Abstract
Docetaxel, a member of the taxane family, promotes cell death by binding β-tubulin and has demonstrated activity against several human malignancies, both as a single agent and in combination therapy. It has been approved in Europe and the US as front-line treatment for advanced gastric cancer in combination with cisplatin and fluorouracil (DCF regimen). This approval was based on the results of a pivotal study (V325) which demonstrated that the addition of docetaxel to the reference regimen of cisplatin and fluorouracil improves overall survival and progression-free survival with a better quality of life despite increased toxicity (mainly haematological). Modifications of DCF regimen have been successfully investigated as a means of making the treatment more tolerable and suitable also for elderly patients or patients with poor performance status. Emerging data from several phase II studies suggest that other docetaxel-based combination regimens with anthracyclines or irinotecan have interesting activity with acceptable toxicity profiles, but the true efficacy of these regimens needs to be assessed in large randomized phase III studies. Thus, the best docetaxel-containing regimen has yet to be identified. Docetaxel also represents a good candidate for combination with novel molecular target agents. In light of the high response rates observed in phase II-III studies, a docetaxel-based chemotherapy regimen might also be considered a treatment option as perioperative or adjuvant therapy in potentially curable gastric cancer and further studies with or without biological agents are eagerly awaited in this setting.
Keywords
Introduction
Despite a decline in incidence in developing countries over the last five decades, 1 gastric cancer remains the fourth most common cancer and the second most common cause of cancer death worldwide. 2 Although surgery is the treatment of choice for early disease, unfortunately about 60% of radically resected patients relapse, 3 and most patients present at diagnosis with unresectable, locally advanced or metastatic disease. For these patients, treatment intent is palliative and therapeutic options are limited, with a median survival time for those who undergo first-line chemotherapy of no more than approximately 10 months in any study. 4
Although the most effective treatment for advanced gastric cancer has not yet been clearly established despite large numbers of studies carried out in this setting, a commonly adopted regimen is the combination of epirubicin, cisplatin and infusional fluorouracil (ECF), which is associated with an overall response rate (ORR) of 30%–35% and a median overall survival (OS) time of 8–9 months.5,6 Recently, docetaxel has been shown to be active against advanced gastric cancer, both as first-line treatment in combination with other cytotoxic drugs, mainly cisplatin and fluorouracil, and as a single agent in pre-treated patients.
Docetaxel in Advanced Gastric Cancer
Docetaxel as a single agent
Based on preclinical data, single agent docetaxel was investigated in advanced or recurrent gastric cancer, both as front-line and salvage chemotherapy, in studies that revealed promising activity.7–12 In these studies, docetaxel was usually administered every 3 weeks at a dose of 60–100 mg/m2, with an ORR of 16%–24% (mean 20%). The most commonly observed toxicities were neutropenia, leukopenia, nausea, vomiting, stomatitis, diarrhoea, fatigue and neuropathy.
To reduce toxicity, some investigators evaluated a weekly schedule of docetaxel at a dose of 35 or 36 mg/m2 in elderly patients or patients with impaired performance status; patients were either pre-treated or chemotherapy naïve.13,14 The ORR was 5%–9%, suggesting that weekly low-dose docetaxel is probably less active than the higher dose administered every 3 weeks. However, the median OS in these studies was 7–7.5 months, which is superior to the survival duration of 3–5 months that might be expected in patients treated with best supportive care alone.15,16 Furthermore, a favourable safety profile was observed in these trials, suggesting that weekly administration could be considered in elderly patients, those presenting with significant co-morbidities or heavily pretreated, all of whom would otherwise have a poor prognosis.
Docetaxel in combination with platinum compounds and fluoropyrimidines
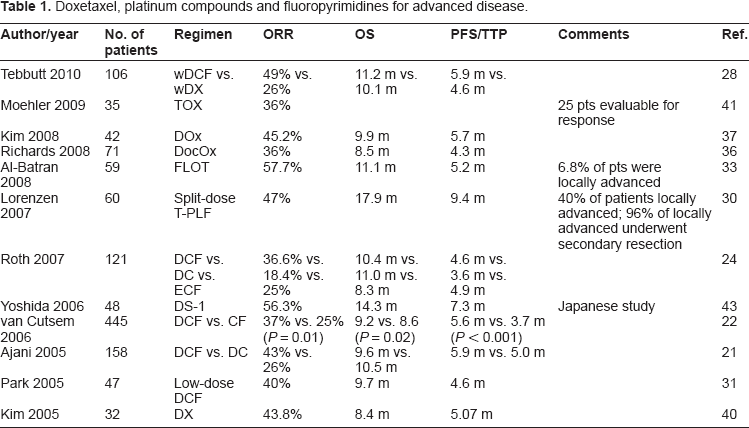
Building on the evidence of docetaxel activity as a single agent in gastric cancer, several clinical trials were designed to investigate the efficacy of docetaxel in combination with other cytotoxic agents (Table 1).
Doxetaxel, platinum compounds and fluoropyrimidines for advanced disease.
Docetaxel in combination with cisplatin and fluorouracil
The first drug studied in combination with docetaxel was cisplatin, which for several decades has been considered the mainstay of treatment of metastatic gastric cancer because of its good activity profile and has had extensive evaluation, mainly in combination with fluorouracil alone (CF regimen) or, as mentioned above, with 5-fluorouracil and anthracyclines (ECF regimen).5,6,17,18
One of the first combination studies was a phase II study carried out to evaluate the feasibility of docetaxel 85 mg/m2 in combination with cisplatin 75 mg/m2 (DC), both administered on day 1 every three weeks, as first-line chemotherapy in patients with advanced disease. 19 In this trial, the ORR in the intent-to-treat population was 52% in 48 patients, with two complete responses. The median time to progression (TTP) of disease was 6.6 months and the median OS was 9 months. The major toxicity was haematological, with grade 3 or 4 neutropenia occurring in 81% and febrile neutropenia in 17% of patients, respectively. However, these adverse effects were expected and manageable; no grade 4 non-haematological toxicity was observed. Other grade 3 toxicities were anaemia, fatigue, mucositis and diarrhoea.
Once the activity and tolerability of the DC regimen had been demonstrated, considerable effort was made to further improve this taxane-based combination. A logical choice was the addition of fluorouracil, given that this agent represents the backbone of most of the effective chemotherapy regimens available for the treatment of advanced gastric cancer (including ECF). Moreover, since fluorouracil has low haematological toxicity when given by continuous infusion, it was considered that this agent could be added to DC without worsening neutropenia rates.
Therefore, a phase I/II dose-finding study was conducted to evaluate the feasibility of adding a protracted continuous infusion of fluorouracil (300 mg/m2/day for 2 weeks) to the previously evaluated DC regimen. 20 As expected, haematological toxicity was no more a problem with DCF than it had been observed with DC. For example, grade 3 or 4 neutropenia occurred in 79% of patients (compared to 81% previously reported for DC). 19 Other grade 3 or 4 non-haematological toxicities were alopecia, fatigue, mucositis, diarrhoea, nausea and vomiting. Moreover, although the limitations of a phase I/II study need to be taken into account, the DCF regimen was shown to be very active, with an ORR in the intent-to-treat population of 51%.
Based on these results, a randomized phase II/III trial (V325), was designed to better define the efficacy of a docetaxel combination treatment for advanced gastric cancer. 21 In the phase II part of the trial, DC was compared with DCF to determine the experimental treatment to be taken forward into the ensuing phase III trial for comparison against cisplatin/fluorouracil (CF). Accordingly, 155 patients received either DCF (docetaxel 75 mg/m2 plus cisplatin 75 mg/m2 on day 1 plus fluorouracil 750 mg/m2/day as a continuous infusion on days 1–5, every 3 weeks) or the previously evaluated DC regimen. The ORR in the intent-to-treat population was 43% for DCF and 26% for DC with an acceptable safety profile, 21 and the former regimen was therefore selected by an independent data monitoring committee for comparison against CF. The choice of this comparator arm in a registration trial for docetaxel in gastric cancer was based on the results of a phase III study with CF. 18 The advantage of this non-anthracycline-containing regimen over the classic ECF regimen was thought to be lower haematological toxicity. However, when the study was initiated, it was not widely accepted that a doublet without epirubicin could be considered standard therapy or that the efficacy of this doublet was equivalent to that of a triplet containing epirubicin.
In the phase III part of the V325 trial, 457 patients with metastatic and untreated gastric adenocarcinoma were randomly treated with either DCF (as described above) or CF (cisplatin 100 mg/m2 on day 1 plus fluorouracil 1000 mg/m2/day as a continuous infusion on days 1–5, every 4 weeks).
22
The median TTP was significantly longer with DCF than with CF (5.6 months vs. 3.7 months, respectively;
Although the V325 study showed a superiority for DCF triplet therapy compared with CF, 22 resulting in FDA approval for use of DCF in the treatment of advanced gastric and gastro-oesophageal adenocarcinoma, as mentioned previously, the question of whether the CF doublet combination can be considered an optimal control arm for comparison with the DCF triplet remains open. Indeed, a population data-based meta-analysis showed that adding epirubicin to cisplatin and fluorouracil resulted in a more prolonged survival, 23 suggesting perhaps that ECF would have represented a better control arm for V325. However, it must be noted that, to date, the relative efficacies of ECF and CF have not been directly compared in a phase III study, and a definitive answer to this question is not available. In a phase II study, 119 patients with gastric cancer were randomized to receive ECF, DC or DCF. 24 The response rate obtained with DCF (37%) was higher than that with DC (18%) or ECF (25%), but no conclusions could be drawn about TTP or survival for DCF versus ECF because the study was not designed to answer this question. A phase III trial could clarify whether there are any differences in superiority between taxane- and anthracycline-containing regimens. It is also possible that data arising from an individual patient data-based meta-analysis, the GASTRIC (Global Advanced/Adjuvant Stomach Tumor Research through International Collaboration) Project, 25 whose interim results have been presented at 2009 ASCO annual meeting, could help to quantify the potential benefit of docetaxel and anthracycline using specific regimens analysis. Preliminary data show no significant difference in 1-year survival rate with anthracycline-based regimens compared with regimens without anthracyclines.
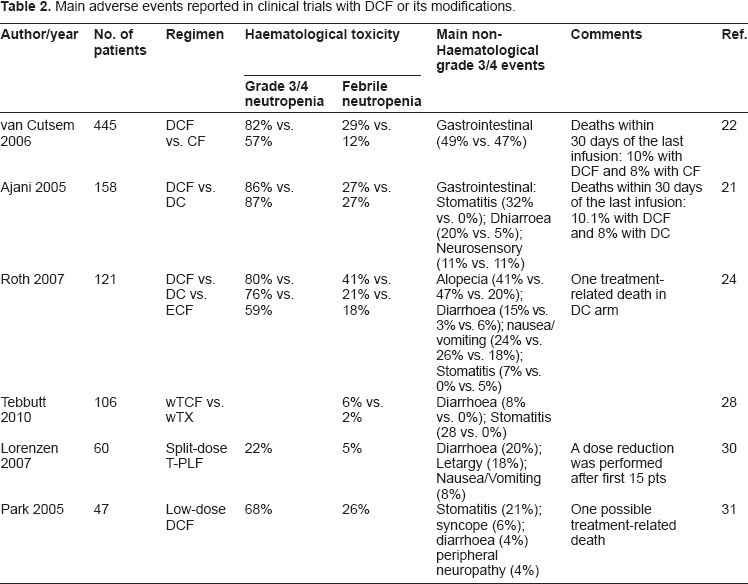
Addition of docetaxel to CF in the V325 trial resulted in improved efficacy but also gave rise to higher rates of grade 3 or 4 neutropenia (82% vs. 57%), complicated neutropenia (29% vs. 12%) and grade 3 or 4 diarrhea (19% vs. 8%) compared with the doublet. 22 The number of deaths occurring within 30 days of the last chemotherapy infusion was 23 (10%) with DCF and 19 (8%) with CF. Of note, complicated neutropenia (i.e. febrile neutropenia or neutropenic infection) was the main cause of toxic deaths in both arms (7 of 8 in DCF; 8 of 12 in CF), and grade 3 to 4 infection related to treatment was more frequently reported with DCF (20%) than CF (9%) in patients age 65 years or older. These findings could influence the feasibility of DCF in elderly patients.
Interestingly, the incidence of complicated neutropenia in the DCF arm was reduced to 12% (the same rate as observed in the CF arm) with the use of secondary granulocyte colony-stimulating factor (G-CSF) prophylaxis. These data suggest both that G-CSF can be beneficial in the management of docetaxel-associated febrile neutropenia and that primary G-CSF prophylaxis should also be considered for patients receiving DCF. Indeed, ASCO recommends routine primary G-CSF prophylaxis when using chemotherapy regimens associated with a risk of febrile neutropenia of more than 20%, 26 which is within the range associated with docetaxel-based regimens and consistent with the incidence reported in the V325 study. 22 Furthermore, the National Comprehensive Cancer Network (NCCN) lists DCF as an example of a chemotherapy regimen associated with a high risk of febrile neutropenia. 27
In addition to utilization of G-CSF to reduce the incidence of febrile neutropenia, some modifications to the original DCF regimen have been pursued as a means of making the triplet a less intense and more generally tolerable treatment.
Based on previously reported findings that weekly docetaxel is associated with a lower incidence of haematological toxicity, a randomized phase II study, the ATTAX trial, evaluated the safety and activity of docetaxel given weekly in combination with cisplatin and fluorouracil (wDCF: docetaxel 30 mg/m2 on days 1 and 8, cisplatin 60 mg/m2 on day 1 and fluorouracil 200 mg/m2/day as continuous infusion for 21 days every 3 weeks) or with capecitabine (wDX; the same schedule of docetaxel combined with capecitabine 1600 mg/m2/day on days 1–14, every 3 weeks). 28 After a median follow-up of 40.7 months, median PFS and OS times were 5.9 and 11.2 months for wDCF and 4.6 and 10.1 months for wDX, respectively. The ORR for wDCF was 47%, comparable to the rate of 37% reported in the V325 phase III study for DCF. 22 Interestingly, both wDCF and wDX had acceptable haematological toxicity profiles with only 6% and 2% febrile neutropenia, respectively, markedly lower than that reported with DCF in the V325 trial, suggesting that further evaluations of weekly docetaxel combination regimens in patients with advanced gastric cancer are warranted. 28
The better tolerability of a weekly schedule of docetaxel has been also confirmed in a retrospective analysis of 95 patients, 29 treated with docetaxel (20 mg/m2 weekly for 6 weeks followed by two weeks break), cisplatin (20 mg/m2, weekly for 6 weeks followed by two weeks break) and 5-FU (350 mg/m2, weekly for 6 weeks followed by two weeks break). Only four patients developed grade 3–4 neutropenia and only nine patients grade 3–4 anemia. The study confirmed an ORR of 34%, with a median TTP of 4.1 months and a median OS of 8.9 months.
A split-dose regimen (docetaxel, cisplatin, leucovorin [folinic acid] and fluorouracil [T-PLF]) was evaluated as a modification of the classic DCF regimen in the GASTRO-TAX-1 phase II trial. 30 In this study, 60 patients with locally advanced (n = 24) or metastatic (n = 36) gastro-oesophageal adenocarcinoma were treated with docetaxel 50 mg/m2 on days 1, 15 and 29, cisplatin 50 mg/m2 on days 1, 15 and 29 and leucovorin 500 mg/m2 and 24-hour infusion fluorouracil 2000 mg/m2, both administered on days 1, 8, 15, 22, 29 and 36, every 8 weeks. After treatment of the first 15 patients, the doses were amended to docetaxel 40 mg/m2, cisplatin 40 mg/m2, leucovorin 200 mg/m2 and fluorouracil 2000 mg/m2 because of frequent grade 3 and 4 toxicities. After the protocol amendment, treatment was generally well tolerated. As expected, haematological toxicity was relatively common and grade 3 and 4 neutropenia occurred in 22% of patients; however, febrile neutropenia was observed only in 5% of patients. The most frequent grade 3 and 4 non-haematological toxicities were gastrointestinal events, including diarrhoea (20%) and nausea and vomiting (8%). After a median follow-up time of 25.5 months, the ORR was 47%, median TTP was 9.4 months and the median OS was 17.9 months. Although the inclusion of patients with locally advanced disease in the trial may have affected the survival data, this evaluation of the T-PLF regimen confirmed the activity of the triplet combination. Moreover, the safety profile of the regimen was encouraging. However, dose modifications were required in 65% of patients and toxicity was the main reason for treatment discontinuation, raising doubts as to whether a simple splitting of the dose represents a substantial improvement in the tolerability of DCF. 30
Another attempt to make DCF more tolerable was evaluated in a phase II study of low-dose docetaxel 50 mg/m2 on day 1 given in combination with cisplatin 80 mg/m2 on day 1 and fluorouracil 1200 mg/m2/day on days 1–3 every 3 weeks as first line chemotherapy in 47 patients with metastatic gastric cancer. 31 The ORR was 40%, similar to that observed with the classic normal dose DCF regimen in the V325 trial, 22 the median TTP and OS were 4.6 and 9.7 months, respectively, and the regimen had an acceptable toxicity profile. Indeed, grade 3–4 neutropenia developed in 68% of patients, febrile neutropenia or neutropenic infection occurred in 26%, and the most common grade 3 or 4 non-haematological toxicity was stomatitis (21%). Thus, both haematological and non-haematological toxicities appeared to occur less frequently with low-dose DCF than with classic DCF as reported in the V325 trial. 22 However, the lower total dose and decreased length of infusion of fluorouracil (3600 mg/m2/cycle and infusion over 3 days for the modified DFC regimen vs. 3750 mg/m2/cycle and infusion over 5 days for classic DCF) might have been just as important reason for the reduction in non-haematological toxicity observed with the modified DCF regimen as the lower dose of docetaxel administered. However, the study showed that modifications of the original DCF regimen can obtain similar efficacy with more favourable toxicity profiles than the classic DCF regimen (Table 2).
Main adverse events reported in clinical trials with DCF or its modifications.
Docetaxel in combinations containing oxaliplatin
A promising attempt to make DCF a better tolerated regimen without affecting its activity is the substitution of another platinum compound for cisplatin. In a two-by-two randomized phase III trial, the REAL-2 (Randomised ECF for Advanced and Locally Advanced Esophagogastric Cancer-2) study, 1002 patients with locally advanced or metastatic oesophageal, gastric or gastro-oesophageal junction cancer were treated with triplet therapy with epirubicin and cisplatin plus either fluorouracil (ECF) or capecitabine (ECX) or triplet therapy with epirubicin and oxaliplatin plus either fluorouracil (EOF) or capecitabine (EOX). 32 This study demonstrated comparable OS and PFS for oxaliplatin and cisplatin containing regimens but, compared with cisplatin, oxaliplatin had a better toxicity profile with lower rates of neutropenia, renal toxicity, alopecia and thromboembolism. Furthermore, treatment with oxaliplatin does not require pre- and post-hydration as is required with cisplatin. On the other hand, however, a slightly higher incidence of grade 3 or 4 diarrhoea and neuropathy was observed in patients treated with oxaliplatin-containing triplets. Since there is no effective prophylaxis or treatment for peripheral neuropathy induced by oxaliplatin, this kind of toxicity is worrying because it could interrupt treatment or reduce the dose-intensity. However, based on the safety data for oxaliplatin-containing triplet therapy observed in the REAL-2 study and on the positive efficacy results for docetaxel in combination with a platinum compound and fluorouracil in the V325 study, 22 a phase II trial was conducted to assess the feasibility of incorporating docetaxel in a tolerable biweekly oxaliplatin-based chemotherapy regimen. 33 In this study, 54 patients with metastatic adenocarcinoma of the stomach or gastro-oesophageal junction were treated with oxaliplatin 85 mg/m2 on day 1, leucovorin 200 mg/m2 on day 1, docetaxel 50 mg/m2 on day 1 and fluorouracil 2600 mg/m2 as a 24-hour continuous infusion, every 2 weeks (FLOT). The reported ORR was 57.7% and the median PFS and OS were 5.2 and 11.1 months, respectively, after a median follow-up of 18.1 months. The predominant toxicity was haematological, with 48.1% of patients having grade 3 or 4 neutropenia, a lower rate than that observed in the V325 trial with DCF (82%). 22 The non-haematological toxicity profile of FLOT was also favourable compared with DCF, with diarrhoea reported in 14.8% of patients and peripheral neuropathy at a relatively low rate (about 9%), suggesting that docetaxel does not enhance this dose-limiting toxicity of oxaliplatin. Similarly, a phase I study was conducted to determine the maximum tolerated dose of docetaxel when given in combination with oxaliplatin 85 mg/m2 and fluorouracil 2200 mg/m2 as a 48-hour continuous infusion, every two weeks (D-FOX). 34 Preliminary results showed that the safety profile of D-FOX was acceptable, with grade 3 and 4 toxicities not being common. The phase I part of the study has been completed and a docetaxel dose of 55 mg/m2 was selected for the D-FOX regimen in the phase II trial, which is ongoing. 35
A phase II trial evaluated the combination of docetaxel 60 mg/m2 and oxaliplatin 130 mg/m2, both given on day 1 every 3 weeks (DOCOX regimen), in 71 patients with metastatic disease. 36 DOCOX has demonstrated activity in advanced gastric cancer, with an ORR of 36%, similar to that reported in the DCF arm of the V325 study. 22 The observed toxicity was mild to moderate and manageable, with grade 3 or 4 neutropenia in 70% of patients and grade 3 neuropathy in 6% of patients. Similar data derived from another smaller phase II study that evaluated a similar regimen with slightly different dosages (docetaxel 65 mg/m2 and oxaliplatin 120 mg/m2). 37 Since triplet regimens appear to be more effective than doublets, it may be that addition of a fluoropyrimidine to the DOCOX regimen could improve its activity. To address this question, a large randomized phase II trial, the GATE study, is ongoing; in this trial, patients will be assigned to DOCOX alone or DOCOX plus fluorouracil or capecitabine. 38 In summary, oxaliplatin and docetaxel combinations seem to be effective and well tolerated treatments, but randomized clinical trials are needed to evaluate whether FLOT, D-FOX or DOCOX with or without fluoropyrimidines may represent valuable alternative treatments to the DCF regimen.
The combination of docetaxel and oxaliplatin has also been evaluated as second-line treatment, after failure of ECF or CF regimens. 39 In this setting the combination of docetaxel (75 mg/mq, day 1 every 3 weeks) and oxaliplatin (80 mg/mq, day 2 every 3 weeks) has shown an acceptable profile of toxicity with no febrile neutropenia reported and only 26% of patients experiencing grade 3–4 neutropenia. Although response rate was low (10%), median TTP (4.0 months) and median OS (8.1 months) are comparable to those of front-line therapies, thus suggesting that this regimen could be beneficial also in pre-treated patients.
Docetaxel in combination with oral fluoropyrimidines
The results of the previously discussed REAL-2 study suggest that capecitabine can substitute for fluorouracil with the same efficacy and a different pattern of side effects, but with a generally better toxicity profile. 32 More specifically, when capecitabine is used instead of fluorouracil, neutropenia occurs less frequently, but hand-foot syndrome is more often observed. In a phase II study, 32 patients with previously untreated advanced gastric cancer received docetaxel 75 mg/m2 on day 1 and capecitabine 1000 mg/m2 twice daily from day 1 to 14, every 21 days. 40 Amongst the 30 patients who were evaluable for response, the ORR was 43.8% and the median OS was 8.4 months. Grade 3 or 4 neutropenia occurred in 9.7% of patients, and grade 2 or 3 hand-foot syndrome in 12.9%.
Preliminary results of an ongoing phase II study suggest that addition of oxaliplatin to a docetaxelcapecitabine schedule is feasible with a low incidence of grade 3–4 toxicities. Grade 3 anemia is reported in 10% of patients, while only one patient experienced febrile neutropenia. Non heamatological adverse events included grade 3–4 diarrhoea (23%), vomiting (14%) and asthenia (9%). Efficacy is encouraging with a 36% response rate and 40% of disease stabilitazion. 41
Among oral fluoropyrimidines, S-1 is widely used in Japan following the results of a phase III study that documented a survival benefits for patients receiving cisplatin and S-1, compared to patients receiving S-1 alone (11 vs. 13 months;
Docetaxel in combination with other cytotoxic drugs
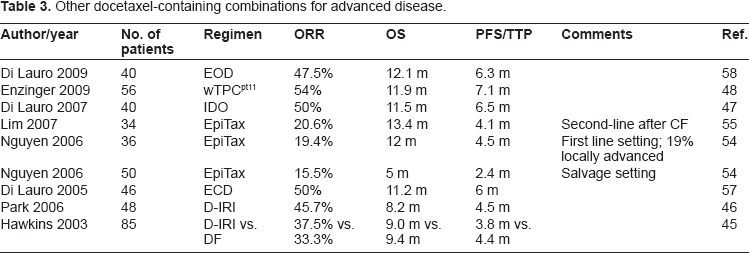
Docetaxel in combinations containing irinotecan Irinotecan, a topoisomerase I inhibitor, has shown activity in the treatment of advanced gastric cancer, with phase II studies reporting response rates of 14%–23% for irinotecan as a single agent and 22%–51% when the drug is given in combination with fluorouracil or cisplatin. 44 Given the activity of both drugs, an irinotecan-docetaxel combination was investigated, but only data from phase II studies are available. In a randomized phase II study, a combination of docetaxel 60 mg/m2 and irinotecan 250 mg/m2, both given on day 1, every 3 weeks, was evaluated as first-line chemotherapy in comparison with docetaxel 85 mg/m2 on day 1 and fluorouracil 750 mg/m2 protracted continuous infusion from day 1 to 5, every 3 weeks. 45 The observed ORR in the docetaxel-irinotecan arm was 37.5% (versus 33.3%), with a median OS of 9 months. However, treatment was associated with considerable toxicity; the most frequently observed grade 3 and 4 toxicities were neutropenia (85%), febrile neutropenia (26.8%) and diarrhoea (42.9%).
Weekly and bi-weekly schedules have been developed to reduce the toxicity of the docetaxel-irinotecan doublets. In a phase II study, docetaxel 30 mg/m2 and irinotecan 70 mg/m2 were administered on days 1 and 8, every 3 weeks, with an ORR of 45.7% and a median OS of 8.2 months after a median follow-up of 15 months. 46 Grade 3 and 4 events were less frequent than those observed with the 3-weekly schedule. More specifically, neutropenia of grade 3 or 4 was observed in 57.4% of patients, and the most frequent non-haematological toxicity (diarrhoea) occurred only in 19.1% of patients.
Building on evidence of the activity of the docetaxel-irinotecan combination, a triplet regimen with the addition of oxaliplatin was evaluated in a phase II study. 47 In this study, 40 patients with advanced gastric cancer received irinotecan 150 mg/m2 and docetaxel 60 mg/m2 on day 1 plus oxaliplatin 85 mg/m on day 2, every 3 weeks. The ORR was 50% with a median OS of 11.5 months; grade 3 or 4 neutropenia occurred in 47.5% of patients, and non-haematological toxicity was moderate with a grade 3 diarrhoea in 10% of patients. Similarly, another phase I/II study evaluated docetaxel and irinotecan in association with cisplatin, administered according to the following weekly schedule: docetaxel 30 mg/m2, cisplatin 25 mg/m2 and irinotecan (initially 65 mg/m2, then reduced to 50 mg/m2 because of a high frequency of diarrhoea). 48 The combination was administered as first-line chemotherapy to 56 patients with advanced gastro-oesophageal cancer enrolled in the phase II trial. The ORR was 54% with three complete responses, the median PFS was 7.1 months and the median OS was 11.9 months. With this reduced dose of irinotecan, grade 3 and 4 toxicities were uncommon, and the most frequently observed toxicities were neutropenia and diarrhoea.
These emerging data suggest that the docetaxel-irinotecan combination, especially if the regimen also contains platinum compounds, could be active in advanced gastric cancer, and it may be that modifications of the classic 3-weekly schedule could make the treatment more tolerable. Phase III studies are required to definitively evaluate the true efficacy of this combination compared with other regimens, particularly DCF, which is the standard docetaxel-based therapy for gastric cancer. However, as observed in phase III studies that evaluated addition of irinotecan to fluoropyrimidine or cisplatin as first-line therapy for advanced gastric cancer, addition of irinotecan does not seem to confer a survival benefit.49,50 Thus, irinotecan combined with docetaxel may not be ideal in the front-line setting, but may be considered an option worth evaluating in the setting of salvage treatment.
Docetaxel in combinations containing epirubicin
Epirubicin is another chemotherapeutic agent that has been and remains widely used in the treatment of advanced gastric cancer. In other malignancies, particularly metastatic breast cancer, epirubicin and docetaxel have synergistic effects and their toxicity profiles do not overlap;51,52 furthermore, combining epirubicin with taxanes does not influence the pharmacokinetic of either compound. 53 These findings, together with a reported high activity of both drugs used separately as single agents in advanced gastric cancer, provided a strong rationale for evaluating a docetaxel-epirubicin combination in metastatic gastro-oesophageal carcinomas.
Two French phase II trials evaluated the tolerability and efficacy of the Epi-Tax regimen, that is, docetaxel 75 mg/m2 on day 1 given in combination with epirubicin 60 mg/m2 on day 1, every 3 weeks, both as first- and second-line therapies. 54 The most common toxicity reported was haematological in both settings, but this was expected and manageable, suggesting that combining these two drugs is feasible. Unfortunately, however, the efficacy data from these studies were disappointing, with ORRs of 19.4% and 15.5%, respectively, for first-and second-line treatment. A more recent Japanese study has reported a similar toxicity profile and comparable ORR (21.8%) for an epirubicin-docetaxel regimen in a salvage treatment setting. 55
Despite these discouraging results, epirubicin-docetaxel combinations have been further investigated, and several attempts to improve the efficacy of this doublet by adding a platinum compound have been undertaken. Two phase II studies evaluated docetaxel (generally 50–60 mg/m2 on day 1) in combination with epirubicin 30–60 mg/m2 on day 1 and cisplatin 60–75 mg/m2 on day 1 or 2, every 3 weeks (ECD regimen).56,57 In these studies, the ORR was 47%–50%, the median TTP 5 months and the median OS 11 months. There was also significant haematological toxicity, mainly consisting of neutropenia. Another similar phase II study was conducted using the EOD regimen, i.e. docetaxel 50 mg/m2 and epirubicin 60 mg/m2 on day 1 combined with oxaliplatin 100 mg/m2 on day 1, every 3 weeks. 58 The ORR was 47.5%, the median OS was 12.1 months, and the main toxicity was haematological with grade 3 or 4 neutropenia in 50% of patients. These data confirm the previously reported observation that in gastric cancer a triplet is generally more active than a doublet of drugs, and suggest that the combination of docetaxel and anthracyclines does not have the same efficacy in gastric cancer as has been reported for other malignancies such as breast cancer. Docetaxel probably has better synergism with platinum compounds or fluoropyrimidines than with epirubicin in gastric cancer; indeed, addition of a platinum compound to the docetaxel-epirubicin doublet seems to consistently increase treatment efficacy, if we compare results from studies with Epi-Tax to those with EOD or ECD regimens. 58 Clearly, the encouraging data about the efficacy of EOD or ECD regimens arising from phase II studies needs further confirmation in larger randomized phase III trials in terms of OS and clinical benefit. Phase III studies may also clarify the possible role of docetaxel combined with platinum compounds and anthracyclines and evaluate the efficacy and toxicity of such a combination compared with DCF (Table 3).
Other docetaxel-containing combinations for advanced disease.
Docetaxel and quality of life
Since patients with advanced gastric cancer are often symptomatic when they receive the diagnosis, the optimal treatment would not only prolong OS and PFS, but would also give rise to an improvement in quality of life (QoL) and clinical benefits. However, it is not always true that response to treatment correlates with a positive effect on patient's QoL or global health status. As previously discussed, docetaxel has demonstrated good activity against advanced gastric cancer, but what data are available about its impact on QoL and clinical benefits? The V325 pivotal study has specifically assessed this issue; indeed, evaluation of QoL and clinical benefit was an important secondary endpoint of that trial. 22
Contrary to expectations, given increase in toxicity when docetaxel was added to CF, in the V325 trial both QoL and clinical benefit were better in the docetaxel-containing regimen.59,60 QoL was primarily assessed using the disease-specific European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire C30 (EORTCQLQ-C30), which incorporates functional, symptom and global health status scales in addition to six single items assessing additional symptoms commonly reported by cancer patients. 59 When available, the generic and relatively simple to administer EuroQOL EQ-5D questionnaire, which comprises five questions and a visual analogue scale, was also administered to patients. Time to definitive deterioration from baseline in global health status, physical and social functioning, appetite loss, nausea and vomiting, pain and EQ-5D visual analogue scale scores consistently favored DCF over CF. DCF also significantly prolonged time to definitive worsening of Karnofsky performance status. Furthermore, time to definitive worsening of appetite and time to weight loss were longer in the DCF arm, albeit not statistically significantly.
Docetaxel and target therapies
To improve efficacy in the treatment of advanced gastric cancer, the contribution of adding molecular target agents to traditional cytotoxic drugs was investigated. Cetuximab and bevacizumab are the molecules for which there is the greatest body of available clinical data relating to their use in combination with docetaxel. However, addition of other drugs such as sorafenib to docetaxel-based regimens has also been investigated (Table 4).
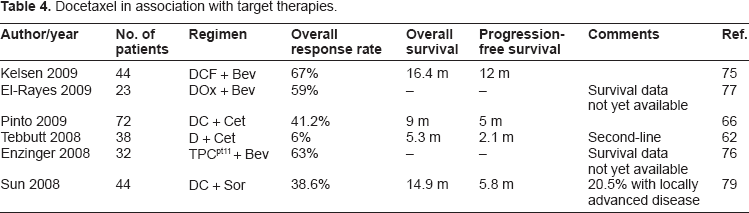
Docetaxel in association with target therapies.
Cetuximab
Cetuximab is a monoclonal chimeric human-murine antibody which binds competitively and with high affinity to the epidermal growth factor receptor (EGFR), thus inhibiting downstream pathways. 61 It is generally administered with an initial loading dose of 400 mg/m2 followed by weekly maintenance therapy of 250 mg/m2. As cetuximab seems to be able to reverse chemoresistance, and preclinical data have shown a synergism with taxanes, a combination of cetuximab with the traditional dosing schedule and docetaxel 30 mg/m2 was evaluated as second-line therapy in the ATTAX-2 phase II trial. 62 Efficacy was modest with an ORR of 6% among 35 evaluable patients, a median PFS of 2.1 months and a median OS of 5.3 months after a median follow-up of 19 months; these survival times are comparable with other salvage therapies for refractory advanced gastric cancer. 63 Furthermore, as reported in other studies of cetuximab,64,65 a landmark analysis correlated an increased grade of cetuximab-related acneiform rash with longer median PFS and OS.
The Italian phase II DOCETUX study evaluated the efficacy of the doublet regimen of docetaxel 75 mg/m2 plus cisplatin 75 mg/m2 every 3 weeks combined with the traditional cetuximab regimen as first-line therapy for advanced gastric cancer. 66 Seventy-two patients were enrolled and treated with this regimen for a maximum of six courses; patients could then continue with cetuximab alone if stability or a response to the therapy was achieved. The ORR was 41.2% with one complete response; the median TTP and OS were respectively 5 months and 9 months. The main toxicity was haematological with 44.4% of patients having grade 3 or 4 neutropenia and 19.4% having febrile neutropenia; less frequent grade 3 or 4 toxicities included anaemia, thrombocytopenia, asthenia, vomiting, stomatitis, diarrhoea, hyperbilirubinaemia and serum electrolyte imbalances. Despite discouraging data for TTP and OS times in comparison with other traditional cytotoxic regimens, the high ORR achieved in this study could suggest the use of this combination in a setting where the response is a crucial end-point, such as the neoadjuvant therapy; however, no data are yet available to support this speculation.
Interestingly, in this study, a subset analysis of 21 patients revealed that cetuximab plus docetaxel and cisplatin induces a modulation in vascular endothelial growth factor (VEGF) circulating levels. Early reduction of VEGF in serum and plasma can be considered an early surrogate marker of response, as a reduction by at least 40% in basal VEGF levels observed after 4 days was statistically correlated with control of the disease. 67 An early marker of efficacy is always desirable, especially for expensive treatments such as biologic agent-including regimens, to avoid needless toxicity in non-responder patients and gain maximum efficacy in responder patients, thereby rationalizing health expenditure. In the DOCETUX study KRAS and BRAF mutational status was also evaluated and only 9.4% of patients were documented to carry a KRAS mutation, while no BRAF mutations were found. These data are consistent with those reported in literature and confirm that KRAS/BRAF mutations are more rare events in gastric cancer than in colorectal cancer where they occur in 40%–50% of tumours. Moreover, differently from colorectal cancer, in DOCETUX study no significant correlation between KRAS/BRAF mutational status and response rate was observed. 66
The same cetuximab plus docetaxel-cisplatin schedule will be further evaluated in another ongoing phase II study. 68 Preliminary results are similar to those reported by Pinto et al. 66
Bevacizumab
Bevacizumab is a recombinant humanized monoclonal antibody which targets VEGF and prevents growth and maintenance of tumour blood vessels, thus acting as an anti-angiogenic drug. 69 As angiogenesis is a fundamental mechanism for tumour growth and metastasis, bevacizumab has been evaluated in several human malignancies and has been shown to improve response and survival in patients with colorectal, lung and breast cancer.70–72 Moreover, laboratory data suggest that VEGF expression is increased in gastric cancer,73,74 providing a strong rationale for using bevacizumab as therapy for this neoplasm. However, the best chemotherapy platform for the addition of bevacizumab has yet to be identified.
A phase II study has evaluated bevacizumab 10 mg/kg on day 1 in conjunction with a modified DCF schedule consisting of docetaxel 40 mg/m2 on day 1, fluorouracil bolus 400 mg/m2 on day 1, leucovorin 400 mg/m2 on day 1, a 2-day infusion of fluorouracil 1000 mg/m2/day starting on day 1 and cisplatin 40 mg/m2 on day 3, every 14 days. 75 Recently presented results showed an ORR of 67% and, after a median follow-up of 12.4 months, a median OS of 16.4 months. The most common grade 3 or 4 haematological adverse event was neutropenia (51%), with a febrile neutropenia rate of 4%, while the most frequent grade 3 or 4 non-haematological toxicity was thromboembolism (31%). These survival data, albeit derived from a phase II study, are very encouraging when compared to those reported in phase III studies with ECF or DCF,5,22 which are generally not longer than 9–10 months.
Bevacizumab 10 mg/kg on day 1 was also added to the previous reported schedule of docetaxel 30 mg/m2, cisplatin 25 mg/m2 and irinotecan 50 mg/m2, administered on days 1 and 8 every 3 weeks, 48 in a phase II study of patients with gastro-oesophageal cancer. 76 Thirty of 32 enrolled patients were evaluable for the response, and the observed ORR was 63%. The regimen was generally well-tolerated, with grade 3 neutropenia reported in 22% of patients and diarrhoea reported in 28% of patients. Also noteworthy was the fact that grade 4 thromboembolic events were reported in 9% of patients.
Molecular structures of Paclitaxel (A) and Docetaxel (B).
Since, as discussed above, the combination of docetaxel and oxaliplatin has a good efficacy with an acceptable toxicity profile, this regimen can be a valid platform for the addition of bevacizumab. An ongoing phase II trial has enrolled 23 patients to receive bevacizumab 7.5 mg/kg, docetaxel 70 mg/m2 and oxaliplatin 75 mg/m2 administered on day 1 of a 21 day cycle. 77 Preliminary results have shown a partial response in ten patients and a stable disease in seven patients. The most commonly reported grade 3 or 4 toxicities were neutropenia, fever, acute neuropathy and hypertension. It is of concern that gastrointestinal perforation occurred in three patients.
In summary, bevacizumab seems to consistently enhance the activity of docetaxel-based regimens but its safety in advanced gastric cancer has yet to be clearly established.
Sorafenib
Sorafenib is a potent oral multikinase inhibitor that targets Raf tyrosine kinase and tyrosine kinases associated with membrane receptors such as VEGFR-2 and -3 and platelet-derived growth factor receptor-β which are involved in the pathogenesis and progression of gastric cancer. 78 Based on this rationale, sorafenib, at the usual dose of 400 mg twice daily orally, has been evaluated in association with a platform doublet of docetaxel 75 mg/m2 and cisplatin 75 mg/m2, both administered on day 1, every 3 weeks, in a phase II study which enrolled 44 patients with metastatic or locally advanced gastric cancer. 79 The ORR was 38.6% (including one complete response) with a median PFS of 5.8 months and a median OS of 14.9 months. The most common grade 3 or 4 toxicity was neutropenia, and other common grade 3 or 4 toxicities included fatigue, anorexia, hand-foot reaction, nausea, diarrhoea and dehydration. However, it should be noted that two patients died during the study, one from infection (despite having only mild neutropenia), and one of gastrointestinal haemorrhage. Although these efficacy data for sorafenib added to a docetaxel-cisplatin doublet are encouraging, careful evaluation of toxicity is necessary.
Docetaxel in Neoadjuvant/Adjuvant Treatment Settings
A standard treatment strategy for localized gastric cancer has not been clearly established. Radical surgical resection remains the fundamental approach for managing early-stage disease, but most patients who undergo curative surgery develop locoregional or distant recurrence, and ultimately die of their disease. Therefore, considerable attention has been paid to neo-adjuvant and adjuvant strategies to improve surgical outcomes in patients with stage II and III disease, and several clinical studies conducted in the US, Europe and Japan have demonstrated improved survival with multimodality strategies compared with surgery alone.80–82 Thus, the most widely used approach in the US is primary surgical resection followed by chemotherapy with fluorouracil and concurrent radiation therapy. In Europe, perioperative chemotherapy is preferred, with three courses of ECF before and after surgery. In Japan, surgery consisting of a gastrectomy with D2 lymph node dissection is generally followed by adjuvant chemotherapy with S-1.
As reported in the section Docetaxel in Combination with Platinum Compounds and Fluoropyrimidines, patients with advanced gastric cancer treated with DCF in a randomized phase II study had a greater response rate (37% vs. 25%) compared with those treated with ECF, which is the most frequently used regimen for perioperative therapy of patients with locally advanced gastric cancer in Europe. 21 These findings suggest that docetaxel-based regimens could be better than ECF in the neoadjuvant setting, where the most important target is to achieve a good response so as to increase the chances of an R0 resection.
The available data from phase II studies in patients with locally advanced gastric cancer suggest a possible role for neoadjuvant taxane-based chemotherapy in downstaging the cancer, obtaining complete surgical resection and probably improving the outcome. For example, in a phase II study, 49 patients with gastric cancer that was clinically unresectable because of local invasion or intra-abdominal metastases in para-aortic lymph nodes or the peritoneum were treated with neoadjuvant chemotherapy consisting of docetaxel 60 mg/m2 on day 1, cisplatin 60 mg/m2 on day 1 and capecitabine 1875 mg/m2 orally on days 1–14, every 21 days (DXP). 83 Seventy-four percent of patients could undergo surgery, and 63% underwent R0 resection. Among the patients who underwent R0 resection, the median OS was 33.4 months and the median PFS was 18.2 months. Similar results have been achieved in patients with T4 gastric cancer, as reported in a Chinese study that evaluated the efficacy of DCF in the neoadjuvant treatment of 24 patients with T4 stage gastric cancer. 84 Seventy percent of tumours were downstaged, 19 patients underwent R0 resection and 3 patients underwent R1 resection; distant metastasis was found in two patients in whom surgery was abandoned. After neoadjuvant chemotherapy, patients who underwent R0 resection had a 3-year survival rate of 78.9%.
Preliminary data are available from a dose-finding study of docetaxel administered at a starting dose of 15 mg/m2, with dose escalations at intervals of 5 mg/m2 at weeks 1, 2, 4 and 5, in combination with fixed doses of oxaliplatin 50 mg/m2 at weeks 1, 2, 4 and 5 and capecitabine 650 mg/m2 twice daily orally from Monday to Friday; radiation therapy (45 Gy in 25 fractions) was also administered. 85 The recommended dose of docetaxel was 25 mg/m2. The interesting finding from this study was that an R0 resection was able to be performed in 90% of 23 enrolled patients. These data attest that docetaxel-based chemotherapy could offer a reasonable chance of curative surgery in patients with initially unresectable disease. More trials are needed to accurately identify the most active docetaxel regimen in the preoperative setting in combination with other chemotherapeutic agents or radiotherapy (Table 5).
Docetaxel in perioperative setting.

In the adjuvant setting, sequential therapy with FOLFIRI (folinic acid, fluorouracil and irinotecan) for four courses followed by three cycles of docetaxel and cisplatin was compared with mitomycin C for six cycles in a randomized phase III trial of 169 patients with radically resected gastric cancer pT3-4 or with nodal positivity.
86
The sequential treatment regimen was feasible and well tolerated, with 76% of patients completing the planned therapy, while in the mitomycin C arm, a protocol amendment was needed to reduce the dose (from 10 to 8 mg/m2 after 13 patients) because of unacceptable toxicity. After a median follow-up of 29 months, the estimated 3-year disease-free survival (DFS) and OS rates were more favourable in the FOLFIRI/DC arm versus mitomycin C arm: 3-years DFS was 67.4% vs. 50.2% (
Although this study demonstrated the feasibility of this sequential treatment regimen, many doubts remain about the true efficacy of adjuvant chemotherapy in gastric cancer. Indeed, in studies that demonstrated an advantage for perioperative chemotherapy for gastric cancer, only about half of the patients were able to restart chemotherapy after the surgical resection, 80 underlining the difficulty of administering adjuvant chemotherapy in gastric cancer; it may be that the therapeutic effect of peri-operative treatment is largely due to the neoadjuvant part of chemotherapy. For this reason, use of docetaxel in the neoadjuvant setting seems to be more interesting than its use in post-operative therapy. A randomized phase II study has been conducted to evaluate the efficacy of neoadjuvant versus adjuvant cisplatin/docetaxel in patients with gastric cancer and, as expected, there was a significantly lower incidence of grade 3 or 4 toxicities in the neoadjuvant arm, but survival data are not yet available to indicate which strategy might be associated with the better final outcome. 87
Similarly, a recently published randomized phase II study assessed safety and efficacy of DCF administered before or after surgery. The study confirmed that neo-adjuvant DCF is effective (with 85% R0 resections) and safe (with no surgical mortality and 28.5% morbidity rate). As previously reported, neoadjuvant chemotherapy appeared to be better tolerated than adjuvant one, with a lower incidence of grade 3–4 adverse events (23% vs. 11%;
As mentioned above, the association between a docetaxel-based chemotherapy and targeted molecular therapies such as cetuximab or bevacizumab seems to further improve the response rates, although consistent data are not yet available in this setting.
Conclusion
In the landscape of therapeutic options for advanced gastric cancer, where a standard of cure is not yet established and the prognosis remains poor, the introduction of docetaxel must be considered an important achievement. Indeed, DCF is an effective regimen for front-line therapy, since it prolongs survival, improves quality of life and has clinical benefits. It is important to recognize, however, that the median age of patients enrolled in the V325 phase III study was 55 years, 22 lower than the age at which advanced gastric cancer is usually diagnosed and treated. Furthermore, the performance status of patients in this study was almost ‘good’, another finding that is uncommon in advanced gastric patients, who often have symptoms of their disease and are prostrated by weight loss or anaemia. Therefore, accurate patient selection is always recommended, and DCF should be considered only for young patients with good performance status; moreover, G-CSF prophylaxis should be considered because of the high incidence of haematological toxicity. Modifications to the classic DCF schedule or substitution of cisplatin with oxaliplatin, or fluorouracil with capecitabine, can make the docetaxel-based regimen more tolerable and able to be administered safely to elderly patients or patients with a reduced performance status, probably without affecting therapeutic efficacy.
Docetaxel-containing regimens other than DCF have been evaluated in phase II studies with somewhat encouraging results, but no definitive data are available about their efficacy because they have not been evaluated in phase III studies. Data from second-line studies also show that docetaxel, both as a single agent and in combination with other agents, can induce responses in patients who have not responded to other agents. Furthermore, docetaxel-based chemotherapy represents a good platform for the addition of novel molecular agents, and several clinical trials are ongoing to determine the best partnership between cytotoxic drugs and chemotherapy in the hope that gastric cancer, like colorectal cancer, could be a disease in which target therapy could improve the impact of chemotherapy. Data from phase II studies (involving the combination with bevacizumab and cetuximab) are encouraging but not conclusive. In the view of the recent results of a phase III study that demonstrated a survival benefit for patients with HER2 positive tumours receiving trastuzumab in combination with cisplatin and 5- fluorouracil/capecitabine, 89 an association between trastuzumab and a docetaxel-containing regimen should be evaluated, also considering the synergism of these agents in breast cancer. 90
Finally, incorporation of docetaxel in neoadjuvant chemotherapy is being evaluated in patients with locally advanced disease and may lead to a better outcome for patients in this setting.
In summary, docetaxel containing regimens may be considered as a valid option both as front-line and salvage treatment for advanced gastric cancer, or in the perioperative setting, and the addition of targeted therapy to docetaxel-based chemotherapy could increase treatment efficacy.
List of Abbreviations for Tables
T, D, Doc or Ta, Docetaxel; C or P, Cisplatin; E or Epi, Epirubicin; F, 5-Fluorouracil; L or Fol, Leucovorin; O or Ox, Oxaliplatin; X, Capecitabine; I, Cpt11 or IRI, Irinotecan; Bev, Bevacizumab; Sor, Sorafenib; Cet, cetuxumab; MMC, mitomycin C; S-1, S-1; w, weekly.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
