Abstract
Lubiprostone is a novel chloride channel 2 activator indicated for the treatment of chronic idiopathic constipation in adults and constipation-predominant irritable bowel syndrome (IBS-C) in adult women. It is effective for chronic constipation at a dose of 24 mcg twice daily and for IBS-C at a dose of 8 mcg twice daily. Lubiprostone is well-tolerated, with nausea and headache being the most commonly reported adverse drug reactions. It also has a low potential for drug-drug interactions. Lubiprostone is a potential treatment for both disorders, which have few treatment options available. Studies of longer duration will help more firmly establish its use for these chronic conditions.
Introduction
Chronic constipation is a commonly occurring ailment that results in 2.7 million physician visits, 38,000 hospitalizations and $750 million in over-the-counter remedy sales annually.1–3 In addition to the burden this disorder places on the health care system, patients’ quality of life is also affected. 4 Constipation is thought to affect up to 27% of the population. 5 The disease is more common in women, the elderly and in non-Caucasian races. 6
The Rome III criteria in Table 1, published in 2006, form a basis of diagnosis for constipation and take both symptoms and symptom duration into account. 5
Rome III diagnostic criteria for functional constipation in adults. 5

Since lifestyle changes and over-the-counter products are appropriate treatment options for constipation, patients often attempt these before seeking physician assistance. Lifestyle changes include increasing water and dietary fiber intake. Over-the-counter and prescription treatments used for constipation are listed in Tables 2 and 3. 1
Over-the-counter treatments for constipation. 1

Over-the-counter polyetheylene glycol is approved for occasional constipation and is not recommended for use beyond 1 week unless directed by a healthcare provider.
Prescription treatments for constipation. 1

Irritable bowel syndrome with constipation (IBS-C) affects 15%–20% of the population and predominantly affects women 20–50 years old. 4 Few patients seek treatment; however, it still accounts for numerous physician visits and results in work absenteeism. 7 The Rome III criteria for diagnosis of IBS are listed in Table 4. 5 Treatment of these patients presents a challenge because there are few options available. The agents presented in Table 5 have few randomized clinical trials that support their use in IBS-C.7,8
Rome III diagnostic criteria for irritable bowel syndrome in adults. 5

“Discomfort” means an uncomfortable sensation not described as pain.
Lubiprostone
Lubiprostone (Amitiza, Takeda Pharmaceuticals) is approved by the FDA for the treatment of chronic idiopathic constipation in adults and for the treatment of constipation-predominant irritable bowel syndrome in adult women. 9
Mechanism of action
Lubiprostone exerts its effects by activating chloride channel 2 (ClC2), which is found in the stomach, small intestine and colon. 10 Activation of this chloride channel increases chloride ion transport into the intestinal lumen, increasing intestinal fluid, and thus promoting motility. 9
Pharmacokinetic profile
Lubiprostone has low oral bioavailability and systemic plasma concentrations are below detectable levels. It is rapidly metabolized in the GI tract by carbonyl reductase into an active metabolite, M3, although this only makes up 10% of radiolabeled lubiprostone. This active metabolite is systemically absorbed; peak plasma concentrations are reached in approximately 1.1 hours and it has a half-life of 0.9–1.4 hours. 9 Gender does not have an effect on the pharmacokinetic profile of lubiprostone. The administration of lubiprostone with a high-fat meal did not affect the AUC, and thus, drug-food interactions are unlikely. 9 The drug is not metabolized via the cytochrome P450 system and is therefore unlikely to have drug-drug interactions.
Efficacy of Lubiprostone
Clinical studies: chronic constipation
To date, 3 clinical studies of lubiprostone for the treatment of chronic constipation have been published in manuscript form.11–13 Other study results have been presented as abstracts and are summarized in Table 6.14–20
Studies of lubiprostone for chronic constipation.
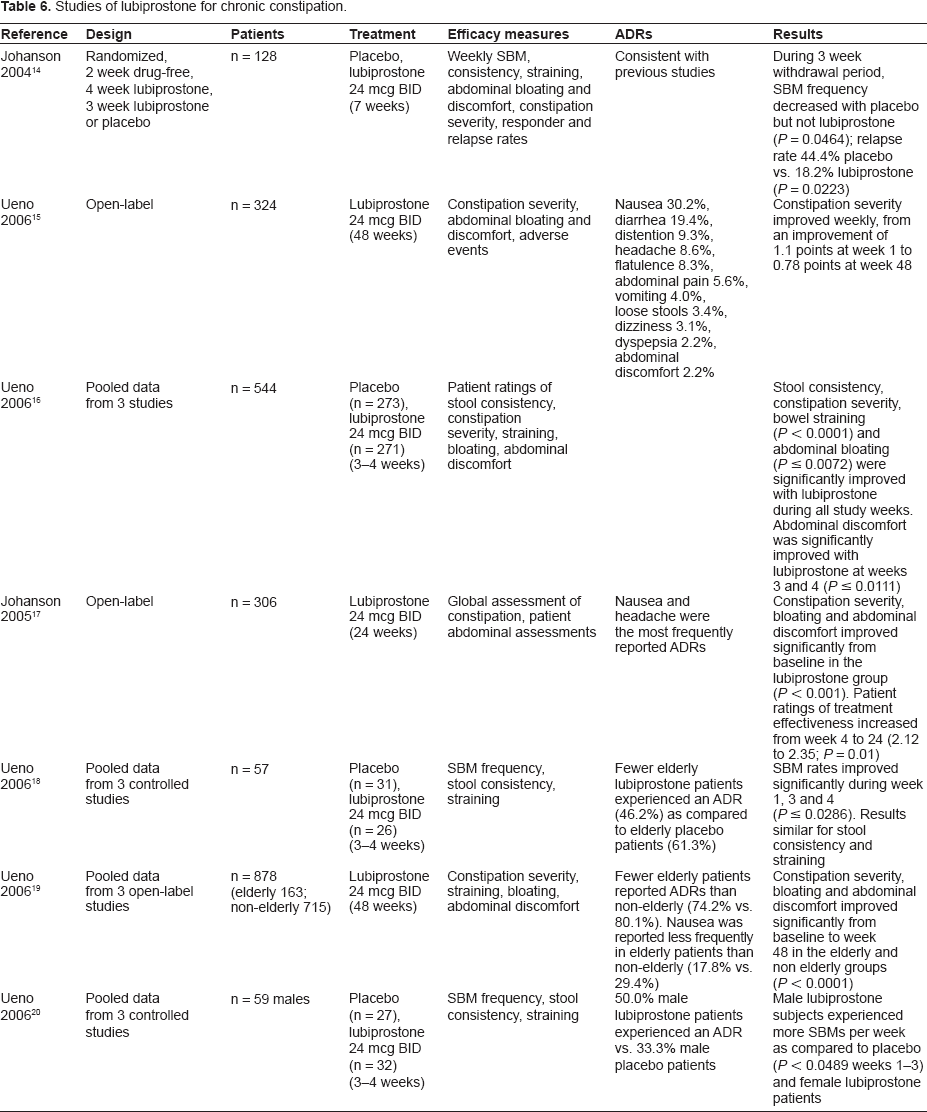
A phase II, randomized, double-blind, placebo-controlled dose-ranging study assessed the efficacy and safety of lubiprostone in 129 patients with chronic constipation. 11 Subjects were randomized to receive either 24 mcg/day (n = 29), 48 mcg/day (n = 32), 72 mcg/day (n = 33), or placebo (n = 33) for 3 weeks. Subjects 18–75 years were included if they had less than 3 spontaneous bowel movements (SBM) per week, a current diagnosis of chronic constipation, symptoms of bloating or discomfort and at least one symptom of constipation (hard stools, sensation of incomplete evacuation, straining) with ≥25% of bowel movements for at least 6 months. Subjects were excluded if they had mechanical bowel obstruction, drug-induced constipation, significant systemic disease, HIV, or known or suspected large bowel disorders. Efficacy measures included daily mean number SBMs, mean number SBMs per week, percentage of subjects experiencing SBMs on day 1, degree of straining, stool consistency, abdominal bloating and discomfort, global assessments and safety. Patients were allowed use of rescue medications of bisacodyl suppositories or sodium phosphate enemas.
Subjects receiving lubiprostone experienced more SBMs per week as compared to placebo during week 1 (P = 0.006), week 2 (P = 0.014) and during the 3 week treatment period (P = 0.046). The 48 mcg/day and 72 mcg/day doses had higher weekly SBM frequency as compared to placebo during the 3 week treatment period. The 42 mcg and 72 mcg per day doses also had higher percentage of subjects experiencing SBM on day 1 of treatment as compared to placebo (59.4%, P = 0.009 and 63.6%, P = 0.003, respectively). Among the subjective measurements, lubiprostone improved symptoms of straining, stool consistency, bloating and discomfort, and global assessments.
The most common adverse events were nausea, headache and diarrhea. There was a positive trend towards increasing adverse events with increasing dose (P = 0.006) and towards increased GI adverse events (P = 0.006).
The authors of this study concluded that the 48 mcg/day dose offered the most clinical improvement in symptoms with balanced adverse events. Thus, 48 mcg/day was chosen as the dose for subsequent phase III studies.
A phase III double-blind, randomized, placebo-controlled study evaluated the 48 mcg/day dose in the treatment of chronic constipation in adults. 12 In this study, 242 subjects were randomized to receive either lubiprostone 48 meg/day (n = 120) or placebo (n = 122) for 4 weeks. The primary endpoint was the number of SBMs during week 1 of treatment. Other endpoints included SBMs during weeks 2–4, straining, stool consistency, severity of constipation, bloating, discomfort, global assessments, and safety. Subjects over 18 years were included if they had less than 3 SBMs per week, and at least one symptom of constipation (hard stools, sensation of incomplete evacuation, straining) with ≥25% of bowel movements for at least 6 months. Subjects were excluded if they had mechanical obstruction, megacolon or megarectum, pseudo-obstruction, known or suspected large or small bowel disorders, secondary causes of constipation, hospitalization for any GI or abdominal surgery during 3 months prior to start of the study, prior bowel resection, significant systemic disease, neuropsychiatric disorders, serum creatinine >1.8 mg/dL, laboratory abnormalities, or cancer within the past 5 years. Subjects were allowed to use rescue medication of bisacodyl suppository, followed by sodium phosphate enema if necessary.
Ninety percent of subjects were female with a mean age of 48.6 years. The mean number of SBMs per week at baseline was 1.42. During the first week, subjects in the lubiprostone group experienced more SBMs than the placebo-treated subjects (5.69 vs. 3.46, respectively; P = 0.0001). More lubiprostone subjects experienced an SBM in the first 24 hours of treatment as compared to placebo (56.7% vs. 36.9%, respectively; P = 0.0024). Lubiprostone-treated subjects also reported more improvements in straining, stool consistency and constipation severity.
More subjects in the lubiprostone group reported adverse events (70.0%) as compared to the placebo group (50.8%; P = 0.0026). The most common adverse events were nausea and headache.
The authors concluded that lubiprostone 48 meg/day was effective in improving symptoms of chronic constipation and was also well-tolerated.
Another phase III double-blind, randomized, placebo-controlled study evaluated lubiprostone 48 mcg/day in adults with chronic constipation. 13 The subjects (n = 237) received the active drug 24 mcg twice daily (n = 119) or placebo twice daily (n = 118) for 4 weeks, preceded by a 2 week washout phase and followed by a 2 week follow up phase. The primary endpoint was the number of SBMs after 1 week of double-blind treatment. Secondary endpoints included the number of SBMs at weeks 2, 3 and 4; percent of subjects with an SBM after one dose; stool consistency; degree of straining; severity of constipation; abdominal bloating and discomfort; global treatment effectiveness; and safety assessments. Subjects were included if they were over 18 years, had a history of constipation, very hard and/or hard stools; sensation of incomplete evacuation; straining at defecation; or did not have flexible sigmoidoscopy or colonoscopy if they were greater than 50 years old. Subjects were excluded if they had a mechanical GI obstruction; megacolon, megarectum or pseudo-obstruction, organic disorder of the large or small intestine; secondary causes of constipation; hospitalization for a GI or abdominal surgical procedure in the preceding 3 months, prior bowel resection, did not have a flexible sigmoidoscopy or colonoscopy in the past 5 years if they were less than 50 years old. Investigators were allowed to prescribe bisacodyl suppository or sodium phosphate enema at their discretion as rescue medication.
Approximately 89% of subjects were female with a mean age of approximately 46 years. The mean number of SBMs per week at baseline were 1.3 ± 0.88 in the lubiprostone group and 1.5 ± 0.80 in the placebo group (P = 0.0382). The primary endpoint, the number of SBMs after 1 week of treatment, was 5.89 ± 5.0 in the lubiprostone group and 3.99 ± 3.5 the placebo group (P < 0.0001). More patients in the lubiprostone group experienced an SBM after one dose as compared to placebo patients (61.3% vs. 31.4%; P < 0.0001). The time to first SBM was also lower in the lubiprostone group (P = 0.003).
Adverse events were more commonly reported among lubiprostone patients (39.5%) as compared to placebo (18.6%). The most common adverse events reported were nausea (24.4% lubiprostone vs. 6.8% placebo) and headache (6.7% lubiprostone vs. 4.2% placebo). Treatment-related adverse events were more common among lubiprostone patients (42.9% vs. placebo 16.1%) with nausea and abdominal pain as the most common.
The authors concluded that lubiprostone 48 mcg/day was an effective treatment for chronic constipation.
Clinical studies: irritable bowel syndrome with constipation
The use of lubiprostone in patients with IBS-C was first evaluated in a post hoc analysis of two phase III constipation trials. 21 Although only a small subgroup of patients with IBS-C was identified, this analysis indicated lubiprostone's potential to alleviate symptoms in patients with IBS-C. SBMs, stool consistency, straining, abdominal bloating, discomfort and constipation severity all improved with lubiprostone compared to placebo.
A phase II trial demonstrated the safety, efficacy, and dosage of lubiprostone for the treatment of IBS-C. 22 This multicenter, double-blind, placebo controlled dose ranging study included 195 patients meeting Rome II criteria for IBS-C who were randomized to receive either placebo or either 16 mcg/day, 32 mcg/day, or 48 mcg/day of lubiprostone for 3 months. Subjects enrolled were predominantly women; however no significant differences in demographics between study arms were identified. The primary endpoint was the change in abdominal discomfort or pain during the first 28 days of treatment. Secondary endpoints included SBM frequency, abdominal bloating, constipation severity, degree of straining and stool consistency.
At the end of the first month, patients treated with lubiprostone demonstrated significant improvement in abdominal discomfort/pain compared to placebo (P = 0.043), with the 48 mcg/day arm displaying the most improvement (P = 0.023). After 2 months of treatment, all lubiprostone arms displayed significant improvement (trend P = 0.0336). Although significance was not achieved at the end of month 3, there was still improvement in each of the lubiprostone arms compared to placebo. There were significant improvements in SBM frequency, stool consistency, degree of straining, constipation severity and abdominal bloating across all three months, except for abdominal bloating at month 3. A dose-response realtionship was displayed in patients receiving lubiprostone and improvement of the various secondary endpoints; however, there was a greater incidence of adverse events with the higher doses. The authors concluded 16 mcg/day was the optimal dose for the treatment of patients with IBS-C.
An analysis evaluating 1171 patients with IBS-C meeting Rome II criteria was conducted from two phase III studies. 23 These multi-centered, double-blind, randomized, placebo-controlled trials were designed to demonstrate the efficacy and safety of lubiprostone 16 mcg/day for 12 weeks vs. placebo for the treatment of IBS-C. Patients were predominantly female Caucasians with a mean age of 46.6 years. The primary endpoint was the overall responder status determined by patient responses to the question “How would you rate your relief of IBS symptoms over the past week compared to how you felt before you entered the study?” Answers were based on a 7-point balanced scale; significantly worse, moderately worse, a little bit worse, unchanged, a little bit relieved, moderately relieved, or significantly relieved. Patients were considered an overall responder if they were monthly responders for at least 2 months of the study. A monthly responder was defined as those patients who reported their symptoms to be at least moderately relieved for the entire month or significantly relieved for at least 2 weeks of the month, with no rating of moderate or severe worsening.
In the pooled analysis, the number of total responders was significantly higher in the lubiprostone group compared to placebo (17.9% vs. 10.1%; P = 0.001), with lubiprostone patients almost twice as likely to experience improvement in symptoms. This same trend was demonstrated in the individual studies as well (P < 0.05). In addition, monthly responders were significantly higher in the lubiprostone group compared to placebo at months 2 and 3 (18.3% vs. 11.4%, P = 0.003 and 22% vs. 14.5%, P = 0.003), as well as weekly responders at weeks 2, 4, 5, 6, 10, and 12 (P < 0.03). Secondary endpoints, including abdominal discomfort/pain, bloating, straining, severity of constipation, frequency of SBM, and stool consistency, all improved in patients receiving lubiprostone compared to placebo. The effects of treatment to a patient's quality of life (QOL) was evaluated using a validated questionnaire for assessing IBS-QOL. Although there was no statistical significant improvement in regards to QOL, there was a trend towards greater improvement with lubiprostone use at week 12 (P = 0.066). There was, however, significant improvement compared to placebo in the domains of body image and health worry after 3 months of treatment (P ≤ 0.025). Lubiprostone also demonstrated improvements in the domains of social reaction, food avoidance, health worry, body image and dysphoria, displaying a 14 point change in the questionnaire from baseline.
Patients who demonstrated a >70% drug compliance in the above two phase III trials were eligible to participate in a follow-up study to assess the efficacy and safety of lubiprostone in a 36-week open-labeled extension period. 24 The primary endpoints were long-term safety and efficacy of lubiprostone defined as monthly responders, identical to the 12-week phase III trials. A total of 476 patients enrolled and received lubiprostone 16 mcg/day, creating two treatment groups: those who originally received placebo (“placebo-lubiprostone”) and those who originally received lubiprostone (“lubiprostone-lubiprostone”). Prior to the start of the open label extension, the monthly responder rate was significantly higher with lubiprostone vs. placebo (15% vs. 8%; P = 0.001. At the end of 36 weeks, both groups experienced an increase in responder rate (lubiprostone-lubiprostone = 37% and placebo-lubiprostone = 31%). The authors concluded that lubiprostone confers long-term efficacy and safety in patients with IBS-C.
A randomized withdrawal trial was conducted as part of one of the above mentioned phase III trials. 25 Following 12 weeks of treatment with lubiprostone, the 436 patients were then randomized to either continue with lubiprostone 16 mcg/day or be switched to placebo for 4 weeks. The primary outcomes were identical to the phase III trial.
The overall responder rate from the 12-week trial was significantly higher in the lubiprostone vs. placebo treated patients (14% vs. 8%; P = 0.029). At the end of the 4 week withdrawal period, both treatment groups displayed significant improvement (P < 0.001) compared to baseline for each of the symptoms evaluated (abdominal discomfort/pain, bloating, SBM, consistency, straining and severity). The lubiprostone-lubiprostone group had a 38% response rate at week 16 compared to 40% for the lubiprostone-placebo group. The authors concluded that lubiprostone is effective in relieving IBS-C symptoms and is not associated with symptom rebound following discontinuation.
Safety and Tolerability of Lubiprostone
Overall, lubiprostone appears to be well tolerated. Most trials indicated gastrointestinal complaints as the most common adverse events.11–13,23,26 In a pooled analysis evaluating 1429 patients (1113 lubiprostone 48 mcg/day and 316 placebo), 31.1% of patients experienced nausea and 13.2% experienced diarrhea; the majority of cases reported were considered to be mild to moderate in severity. 26 Similarly, in a phase III clinical trial evaluating lubiprostone 16 mcg/day in patients with IBS-C, nausea (8% vs. 4%) and diarrhea (6% vs. 4%) were the most frequently reported adverse events. 23 In a phase III clinical trial evaluating the safety of lubiprostone 48 mcg/day in patients with chronic constipation, nausea (21% lubiprostone and 4.2% placebo) and abdominal pain (6.7% lubiprostone and 4.2% placebo) were the most reported treatment adverse events experienced by patients. 13
Although nausea was experienced in up to 30% of all patients studied, analysis of pooled data indicates the incidence of nausea is decreased in the elderly (patients aged >65 years) and male populations.19,20 One analysis revealed that the elderly population had a markedly decreased incidence rate for nausea when compared to their non-elderly counterparts (17.8% vs. 29.4%). 19 In addition, adverse events in general were reported less frequently by elderly subjects as compared to non-elderly subjects (74.2% vs. 80.1%). In another analysis, the authors reported that nausea occurred in only 18.8% of elderly patients (overall rate of nausea = 31.1%) and 8.2% of male patients, compared with 34.5% of female patients. 26 The incidence of nausea decreased when lubiprostone was administered with food. 9
Initially, the safety of lubiprostone was demonstrated in a multiple, escalating dose study. 27 Twenty-six healthy volunteers were monitored for changes in vital signs, ECG parameters, general adverse events, and clinical laboratory tests. No clinically significant changes were identified. These findings have been confirmed in a phase II dose ranging study and in analysis of pooled data from previous trials.11,28,29 The phase II trial evaluated the safety of lubiprostone 24 mcg once, twice or three times daily in 129 constipated patients for 3 weeks. 11 No consistent changes were reported in blood chemistry, laboratory values and ECG parameters. In addition, a retrospective review analyzing the ECGs collected during this phase II trial concluded there was no evidence of QTc prolongation on ECG monitoring. 29 Lubiprostone has also been studied in a thorough QT/QTc study in 17 healthy volunteers. 29 Patients received a single dose of either 24 or 144 mcg. Again, lubiprostone demonstrated no QTc prolongation, consistent with previous data supporting lubiprostone's safety profile. Pooled data from three short-term, placebo-controlled trials and three long-term, open-labeled trials were evaluated to determine lubiprostone's effect on serum electrolyte levels (sodium, potassium, chloride, calcium, phosphorous, and magnesium). 28 A total of 1136 patients receiving lubiprostone 48 mcg/day were included in this analysis. No significant changes in serum electrolyte concentrations from baseline were observed over the course of treatment, which ranged from 3 to 48 weeks.
Lubiprostone has been designated pregnancy category C. This agent should only be used in pregnancy if the potential benefit clearly outweighs the risks to the fetus. Teratology studies have been conducted in rats, rabbits and guinea pigs. 9 Lubiprostone was not teratogenic in rats receiving 332-times the recommended human dose or rabbits receiving 33 times the recommended human dose; however, in guinea pigs receiving 2 and 6 times the recommended human dose, fetal loss was observed. There are no clinical trials available assessing lubiprostone's safety profile in pregnant women. The manufacturer did report of 6 women who became pregnant during clinical testing. The women were instructed to discontinue lubiprostone. Four pregnancies resulted in the birth of healthy babies. One woman was lost to follow-up and the sixth pregnancy was electively terminated. Prior to the initiation of lubiprostone in women who are of child-bearing age, a pregnancy test should be conducted to exclude pregnancy and the use of effective contraceptive measures should be implemented.
Place in Therapy
Currently, there are few therapies available for the treatment of chronic idiopathic constipation and IBS-C. For the treatment of both disorders, tegaserod remains an option; however, its use is restricted to a specific patient population (women under 55 years) and is only available to patients in emergency situations after FDA review of the patient cases.
In patients with chronic constipation, lifestyle modifications should be attempted initially, followed by the numerous over-the-counter treatments available. Lubiprostone, at a dose of 24 mcg twice daily, is an appropriate option for both men and women who fail those treatments. Chronic constipation is more prevalent in the elderly population; lubiprostone has shown not only efficacy and safety in this population, but also that adverse events were fewer in elderly patients as compared to younger patients.
Patients with IBS-C have few treatment options; lubiprostone 8 mcg twice daily is the only FDA-approved agent for this indication. Although lubiprostone is FDA-approved for the treatment of IBS-C only in women, the lack of age restriction and FDA case review makes it an appealing choice.
Lubiprostone is a viable therapeutic option for both chronic idiopathic constipation and IBS-C. It lacks drug-drug and drug-food interactions. It is well-tolerated, with nausea as the most common and bothersome adverse effect, although, administration of lubiprostone with food may help reduce the incidence of nausea. Patients with chronic constipation who are unable to tolerate the 48 mcg/day dose due to nausea may tolerate a lower dose that still offers symptom relief.
Conclusion
Lubiprostone is a novel agent with a unique mechanism of action. Its efficacy and safety have been demonstrated in short term trials for the treatment of chronic idiopathic constipation and IBS-C. Lubiprostone has been studied for up to 48 weeks in both chronic constipation and IBS-C; these long-term studies of lubiprostone in the treatment of chronic constipation have been published in abstract form only and are not discussed in this review because adequate analysis and review of those data can not be performed.
The studies of both chronic constipation and IBS-C showed that lubiprostone increased the number of SBMs, decreased the time to first SBM, decreased straining, improved stool consistency, and decreased constipation severity. Lubiprostone was overall well-tolerated, with nausea and headache being the most commonly reported adverse events. More long-term studies, especially in patients with IBS-C, will firmly establish its role in treatment of these conditions.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.

