple comparisons. Demographic and baseline clinical characteristics were summarized using descriptive statistics.
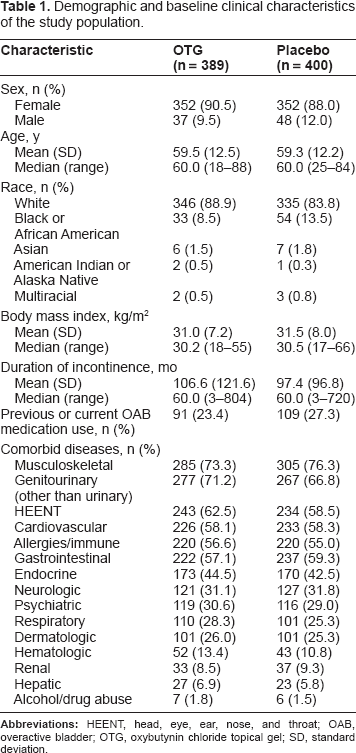
Demographic and baseline clinical characteristics of the study population.
Results
Of 789 participants, 389 received OTG and 400 received placebo. Large majorities of participants were white (681/789; 86.3%) and female (704/789; 89.2%). The mean age was 59 years, with 35.9% of participants aged 65 years or older. The median duration of incontinence in the study population was 5 years; the mean number of daily incontinence episodes was 5.4, indicating that participants had fairly severe urinary incontinence at baseline. A quarter of the study population (200/789; 25.3%) had taken medication for OAB previously. Many participants had comorbid conditions, most commonly musculoskeletal disease (590/789; 74.8%); genitourinary disease unrelated to a urinary condition (544/789; 68.9%); head, eye, ear, nose, and throat abnormality (477/789; 60.5%); cardiovascular disease (459/789; 58.2%); and gastrointestinal disease (459/789; 58.2%). Participants were taking concomitant medications for these medical conditions.
IIQ scores
At baseline (Table 2), participants indicated that the negative impact of their bladder control problems was greatest on the IIQ subscale of Travel followed by Emotional Health, Physical Activity, and Social Relationships. Small intergroup differences in baseline total scores (P = 0.0342) and baseline scores for Social Relationships (P = 0.0119) were observed. Subsequent treatment effect analyses were corrected for these differences by including baseline as a covariate in the analysis of covariance model. 20
OTG treatment compared with placebo had a highly significant positive effect on HRQoL as assessed by IIQ (Fig. 2A). Scores for IIQ (total score) and its four subscales (Emotional Health, Social Relationships, Travel, and Physical Activity) showed significantly greater decreases (indicating HRQoL improvement) with OTG than placebo (P < 0.01). Mean change from baseline to study end in IIQ total score was -72.1 points for OTG compared with -49.5 points for placebo (P = 0.0005).
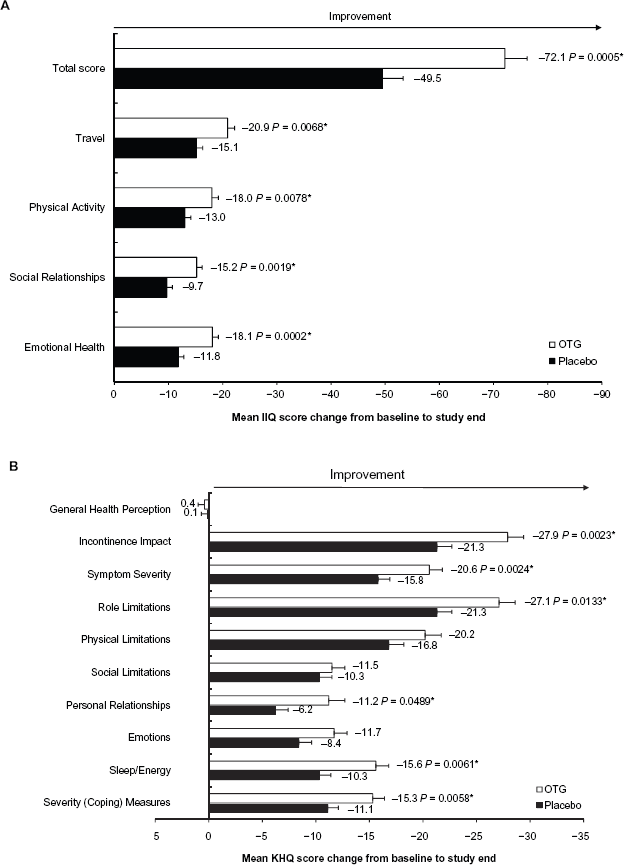
Mean changes in Incontinence Impact Questionnaire (IIQ) total score and subscale scores (A) and in King's Health Questionnaire (KHQ) domain scores (B) from baseline to study end. Error bars indicate standard errors of the mean.
P < 0.05, analysis of covariance.
KHQ scores
Baseline KHQ scores (Table 2) indicated mild HRQoL impairment in the domains of General Health Perception (mean score in OTG group, 21.4; placebo group, 21.3) and Symptom Severity (OTG group, 18.6; placebo group, 18.2). However, participants reported that bladder control problems had a major negative impact on their HRQoL (mean Incontinence Impact scores: OTG group, 79.9; placebo group, 78.3). Participants experienced the greatest negative effect of OAB on HRQoL in the domain of Severity (Coping) Measures, followed by Sleep/Energy, Role Limitations, Physical Limitations, Emotions, Social Limitations, and Personal Relationships.
Summary of health-related quality of life scores at baseline.
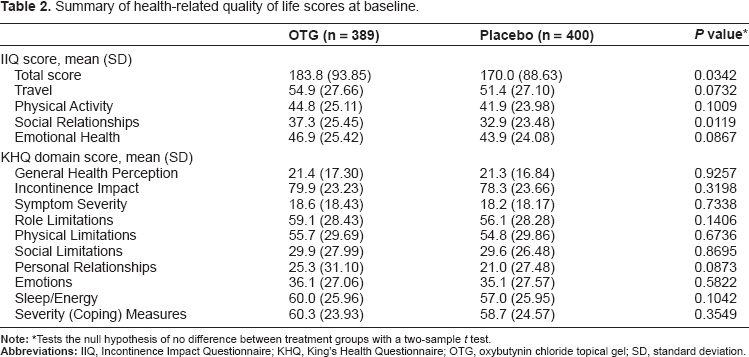
Tests the null hypothesis of no difference between treatment groups with a two-sample t test.
Compared with placebo, OTG was associated with significantly greater reductions in scores for the incontinence-specific KHQ domains of Incontinence Impact, Symptom Severity, Severity (Coping) Measures, and Sleep/Energy (P < 0.01) (Fig. 2B). In addition, OTG provided significantly greater improvement in Role Limitations and Personal Relationships than placebo (P < 0.05). No statistically significant differences between treatment groups were observed for changes in Emotions, Physical Limitations, or Social Limitations, although improvements in these domains were numerically greater for OTG than placebo. Neither treatment improved General Health Perception (Fig. 2B).
Discussion
As previously reported, the results of this randomized, double-blind, placebo-controlled phase III study established the safety and efficacy of OTG in reducing the number of incontinence episodes, decreasing urinary frequency, and increasing voided volumes in adults with OAB. 11 The present analysis of additional subject-reported outcomes of this study further demonstrates that OTG generally alleviated the negative impact of OAB on quality of life, as indicated by significant reductions in scores for IIQ and highly incontinence-specific domains of the KHQ among study participants receiving OTG versus placebo. Thus, the previously reported treatment-related improvements in objective outcomes now have been corroborated by improvements in subjective outcomes. Our findings are consistent with those of a recent uncontrolled, open-label, community-based study of transdermally administered oxybutynin in adults with OAB that reported clinically significant improvements in HRQoL after a treatment period of at least 6 months. 21
Individuals with OAB tend to avoid daily activities such as travel, physical activity, and social activity because of fear of urine leakage.12,19 These limitations have a more significant impact on individuals with more severe urinary incontinence. When urinary incontinence does occur, common reactions include embarrassment, frustration, anxiety, and depression. 12 Because of restricted physical and social function, persons with incontinence often find themselves limited in performing activities such as work, household chores, and shopping. 19 Compared with placebo, OTG improved these aspects of participants’ lives in our study, as demonstrated by significantly reduced scores in the IIQ subscales of Travel, Physical Activity, Social Relationships, and Emotional Health and in the KHQ domain of Role Limitations. Scores in the KHQ domains of Physical Limitations, Social Limitations, and Emotions also were reduced to a greater extent by OTG than by placebo, although the differences between treatments were not statistically significant. The placebo group received behavioral interventions, which have been shown to improve quality of life in OAB patients; these interventions may have affected participants’ QoL scores.
To cope with OAB symptoms, many persons with OAB develop habits such as mapping out bathrooms, wearing absorbent products (eg, perineal pads), and avoiding sexual intimacy. 12 Such coping measures may negatively affect personal relationships. In addition to urinary incontinence, urgency, and frequency, persons with OAB may experience nocturia, which contributes to lack of sleep and reduced energy. 22 In our study, OTG improved these aspects of participants’ lives, as indicated by significant reductions in score for the KHQ domains of Severity (Coping) Measures, Personal Relationships, and Sleep/Energy compared with placebo.
Overall, our results showed that OTG was significantly more effective than placebo in reducing the impact and severity of OAB symptoms (KHQ domains of Incontinence Impact and Symptom Severity). The greater improvements in more incontinence-specific domains of the KHQ likely are due to the fact that study participants had relatively severe urinary incontinence at baseline. Patients’ perception of improvement in their OAB symptoms may be expected to result in better treatment adherence and, consequently, better clinical outcomes. 23
Our study had several limitations. Because statistical comparisons were not corrected for multiple comparisons, some of the observed differences may be the result of chance. However, it is remarkable that all IIQ comparisons and most of the comparisons for highly incontinence-related KHQ domains yielded highly significant results. This strongly suggests that the documented efficacy of OTG in symptom improvement 11 indeed is experienced by patients as improvement in HRQoL. A substantial placebo effect was observed in this study, both for previously reported efficacy end points 11 and for HRQoL. The placebo effect may be attributable in part to the behavioral instructions given to the subjects during screening. Mean changes from baseline in individual KHQ domain scores observed in this study suggest that participants who received placebo on average experienced small to moderate improvement in HRQoL (5–15 points), whereas those treated with OTG experienced at least moderate improvement (.10 points). 24
Many participants had comorbid medical conditions, which may have affected the less disease-specific portions of the KHQ. Although participants reported little impairment in their General Health Perception at baseline and overall experienced no clinically significant changes attributable to treatment, 24 it is conceivable that generic questionnaires might have revealed effects of OAB and its treatment with OTG on additional QoL-related health dimensions. For example, the potential psychological effects of OAB were not assessed. Moreover, because our study included few racial and ethnic minorities, men, and persons younger than 45 years, effects of OTG on HRQoL in these subgroups are unknown and need to be studied in future clinical trials.
Conclusion
OAB is a medical condition defined by its urinary symptoms; therefore, it is important to measure treatment efficacy through patient-reported outcomes. In the OTG phase III study, OTG compared with placebo showed a positive effect on OAB-related symptoms from the patient's perspective, as indicated by significant improvements in incontinence-specific HRQoL measures. The observed effects of OTG on these patient-reported outcomes help to validate the clinical significance of the previously reported improvement in primary and secondary efficacy outcomes of the phase III study. The positive effects of OTG on patient- reported outcomes combined with its previously demonstrated efficacy and excellent safety profile should make it an attractive treatment option for adults with OAB.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. Diane Newman has served on the advisory boards of Novartis, Astellas, GSK, and Watson Pharma; has served as a speaker for Procter and Gamble, Novartis, Allergan, Astellas, GSK, Pfizer, Allergan, and Watson Pharma; and has conducted clinical research for GSK, Allergan, and Watson Pharma. Philip M. Hanno is a consultant for Watson. Roger R. Dmochowski is a consultant for Allergan, Astellas, Watson, Pfizer, and Novartis. Delbert C. Rudy is a speaker and study investigator for Watson. Heather Thomas, Kim E. Caramelli, and Gary Hoel are employees of Watson. The peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgments
The study was supported by Watson Laboratories, Inc., Salt Lake City, Utah, USA; Watson Laboratories, Inc., was involved in study design, data analysis, manuscript preparation, and publication decisions and, thus, three Watson employees are included as authors. Editorial assistance funded by the study sponsor was provided by Jinling Wu, PhD, MD, and Candace Lundin, DVM, MS, of Scientific Connexions, Newtown, Pennsylvania, USA.
