Abstract
Small cell lung cancer (SCLC) represents approximately 15% of all lung cancers, and is the most aggressive form of lung cancer. Left untreated, the time from diagnosis to death is 2–3 months. With current treatment, expected survival is 7–20 months, depending on the stage of disease. A new drug, amrubicin, is approved in Japan for lung cancer and has demonstrated efficacy in U.S. and European phase II trials of SCLC patients with either untreated disease or relapsed refractory illness. In a phase II study of amrubicin in previously untreated patients, response rates reached 75% with a median survival time of almost 1 year. Amrubicin is a fully synthetic 9-aminoanthracycline, and an analog of doxorubicin and epirubicin. The major mechanism of action of amrubicin is inhibition of topoisomerase II. Unlike doxorubicin, however, it exhibits little or no cardiotoxicity in clinical studies and preclinical models. In preclinical rodent tumor models, it is selectively distributed to tumour tissue and is not detected in the heart when compared with doxorubicin, which is distributed equivalently to these sites. The primary metabolite of amrubicin, amrubicinol, is up to 100 times more cytotoxic in vitro than the parent compound. This review describes the mechanisms of action of amrubicin as well as clinical studies which demonstrate the potential of this drug in future SCLC treatment. The review also puts forward hypothetical considerations for the use of other drugs such as lenalidomide, an immunomodulatory drug acting on multiple signalling pathways, or histone deacetylase inhibitors, in combination with amrubicin in SCLC.
Introduction
Small cell lung cancer (SCLC) represents approximately 15% of all lung cancers, and is the most aggressive form of lung cancer. 1 Despite its relatively low incidence, SCLC is responsible for approximately 4% of all cancer deaths. 2 In the absence of treatment, the time from diagnosis to death is 2–3 months. With treatment, expected survival is 7–20 months, depending on the stage of disease.
Although the modified Veterans’ Administration Lung Study Group staging system is frequently used, classifying SCLC as extensive or limited stage disease, more recently, it has been suggested that current TNM staging would provide more useful prognostic information 3 SCLC is graded as limited disease (defined as affecting one hemithorax with regional lymph nodes, contralateral mediastinal lymph nodes and ipsilateral supraclavicular lymph nodes or extended disease which is in areas exceeding the limited disease grading. 4 ) SCLC is not a surgically resectable disease unless infrequent primary peripheral SCLCs are identified. Instead chemo-radiotherapy or chemotherapy form the mainstay of initial treatment of SCLC (reviewed in. 5 ) Cisplatin (or carboplatin) combined with etoposide has been the gold standard first line chemotherapy since the 1980s, with response rates as high as 90% in limited stage disease. However, although SCLC is extremely chemosensitive, the disease relapses rapidly, and options for treatment of recurrent disease, or for disease that is refractory to first line therapy are very limited. Prophylactic cranial irradiation (PCI) treatment for brain metastases, which are frequent in lung cancer and are responsible for shortened survival, is also a key therapeutic tool for patients with limited disease and patients with extended disease that responds to first line therapy. 6 Topotecan is approved in the US and EU for patients with sensitive disease after failure of first-line chemotherapy. Sensitive disease was defined as disease responding to chemotherapy but subsequently progressing at least 60 days after chemotherapy. 7 Other agents used in second line setting include C AV (cyclophosphamide, doxorubicin, vincristine), epirubicin, or retreatment with the first line regimen.
Despite available therapies, the 5 year survival rate is less than 20% in limited stage disease, and approximately 2% in extensive stage disease. The requirement for improved therapies is therefore critical in this disease. In recent years, a number of new drugs have come to the forefront in SCLC treatment. Both single and multi-modal drugs have been evaluated and novel treatments for the disease are being designed based on the understanding and targeting of the multiple signalling cascades involved. Among these, a promising drug is amrubicin, a fully synthetic anthracycline which inhibits topoisomerase II. This review will discuss the mechanisms of action of this drug and its clinical efficacy, while also discussing other potential drugs such as immunomodulatory IMiDs®, which have multi-modal activities, or histone deacetylase inhibitors that could be used in combination with amrubicin and earlier generation anthracyclines.
Mechanisms of Action of Amrubicin
Amrubicin is an inhibitor of DNA topoisomerase II (topo II), an enzyme in the nucleus which regulates DNA structure through double strand breakage and religation, thus controlling replication and transcription. Inhibition of the enzyme will inhibit DNA replication and halt cell growth with an arrest of the cell cycle at G2/M. The mechanism by which amrubicin inhibits DNA topo II is thought to be through stabilization of the cleavable DNA–topo II complex, resulting in religation failure and DNA strand breakage. 8 DNA damage subsequently triggers activation of caspase-3 and -7 and cleavage of the enzyme PARP, initiating apoptosis and a loss of mitochondrial membrane potential. 9 Amrubicin, like all anthracyclines, also intercalates into DNA and generates reactive oxygen free radicals via interaction with NADPH, leading directly to cell damage.
Compared to doxorubicin, amrubicin binds DNA with approximately 7-fold lower affinity and concomitantly, higher concentrations of amrubicin are necessary to produce a DNA unwinding effect. The two drugs inhibit topo II activity to a similar extent; however, amrubicin is much more potent than doxorubicin in inducing double-stranded DNA breaks and formation of DNA-protein complexes, suggesting that the two drugs potentially act via different mechanisms. 10 Furthermore, in vitro, doxorubicin produces more oxygen free radical and alcohol metabolites than amrubicin. 11 Secondary alcohol metabolites of earlier generation anthracyclines have been linked to cardiac toxicity which is a major toxicity of conventional anthracyclines and limits the amount that can be delivered safely to patients. Preclinical studies of amrubicin demonstrate a lack of cardiac toxicity in beagle dog and rabbit, and cardiotoxicity of doxorubicin in rabbits is not exacerbated with further amrubicin administration.12,13 Little or no cardiotoxicity has been observed in patients treated with amrubicin.14,15
Amrubicin is cytotoxic in vitro for a wide range of cell lines, including those of SCLC origin, and the EC50s of amrubicin are approximately 10-fold higher than doxorubicin. 16 However, amrubicinol, one of the primary metabolites of Amrubicin, is approximately 10-fold more potent than doxorubicin in inducing cytotoxicity in many of the same tumor cell lines. The antitumor activity of amrubicin in comparison to doxorubicin was also evaluated using human tumor xenograft models. Various tumor cell lines of breast, lung and gastric origin were implanted subcutaneously in nude mice and treatment initiated following establishment of tumor growth. A single administration of amrubicin resulted in a significant reduction of tumor growth rates and furthermore, the rate of inhibition observed following amrubicin treatment was comparable or higher than that observed following treatment with doxorubicin. 17 As previously mentioned, intra-tumor concentrations of amrubicinol, a primary metabolite of amrubicin, were high and correlated closely with the antitumor effect of the drug. 18
Combination treatment of tumor cell lines including SCLC with amrubicin and other agents such as 5-FU, trastuzumab, gefitinib, cisplatin and irinotecan has demonstrated additive or synergistic in vitro cytotoxicity and in vivo tumor growth inhibition.17,19,20 Additionally, amrubicin treatment has been shown to radiosensitize A549 lung adenocarcinoma cells, which initially have a low level of radiosensitivity to x-ray radiation. 21 Amrubicin can also increase thermosensitivity of A549 cells by approximately threefold. 22 Studies with earlier generation anthracyclines demonstrate additive or synergistic preclinical antitumor activity with histone deacetylase inhibitors. 23 A Phase 1 study with epirubicin and valproic acid in heavily pre-treated patients demonstrated activity. 24
Amrubicin Activity in Drug-resistant Tumor Cells
Amrubicin has also been shown to be effective against tumour cells which display resistance to doxorubicin, etoposide, 25 cisplatin and irinotecan.20,26 In these in vitro models, where resistant cell lines are derived from their parental counterparts via long-term culture in various drugs, amrubicin is either equally effective in the parent (sensitive) and drug resistant cell lines or has only a partially reduced sensitivity in the drug resistant lines. Although amrubicin and amrubicinol are substrates for the P-glycoprotein drug efflux pump, overexpression of P-glycoprotein does not appear to prevent amrubicin accumulation in resistant cell lines to the same extent as it prevents accumulation of doxorubicin.14,18
Metabolism of Amrubicin
Amrubicin is converted to the C-13 alcohol metabolite amrubicinol upon entry into the body27,28 (see Fig. 1). Metabolism of amrubicin in cells is mediated by NADPH-cytochrome P450 reductase, NADPH: quinone oxidoreductase and carbonyl reductase. 29 In contrast to doxorubicin and daunorubicin, which have alcohol metabolites that are equally or less efficacious than the parent drug, amrubicinol has 50 times the antitumor activity of the parent drug in vitro (in cell growth assays with CCRF-CEM cells or KU-2 cells) and is 10–100 times more effective than the parent drug at inducing formation of DNA-protein complexes. 30 However, amrubicinol and amrubicin bind similarly to DNA and inhibit purified topo II to the same extent. Remarkably, the administration of amrubicin is more effective than the metabolite in studies of tumour growth in murine xenograft models: Whereas administration of amrubicinol results in an even distribution of the drug in normal and tumour tissue, administration in the form of amrubicin results in a preferential conversion to amrubicinol in tumour tissues compared with normal tissues. 28 This selective metabolism in tumour tissues predicts a less toxic profile compared to doxorubicin which is distributed evenly in tumour and normal tissue.
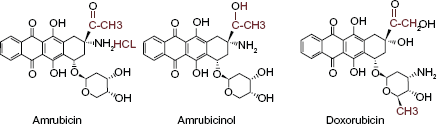
Structures of amrubicin, amrubicinol and doxorubicin.
Pharmacokinetic Profile of Amrubicin and Amrubicinol
Pharmacokinetic studies in patients with advanced lung cancer show that amrubicin and amrubicinol have areas under the plasma concentration versus time curve ranging from 8,601 to 16,706 ng h/ml and from 1361 to 4097 ng h/ml, respectively (measured from 0 to 72 hours at doses of 40–45 mg/day). The ratio of AUCs of amrubicinol to amrubicin (at 0–24 hr and 0–72 hr) have been found to be approximately 15%.31,32 After administration, the plasma amrubicin concentration decreases in a biphasic manner, with a terminal half-life of approximately 4–7 hrs. Amrubicinol shows a slow, monophasic elimination, and its half-life ranges from 17.6 to 24.5 hrs. 33 The current administration schedule for amrubicin is once daily for 3 consecutive days of a 21-day cycle, and while AUCs for amrubicin do not vary over this time period, amrubicinol has a tendency to accumulate, exhibiting 2–6 times higher AUCs on day 3 than day 1. 33
Clinical Studies of Amrubicin in Small Cell Lung Cancer
Amrubicin (CALSED™) was approved in Japan in 2002 for the treatment of non-small cell and small cell lung carcinoma. Several Phase 2 studies have now been published using amrubicin as a single agent in patients either as a first line therapy, or in patients with refractory disease who have been previously treated with platinum containing chemotherapies. The following sections describe these studies and Table 1 shows a summary of all phase II trials described in this report.
Summary of phase II studies with amrubicin in patients with untreated or previously treated extensive disease. Very similar overall response rates (75.9%–89%), and survival rates (11.7–15.4 months) are obtained in the studies on untreated patients. In the studies with previously treated patients, response rates (36%–52%) and survival (9.3–11.6 months) are at similar levels for patients with sensitive relapse. In previously treated patients with refractory relapse, three studies have comparable response rates (17%–25%) using patients with extensive disease on 40 mg/m2 amrubicin. Where survival is compared in refractory patients with extensive disease, treated with 40 mg/m2 amrubicin, rates are also similar (5.4–6 months). Results with refractory patients are more promising when greater levels of amrubicin (45 mg/m2) are used (53% response rate) and 8.8 months median survival) and when patients with limited disease are included (50% response rate, and 10.3 months overall survival).

Japanese Studies of Amrubicin in Untreated SCLC
In Japan, amrubicin has been studied as monotherapy or combined with other cytotoxic chemotherapy in the first line treatment of SCLC.
Safety and tolerability of amrubicin combined with cisplatin 34 carboplatin35,36 irinotecan 37 and topotecan 38 were evaluated in phase 1 studies. Combined with cisplatin, 34 the main dose limiting toxicity was neutropenia or febrile neutropenia at amrubicin 45 mg/m2 on days 1–3 and cisplatin 60 mg/m2 on day 1 (N = 3). However, at the recommended dose of amrubicin 40 mg/m2 iv days 1–3 and cisplatin 60 mg/m2 iv day 1 of a 21-day cycle, no dose limiting toxicity was observed (N = 4). The safety and tolerability of amrubicin and carboplatin was assessed in two phase 1 studies. In the first, 35 15 male subjects aged 49–75 received one of 2 dosing regimens. Amrubicin 35 mg/m2 iv days 1–3, the lower of the two tested dose levels (35 and 40 mg/m2), and carboplatin AUC5 day 1 of a 21-day cycle was deemed safe and well tolerated. The main toxicity was myelosuppression, and all subjects developed grade 3/4 neutropenia. In the second study 36 of 12 subjects aged 71–77 years, the recommended dosing regimen was amrubicin 35 mg/m2 iv days 1–3 and carboplatin AUC4 day 1 of a 21-day cycle. Combined with irinotecan, 37 the recommended phase 2 regimen was amrubicin 35 mg/m2 iv days 1–3, irinotecan 60 mg/m2 iv day 1 of a 21-day cycle together with G-CSF support. There were no dose-limiting toxicities at this dose level, however one subject discontinued after 2 cycles because of pneumonitis. Shibayama et al. 38 investigated amrubicin combined with topotecan in subjects with either treatment naïve or relapsed SCLC. Although detailed results in the 2 patient subsets are not available, the recommended phase 2 dose was amrubicin 35 mg/m2 iv days 1–3, and topotecan 0.75 mg/m2 days 1–5 of a 21 day cycle, and the authors commented that the combination was particularly well tolerated in patients who were treatment-naïve.
In Japan, the activity of amrubicin in first line extensive stage SCLC has been reported in 4 single-arm, open-label, phase 2 studies. In a study of single agent amrubicin 45 mg/m2 iv days 1–3 of a 21-day cycle, 39 the overall response rate was 75.8% (25/33 subjects (95% CI, 57.7%–88.9%)) with a median survival of 11.7 months. The amrubicin-cisplatin doublet regimen described above 34 was studied in 41 subjects by the same group giving an overall response rate of 87.8% (95% C.I. 73.8% to 95.9%) with a median survival of 13.6 months. Thirty six elderly (age ≥ 70 years) subjects participated in a study of amrubicin 35 mg/m2 given on days 1–3 plus carboplatin AUC4.36,40 Of these patients, 15 had limited disease and 21 had extensive disease. The overall response rate was 89% with a median overall survival of 18.6 months for patients with limited disease and 12.8% for patients with extensive disease. There is a preliminary report of a study of cisplatin-irinotecan (3 cycles) followed by amrubicin single agent (3 cycles). 41 Forty three subjects were eligible for assessment, with an overall response rate of 79% and a median survival of 15.4 months.
The Japanese first line data suggest that amrubicin appears to be well tolerated and active as a single agent or combined with other cytotoxic drugs as therapy for treatment-naïve extensive stage SCLC. The main toxicity is myelosuppression, predominantly of white blood cells. However, larger randomized studies that compare amrubicin-containing regimens with the standard platinum-etoposide doublet will be required to confirm these findings.
Japanese studies of amrubicin in previously treated SCLC
Most Japanese reports of amrubicin in previously treated SCLC describe monotherapy protocols. There is one phase 1 evaluation of amrubicin combined with topotecan described above. In addition, there are a number of small, retrospective analyses describing institutional experiences.42–44 Of the 5 reported phase 2 single agent studies, there is one randomized study. 45 In this trial, patients who had received one previous line of platinum containing chemotherapy were randomized to receive either amrubicin 40 mg/m2 iv days 1–3 or topotecan 1.0 mg/m2 iv days 1–5 of a 21 day cycle, with treatment allowed until disease progression. Fifty nine subjects 36 patients with sensitive relapse and 23 patients with refractory relapse received amrubicin (n = 29) or topotecan (n = 30). Crossover to the other treatment arm, or additional 3rd line therapy was permitted after disease progression. The overall response rate was 38% (95% CI 20%–56%) in the amrubicin arm, with 53% response for patients with sensitive relapse and 17% response for patients with refractory relapse. This compared favourably to topotecan where overall response rate was 13% (95% CI, 1% to 25%), with 21% response in sensitive relapse and 0% response in refractory relapse. Median progression free survival for amrubicin was 3.5 months, and 2.2 months for topotecan. Median overall survival was 8.1 months in the amrubicin arm and 8.4 months in the topotecan arm. Exploratory analyses suggested that crossover to amrubicin in subjects initially randomized to topotecan could have had a confounding effect on the overall survival time in the topotecan arm. Myelosuppression was the predominant toxicity in both arms; 93% and 87% (amrubicin vs. topotecan) participants with ≥ grade 3 neutropenia, 28% and 40% (amrubicin vs. topotecan) participants with ≥ grade 3 thrombocytopenia.
These data from the Japanese randomized study are supported by a single arm phase 2 study of 60 subjects (16 refractory and 44 sensitive) who had received previous chemotherapy, either one or 2 lines including platinum. 46 In that study the overall response rate was 52% (95% CI, 37% to 68%) in the sensitive group and 50% (95% CI, 25% to 75%) in the refractory patients with a median overall survival of 10.3 and 11.6 months respectively. Eighty three percent of subjects developed ≥ grade 3 neutropenia. Two smaller (n < 35) Japanese single arm studies47,48 in second or third line SCLC were also reported.
Western studies of amrubicin
Celgene Corporation (Summit, NJ) has completed 2 phase 2 studies of amrubicin in previously treated SCLC14,15 and there is an ongoing sponsored development program in Europe, US, Canada and Australia. Currently a randomized phase 3 study in previously treated SCLC is ongoing (clinicaltrials.gov NCT00547651), as well as a randomized phase 2 study in a treatment-naïve population comparing amrubicin, amrubicin-cisplatin, and etoposidecisplatin (NCT00388960) and a phase 1 study to evaluate the effect of amrubicin on the QT interval of the electrocardiogram (NCT00915083).
The phase 2 studies were conducted in the US and Europe, and near final data have been reported in abstract form.14,15 The first study 15 was a randomized phase 2 study in subjects with sensitive disease (tumor progression/recurrence ≥90 days after completing first line platinum containing chemotherapy). Subjects were randomized 2:1 to amrubicin 40 mg/m2 iv days 1–3 (n = 50) or topotecan (n = 26) 1.5 mg/m2 iv days 1–5 of a 21-day cycle. The primary endpoint was overall response rate (ORR) with the aim of excluding an ORR in the amrubicin arm of ≤25%. Treatment could continue until disease progression or unacceptable toxicity. Four subjects discontinued before receiving study drug. The study met its primary endpoint. An intent-to-treat analysis revealed an overall response rate in the amrubicin arm of 22/50 (44% 95% confidence interval 31.2, 51.7 Wilson score method). The ORR in the topotecan arm was 3/26 (11.5%). The median progression free survival was 4.6 vs. 3.3 months (amrubicin vs. topotecan) and the median overall survival was 9.3 vs. 7.7 months (amrubicin vs. topotecan). Four amrubicin patients (5%) died of neutropenic infection, and the most common ≥ grade 3 adverse events with amrubicin vs. topotecan were neutropenia (61% vs. 78%) and thrombocytopenia (39% vs. 61%).
The second study 14 was a single arm phase 2 study in patients with refractory disease (progression during therapy or within 90 days of completing first line platinum-containing chemotherapy). Seventy five patients were enrolled, of whom 69 were treated with amrubicin 40 mg/m2 iv days 1–3 of a 21-day cycle. The primary endpoint was ORR with the aim of demonstrating an ORR of ≥18%. Treatment could continue until disease progression or unacceptable toxicity. The study met its primary endpoint. An intent-to-treat analysis revealed an overall response rate of 21% (16/75, 95% Clopper-Pearson lower bound 13.9%). The median progression free survival was 3.3 months and the median overall survival was 6.1 months. The most common grade 3 or 4 adverse events were neutropenia (67%) and thrombocytopenia (41%).
Cardiac safety of amrubicin
In contrast to other anthracyclines, there has been little if any evidence for cardiotoxicity of amrubicin. In Japan, there is no cap on the maximum cumulative dose of amrubicin that may be administered. Although not fully understood, the lack of cardiotoxicity may be explained by the decreased levels of oxygen free radical production, by lower plasma levels of amrubicin/amrubicinol or decreased accumulation in cardiac tissue following amrubicin administration. 11 In preclinical cardiotoxicity studies, Amrubicin treatment did not change electrocardiogram (ECG) profiles or blood pressure, and no progress in the grade of cardiomyopathy was seen upon histopathological examination of heart tissue in dogs. In dogs previously treated with doxorubicin to induce low grade cardiomyopathy, no further increase in cardiomyopathy was seen after continued amrubicin treatment. 13
In Japanese clinical pre- and post-marketing studies, amrubicin was not associated with significant cardiotoxicity. In order to investigate the cardiac effects of amrubicin in Western patients, left ventricular ejection fraction (LVEF) measurements from the Western phase 2 studies of amrubicin in second line SCLC were pooled. 49 One hundred and twelve patients who were previously anthracyclinenaïve received amrubicin and had at least one LVEF assessment. Median LVEF did not decrease and were similar at every time point measured, including 15 patients who received a cumulative dose of > 1000 mg/m2 amrubicin. Although these data are preliminary, they demonstrate that in the second line SCLC setting, amrubicin does not appear to cause anthracycline-related cardiotoxicity, which is a limiting factor for other anthracyclines in second line.
Novel Therapies for Use in Combination with Amrubicin?
Research focused on the mechanisms of SCLC development has led to the proposition of new molecular targets for the disease. Signalling pathways involving angiogenesis, apoptosis and proliferation have critical roles in SCLC progression (reviewed in). 50 Three of the most commonly studied targets in SCLC are the VEGF, IGF and Bcl2 receptors, respectively. VEGF receptors and IGF receptors signal through the PI3K/AKT and RAF/MEK/MAPK signaling cascades to promote cancer progression. Bcl-2 is an anti-apoptotic protein which inhibits the caspase cascade through the Bak/Bax complex, and is overexpressed in the majority of SCLC cases.51,52 A number of agents have been identified that act on specific points in these pathways. The best studied anti-angiogenic drugs are sunitinib,53,54 which blocks VEGFR-1 and -2, platelet-derived growth factor receptors α and β, CSF-1R, c-KIT, FLT3 and RET, and sorafenib, 55 which inhibits VEGFR-2, Raf-kinases, and also targets platelet-derived growth factor-β and c-KIT. Other anti-angiogenic agents such as bevacizumab and aflibercept target the VEGF molecule directly. 56 Drugs targeting IGFR include the humanized antibodies AMG-479, IMC-A12 and MK-0646, 57 which have been shown to be well-tolerated in SCLC patients in recent phase I trials. IGF receptor inhibitors can sensitize cells to the effects of etoposide, and may potentially have a role in combination with amrubicin. 58 Inhibitors of Bcl-2 have been recently developed and include the non-peptidic small molecules inhibitors (SMIs) that act by blocking Bcl-2 heterodimerization with other pro-apoptotic members of the Bcl-2 protein family. 59 The Bcl-2 inhibitor Obatoclax has been used in a recent phase one trial combined with carboplatin and etoposide and results showed that the combination was tolerable and resulted in a partial response in one patient out of seven with a median time to progression of 11 weeks. 60 Also interesting, is the combination of anthracyclines and histone deacetylase inhibitors and encouraging activity in heavily pre-treated patients has been observed. 23
A drug which can act on the angiogenic, apoptotic and proliferative pathways is the immunomodulatory compound lenalidomide. 61 This drug has been FDA approved in multiple myeloma, when used together with dexamethasone, and data from two phase III trials have shown a doubling of time to progression and a 30% increase in survival times62,63 and in myelodysplastic syndromes in cases where there is a mutation in the q region of chromosome 5. In the latter disorder, there was an 83% response rate and 5q mutation was found to be undetectable in 50% of the patients treated with lenalidomide. 64 The 5q mutation is also commonly seen in many SCLC cell lines and in tumours of patients with extensive SCLC (compared with patients with limited disease), suggesting that this is a pathologically significant mutation.65,66 Lenalidomide has been evaluated in phase I patients with solid tumours including NSCLC patients where it was shown to be tolerable at the dose given for multiple myeloma (25 mg/day). 67
Lenalidomide affects tumour angiogenesis and metastasis,68,69 but is also a potent immunomodulator and has activating effects on T cells, NK cells, and gamma delta cells, combined with an inhibitory effect on T regulatory cells, resulting in inhibition of the anti-tumour immune response.61,70,71 Although there is not yet any clinical or preclinical data to support combination therapy with lenalidomide and amrubicin, there may be a hypothetical potential for the two drugs to act in a complimentary manner to inhibit tumor growth and metastasis.
Conclusions
Amrubicin is a highly effective drug for SCLC with response rates up to 78% in untreated patients with extensive disease, and up to 53% for previously treated patients with refractory relapse. The studies described here, together with the summary table (Table 1) demonstrate that trials which use the same concentration of amrubicin, in patients with similar disease status (i.e. extensive disease, vs. limited disease, and refractory vs. sensitive relapse) show similarities in both overall response rates and median survival. Amrubicin is currently in phase III clinical trials for both relapsed and untreated cases. Its current use as a single agent has proven successful with populations of all ethnicities and its future use in combination with both existing standard therapies, and with potent immunomodulatory agents such as lenalidomide, may render SCLC a more treatable and even curable cancer.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
