Abstract
The management of chronic hepatitis C (CHC) is a rapidly evolving field. New treatment paradigms are emerging due to an improved understanding of the host-viral interaction and the recent development of novel anti-viral therapies. Nevertheless, combination therapy with pegylated interferon (PEG-IFN) and ribarivin (RBV) remains the cornerstone of the treatment of CHC and will likely remain so for the foreseeable future. Over the past decade treatment regimens involving peginterferon plus ribavirin therapy have evolved from initial fixed dosing schedules to more recent individualized response-guided schedules that utilize on-treatment virological responses to determine treatment duration. The future of CHC treatment in the near to medium term is likely to involve the use of PEG-IFN and RBV in conjunction with potent and specific direct acting anti-viral agents (DAA) (protease +/- polymerase inhibitors). This review will focus on the use of PEG-IFN and RBV in the treatment of CHC as well as provide insight into how DAA agents may be used with this therapy as we enter the era of specifically targeted antiviral therapy for HCV.
Introduction
The hepatitis C virus (HCV) is a small, single stranded RNA virus belonging to the Flaviviridae family of viruses. There are 6 major HCV genotypes with genotype 1 accounting for about 60% of infections worldwide. 1 The vast majority of individuals infected with HCV develop a chronic infection that typically results in chronic hepatitis C (CHC). On a global scale the World Health Organisation (WHO) estimate that 170 million people worldwide are infected with hepatitis C while CHC represents the leading cause of end-stage liver disease and liver transplantation in developed countries.
Initially referred to as non-A, non-B hepatitis Choo and colleagues, 2 first characterized the HCV in 1989. The treatment of chronic hepatitis C subsequently evolved over the ensuing decade from interferon monotherapy to the current standard of care consisting of a combination of PEG-IFN (as either PEG-IFNα-2a, 180 μg/week subcutaneously [PEGASYS, F. Hoffmann-La Roche Ltd, Switzerland] or PEG-IFNα-2b, 1.5 μg/kg/week subcutaneously [PEGINTRON, Schering-Plough corporation, Kenilworth, NJ, USA]) in combination with RBV.3–13 This strategy is based upon the results of three pivotal studies demonstrating the efficacy of this regime over both standard IFN and RBV and PEG-INFα monotherapy.14–16 In this review we discuss in detail the current standard treatment regime as well as provide insight into likely treatment strategies in the short to medium term involving the combination of direct acting anti-viral agent(s) (DAA) with PEG-IFN plus RBV.
Aim of Antiviral Therapy for CHC
Chronic hepatitis C progresses to cirrhosis in 20%–30% of individuals over a period of 20–30 years with affected subjects at high risk for liver-related morbidity and mortality. Furthermore, the rate of hepatic fibrosis progression is often accelerated in individuals with additional risk factors for liver disease such as co-infection with hepatitis B or HIV, excess alcohol consumption, obesity, insulin resistance or advancing age. Consequently, the primary reason for commencing treatment for CHC is to prevent the development of advanced liver disease and/or related complications of hepatic decompensation, portal hypertension, variceal haemorrhage and hepatocellular carcinoma. Even in those subjects with established cirrhosis, the incidence of these clinical endpoints can be reduced by achieving viral eradication.17,18 Despite the importance of these clinical end points, in practice virological end points are used to define the success of anti-viral therapy (Table 1). In particular, the definition of treatment success is a sustained virological response (SVR) that is defined by having undetectable HCV RNA 24 weeks after the cessation of treatment. 19 This is invariably associated with an end of treatment response (ETR) that refers to having undetectable HCV RNA at the cessation of therapy. SVR is a durable end point with over 98% sustained responders remaining PCR negative at 5 year. 20 There are also several other important virological endpoints that occur early on during therapy. These include rapid virological response (RVR), defined as undetectable HCV RNA at week 4 of treatment, partial early viological response (pEVR), defined as a ≥2-log10 reduction in viral load from baseline at 12 weeks, and complete early virological response (cEVR), defined as undetectable HCV RNA at week 12. Each of these on-treatment virological responses may assist clinicians in making decisions related to treatment duration as part of response guided therapy (see below).
Virological response definitions. 19
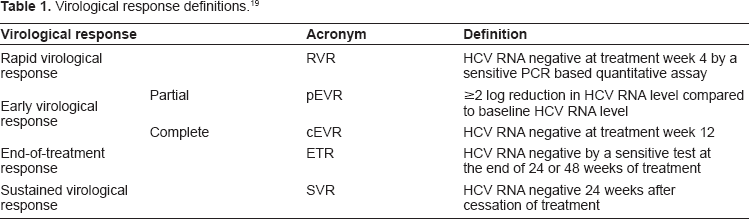
Hepatitis V Viral Kinetics on Treatment
Monitoring the viral kinetics on treatment can provide useful information on the predictability of achieving a SVR. In the clinic setting, fluctuations of hepatitis C viral load can often be categorized into one of a finite number of patterns (Fig. 1). Typically the reduction in hepatitis C viral load on PEG-IFN and RBV therapy follows a biphasic response. The first phase consists of the rapid reduction in the viral load. This phase of viral decline reflects the ability of IFN to block viral replication within infected hepatocytes. This rapid reduction in viral load typically lasts only a few days, however the magnitude of the first phase response is an important factor determining the chance of an RVR and subsequent SVR. 21 The second phase response is characterized by a slower decline in viral load and is thought to relate to the clearance of infected cells. However not all individuals exhibit this classic biphasic response to therapy. Patterns such as a triphasic response (consisting of a rapid first phase decline in viral load followed by a plateau phase then a subsequent more rapid decline) or a null response (no first and second phase decline) are also seen. A variety of both host and viral factors may be important in accounting for the variable responses observed. Viral genotype is one such factor whereby the magnitude of the first phase response is typically greater in genotypes 2/3 compared to genotype 1. The recent identification of the importance of polymorphisms in the interleukin (IL)-28B gene in predicting treatment outcome22–24 may also explain differences in the early virological response patterns between individuals. In particular, improved early viral kinetics are observed in those possessing the favorable polymorphism (CC genotype) compared to the less favorable CT or TT genotypes. 25
Potential virological responses observed of PEG-IFN and RBV treatment for CHC.
Pegylated Interferons
The development of PEG-IFN was a major advance in the way CHC is treated. It not only allowed IFN to be administered weekly rather than thrice weekly as subcutaneous injection but also improved clinical efficacy without increasing treatment side-effects.
The pegylation of IFN (either IFNα-2b or IFNα-2a) is a result of the conjugation of a polyethylene glycol (PEG) molecule to an IFN molecule. There are currently two PEG-IFN's used in widespread clinical practice: peginterferon-α2a (PEG-IFNα-2a) [PEGASYS, F. Hoffmann-La Roche Ltd, Switzerland] and peginterferon α2b (PEG-IFNα-2b) [PEGINTRON, Schering-Plough corporation, Kenilworth, NJ, USA]. While either preparation can be used in combination with RBV for the treatment of CHC, fundamental differences exist between these two preparations. PEG-IFNα-2a has a molecular weight of approximately 60 KD due to the conjugation of a 40 KD single branched PEG chain to IFNα-2a. Peak serum concentrations are reached 72 to 96 hours after dosing and serum concentrations are maintained throughout 7 days supporting weekly dosing. By comparison PEG-IFNα-2b is the construct of a linear 12 KD PEG chain linked to IFNα-2b. Thus the two PEG-interferon molecules significantly differ in their molecular mass. Also different is the nature of the chemical bond between the PEG molecule and IFN. PEG-IFNα-2b utilises a urethane linkage that is susceptible to hydrolysis. After an injection the native interferon molecule is released from the PEG and circulates in the body. The branched chain PEG moiety used in PEG-IFNα-2a is bound by a stable, amide bond that is not subject to hydrolysis and hence the entire compound circulates intact, and interacts with the receptor, not the free native IFNα. These differences mean that PEG-IFNα-2b is absorbed more rapidly (4.6 hours compared to 50 hours) and has a larger volume of distribution (0.99 L/kg compared to 8 L) compared to PEG-IFNα-2a. 26 The clinical implication of these differences is that PEG-IFNα-2a can be given as a fixed non-weight dependent dose of 180 μg/wk where as weight-based dosing is required for PEG-IFNα-2b.
The antiviral activity of IFN is complex. The interferons are named after their ability to “interfere” with viral replication within a host cell. IFNα is a type 1 interferon that exerts its anti-viral activity through binding to a specific cell surface receptor complex known as the IFN-α receptor (IFNAR). These cell surface receptors are able to induce transcriptional activation of a large number of interferonstimulated genes (ISGs). These genes encode a variety of proteins that alter cell metabolism and interfere with virus replication, protein synthesis, and assembly. The intracellular signalling used by the IFNAR is a system of Janus-activated and tyrosine kinases that phosphorylate the cytoplasmic signal transducers and activators of transcription (STAT) proteins. 27
Ribavirin
Ribavirin is a synthetic nucleoside (guanosine) analogue that has modest anti-viral activity against HCV, although our understanding of the mechanism of this is limited. RBV is ineffective against CHC when used as monotherapy, however when used in conjugation with PEG-IFN, there is a marked improvement in SVR. It is likely that the antiviral activity of RBV occurs through several mechanisms including an effect on the host immune system as well as impairing viral RNA synthesis by competitive inhibition of inosine monophosphate dehydrogenase (IMPDH), which results in depletion of the GTP required for this RNA synthesis to occur.28,29
Standard of Care Treatment of Chronic Hepatitis C
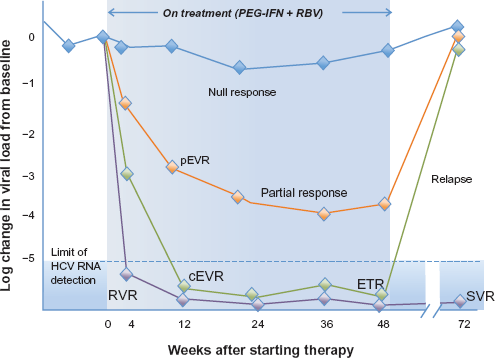
The standard of care for the treatment of CHC consists of the combination of PEG-IFN (either PEG-IFNα-2a 180 μg/wk or PEG-IFNα-2b 1.5 μg/kg/wk by subcutaneous injection) plus oral RBV 800 mg–1400 mg daily which is dosed according to body weight and genotype 19 (Table 2). Fried et al 14 convincingly demonstrated the superiority of the combination of PEG-IFN and RBV over standard IFN plus RBV in both genotype 1 and genotype 2/3 infection. In this study combination PEG-IFN therapy improved SVR from 45% to 56% across all genotypes (37% to 46% for genotype 1; 61% to 76% for genotype 2/3; 36% to 77% for genotype 4). Hadzyannis et al 16 subsequently showed that for genotype 2/3 infection PEG-IFNα-2a 180 μg/wk plus RBV 800 mg/d for 24 weeks resulted in a similar SVR to that achieved with a higher RBV dose (1000–1200 mg) and longer 48 wks duration, thereby establishing the 800 mg RBV dose for 24 weeks as the standard of care for genotype 2/3. This study also confirmed the importance of treating patients with genotype 1 infection with a higher dose of 1000–1200 mg/d of RBV for 48 weeks to maximize SVR (Fig. 2).
Treatment of CHC with fixed dosing schedule. 19

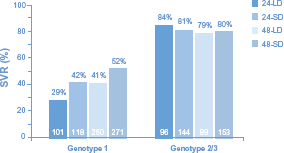
SVR rates for genotype 1 and 2/3 according to genotype and RBV dose. 16
Early virological endpoints can assist in the prediction of success to standard therapy, and/or alternatively identify poorly responding individuals in whom ongoing treatment is futile. These so called stopping rules include the failure to achieve an EVR at week 12 as these individuals have less than a 5% chance of an SVR.14,30 Also genotype 1 individuals who fail to achieve PCR negativity after 24 weeks of treatment should cease therapy due to futility.
Individualized Dosing for CHC
Since the initial phase III registration trials of PEG-IFN plus RBV, the focus of PEG-IFN therapy has shifted from the concept of a fixed treatment schedule in all patients to a tailored ‘response-guided’ regimen that uses an individual's early on-treatment responses to determine optimal treatment duration and/or maximize SVR. In particular, recent data has explored the possibility of shortening the treatment duration of both HCV-1 and HCV-2/3 individuals with a rapid treatment response. Several post hoc analyses and prospective studies of response-guided therapy suggested that it may be possible to shorten treatment duration to 24 weeks in HCV-1 patients with a low baseline viral load who achieve an RVR at week 4. 31 – 33 Based on these data the European Union recently approved a 24-week course of treatment of genotype 1 infected patients in such patients.
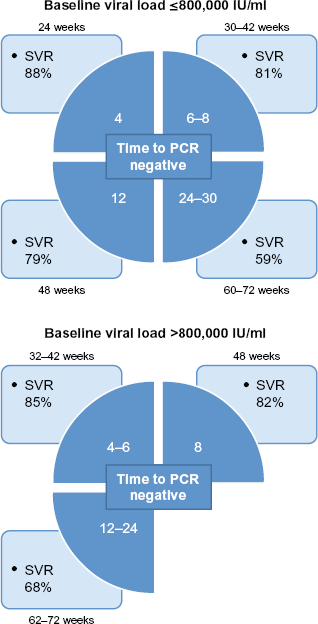
SVR results reported in the INDIV-2 study using individualized treatment duration according to time to PCR negativity. Data stratified by baseline viral load. 35
Mangia and colleagues 33 explored the response-guided concept further by tailoring treatment duration in HCV-1 subjects according to the time taken to achieve PCR negativity. Treatment was administered for 24, 48 or 72 weeks for individuals who were PCR negative at weeks 4, 8 and 12 respectively. While overall SVR rates were similar between the fixed and variable dosing groups, the SVR in RVR subjects was higher in those treated for 48 weeks compared to 24 weeks (87% vs. 77%; P = 0.12); however the results were similar in those with low baseline viraemia <400,000 IU/ml (84% vs. 83%). Moreover among those with a cEVR at week 12 SVR appeared higher in subjects receiving 72 weeks therapy compared to those in the fixed 48-week treatment group (31.8% vs. 63.5%, P = 0.07). Subsequent to this, the results of the INDIV-1 study 34 indicated that an individual treatment schedule (treatment duration from 18–48 weeks according to time of PCR negativity) was inferior to standard treatment. The likely reasons for this discrepancy include: 1) the HCV RNA assay used initially was not sensitive enough, and 2) patients with high baseline viral load (≥800,000 IU/mL) had higher relapse rates and therefore needed longer treatment durations. In comparison patients with a baseline viral load ≤ 800,000 IU/ml and RVR had SVR rates comparable to the standard 48-week treatment group. The results of the INDIV-2 study 35 that has a study design similar to the INDIV-1 study were recently presented. A total of 398 HCV-1 patients were treated with PEG-IFNα-2b and RBV for a minimum of 24 weeks and a maximum of 72 weeks according to baseline and on-treatment HCV RNA levels measured using a sensitive real-time quantitative HCV RNA assay. Patients with baseline HCV RNA ≥ 800,000 IU/mL were treated for 6–12 weeks longer than patients with baseline HCV RNA < 800,000 IU/mL. The overall SVR was 55% of subjects in the INDIV-2 study, compared to 48% in the control arm of INDIV-1. The numerically higher SVR rate in INDIV-2 likely resulted from the additional prolongation of therapy in the slow responders. Using an individualized approach to therapy, approximately one third of patients required shorter than 48 weeks of therapy and approximately one third are treated for longer than 48 weeks.
Several trials have also explored the possibility of shortening treatment duration in HCV-2/3 subjects however the results of these have been discordant. The NORDynamIC trial 36 found that twelve weeks of therapy for both genotype 2 and 3 subjects was inferior to 24 weeks (SVR rates: 59% versus 78%, P < 0.0001). Still subgroup analysis indicated that rapid virological responders could maintain an acceptable SVR although this was most evident in those <40 years. Mangia et al 37 also examined the utility of a 12 week treatment regime (PEG-IFNα-2b 1.0 μg/kg/wk + RBV 1000–1200 mg dose on body weight). Amongst the HCV-2 population who achieved an RVR there was no increase in the SVR rate amongst the 24 week group (89%) compared to the 12 week group (87%). The large ACCELERATE study 38 used standard dose RBV (800 md/day) in conjunction with PEG-IFNα-2a for either 16 or 24 weeks. Overall SVR rates were lower among those who received 16 weeks versus 24 weeks of treatment (62% vs. 70%, P < 0.001). This was predominantly related to a relapse rate of 31% in the 16-week arm versus 18% in the 24-week treatment arm (P < 0.001). However, in patients with low baseline viral load ≤400,000 IU/mL, SVR was similar between the 16-week and 24-week treatment arms (82% vs. 81%). Notably in those who achieved an RVR, SVR rates were consistently higher in the 24-week group than in the 16-week group, both overall (85% vs. 79%, P = 0.02) and within each genotype group. The conclusion from this large study was that the duration of treatment for patients with genotype 2 or 3 should continue to be 24 weeks. Other studies39,40 have however found that a 16 week treatment duration can achieve comparable SVR rates to 24 weeks. It is important to note that the success of shortened treatment regimes appears to be related to the dose of RBV used with studies using weight based RBV more successful. Nevertheless the results from the above studies support response-guided therapy with a shortening of treatment in RVR G2/3 subjects who have low baseline viral load and no adverse prognostic factors such as advanced liver fibrosis.
Predictors of an SVR
Various host, viral and disease factors predict a favorable response to therapy. Still an SVR is possible even in individuals who appear to have no good prognostic features and for that reason all patients are potential candidates for PEG-IFN + RBV therapy. Of the known pre-treatment prognostic factors, viral factors particularly HCV genotype and to a lesser extent viral load are particularly important. 19 Other factors such as female, gender age <40 years, being of non-African American ethnicity, lower body weight (≤75 kg), absence of insulin resistance, elevated ALT levels, and absence of advanced hepatic fibrosis/cirrhosis are also associated with improved treatment response. Recently host genetics related to IL-28B genotype status have also been shown to strongly predict treatment success. 25 Treatment naïve genotype 1 subjects with the CC genotype have a 2–3 fold increased likelihood of achieving an SVR compared to those with the CT or TT genotype. 22 The importance of the CC genotype in predicting SVR also holds true for genotype 2 and 3 patients who fail to achieve an RVR with standard therapy. 41 Once treatment is commenced however, the strongest predictor of an SVR is the presence of an RVR; SVR rates in RVR patients are 80%–90% across all HCV genotypes. However an RVR is only achieved in 15%–20% of patients with HCV-1 and up to 65%–70% in HCV-2/3 infection.
Special Patient Populations
Cirrhosis
Although treatment success occurs less frequently in patients with compensated cirrhosis14–16,42 there are several benefits associated with achieving an SVR in this cohort. These include a reduction in mortality,43–45 a lower risk of progression to advanced liver disease complications43,46 including hepatocellular carcinoma 18 and development of esophageal varices, 17 and an improvement in liver fibrosis and/or reversal of cirrhosis.47,48 Treatment studies focusing exclusively on subjects with advanced fibrosis have demonstrated an SVR of only 44%–52% with the combination of standard dose peginterferon alfa-2a/2b and ribavirin for 48 weeks.49–52 Results are even lower in genotype 1 patients with cirrhosis with only 10%–33% achieving an SVR.42,49–54 In contrast SVR rates in HCV genotype 2/3 subjects with cirrhosis are 57%–73% when treated with 24 weeks of combination peginterferon plus ribavirin49–53 and are not improved by extending therapy to 48 weeks. 16 Several factors contribute to the poor SVR in cirrhosis subjects including lower rapid and early viral responses and higher relapse rates.53,55 Inadequate therapeutic dosing of either peginterferon or ribavirin in cirrhosis patients is not however an explanation for the lower SVR even though dose reductions due to cytopenias are more common.50,55
Patients with decompensated cirrhosis tolerate interferon-based treatment poorly due to increased side-effects. These patients should therefore be referred to an experienced liver transplant centre where treatment could be considered preferably with initiation of a lower dose of interferon and after acceptance on to the transplant program. 19 Those with compensated liver disease can however receive standard therapy but require close monitoring for adverse events. 19 The recommended treatment duration for HCV genotype 1/4 patients is 48 weeks while genotype 2/3 patients should receive 24 weeks. During treatment growth factors may be useful to reduce the severity of anemia and leucopenia, to improve quality of life and to limit dose-reductions but have not been shown to improve SVR. The use of the thrombopoietin receptor agonist, eltrombopag olamine (GlaxoSmithKline, Collegeville, PA) before and/or during peginterferon therapy also holds promise in enhancing SVR rates in this patient population. 56
HIV co-infection
Approximately 25% of HIV infected subjects are co-infected with HCV, 57 with liver disease a leading cause of death in co-infected patients particularly in the era of highly active anti-retroviral therapy (HAART). 58 HCV-associated liver disease appears to be accelerated in co-infected patients with a significant increase in progression of hepatic fibrosis. 59 For these reasons there is a greater urgency to consider treatment of HCV in co-infected individuals even though the HCV viral clearance rate of 27%–44% is lower than that observed in HCV-monoinfection.60–63 Treatment should be directed at those whose severity of liver disease and likely response to therapy likely outweigh the morbidity of therapy, and should involve combination therapy with PEG-IFNα and RBV for 48 weeks at doses used for HCV mono-infection. This approach is based on results of the large APRICOT trial 60 and others61–63 in which SVR rates were 40% overall with 29% for genotype 1 subjects and 62% for genotype 2/3. Patients taking the anti-HIV drugs, zidovudine and didanosine, should be switched to equivalent antiretroviral agents prior to commencing therapy to reduce the risk of treatment-related toxicities including enhanced ribavirin-induced anaemia and lactic acidosis respectively.64,65
Chronic kidney disease
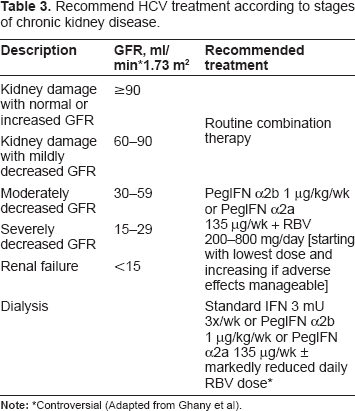
Hepatitis C viral infection is relatively common (~15%) among haemodialysis subjects in the USA 66 and represents a significant health burden in this patient group with a higher mortality rate67,68 and worse morbidity and patient and graft survivals following renal transplantation69–72 compared to non-infected patients. Consequently interferon-based treatment is generally recommended in patients with chronic renal disease and hepatitis C prior to the need for kidney transplantation, but not in those who have undergone renal transplantation unless HCV-related fibrosing cholestatic hepatitis develops.19,73 The recommended treatment regime varies according to the expression and severity of the underlying kidney disease (see Table 3 modified from Ghany MG Hepatology 2009). This is because the kidney is important for the filtration and catabolism of both (peg) interferon alfa-2a/2b74–76 and ribavirin 29 and hence the clearance or excretion of these compounds may be reduced by worsening renal function. As renal disease progresses reduced doses of both peginterferon and ribavirin are warranted to avoid increasing adverse events, and in particular ribavirin-induced haemolytic anaemia. Indeed in patients with renal failure and/or on dialysis ribavirin should be used with great caution and close subject monitoring with/without erythropoietin due to pre-existing significant anemia. In treating dialysis patients (peg)interferon monotherapy is generally preferred with current data suggesting standard interferon 3 MU tiw may deliver SVR rates of ~35% that are comparable to those achieved with peginterferon qw therapy.77–80 Thus the decision to treat hepatitis C in patients with renal disease is complex and should take into consideration several factors including: a) the severity of both the underlying renal and liver disease, b) current or future need for dialysis and/or kidney transplantation, and c) the presence significant co-morbidities such as cardiovascular disease that may affect treatment tolerability.
Recommend HCV treatment according to stages of chronic kidney disease.
Acute Hepatitis C
An estimated 75% of subjects acutely infected with HCV develop a chronic infection and are at increased risk of progressive liver disease. IFN-α based therapy of acute HCV infection yields high SVR rates81–84 and reduces the risk of evolving chronic infection compared to observation alone. 85 Therefore in the absence of contraindications all patients with acute HCV infection should be considered for treatment although the optimal timing, type and duration of therapy are unclear. Current data suggests it is reasonable to monitor for 8 to 12 weeks after the acute onset before initiating treatment to allow for spontaneous HCV resolution as this strategy still results in high (80%–95%) rates of treatment-related viral clearance.82,86 Treatment of acute HCV should be with a course of standard IFN or PEG-IFN monotherapy for a minimum of 12 weeks and up to 24 weeks in duration.81,83,84 Limited data supports a role for higher doses of PEG-IFN87,88 to improve SVR although the role of concurrent RBV use in the treatment of acute HCV remains unclear. 19
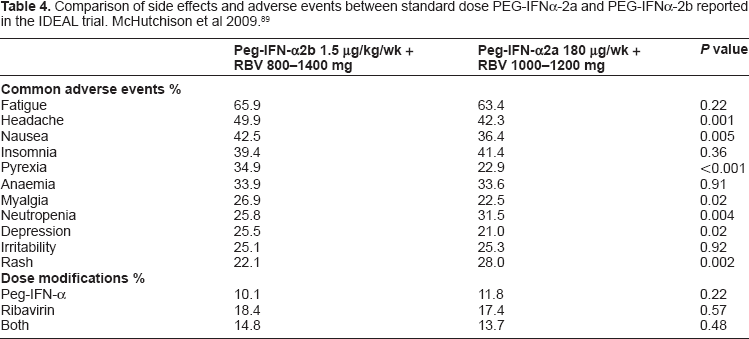
Comparison of side effects and adverse events between standard dose PEG-IFNα-2a and PEG-IFNα-2b reported in the IDEAL trial. McHutchison et al 2009. 89
Does the Type of PEG-IFN Matter?
Despite clear differences in molecular structure, both PEG-IFNα-2a and PEG-IFNα-2b are generally thought to have similar clinical efficacy. However several recent data challenge this assumption. The industry-sponsored IDEAL study was the largest head-to-head trial comparing the efficacy of PEG-IFNα-2a and PEG-IFNα-2b in genotype 1 subjects. 89 It enrolled 3070 treatment naïve participants and evaluated three different regimes (PEG-IFNα-2b at a standard dose of 1.5 μg/kg per week or a low dose of 1.0 μg/kg per week, plus ribavirin at a dose of 800 to 1400 mg per day, or PEG-IFNα-2a at a dose of 180 μg/kg per week plus ribavirin at a dose of 1000 to 1200 mg per day). Whilst ETR rates were higher in PEG-IFNα-2a recipients compared to low and standard dose PEG-IFNα-2b regimes (64.4% vs. 49.2% and 53.2%) SVR rates were similar (40.9% vs. 38.0% and 39.8%). Several prospective randomized studies published since then comparing PEG-IFNα-2a to PEG-IFNα-2b have produced mixed results.33,89–95 Two moderate sized investigator-initiated Italian studies compared standard dose PEG-IFNα-2a and PEG-IFNα-2b in treatment naïve subjects.93,94 Rumi et al found SVR rates were higher in PEG-IFNα-2a compared to the PEG-IFNα-2b treated patients (66% vs. 54%, respectively, P = 0.02), being 48% vs. 32% in the 222 HCV-1 and −4 patients (P = 0.04), and 96% vs. 82%, respectively, in 143 HCV-2 patients (P = 0.01). Ascione et al reported similar findings in a study of 320 treatment-naïve patients infected with HCV genotypes 1/4. No differences were observed in either study in the frequency of serious adverse events or treatment discontinuation rates for adverse effects. A subsequent systematic review that included data on more than 5000 patients concluded that PEG-IFNα-2a results in a significantly higher SVR compared to PEG-IFNα-2b (47% versus 41%). 96 Still both PEG-IFNα-2b and PEG-IFNα-2a are currently considered appropriate first-line treatment options in combination with RBV for chronic hepatitis C. This may change however in the future as we enter the new era of triple or possibly quadruple therapy that incorporates direct acting anti-viral agents.
Side Effects of PEG-IFN and RBV
In excess of 80% of patients will tolerate the combination of PEG-IFN and RBV therapy without the requirement for dose-reduction(s) or treatment discontinuation. Nevertheless side effects to either PEG-IFN and/or RBV are commonly encountered that may impact on a patient's quality of life (Table 3). The side effect profile of PEG-IFNα-2a and PEG-IFNα-2b are similar.89,96 The most common side effects attributed to the PEG-IFNs are flu-like symptoms involving myalgias, arthralgias, febrile symptoms and fatigue, as well as depression. Hematological effects also commonly occur and in particular significant neutropaenia (≥500 mm3 but ≤750/mm3) may develop in around 20% of subjects receiving standard-dose PEG-IFNα-2a/2b. 89 The bone-marrow suppressing effects of PEG-IFN are dose-related and typically respond to dose-reduction or temporary drug withdrawal. Overall treatment-related serious adverse events are infrequent occurring in less than 5% of subjects. A recent meta-analysis 96 concluded that the two PEG-IFN's are likely to be comparable with regard to adverse events leading to treatment discontinuation.
Ribavirin causes a dose-related decline in haemoglobin that may lead to anemia and need for dose reduction or infrequently discontinuation. In the IDEAL study 26%–28% of subjects required RBV dose reduction (haemoglobin level <10 and ≥8.5 g per deciliter) while anemia requiring drug discontinuation occurred in less than 4% of subjects. Cough, metallic taste and rash are also relatively common side–effects of RBV. In addition, RBV is strongly teratogenic and therefore contraindicated in pregnancy, with adequate contraception required in females during and for 6 months after treatment.
Future Directions
Novel interferons
Drug toxicity represents a major limitation of the current standard-of-care regimes. Novel interferons such as PEG-IFN lambda (IL-29) have therefore been developed in order to improve the tolerability of therapy by reducing the systemic and hematological side-effects commonly observed with PEG-IFNα-2a/α-2b without compromising treatment efficacy. Peg-IFN-λ1a (Zymogenetics) is a type III interferon that binds to a unique receptor with a more limited distribution than the type I interferon receptor. This receptor is not found on bone marrow CD34+ progenitor cells hence explaining the decreased incidence of neutropenia observed with this type of interferon. Peg-interferon-λ1a is currently being evaluated in clinical trials to establish its efficacy compared to the current HCV treatment regimes with the results of these studies eagerly anticipated.
Direct acting antiviral agents
The development of novel direct acting antiviral agents (DAA's) that target specific HCV enzymes/proteins linked to viral replication including HCV protease, NS5B polymerase and NS5A inhibitors holds great promise for the treatment of CHC. Several recent studies have reported enhanced SVR rates with the addition of a DAA to PEG-IFN plus RBV in HCV-1 infected subjects. Of the DAA's in clinical development, the HCV protease inhibitors telaprevir and boceprevir that target primarily HCV-1 are the most advanced with phase III studies concluded. The telaprevir phase II (PROVE) clinical development program conducted in the USA and Europe provided proof of concept that the addition of telaprevir to standard therapy increased SVR rates both in treatment naïve97,98 and relapser/non-responder genotype 1 subjects. 99 In the treatment-naïve population (PROVE 1 and 2) telaprevir 750 mg orally tid for 12 weeks in combination with PEG-IFNα-2a plus RBV for 24 or 48 weeks achieved higher 61%–69% SVR rates compared to 41%–46% in those receiving standard therapy.97,98 The large multicentre phase III ADVANCE study involving 1066 treatment naïve genotype 1 patients confirmed these findings. Treatment with telaprevir for 12 weeks in combination with PEG-IFNα-2a plus RBV for 24 or 48 weeks (depending on whether an extended RVR (eRVR) as defined as undetectable HCV RNA at week 4 through to week 20 was achieved) resulted in an SVR of 75% versus 44% (P < 0.0001) in those receiving standard of care. Importantly, almost 60% receiving telaprevir-based therapy in this study were eligible for the shorter 24 weeks duration of therapy while the SVR was 89% in those achieving an eRVR in the 12-week telaprevir treatment arm. 100 Boceprevir also enhances SVR rates in treatment naïve HCV-1 patients when combined with PEG-IFNα-2b plus RBV. In the open-label phase II SPRINT 1 study in which there was a 4 week lead-in phase of standard therapy, the addition of boceprevir to PEG-IFNα-2b plus RBV in doses up to 800 mg orally tid for up to 48 weeks duration had the potential to nearly double SVR rates compared to those receiving standard of care. 101 These results were confirmed in the randomized controlled phase III SPRINT 2 trial involving 1097 treatment-naïve HCV genotype 1 patients. In this study that also included a 4 week lead-in phase of standard therapy as well as a response-guided arm, patients received either boceprevir 800 mg orally tid in combination PEG-IFNα-2b plus RBV for 24 to 44 weeks, or 48 weeks of standard therapy. SVR rates were significantly higher and relapse rates significantly lower in both boceprevir arms compared to control with the highest SVR rates of 57% and 62% in black and non-black patients respectively achieved in those receiving triple therapy for 44 weeks. 102 In addition both telaprevir and boceprevir based triple therapy increase treatment response rates by as much as 3–4 fold in genotype 1 relapsers and non-responders to PEF-IFN plus RBV including null-responder subjects;99,103 the results of phase III studies in this patient population are anticipated soon. However, the increased efficacy of both telaprevir and boceprevir is not without cost. Rash, anemia and gastrointestinal complaints are significantly more common in those receiving telaprevir, while significant anemia and dysgeusia are commonly observed with boceprevir therapy. Because of this a number of potent “second generation” protease inhibitors have been developed including TMC435, danoprevir (RG7227), and vaniprevir (MK-7009) and these are currently in phase II/III clinical development. Common to all in this class of compounds however is the development of viral resistance that while partly mitigated by concomitant PEG-IFN based therapy may still lead to viral breakthrough and diminished efficacy in some patients.
Polymerase inhibitors
In contrast to protease inhibitors, there are fewer NS5B polymerase inhibitors in clinical development in combination PEG-IFN plus RBV. While NS5B inhibitors are generally less potent than protease inhibitors they do have the important class advantage of being active against all genotypes. Mericitabine (RG7128) is the most promising and advanced of the polymerase inhibitors under evaluation as triple therapy with PEG-IFN plus RBV. In the phase IIb PROPEL study currently underway in HCV-1 and HCV-4 treatment naïve patients, the addition mericitabine 500 mg/d or 1000 mg/d for 8–12 weeks to PEG-IFNα-2a plus RBV resulted in cEVR rates of 80%–88% of patients compared to 49% in the control arm. 104 It remains to see whether these higher early virological responses translate to higher SVR rates like they do with protease inhibitor based triple therapy.
Conclusion
The current standard-of-care treatment for chronic hepatitis C is the combination of PEG-IFN and RBV. The intent of treatment is viral eradication defined as an SVR to prevent advanced liver fibrosis and its related complications. The duration of therapy is determined not only by the viral genotype but also by early on-treatment viral response and potentially IL-28B genotype status. Treatment regimes for hepatitis C are however rapidly evolving and it appears inevitable that in the near future treatment will incorporate the addition of one or more direct acting antiviral agents to PEG-IFN and RBV. These new regimes will likely offer not only improved treatment efficacy but also the potential to further shorten treatment duration. Future developments in this area are eagerly awaited.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
