Abstract
Donepezil is a potent, selective, noncompetitive, and rapidly reversible inhibitor of acetylcholinesterase (AChEI) licensed for the treatment of Alzheimer disease (AD); and is the first and only AChEI licensed for the treatment of severe AD. Its efficacy as monotherapy, or in combination with the NMDA-agonist, memantine, has been documented in several randomised double-blind, placebo-controlled, short-term clinical trials, as well as long-term extension trials and observational studies. Donepezil is a well tolerated drug that is generally safe as demonstrated even in patients with multiple co-morbidities receiving polypharmacy. It has been shown that donepezil improves cognition and global function in patients with mild-to-moderate AD; and long-term efficacy is maintained for up to 50 weeks. There is a dose-response relationship, with higher doses more likely to produce symptomatic benefit. Furthermore, donepezil-treated patients may improve cognitively and show global clinical improvement in all disease stages, including severe AD. Less consistent results in all disease stages were obtained on measures of function and behavior, and observations of mood. No effect on transition to AD has been found in long-term, randomized clinical trials in mild cognitive impairment (MCI). Cost-effectiveness of the treatment has been questioned by one long-term open-label societal study of 2-years duration. This study reported modest improvement of cognition but no statistically significant benefits during donepezil treatment as compared to placebo, in terms of rates of institutionalization and progression toward greater disability. However, there is a need for further research on clinically meaningful outcomes and treatment benefits favored by patients and caregivers, which are traditionally not defined as outcomes in clinical trials. Likewise, we need to know how to select responders, what is an optimal AChE inhibition particularly during the long-term treatment, in which patients the dosage should be increased for a sustained benefit, what is the optimal duration of treatment and when is meaningful to stop the treatment. After almost two decades of donepezil use in everyday clinical practice these issues are still unresolved.
Introduction
Alzheimer disease (AD) is a neurodegenerative disease that affects primarily the elderly. It is characterized by progressive dementia exhibiting typical neuropathological findings and neurochemical deficiencies in selectively vulnerable regions of the brain.
Numerous neurotransmitter systems are affected in the disease, but the most consistent and pronounced changes are those described in relation to the cholinergic nervous system.1,2 Cholinergic deficiency contributes to cognitive decline and possibly behavioral symptoms in AD and is shown to be the best neurochemical correlate of dementia severity in AD.3–5 Against this background, four acetylcholinesterase inhibitors (AChEIs) had been developed during the nineties for therapeutical intervention in AD. Among the three AChEIs remaining on the market donepezil, first second-generation non-competitive inhibitor of AChE introduced in 1997, is the most widely used in the treatment of AD patients with more than three billion patient days of Aricept, marketing name for donepezil (www.emaxhealth.com/91/7904.html). It is efficacious as a monotherapy or in combination with NMDA-agonist memantine as documented in several randomized double-blind, placebo-controlled, short-term clinical trials.6–10 They have consistently reported either improvement or stabilization on the basis of cognitive and functional performance measures or by means of a measure of global clinical change across the severity spectra of AD.
Furthermore, a positive effect of long-term donepezil treatment was also reported on functional and structural neuroimaging surrogate markers of a disease process, such as reduced decline in regional cerebral blood flow as measured by SPECT and slowing of progression of hippocampal atrophy as measured by MRI volumetric analysis after one year of continuous treatment.11,12 These findings suggest both neuroprotective effect and preservation of functional brain activity. Extended post-marketing studies have shown some long-term benefits and good safety profile of AChEIs in general, and donepezil in particular. 13 Donepezil has also been approved for a treatment of severe AD in the USA, being the first AChEI used for the treatment of the entire clinical spectra of the disease. However, some regulatory bodies still favor donepezil for a treatment of a subgroup of AD patients with mild to moderate AD (www.nice.org.uk/TA111).
In this review authors would like to present a pharmacological profile of the drug, including its mechanisms of action, efficacy and safety, as demonstrated in pivotal and observational open-label trials, as well as cost-effectiveness. Furthermore, problems in translation of positive findings from clinical trials (efficacy) to desirable effects in clinical practice in individual patients (effectiveness) will be discussed.
Mechanism of Action
Donepezil is a potent, selective, noncompetitive and rapidly-reversible inhibitor of AChE. For comparison, galantamine and phenserine seem to be quite selective and reversible AChE inhibitors, with weak-moderate potency, while rivastigmine is a pseudo-irreversible inhibitor of both AChE and butylcholinesterase (BuChE) enzymes. 14 Tacrine, also a rapidly-reversible inhibitor of both enzymes, is no longer used in clinical practice due to poor tolerability.
The clinical importance of the selectivity of ChEIs for AChE over BuChE is not clear. Based upon recent data it is evident that BuChE is capable of compensating for some function of AChE in the brain, particularly in AD patients.15,16 There are also reports demonstrating that selective BuChE inhibitors may elevate extracellular levels of cortical ACh, augment learning and lower AD Aβ peptide in rodent brain.17,18
The rapidly-reversible inhibitors, ie, donepezil, galantamine and tacrine inhibit AChE by binding to hydrophobic binding sites, while leaving the enzyme intact. In this dynamic process, neither the inhibitors nor the enzyme are transformed by their interaction. Thus, for a rapidly-reversible inhibitor, the drug concentration and enzyme inhibition correspond negatively to each other, showing graphically as mirror images. The presence of the drug causes inhibition, and consequently, its elimination rate is a key aspect of the pharmacokinetics. 14
Pharmacokinetic Profile
The profile of donepezil concerning its absorption, metabolic pathways, drug-drug interaction and clearance is well established (for a review see 19 ). We hence focus here on the long-term concentration profile of donepezil in plasma and CSF of AD patients. Due to its half-life of 70 hours, donepezil has the convenient once-daily administration dosage. Donepezil plasma concentration reaches its steady state within three months of treatment and remains unchanged. 20 Long-term donepezil concentration is dose-proportional in the plasma, 20 which is in agreement with its short-term concentration profile.21–23 The plasma concentrations of donepezil are 30 ng/mL in patients receiving 5 mg/day, and 60 ng/mL in patients on 10 mg/day of the drug.20,21
Reports pertaining to the distribution of donepezil in CSF as a surrogate of the concentrations in the CNS are very scarce. However, one report shows that the long-term donepezil concentration profile in CSF differs from that in the plasma. 20 The CSF-donepezil concentration appears approximately ten times lower than the plasma level, but shows a similar dose-proportional pattern, namely 4–5 ng/mL in patients taking 5 mg/day and 7–8 ng/mL in those receiving 10 mg/day. 20 In addition, there are indications of time-dependent pharmacokinetics for donepezil in CSF but not in plasma. 20 Donepezil concentration in CSF is found to increase by 50% between the 12 and 24 months treatment in AD patients receiving the same dose during both intervals. 20
Pharmacodynamics
Few studies are available that describe long-term pharmacodynamic profiles of donepezil either in blood or, particularly, in CSF. To our knowledge, only one report is available in which the effects of up to two years of chronic donepezil treatment on blood and CSF AChE activities, as well as the CSF AChE protein expression have been evaluated in relation to the drug concentration and cognitive changes of AD patients.20,24
Most studies utilize a measurement of blood-AChE inhibition as a surrogate for inhibition in the brain. The main reason is that no simple method for estimating CSF-AChE inhibition in donepezil-treated patients is available due to its rapidly-reversible nature, as well as due to the necessity of diluting the samples during the currently available enzyme activity assay of AChE.20,24–25 However, it has been shown that by combining enzyme data derived from a direct colorimetric assay and an AChE protein level assay, an estimate can be obtained that rather accurately approximates the enzyme's inhibition level to the in vivo brain-AChE inhibition, as assessed by PET-tracer following administration of the rapidly-reversible inhibitors, donepezil and galantamine.20,25
Red blood cell (RBC) AChE inhibition following short-term (up to three months) donepezil treatment appears to be dose-dependent and quite strong (about 19%, 44%, 64% and 77% inhibition in patients receiving 1, 3, 5 and 10 mg/day of the drug, respectively).21–23
In a long-term report, the inhibition of RBC-AChE activity after donepezil treatment has been estimated to be about 20%–30% in a dose-dependent manner, which strongly correlates with an approximately 20%-higher degree of CSF-AChE inhibition. 20 This moderate RBC-AChE inhibition is hence generally lower than earlier reports that suggested 50%–75% RBC-AChE inhibition.21–23 Although, methodological differences may to some degree account for these discrepancies, due to the lack of assay's details in these reports we cannot deduce the reason for these apparent discrepancies.
The moderate level of RBC-AChE inhibition reported by Darreh-Shori et al 20 however, is more compatible with reported levels in CSF and with the 20%–30% cortical-AChE inhibition in AD patients after short-term donepezil treatment measured by PET.26,27
As mentioned above, the long-term donepezil concentration in CSF of AD patients show a time-dependent profile in CSF, which may imply a lower short-term CSF-donepezil concentration compared to after 1–2 years of chronic donepezil treatment. This in turn may explain the discrepancy between the 20%–30% in vivo brain-AChE inhibition after short-term donepezil treatment, as measured by PET,26,27 and the observed 35%–55% CSF-AChE inhibition following the chronic donepezil treatment. 20
Protein levels of AChE in CSF increase after long-term treatment with all rapidly-reversible AChEIs used in AD patients (50% increase after tacrine treatment; four-fold increase after donepezil treatment and two-fold increase after galantamine treatment.20,25,28–31 However, it is unlikely that this increase in protein expression reflects a tolerance effect because as mentioned above the estimated long-term AChE inhibition in CSF in AD patients receiving 5–10 mg once daily ranges between 35%–55% in a dose-dependent manner, which strongly correlates with the CSF donepezil concentration. 20
Nonetheless, there are also some indications that a tailored dosage may increase efficacy of the donepezil. This is because not all the patients that receive 10 mg/day of donepezil will reach higher CSF donepezil concentration than those receiving 5 mg/day. Furthermore, inhibition levels of AChE in CSF show inhibition-response relationship with the patients’ cognitive performance in the MMSE test after 1 and 2 years of treatment. AD patients with 45% or stronger CSF AChE inhibition show preserved cognition up to two years of donepezil treatment, while patients having 30% or less display cognitive deterioration regardless of the dosage. 20
A similar threshold for the AChE inhibition which is needed for detection of a therapeutic response to donepezil has also been reported after short-term treatment of AD patients with this drug. 27
Other pharmacodynamic properties have been suggested recently, which seem to be related to the inhibitory action of ChEIs on their target enzyme, ie, an increase in the cholinergic tone on immune cells such as suppression of microglial activation.32–34 There are also reports suggesting that donepezil may stimulate neuroprotective and/or neurogenic processes by elevating back the plasma concentration of the hematopoietic growth stem cell factor in AD patients to the levels observed in control subjects. 35
In summary, a high variation in pharmacodynamic responses is expected within the patients in both the 5- and 10-mg treatment groups, which calls for individualization of (and perhaps increasing) the drug regimen based on the AChE inhibition level, particularly following long-term treatment. A CSF-AChE inhibition level of 45%–55% seems to be the optimal level in donepezil-treated AD patients, as lower levels seem ineffective; whereas higher levels may cause hyper-excitation of the vulnerable but still functional cholinergic neurons in the AD brain.
Clinical Efficacy
The Table 1 provides an overview of randomized, double-blind, placebo-controlled, parallel-group clinical studies of at least 12-weeks duration. The review begins with pivotal trials in patients with mild-to-moderate AD, which led to the registration of donepezil. In Table 2, open label extension clinical trials or observational studies are presented. A literature search in Pub Med, by the terms, “donepezil” and “clinical trials”, enabled the selection of the reviewed studies. The basis of each selection is noted in the table headings. Since mild cognitive impairment (MCI), and amnestic MCI (aMCI) in particular, are considered preclinical-AD, 36 trials including this patient group were also included in this review.
Randomized double-blind placebo-controlled clinical trials of donepezil in AD and MCI.
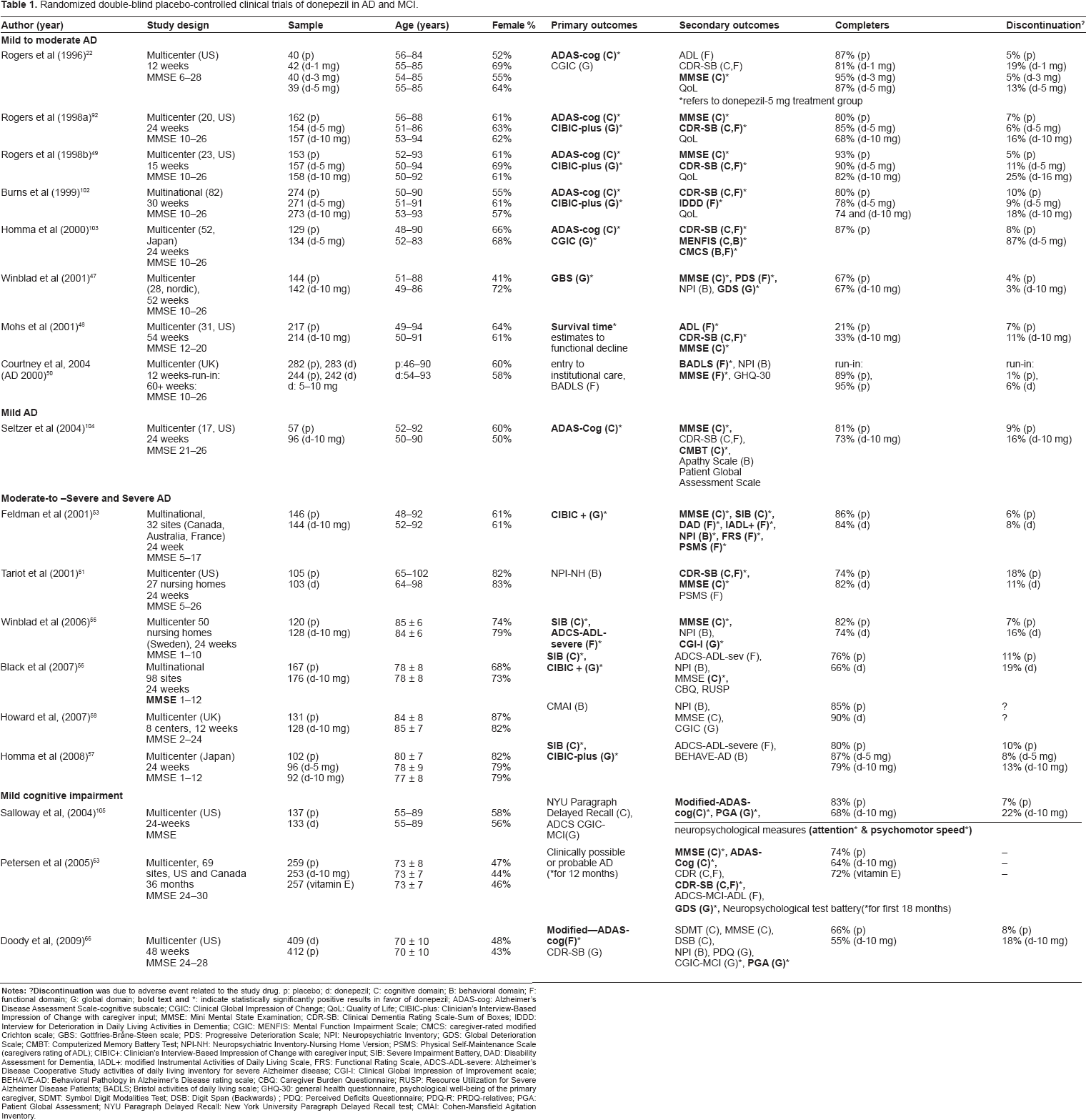
Clinical trials methods routinely employ a variety of scales that measure outcomes that represent four key symptom domains, including cognitive, functional, and behavioral, as well as a clinical global assessment of change (Table 1). The latter is a subjective integrative judgment by an experienced clinician. Although the aim of this review is not to describe in detail the efficacy measures used in clinical trials, it is important to emphasize that the choice of scales, as well as the assignment of primary and secondary outcome variables is determined by the disease severity of the study population.
Mini Mental State Examination (MMSE) and Alzheimer Disease Assessment Scale-Cognitive Subscale (ADAS-cog) are the most widely used cognitive scales that assess change in mild-to-moderate stages of the disease. Lowered scores on the 30-point MMSE and elevated scores on the 70-point ADAS-cog scales denote performance deficits and cognitive deterioration.37,38 However, these measurements reach plateau in severe disease stages, at which the Severe Impairment Battery (SIB) is a more appropriate measure of cognitive deterioration. 39
Progression of the disease broadens the spectrum of symptom domains, including changes in behavior and activities of daily living (ADL) which has consequences on quality of life, service utilization and caregiver burden.
The Clinician Global Impression of Change-Plus (CIBIC-plus) provides a 7-point rating scale of global change in cognitive ability based on patient and caregiver interviews by a clinician, 40 from marked improvement to marked worsening. This instrument is required by FDA for evaluation of efficacy in clinical trials for AD. 40
In the absence of a placebo group the estimates of long-term benefits of donepezil-treated patients are derived from comparison of their annual decline on primary outcome measures compared to annual decline on same measures in historical cohorts of untreated patients. For example, the estimated annual lowering of the MMSE score in untreated patients with mild-to-moderate AD has been reported to be 2.8 points.41,42 The annual increase in ADAS-cog in patients with untreated moderate AD has been estimated to be as much as 9–11 points per year. 43
The global clinical measure, Clinical Dementia Rating-Sum of Boxes (CDR-SB), has been estimated to increase by approximately 2.4 points per year. 44 The expected decline without treatment on the Neuropsychiatric Inventory (NPI) total scale, measuring behavioral and psychiatric symptoms in patients with mild-to-moderate AD, has been shown to be 3.9 points at 6 months. 45
Double-blind, placebo-controlled randomized clinical trials
Most of the trials included in this category have satisfied at large requirements for internal validity assessment such as randomization, allocation concealment, similarity of compared groups at the baseline in terms of demographics and clinical characteristics, and in a few studies intention-to-treat (ITT) analyses were conducted as well as monitoring overall and differential loss to follow-up. 46
Mild to moderate AD
Eight trials that included patients with mild-to-moderate AD have been reviewed in the Table 1. Most of these short-term trials were pivotal phase III-studies that reported small but consistent improvements in scores on global, cognitive and functional measures. One-year-studies by Winblad 47 and Mohs 48 showed that benefits of donepezil treatment on global and cognitive measures and instrumental and basic activities of daily living (ADL) were maintained for at least 1-year. Furthermore, Mohs et al 48 demonstrated the median time to attain clinically evident functional decline was delayed by 5 months for patients on donepezil when compared to placebo. In the pivotal trial of Rogers et al 49 a beneficial effect on cognition in a donepezil-treated subjects fell to the baseline values after a 6-week wash-out period, indicating either the importance of continuous treatment or the possibility of a wearing-off phenomenon. A very much debated the AD 2000 study, 50 not sponsored by industry but which recruited typical patients representative of everyday clinical practice, claimed that minimal and short-term lasting benefits, as indicated by marginal improvement on MMSE and ADL measures after 3 months, were not cost-effective given the absence of risk reduction of progression of disability or entering institutional care. The study faced a lot of criticism due to its design and being underpowered for most of its primary end points. While recruitment of 3000 patients was planned only 565 were randomized and only 20 patients completed phase-III treatment.
Moderate- to- severe and severe AD
Two prospective randomized-controlled trials (RCT) with moderate-to-severe AD patients, one study reporting post-hoc analysis on a more severe subgroup of patients, one 12-week study of donepezil treatment of agitation in mostly severe AD patients, and three studies of patients with severe AD treated with donepezil were identified through a literature search (Table 1). The first clinical trial reporting efficacy and safety of donepezil in a vulnerable patient population residing in nursing home settings was that of Tariot et al 51 (Table 1). These generally older patients exhibited more severe dementia, more frequent presence of behavioral and psychiatric symptoms of dementia (BPSD) and had more co-morbid illness than patients with mild to moderate AD studied in previous trials. The primary outcome was not showing significant difference between the placebo and donepezil group in scores on NPI due to the relative improvements in both groups. Secondary sub-item analysis showed that donepezil produced beneficial effect in 67% of patients with agitation and aggression which were the most common symptoms at baseline. 52 Furthermore, effects on cognition, overall dementia severity, safety and tolerability were similar to those reported in studies performed in outpatient settings on mild-to-moderate AD, suggesting that advanced age, co-morbidity and concomitant use of other medication probably should not limit donepezil treatment.
A following study reported a post-hoc analysis on a subgroup of patients with more severe AD from a 24-weeks randomized, placebo-controlled trial in moderate to severe AD (MSAD). 53 Analysis of these results showed small benefits across all three key symptom domains, including: global function (CIBIC-plus), cognition (MMSE and SIB), functional disability (DAD) and behavior (NPI). 54
Three large RCT of 24-weeks duration included exclusively patients with severe AD: the Swedish nursing home study, 55 the multinational study 56 and the Japanese study 57 (Table 1).
All 3 studies demonstrated consistently improvements in cognition and global function. However, a statistically significant positive difference on measure of function (ADCS-ADL-sev) was reported only in the Swedish study which led to the FDA approval of donepezil for treatment of severe AD. None of the three studies could demonstrate positive effects on behavioral and psychological symptoms of AD as measured by NPI or BEHAVE-AD. This symptom cluster occurs invariably and frequently in severe AD, increasing caregiver stress and the likelihood of institutionalization. A 12-week UK trial investigated treatment of agitation but was unable to obtain a pre-specified 30% or greater reduction in agitation, since a similar proportion of patients (20%) from both treatment groups were found to be treatment responders. 58 The mean reduction in NPI score was 3.8 in the donepezil treated vs. 3.6 in the placebo group. It is not clear at present if lack of behavioral benefits means lack of efficacy of donepezil on this symptom domain or if behavioral outcome measures lack sensitivity in a presence of psychotropic medication. 59 A pooled-efficacy analysis of the three RCT in severe AD, including only patients receiving 10-mg donepezil daily, showed that donepezil-treated patients were 2–3 times more likely to achieve positive combined-domain response. 60 Positive combined-domain response is defined as an observable benefit across more than one domain with varied stringency across 3 definitions–-each patient having a unique profile–-relative to placebo-treated patients by the endpoint of the study.60,61 Cognitive responders defined as improvement of ≥4 or ≥8 points of SIB showed significantly greater reduction in total NPI scores (-5.5 and 6.4, respectively), indicating positive relation between cognitive and behavioral improvement. A 12-week, multicenter, open-label study in 103 patients with moderate-to-severe AD focused on switch to donepezil therapy after discontinuation of memantine monotherapy due to poor tolerability or lack of efficacy. 62 The study reported that re-initiation of donepezil treatment in patients with moderate-to-severe AD showed benefits on cognition measuring an approximate increase of 2 points on MMSE, on global assessment of change measured by CGI-I, and in patient social behavior as rated by a caregiver; but no change in behavior was indicated in NPI scores. Forty-two percent of the patients in this study received before study entrance a long-term therapy with AChEI which was discontinued due to lack of efficacy This implies that in clinical settings, a decision for discontinuation of the treatment might have been done prematurely and that realistic evaluation of treatment benefits should include not only improvement and stabilization but also less-than-expected decline.
Open-label clinical and observational studies of donepezil in AD.
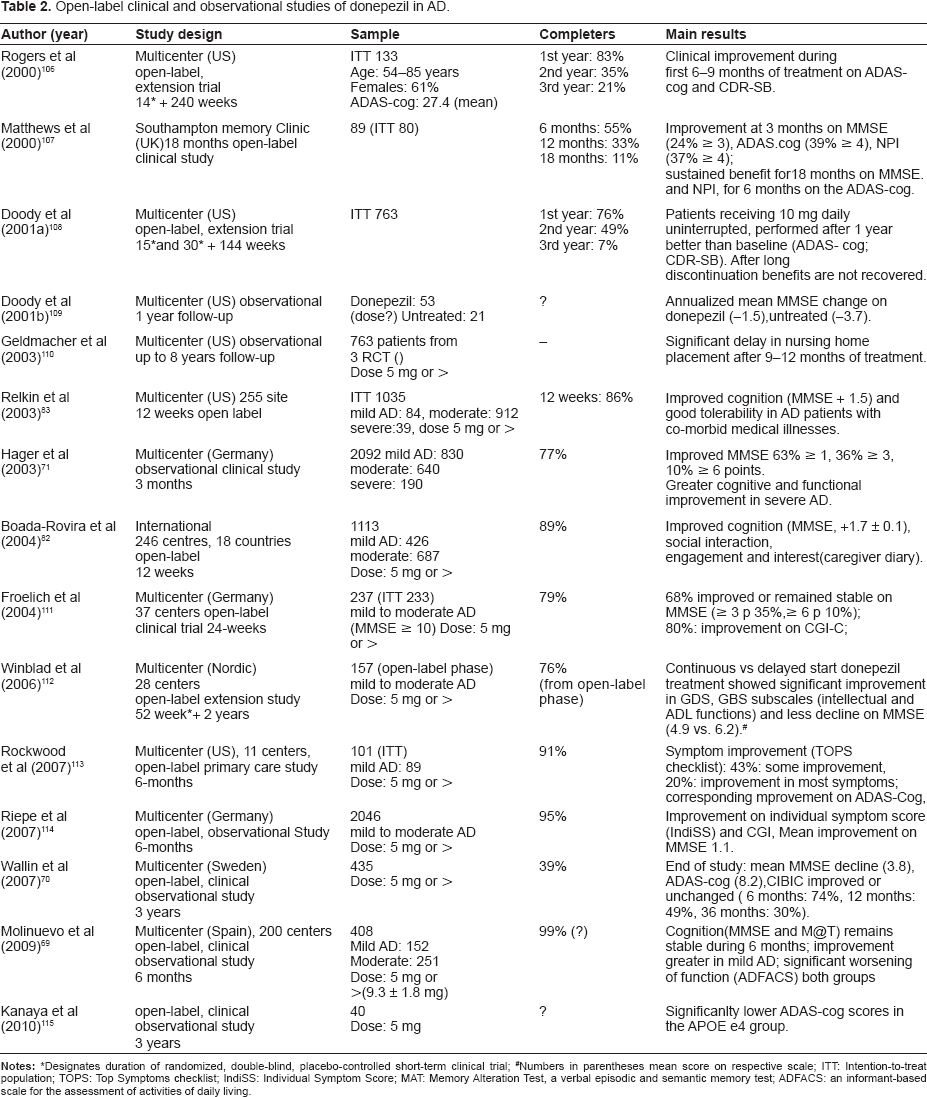
Designates duration of randomized, double-blind, placebo-controlled short-term clinical trial;
Numbers in parentheses mean score on respective scale; ITT: Intention-to-treat population; TOPS: Top Symptoms checklist; IndiSS: Individual Symptom Score; MAT: Memory Alteration Test, a verbal episodic and semantic memory test; ADFACS: an informant-based scale for the assessment of activities of daily living.
Mild cognitive impairment
So far three randomized, placebo-controlled clinical trials with donepezil treatment of this particular patient group were reported: a short 6-month trial, 3-year secondary prevention study (the Memory Impairment Study, MIS), and 1-year study (Table 1). While no effect on disease progression to AD has been found in long term, all three studies consistently showed small but consistent benefit on ADAS-cog in favor of donepezil. Moreover, the effect may be modified by the presence of APOE ε4 genotype. 63 Interestingly, although it had a weak effect, these data appeared to contradict observations from earlier trials in AD that showed no effect, 64 as well as a stronger treatment effect obtained among e4 non-carriers. 65 Only the one-year study by Doody et al 66 demonstrated short-lasting effect on a global clinical measure at week 6 of the trial.
Inefficacy of AChEIs at an early stage of the disease is one possible explanation for these negative results on primary outcomes and the appearance of relatively weak benefits in secondary outcomes; however other explanations have been suggested, including, for example, insensitivity of traditional measurement instruments. 67 High order instrumental tasks that have predictive validity for future conversion from MCI to AD could not be reliably measured using functional measures typically used in AD trials. Further criticism is that a duration of the MCI-trials results in a higher drop-out rates, as it was around 40% in the 3-year-study. Drop-out bias could not be understood by simply analyzing a random selection of drop-out patients, rather a retrieved drop-out analysis should be considered while interpreting the results of the trials.
Open-label and observational studies
Extended placebo-controlled studies addressing long-term benefits, safety and tolerability of a drug intended for use over a prolonged period of time are not ethical. Pivotal studies of clinical efficacy usually exclude the patients from the “real world” clinical practice with a broader range of co-morbid illnesses and concomitant therapy. 68 These issues are addressed by pre- and post-marketing, long-term, open-label, extended clinical studies, which follow short-term, placebo-controlled, double-blind clinical trials, and additionally, by observational surveillance studies of patients treated by prescription.
Fifteen open-label studies have been included in this review; four of them were open-label extension studies of pivotal RCTs (Table 2). All of these studies have reported findings consistent with previous short-term studies in terms of stabilization or marginal improvement on cognitive measures; and with less consistent results regarding improvement on functional and behavioral measures. Mean MMSE change during continuous donepezil treatment ranged from 0.5 in mild AD and 0.0 in moderate AD in a 6-month Spanish study reported by Molinuevo et al 69 -3.8 in a 3-year Swedish study of Wallin et al 70 to -4.9 in a 3-year open-label Nordic extension trial of Winblad et al. 70 While the 6-month Spanish study suggested greater cognitive improvement in mild AD, the 3-month German study reported intriguingly greater cognitive and functional improvement in more severe AD, reporting higher scores on MMSE of +0.8 in mild, +1.9 in moderate and +2.3 in severe AD. 71 Similarly, in the same group of patients donepezil showed beneficial effects on functional ability. The latter study also showed that donepezil treatment in AD patients exhibiting parkinsonian symptoms was equally effective and as well tolerated as in the overall AD population.
Head-to-head evidence of comparative efficacy of AChEI
There were very few direct comparisons among candidate AChEI. Four comparative studies were identified in the literature, two comparing donepezil with galantamine and two with rivastigmine.72–75 In a 12-week, multinational study comparing the tolerability and cognitive effects of donepezil and rivastigmine in patients with mild to moderate Alzheimer disease, both treatment groups showed comparable improvement on the ADAS-cog but relative to the donepezil treated group fewer subjects in the rivastigmine-treatment group completed the study (69% vs. 89%), and more rivastigmine-treated patients discontinued the study due to AEs (22% vs. 11%, respectively). 75 Another 2-year study comparing donepezil with rivastigmine found no significant differences on measures of cognition and behavioral symptoms, but rivastigmine provided more benefits on measures of global function and activities of daily living. 73 These positive effects were in secondary analyses demonstrated in apolipoprotein e4 carriers, patients with vascular risk factors and those suggestive of concomitant Lewy body disease, women and those younger than 75 years. However, while the magnitude of severe AEs did not differ between the patient groups, rivastigmine treated patients experienced more gastrointestinal side effects such as nausea and vomiting, in both titration (33% vs. 15%; 28% vs. 6%), and maintenance phase (13% vs. 5%; 15% vs. 4%).
A long-term comparison of galantamine and donepezil treatment in a 52-week multicenter UK RCT reported similar safety and tolerability figures for both drugs. 74 However, in a subgroup of moderate-AD patients there were significant differences in scores on cognitive measures, MMSE and ADAS-cog, favoring galantamine. No difference in NPI scores was observed in this study but more caregivers of patients receiving galantamine reported reductions in burden compared with donepezil treated patients.
Different pharmacological and pharmacokinetic profiles of AChEI could influence efficacy and result in different safety profiles. Large scale, randomized, head-to-head comparative studies of AChEI efficacy are needed to establish relative drug superiority.
Combination therapy
Several studies have focused on a common clinical approach employing combination therapy with memantine, an N-methyl-d-aspartate (NMDA) receptor antagonist, which is established alternative therapy for AD, and which has been approved for patients with moderate-to-severe stages of disease. 76 Patients receiving stable dose of donepezil with on average 2.5 years history of treatment are either declining or worsening on individual ADL items over 6 months, while those also receiving memantine showed better functional outcomes with a number of ADL items that remained unchanged or improved. 77 Another exploratory reanalysis of a pivotal trial of Tariot et al 78 considered cognitive effect of a combination therapy and showed that memantine additionally affects key areas of cognition, namely memory, language and praxis, as assessed by sub-items from SIB. 79 Although these post-hoc analyses suffer from some weaknesses and were not corrected for multiple comparisons, they suggest nevertheless that effects on cognition, language skills and praxis, could at least partly explain improvement in ADL. Indeed, a subsequent responder analysis using combined outcome measures showed that best risk reduction is achieved by using SIB and NPI together in the responder definition, consistent with finding that a higher percentage of those in the donepezil + memantine group (42%) were identified as responders compared to those in the placebo (donepezil only) group (26%). The combination of cognitive, functional and global abilities as indicated by stabilization of a triple outcome index (SIB+CIBIC-Plus+ADCS-ADL) showed higher response rates in the memantine + donepezil group (25%) vs. the placebo (donepezil only) group (15%).
Taken altogether, the data from several studies suggest that the addition of memantine to patients already receiving stable dosing of donepezil is associated with larger benefits than by donepezil mono-therapy alone, with regard to cognitive function, ADL and some aspects of behavior.
Safety and Tolerability Profile
In general donepezil treatment has been found to be safe and well tolerated in numerous studies. 80 Overall reported AEs are around 80% across the RCTs. Observed AEs include those that are cholinergic in nature, such as nausea, vomiting, diarrhea, insomnia, asthenia, weight loss, fatigue, and anorexia. Due to the cholinergic innervations of parasympathetic nervous system, the gastrointestinal (GI) system is most frequently affected. However, in general GI side effects have been transitory in most patients and disappear during 3–4 weeks of treatment. In pivotal trials nausea was reported in 21% and diarrhea in 16% of patients treated with 10 mg per day, and in 6% vs. 4%, respectively in the placebo group. 80 Lengthening the period of time during which the patients are receiving 5 mg before introduction of higher 10 mg dose, could reduce AE. 81
The discontinuation rate due to adverse events in mild-to-moderate and severe-AD trials is around 10%, while rates are higher in MCI trials (22% in the 24-week-terial and 18% in the 48-week-trials) (Table 1). Probably a better cognitive status in MCI make these subjects more likely to report adverse events and less willing to tolerate side effects in the absence of an established dementia diagnosis.
Concerns about drug tolerability must also consider patients with co-morbidity and those receiving polypharmacy, the type of patient not represented in early pivotal clinical trials. Several post-marketing observational studies designed to evaluate both safety and efficacy in “real-life” clinical or community-based settings did not demonstrate higher occurrence of severe AE than in pivotal RCTs.71,82,83 Similar incidence of total adverse events despite the more complex medical background of patients might be partly due to the naturalistic character of these studies where adverse events are not specifically monitored as in RCTs.
In a large 255 site, nationwide US, open-label study of 1035 patients with a high concomitant medication and extensive co-morbidity, Relkin et al demonstrated that the risk ratios for gastrointestinal side effects were not significantly increased by the concomitant use of aspirin or non-steroidal, anti-inflammatory drugs. 83 Risk ratios for cardiac events, including bradycardia were not significantly increased by concomitant use of beta-blockers, nondihydropyridine calcium-channel blockers or digoxin. One or more dose adjustments during the study were however related to a higher incidence of AEs. German observational study by Hager et al showed similar low risk for cardiovascular events, but reported that patients on concomitant treatment with SSRIs showed moderately increased risk of experiencing any AEs during donepezil treatment. 71 In addition, a large double-blind, placebo-controlled RCT on patients with vascular dementia and cardiovascular co-morbidity receiving donepezil and multiple concomitant medications, reported neither greater occurrence of AE in general nor bradycardia in particular.84,85
In summary, both RCTs and observational naturalistic studies have consistently shown a good tolerability and maintained long-term safety profile for donepezil.
Clinical Effectiveness and Patient Preference
It has been proposed that a treatment is likely to be clinically meaningful if the effect size is large enough to be clinically detectable, if there is a dose response to treatment in question and if measures from different outcome domains converge both within and across the trials. 86 Main efficacy results in clinical trials are based on differences in mean scores or mean change scores in cognitive, behavioral or functional rating scales. Therefore the clinical response in individual patients is difficult to extrapolate from data obtained from this group of highly representative patients. 87 As a result many regulatory bodies also require a responder analysis, which determines actual proportion of patients who benefit from the treatment according to some a priori set criteria. Furthermore, due to progressive nature of the disease, benefits should not only be seen as improvement in single domains but also stabilization or reduced worsening of symptoms preferably analyzed as a combination of outcomes.61,88 This definition of outcome was used in a pooled analysis of 3 pivotal clinical trials, including a 24-week RCT and a one 1-year RCT in mild-to-moderate AD, and a 24-week RCT sampling patients with moderate-to-severe disease, altogether including 906 patients. 88 The odds of declining were significantly reduced in donepezil-treated patients as compared to placebo, and results suggested that many patients initially characterized by improvement on traditional outcomes from single domains as non-responders could still benefit from treatment.
The treatment benefits favored by patients and caregivers are not defined as outcomes of clinical trials. This has been demonstrated in a large survey of careers and patients. 89 Secondary analysis of a 12-month open-label study of 100 patients with mild-to-moderate AD showed that while those donepezil-treated patients who improved on ADAS-cog were less likely to decline on clinical measures, 43% of patients who declined on the ADAS-cog, nevertheless, improved on at least two of clinical measures. 90 Recent FDA guidelines promote patient-reported outcome (PRO) instruments as effectiveness endpoints in clinical trials. 91
Quality of life (QoL) is defined as a patient's and/or caregiver's perception of the multidimensional effects of treatment such as physical, emotional, cognitive and social functioning, and it is not captured by typical outcome measurements such as psychometric tools. In short-term pivotal studies on donepezil treatment by Rogers et al the QoL assessment scale was used as secondary outcome with variable results.22,92 In contrast, an open-label societal study from Germany reported increased QoL of patients and/or their family in 70% of cases during a 3-months observational period. 93
Cost-Effectiveness
Analyses of “cost-effectiveness” consider treatments in terms of health outcomes in nonmonetary units, such as progression on cognitive measures, decline in functional abilities or institutionalization. 94 While this type of analysis cannot determine whether benefits of treatment exceed costs, it takes into consideration quality of life measures. Cost-benefit analyses measure costs and health benefits of interventions in monetary units. Both aspects of treatment, cost-effectiveness and cost-benefit, are of particular interest for health care financing systems and regulatory bodies who stand behind treatment guidelines.
Two earlier studies reported on reduction in time that patients spend in the severe-AD stage: A UK study reported treatment to be cost neutral for both treatment doses of 5 and 10 mg of donepezil, 95 and a Canadian study reported cost-saving over 5-years. 96 An industry-sponsored Swedish study reported a lengthening of time spent in non-severe AD and cost-saving over 5-year period. 97 A study from Japan showed quality-adjusted life-year (QUALY) gains and savings over 2 years. 98
A majority of studies on cost-effectiveness of AChEI treatment in AD, as well as economic modeling studies, utilize MMSE as measurement of cognitive performance while tracking disease progression and costs related to increased health care system utilization.8,99 However cognitive function alone is not an accurate predictor of disease progression, dementia severity and costs of care. 100 In a 24-month, prospective, multicenter, double-blind, community-based RCT secondary analyses of efficacy, safety and tolerability of donepezil and rivastigmine used survival analysis to predict time to nursing home placement. 101 In Cox regression models, older age, female gender, lower ADL at baseline and deterioration in ADL increased risk of nursing home placement. Risk of nursing home placement increased by 3% for each 1-point deterioration in ADL as measured by ADCD-ADL, and was independent of cognition since addition of MMSE to the regression model did not significantly change the outcome. Approximately 6% of patients with AD who show a mean total decline of 15 points in ADL will be institutionalized in 1 year time.
The AD 2000 Collaborative Group, a non-industry sponsored, societal study from UK, 51 reported that improvements in functional ability with donepezil treatment as observed in the study would not delay institutionalization sufficiently to justify the costs of the drug. The Health Technology Assessment Program reported (2006) after extensive review that donepezil treatment has QUALY in excess of £80 000, and that it reduces time spent in full-time care or progression to disease by 1.42–1.59 months. 8 Cost-savings that are gained by this reduction do not sufficiently overcome treatment costs to satisfy cost-effectiveness required by policy-makers in UK.
Place in Therapy, General Summary
Based on the current evidence donepezil is a symptomatic treatment for patients with mild to moderate AD. It is safe and well tolerated even by elderly patients with multiple co-morbidities and polypharmacies when escalation of daily dose from 5–10 mg is titrated over 4–6 weeks. Donepezil-treated patients may improve cognitively and show global clinical improvement in all disease stages, with possible improvement in overall functioning reported even in severe AD. Effect on behavioral and psychiatric symptoms of the disease is more controversial. In moderate-to-severe dementia larger clinical benefit could possibly be achieved in combination with memantine. Cost-benefit data are limited and impact of donepezil on patient-relevant outcomes remains poorly understood. There is a need for further research on clinically meaningful outcomes and treatments benefits, which are favored by patients and caregivers, but which have traditionally not been considered as measurements of outcome in clinical trials. Likewise, we need to know how to select responders, what is an optimal AChE inhibition appropriate in long-term settings, which patients respond to increased dosing for a sustained benefit, what is the optimal duration of treatment and when is meaningful to stop the treatment. After almost two decades of routine clinical use of donepezil these issues are still unresolved.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
