Abstract
Lapatinib is an oral dual inhibitor of epidermal growth factor receptor (HER1/ErbB1/EGFR) and human epidermal growth factor receptor 2 (HER2/ErbB2) which was approved for use in patients with metastatic breast cancer in 2007. In this review, we discuss the quality of life (QOL) results for patients on clinical trials of lapatinib. Six clinical trials, including 4 phase III and 2 phase II trials, were identified for which QOL outcomes have been reported. The trials generally showed stability of QOL during lapatinib therapy with no trial showing a detrimental effect of lapatinib on QOL. With these results, a discussion of the role of QOL assessments in patients with breast cancer is presented.
Keywords
Introduction
Lapatinib is an oral competitive and reversible dual inhibitor of epidermal growth factor receptor (HER1/ErbB1/EGFR, hereafter referred to as EGFR) and human epidermal growth factor receptor 2 (HER2/ErbB2, hereafter referred to as HER2) at low nanomolar concentration, which is approved for use in patients with metastatic breast cancer (MBC). In addition, lapatinib has been investigated in earlier stage breast cancer and other malignancies such as lung, genitourinary, gastrointestinal, head and neck, and salivary gland cancers. Its pharmacology, safety, and efficacy have been recently reviewed. 1
The pivotal trial leading to the 2007 approval of lapatinib by the US Food and Drug Administration (FDA) was presented and published in 2006 by Geyer and colleagues. 2 This study (EGF100151) was an open label, randomized, phase III trial comparing lapatinib plus capecitabine with capecitabine alone in patients with HER2 positive breast cancer previously treated with an anthracycline, a taxane, and trastuzumab. The trial was reported early after a planned interim analysis met criteria for superiority of the combination treatment arm. The results demonstrated a statistically significant improvement in the primary endpoint of time to progression (TTP) from 4.4 months in the capecitabine alone arm to 8.4 months in the lapatinib plus capecitabine arm. An updated analysis reported a non-statistically significant trend toward improved overall survival in the combination arm. 3
Quality of life (QOL) results from the Geyer study have since been reported, 4 and several other recently completed or ongoing trials of lapatinib also include QOL assessments. QOL measurement in breast cancer trials has becoming increasingly common in recent years and has been the subject of several reviews.5–7 This article summarizes and evaluates the available quality of life results from clinical trials of lapatinib in patients with breast cancer and other malignancies.
Methods
English-language articles and abstracts reporting quality of life results for patients on lapatinib were identified through: (1) MEDLINE search (1950-July 2009 including in-process and non-indexed citations) with keyword lapatinib (no articles retrieved using lapatinib AND quality of life), (2) search of American Society of Clinical Oncology (ASCO) abstracts (2006-09) for lapatinib and for lapatinib AND quality of life, (3) search of San Antonio Breast Cancer Symposium abstracts (2006-08) for lapatinib, and (4) search of active, recruiting, and completed clinical trials as of July 29, 2009 at clinicaltrials.gov for lapatinib AND quality of life and for lapatinib AND phase III. Citations clearly identified by title as review articles, case reports, pre-clinical studies, phase I trials, cost-effectiveness analyses, or pediatric studies were excluded. Remaining abstracts were reviewed to identify studies reporting QOL assessments.
Results
Search results
The search strategy identified 459 citations and 51 active, recruiting, or completed studies. After exclusion criteria were applied, 105 citations remained, including duplicate reports of individual studies. Only 3 studies retrieved via clinicaltrials.gov were identified as completed. Review of abstracts identified quality of life data from 6 studies,4,8–12 including published results from 1 phase III trial and results in abstract form from 3 phase III and 2 phase II trials (Table 1).
Studies reporting QOL results for patients on lapatinib.
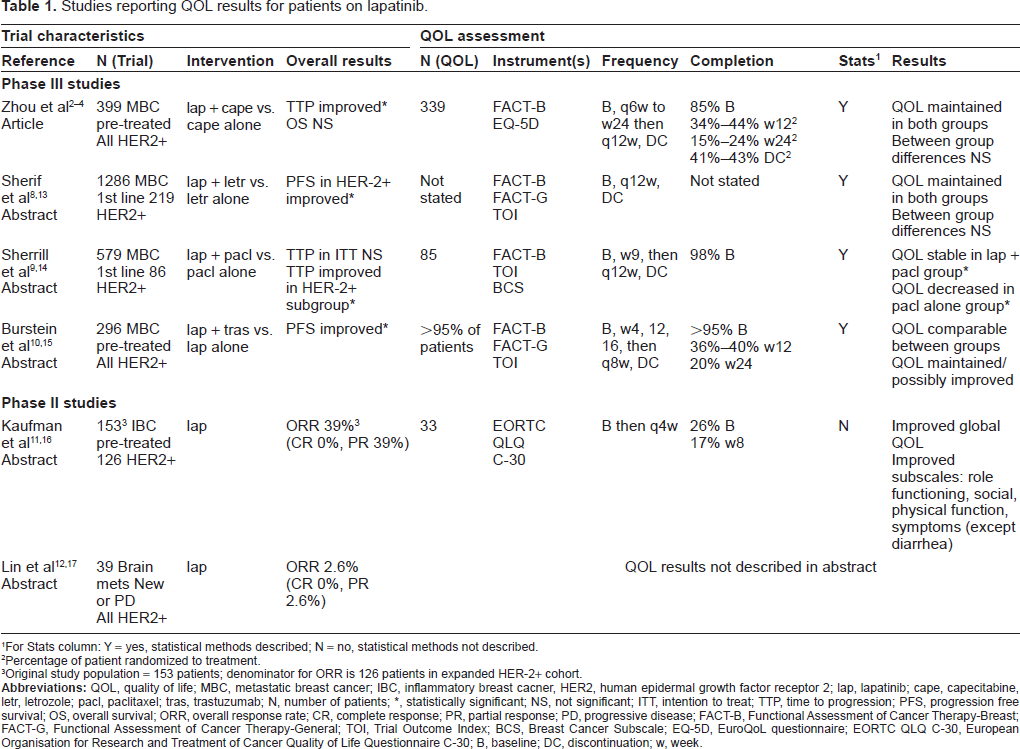
For Stats column: Y = yes, statistical methods described; N = no, statistical methods not described.
Percentage of patient randomized to treatment.
Original study population = 153 patients; denominator for ORR is 126 patients in expanded HER-2+ cohort.
Patient population
All 6 six studies were conducted in patients with breast cancer. No studies were identified which reported quality of life in patients taking lapatinib who had other malignancies.
The 4 phase III trials evaluated lapatinib in combination with other approved systemic agents used in the management of patients with MBC. Two of these trials were conducted in the first line setting8,9 and 2 trials were in pre-treated patients.4,10 Of the phase II trials, 1 was limited to patients with brain metastases 12 and 1 was conducted in patients with relapsed or refractory inflammatory breast cancer. 11
Two of the phase III reports were analyses of HER2+ subgroups in trials which included both HER2+ and HER2-/undetermined patients.8,9
QOL instruments
All four phase III trials used the FACT-B questionnaire. One phase II study used the EORTC QLQ C-30 instrument. 11 Of the trials using FACT-B, 1 also used EQ-5D, 4 3 included TOI (Trial Outcome Index) outcomes,8–10 2 included FACT-G outcomes,8,10 and 1 included BCS (Breast Cancer Subscale) outcomes. 9
Completion rate
All studies except 1 described the percentage of patients who completed at least a baseline QOL evaluation. 8 Three phase III trials obtained at least a baseline quality of life assessment in >85% of patients.4,9,10 In two of these studies, completion rates at 12 weeks were 34%-44% and at 24 weeks 15%-24%.4,10 One phase II study added QOL assessments late in the trial and, therefore, only had data on 26% of patients in the 126-patient HER2+ expanded cohort from an original study population of 153. 11
Frequency of assessments
All trials included QOL assessments at baseline and at study discontinuation. The frequency of assessments while on study varied from as often as every 4 weeks up to 12 weeks between assessments.
Description of statistical methods
All of the phase III trials described statistical methods for QOL analysis. Of the phase II trials, one abstract reported only 95% confidence intervals 11 and the other did not describe methods. 12 Statistical methods for individual studies are described below.
QOL results from individual studies
Lapatinib plus capecitabine versus capecitabine alone
Of the studies reviewed in this article, only Zhou et al have published QOL results, 4 whereas other groups have thus far reported in abstract form only. Zhou et al analyzed QOL data from the EGF100151 study, which evaluated lapatinib plus capecitabine versus capecitabine alone in pre-treated patients with HER2+ metastatic breast cancer.2,3 FACT-B and EQ-5D were used to assess QOL. The primary QOL analysis compared treatment groups based on change from baseline. Adjusted mean change from baseline was calculated using parametric analysis of covariance with baseline score as a covariate. A last observation carried forward (LOCF) method was used to account for missing data. Eighty six percent of patients in the combination arm and 84% in the capecitabine alone arm completed baseline questionnaires; by week 24, completion rate had decreased to less than 20%. Primary analysis demonstrated that QOL was maintained in both groups during 24 weeks of follow-up. Between-group differences in adjusted mean change from baseline (0.7 to 2.2 for FACT-B total and 0.3 to 1.8 for EQ-5D visual analogue scale) were in favour of the combination arm but were not statistically significant. Exploratory analyses, which evaluated the percentage of patients with minimum important differences and the relationship between tumour response and QOL outcomes, also favoured lapatinib and capecitabine in combination.
Lapatinib plus letrozole versus letrozole alone
In a 2009 abstract, Sherif et al reported QOL outcomes from the HER2+ subgroup of a trial comparing lapatinib plus letrozole to letrozole alone for hormone receptor positive MBC. 8 QOL outcomes included FACT-B, FACT-G, and TOI. Analyses included change from baseline evaluated by analysis of covariance and percentage of patients with minimally important differences compared by Fisher's exact test. There were 219 HER2+ patients (17% of study population). Information on completion of QOL assessments was not provided. Mean changes in total and subscale scores were reported as stable over time in both combination and letrozole alone treatment groups. Up to week 48, average change from baseline FACT-B score was positive in both groups with a maximum between-group difference of 2.6. No significant differences were found in percentage of QOL responders with ranges for all outcomes of 33%-38% in both groups.
Lapatinib plus paclitaxel versus paclitaxel alone
Sherrill et al evaluated the HER2+ subgroup of a trial comparing lapatinib plus paclitaxel versus paclitaxel alone for MBC. 9 QOL outcomes were FACT-B total, TOI, and BCS. Analysis of changes from baseline was performed using repeated measures models with baseline score as a covariate. Discontinuation visit assessment was applied to the next scheduled visit. Eighty six patients were HER2+ (15% of study population) and all but 1 of these patients completed a baseline assessment. In the first year, FACT-B scores were stable in the lapatinib plus paclitaxel arm but decreased in the paclitaxel alone group. Statistically significant between-group differences were shown with FACT-B, TOI, and BCS.
Lapatinib versus lapatinib plus trastuzumab
In 2008, Burstein et al reported QOL results from a trial in which patients with HER2+ MBC who had previously received trastuzumab were treated with either lapatinib or lapatinib plus trastuzumab. 10 This study differs from the others in that both arms included lapatinib. QOL outcomes were FACT-B total, FACT-G, and TOI. Changes from baseline were analyzed using analysis of covariance with baseline score as a covariate. Analyses were performed using both observed data and a LOCF method. Over 95% of patients completed an assessment at baseline, 36%-40% at week 12, and 20% at week 24. Only differences between groups in adjusted mean change from baseline were reported and ranged from 0 to 4.1 on FACT-B, 1.0 to 4.0 on FACT-G, and 0.5 to 2.7 on TOI. Only the week 12 FACT-G scores showed a significant difference, which was in favour of the combination arm.
Lapatinib monotherapy (Phase II studies)
In 2008, Kaufman et al presented QOL results from a phase II trial of pre-treated patients with relapsed/refractory inflammatory breast cancer (IBC). 11 All patients were treated with lapatinib monotherapy. QOL assessment was added late in the trial and only 33 of 126 patients in the expanded HER2+ cohort completed at least a baseline assessment. Only 21 patients completed a week 8 questionnaire. EORTC QLQ C-30 was used and mean change in scores with 95% CI were reported. At week 8, there was a moderate improvement (10-20 points) in average scores for global QOL, role functioning, and social subscales, and a minor improvement in physical function. Symptoms subscales, except diarrhea, also showed improvement.
Lin et al presented results of a phase II trial of lapatinib monotherapy in patients with brain metastases from breast cancer. 12 The abstract stated that analysis of QOL would be presented but did not actually describe the QOL results.
Discussion
Quality of life is a measure of physical, emotional, social, cognitive, and global functioning as perceived by the patient. 5 Studies of QOL in patients with breast cancer first appeared in the literature in the 1970s and have become increasingly common, particularly in the past 15 years. 7 Several reviews have been published and provide an overview of the use of QOL assessments in patients with breast cancer.5–7
Emerging theme
Since lapatinib is a relatively new addition to the arsenal of breast cancer therapies, results of QOL assessments for patients on lapatinib are only beginning to be reported. However, the emerging theme appears to be that the addition of lapatinib to standard systemic therapy for MBC results in stability in QOL outcomes. Reports by Zhou, Sherif, and Sherrill provide the strongest evidence for this maintenance of QOL. All three evaluated the addition of lapatinib to a standard treatment (capecitabine, letrozole, or paclitaxel). All employed FACT-B, which is a validated and commonly used QOL instrument in breast cancer. Sherrill et al found that QOL remained stable with lapatinib plus paclitaxel but declined with paclitaxel alone; the other studies saw QOL maintained in both treatment groups.
Phase III studies
Zhou et al reported the largest number of patients and had baseline data on 85% of the entire study population. 4 The analyses by Sherif 8 and Sherrill 9 included HER2+ subgroups of larger trials, which introduced certain limitations. For example, the HER2+ patients in the two treatment arms may not necessarily have been balanced with regard to baseline characteristics. Sherif 8 reported that baseline QOL scores were comparable between arms, while Sherrill 9 identified that more patients in the combination arm had visceral disease and more had stage IV disease.
Another limitation of the studies by Sherif and Sherrill is the lack of information on completion rates for QOL assessments. This limitation is likely due to availability of results in abstract form only and may be clarified upon full publication. Zhou et al detailed completion rates at various time points and acknowledged that attrition could be a limitation to their analysis. 4
The study reported by Burnstein et al, which compared lapatinib plus trastuzumab to lapatinib monotherapy, differed from the other studies in that all patients received lapatinib. Therefore, had any difference been found in QOL, it could be attributed to the use of trastuzumab. However, at all but one time point reported, differences in adjusted mean change from baseline were not statistically significant.
Phase II studies
The 2 phase II studies added little additional evidence. One trial did not start collecting QOL information until late in the trial and only reported QOL results at week 8 from approximately one quarter of patients in the cohort. Small numbers and short duration of follow-up limit the impact of these results. The other phase II trial did not include QOL results in the abstract.
Trastuzumab and QOL
The monoclonal antibody trastuzumab was the first anti-HER2 agent to be approved for treating patients with breast cancer in the metastatic and adjuvant settings. In a phase III trial showing improved time to disease progression and overall survival with the addition of trastuzumab to chemotherapy in the first line treatment of MBC, more patients who received the combined therapy had an improvement in global QOL.18,19 This statistically significant result occurred despite the relatively high incidence of cardiac dysfunction observed in patients receiving trastuzumab, at times concurrently with an anthracycline. Direct comparison of the effects of lapatinib and trastuzumab on QOL will be available after the results of the National Cancer Institute of Canada-led study comparing lapatinib with trastuzumab in combination with taxanes in first-line treatment for Her-2 positive or amplified metastatic breast cancer, and that led by Glaxo SmithKline comparing lapatinib and trastuzumab in combination with capecitabine in Her-2 positive or amplified metastatic breast cancer patients who have been previously treated. Currently, in the individual studies reviewed, neither has been associated with deterioration in QOL. QOL has generally been maintained with lapatanib and was improved with trastuzumab in one study.
Role and contribution of QOL assessments in breast cancer
As evidence regarding QOL in clinical trial patients on lapatinib becomes available, it is worthwhile reflecting on the role of QOL assessment using breast cancer as a model. Along the spectrum from diagnosis to treatment to survivorship or recurrent disease, breast cancer has the ability to impact patients’ QOL. 20 Knowledge gained from assessing QOL has the potential to assist in determining preferred treatments, add prognostic information, and describe the experience of the patient. 6 These roles and other potential benefits have enabled QOL research to expand and be commonly included in current clinical trials.
However, the contribution of QOL information to clinical decision making has been both lauded and questioned. In a bibliographic review of QOL assessment in breast cancer, Montazeri concluded that the QOL literature has made a considerable contribution to improving breast cancer care and cited clinical decision-making, communication, resource allocation, and research prioritization as areas in which QOL data has been helpful. 7 However, in another review, Goodwin et al more specifically concluded that the contribution of QOL results varied depending on the clinical setting. While they found QOL results did not influence decisions in the adjuvant setting and provided little information beyond medical outcomes (including toxicity) in the metastatic setting, QOL did influence management in situations where medical outcomes were equivalent. 6
QOL results may also be valuable when the improvement in survival outcome is modest or in the presence of significant short or long term toxicities. This may be especially true in the metastatic setting, as evidenced by an example from the randomized phase II study of docetaxel/cisplatin/5-FU (DCF) versus cisplatin/epirubicin/5-FU (CEF) versus docetaxel/cisplatin (DC) in patients with recurrent or metastatic gastric cancer. In this study, despite a trend towards improvement in response rate (36.6% versus 25%) and overall survival (10.4 versus 8.3 months) for the DCF arm over CEF, patients in the CEF arm had a substantial improvement in the QOL while there was only maintenance in QOL in the DCF arm. 21 In such circumstance, should QOL trump modest improvement in survival outcome or vice versa?
Methodological considerations
For any research, including QOL research, to have the potential to impact clinical care, it must be conducted in a methodologically sound manner. Methodological issues arising in the QOL literature include the formulation of a hypothesis, choice of instrument, determination of frequency of assessment, compliance, handling of missing data, and statistical analysis.5,6 Only 32% of studies reviewed by Bottomley and Therasse identified a hypothesis. Goodwin et al listed 9 different instruments among the most commonly used in studies of QOL in breast cancer. 6 Even when the same tool is used in different studies, it may be applied at different frequencies. The use of multiple instruments in the same study can be problematic, and one recommendation suggested no more than 3 to 5 scales as endpoints. 5 Despite the large number of currently available tools, a need has been expressed for further research and development of targeted instruments specific to breast cancer issues at various stages along the disease spectrum. 6 Compliance with QOL questionnaires has been cited as a limitation in several studies. Avoidance of overly burdensome assessments in terms of length and frequency and employment of strategies to streamline data collection have been suggested to help optimize completion of QOL evaluations. 5
Questions remaining
Several ongoing clinical trials of lapatinib include QOL assessments. A logical question is: will they add useful information? Although the data in the metastatic setting is young and mostly in abstract form to date, the results have been fairly consistent in showing that QOL is maintained and is comparable to the control arm when lapatinib is used in MBC. What is the likelihood that this trend will change? Although not specific to lapatinib, Goodwin et al suggested that QOL results contribute little to decision-making in the adjuvant setting. If lapatinib were to improve survival results in the adjuvant setting, would QOL outcomes alter recommendations by physicians or acceptability for patients? Perhaps a more important issue, particularly for an oral medication which requires a daily commitment on a patient's behalf to achieve compliance, is whether potential effects on QOL for the otherwise healthy adjuvant patient could impact compliance and, possibly, efficacy.
Conclusions
In summary, early results from randomized phase III trials suggest that the addition of lapatinib to standard systemic therapies for MBC allows for a period of stability in QOL which is comparable to non-lapatinib-containing treatment. Potential limitations of the studies may be clarified further upon full publication of results which are currently in abstract form. Additional QOL information for lapatinib in earlier stage breast cancer and for other malignancies will become available once ongoing studies are completed. The contribution of these results to clinical care remains to be seen.
Disclosure
Research funding from Glaxo SmithKline.
