Abstract
Anemia resulting from a relative deficiency of erythropoietin commonly complicates chronic kidney disease (CKD). With the introduction of recombinant erythropoietin two decades ago, there has been a dramatic reduction in the need for blood transfusions in CKD patients. Epoetin alpha is a first generation erythropoiesis stimulating agent (ESA) that needs to be administered frequently because of its short half-life. Methoxy ethylene glycol-epoetin beta is a longer acting ESA that acts as a continuous erythropoietin receptor activator and is given once every 2 to 4 weeks. In short term clinical studies, Methoxy polyethylene glycol-epoetin beta has been observed to be comparable with other ESAs with regard to safety and efficacy in maintaining hemoglobin levels in the target range. However, hemoglobin is no longer a clinically valid surrogate for safety i.e. correction of anemia does not translate into better clinical outcome. Long term studies evaluating hard end-points such as death and cardiovascular events are lacking. Methoxy polyethylene glycol-epoetin beta has been approved for use in the European Union since 2007 while its use in the United States is prohibited on legal grounds.
Introduction
Anemia is a common complication of chronic kidney disease (CKD) and is inversely related to erythropoietin production by the peritubular interstitial cells in the kidneys. 12 Anemia is associated with higher morbidity and mortality in CKD patients. 3 The hormone was successfully synthesized by recombinant technology and has been used clinically for more than two decades. The use of recombinant erythropoietin has dramatically reduced the need for blood transfusion. 4 Epoetin alpha and beta have short half-lives, 7 to 9 hours when administered intravenously and 24 hours when administered subcutaneously. These short half lives require frequent dosing–-up to three times in dialysis CKD patients (CKD-D) and once a week in non-dialysis CKD (CKD-ND) patients. 5 Frequent dosing has been reported to be associated with higher costs. 6 A next generation ESA, darbepoetin, has a N-linked glycoprotein site resulting in three-times longer half-life, allowing administration every week or every other week. 7 Darbepoetin has also been shown to be successful in studies of CKD anemia using once a month dosing for maintenance of hb levels. 8 Newer erythropoietin receptor stimulators such as methoxy polyethylene glycol-epoetin beta (Methoxy polyethylene glycol-epoetin beta) (Mircera®, Hoffman-La Roche Inc., Basel, Switzerland) stimulate the erythropoietin receptor for up to 130 hours. This molecule is referred to as a continuous erythropoietin receptor activator (CERA). It can be administered less frequently, every 2 to 4 weeks, and may have smoother hemoglobin (Hgb) maintenance. This review covers the pharmacology and clinical studies related to Methoxy polyethylene glycol-epoetin beta.
Pharmacology
Methoxy polyethylene glycol-epoetin beta is a covalent conjugate of epoetin beta and methoxy polyethylene glycol. The molecular weight is 60 kD, twice that of epoetin and differs from recombinant epoetin by integration of an amide bond between polyethylene glycol-butanoic acid and either the N-terminal amino group or the σ-amino group of lysine present in epoetin beta. 9
In mice, methoxy polyethylene glycol-epoetin beta produced comparable but more sustained reticulocyte response with almost half the dose compared to epoetin. 10 In vitro studies showed that methoxy polyethylene glycol-epoetin beta has a slower association with but a slightly faster dissociation from the erythropoietin receptor than epoetin beta. 11 Comparative receptor binding analysis revealed methoxy polyethylene glycol-epoetin beta had up to 100-fold lower affinity for erythropoietin receptor and 1.5 fold faster dissociation from the receptor compared to epoetin beta. 12
Thirteen Phase I clinical studies have been conducted: 11 in healthy subjects (n = 499) and 2 in CKD patients. Methoxy polyethylene glycol-epoetin beta produced dose-dependent reticulocyte response in healthy and CKD patients. In phase I trials, healthy male volunteers given single doses of intravenous or subcutaneous Methoxy polyethylene glycol-epoetin beta had dose-dependent increases in reticulocyte response that peaked within 10 days of administration and returned to baseline values by day 20. The intravenous route had higher reticulocyte response. A Phase I open-label, single dose, 3-way crossover study in 42 healthy volunteers compared the pharmacokinetic and pharmacodynamic profile of methoxy polyethylene glycol-epoetin beta 3.0 μg/Kg after subcutaneous administration into the abdomen, arm and thigh. The pharmacokinetic profile was similar at all the three injection sites with a prolonged elimination half life up to 164 hours and sustained erythropoietic response. Interestingly, a study in 12 healthy individuals, SC methoxy polyethylene glycol-epoetin beta produced less pain compared to darbepoietin alpha as documented by visual analog scale. 13 Compared to healthy subjects, severe hepatic insufficiency did not affect the pharmacodynamic and pharmacokinetic properties of methoxy polyethylene glycol-epoetin beta. 14
Four phase II dose finding studies were performed which included a total of 354 patients. The starting dose was based on the reticulocyte response determined in clinical pharmacology studies. The primary efficacy point was the analysis of the regression slopes of Hgb values over time between baseline and the end of initial treatment period. Dosage schedule did not appear to have an effect on the stability of Hgb levels. Methoxy polyethylene glycol-epoetin beta had a dose dependent reticulocyte response irrespective of frequency or route of administration. 15 MacDougall et al conducted an open-label, crossover study in 16 ESA naïve patients that demonstrated comparable half lives between a single administration of intravenous (0.4 μg/kg) or subcutaneous (0.8 μg/kg) methoxy polyethylene glycol-epoetin beta, mean 134 hours and 139 hours respectively.
Efficacy of Methoxy Polyethylene Glycol-Epoetin Beta
Clinical studies can be divided into anemia correction and Hgb maintenance.
Anemia correction studies
Correction studies conducted in CKD-D (AMICUS, intravenous methoxy polyethylene glycol-epoetin beta) 16 and CKD-ND (ARCTOS, subcutaneous methoxy polyethylene glycol-epoetin beta) 17 had initial 2-week run-in periods, followed by randomization to methoxy polyethylene glycol-epoetin beta or another ESA. The patients were followed for 18 to 24 weeks followed by an evaluation phase. In both the studies, enrolled patients had baseline Hgb levels of 8-11 g/dL. Patients were excluded if they had of evidence of infection, inflammation or elevated C-reactive protein. Target Hgb of 11-13 g/dL and ±1 g/dL of the response level defined as Hgb > 11 g/dL and an increase in >1.0 g/dL from baseline. The primary end point of Hgb response rate was significantly higher; the Hgb rose more predictably and there was less chance of Hgb over shoot in methoxy polyethylene glycol-epoetin beta than the other ESA. Furthermore, blood transfusion was comparable in both the groups in both the studies.
Maintenance studies
There have been four maintenance studies evaluating safety and efficacy of Methoxy polyethylene glycol-epoetin beta.18–21 Each study design was similar except for the MAXIMA trial (see below).
All the trials had about a 4-week run-in period during which patients were either on their usual ESA or methoxy polyethylene glycol-epoetin beta every 2 to 4 weeks and either IV or SC depending on the protocol. This was followed by about 24 week dose titration period and then 4 to 12-week evaluation period with a goal to maintain Hgb within 10-13 g/dL and within ±1 g/dL of the baseline Hgb. The inclusion criteria were similar: patients should be on dialysis for at least 3 months, have baseline Hgb 10.5-13 g/dL and have adequate iron stores. Patients with features that predict epo-resistance/unresponsiveness were excluded, e.g. infection, inflammation or uncontrolled hypertension. The primary endpoint was the mean change in Hgb levels between baseline and the evaluation period (week 29-36). The salient findings were that methoxy polyethylene glycol-epoetin beta was non-inferior to the comparator ESA epoetin alpha epoetin beta or darbepoetin. Methoxy polyethylene glycol-epoetin beta had smoother Hgb level control (Fig. 1). For instance, in non-dialysis CKD patients, methoxy polyethylene glycol-epoetin beta corrected anemia comparable to darbepoetin (97.5% versus 96.3%). However, proportion of patients overshooting the upper limit of Hgb (>13 g/dL) was lower in the Methoxy polyethylene glycol-epoetin beta than darbepoetin (12.4% versus 33%). Further, fewer patients received blood transfusion in the Methoxy polyethylene glycol-epeotin beta group. 17 Similarly, upon discontinuation due to higher Hgb, the rate of decline in Hgb was comparable to other ESAs (Fig. 2), suggesting rate of decline is dependent on erythrocyte half-life and not drug half-life. 22 Details of the studies are outlined in Table 1.
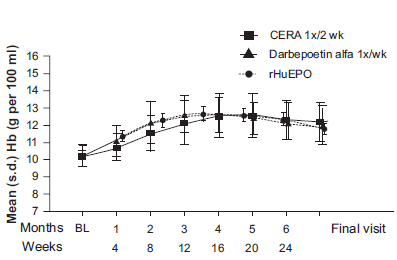
Hgb concentration during correction phase with methoxy polyethylene glycol-epoetin beta (CERA), epoetin alfa and darbapoietin alpha in EPO-naïve non-dialysis CKD patients. The observed rise in Hgb concentration is smoother compared to other ESA.
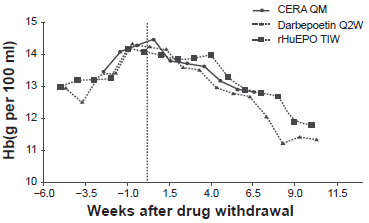
Hgb rate of decline after drug withdrawal in patients exceeding Hgb levels of 14 gm/dL with different ESAs. The rate of decline is dependent on erythrocyte half-life and not drug half-life.
Phase III clinical studies evaluating the safety and efficacy of methoxy polyethylene glycol-epoetin beta in CKD patients.

Methoxy polyethylene glycol-epoetin beta: methoxypolyethylene glycol-epeotin beta; q2wk: every 2 weeks; q4wk: every 4 weeks; tiw: three times a week.
MAXIMA, the largest of the Phase III studies for maintenance of Hgb was recently published. 21 Six hundred seventy three hemodialysis patients were randomized to methoxy polyethylene glycol-epoetin beta every 2 weeks, methoxy polyethylene glycolepoetin beta every 4 weeks or epoetin beta. Mean Hgb levels were similar in the three treatment arms at baseline. The methoxy polyethylene glycol-epoetin beta arms did not differ significantly from the epoetin arm with regard to the rates of red blood cell transfusions or adverse events. The study demonstrated that patients selected to receive methoxy polyethylene glycol-epoetin beta every 2 or 4 weeks maintained similarly stable Hgb levels to patients receiving standard epoetin therapy 1-3 times weekly. Patients’ Hgb levels were maintained at a mean of 11.8-12.0 g/dL. Although mean Hgb levels were similar in the two treatment groups, the wide standard deviation in values at each time point highlighted the heterogeneity of the hemodialysis population. 23 An accompanying editorial argued that the study had limited strength and uncertain clinical relevance as non-inferiority does not validate comparable clinical efficacy. Mean Hgb achieved included targets which are associated with increased deaths in CKD. 24 Thus, a caution is advised in interpreting the results of non-inferiority studies, such as MAXIMA.
Safety of Methoxy Polyethylene Glycol-Epoetin Beta
A pooled analysis of more than 2737 patients with anemia of CKD was evaluated. 11 Adverse event rates were comparable in patients receiving methoxy polyethylene glycol-epoetin beta and other ESAs (88.8% versus 90%): treatment related adverse event occurred in 6% of patients receiving methoxy polyethylene glycol-epoetin beta compared to 3% in other ESAs. Death rate was similar in both groups (5.7% in methoxy polyethylene glycol-epoetin beta versus 6.1% in other ESAs). Serious adverse events occurred in 37% of the study drug compared to 40% of patients receiving other ESAs. Hypertension, nasopharyngitis, diarrhea, headache and upper respiratory tract infection were most commonly reported adverse events in patients receiving methoxy polyethylene glycol-epoetin beta. 25 Majority of adverse events were mild to moderate in intensity. Severe events were <1% in each group except myocardial infarction, which was 1.5% in methoxy polyethylene glycol-epoetin beta and 1.4% in the conventional group. Incidence of vascular disorders was higher (2% versus 1%). Finally, related cardiac disorders were reported in four patients in methoxy polyethylene glycol-epoetin beta arm and none in the conventional arm. No case of pure-red cell aplasia has been reported. Since the analysis was from short term studies, long-term safety cannot be ascertained.
Non-inferiority trials in CKD patients are not sufficient especially when Hgb is not a validated surrogate for mortality in CKD. Recent randomized control trials and meta-analysis have reported increased mortality in CKD patients receiving ESA targeting higher Hgb.26,27 In a post-hoc analysis higher epoetin dose has been associated with higher mortality in CKD patient. 28 Further, since erythropoietin receptors are present on some of the malignant cells, 29 it is of theoretical concern that continuous erythropoietin receptor stimulation may have potential for untoward side-effects such as increased angiogenesis and higher rate of malignant transformation or growth. A Phase II study by Roche examining the safety and efficacy of methoxy polyethylene glycol-epoetin beta in non-small cell lung cancer patients receiving chemotherapy was suspended early. 30 Thus, we need hard end-point to assess the safety of methoxy polyethylene glycol-epoetin beta.
Methoxy Polyethylene Glycol-Epoetin Beta in Clinical Practice
Methoxy polyethylene glycol-epoetin beta may be considered in CKD patients who self-administer their ESA treatment, such as transplant recipients, CKD-ND and peritoneal dialysis patients. Patients with stable clinical condition may also benefit from a long acting ESA such as methoxy polyethylene glycol-epoetin beta. However, a potential drawback could be reduced compliance because of longer times between self-administrations. No information is available in unstable CKD patients and at extremes of age. Methoxy polyethylene glycol-epoetin beta may be used effectively is anemic patients with higher than expected fluctuation in Hgb levels, a phenomenon called Hgb cycling. 31 Since there is a high prevalence of Hgb cycling in CKD-D patients, methoxy polyethylene glycol-epoetin beta may be utilized to potentially reduce the fluctuation. Unlike epoetin, 32 route of administration in patients treated with methoxy polyethylene glycol-epoetin beta should not influence Hgb cycling.
Dosage of Methoxy Polyethylene Glycol-Epoetin Beta
Methoxy polyethylene glycol-epoetin beta was approved by the European Union for sale on July 20, 2007 and by the US FDA on November 15, 2007. 33 Methoxy polyethylene glycol-epoetin beta is approved to treat anemia in patients with chronic kidney disease is in the European Union. 11 In ESA naïve CKD patients, the recommended initial dose is 0.6 μg/kg every 2 weeks by subcutaneous or intravenous route. Hgb levels should be monitored every 2 weeks until they are stabilized and then on a periodic basis. If Hgb increase is <1 g/dL after 1 month, the recommendation is to increase the dose by 25%. Patients already receiving ESA can receive methoxy polyethylene glycol-epoetin beta once a month either subcutaneously or intravenously. The monthly starting dose of methoxy polyethylene glycol-epeotin beta depends on the weekly darbepoetin or epoetin dosage: 120 μg/month if epoetin dose is <8000 IU/week or darbepoetin is <40 μg/week; 200 μg/month if epoetin dose is 8000-16000 IU/week or darbepoetin is 40-60 μg/week; and 360 μg/month if epoetin dose is > 16000 IU/week or darbepoetin is >80 μg/week. Dosage change by 25% is recommended to keep Hgb between 11 and 12 g/dL. 11
Legal Issues with Methoxy Polyethylene Glycol-Epoetin Beta
The sale of methoxy polyethylene glycol-epoetin beta in the United States is blocked by a patent infringement. 34 To our knowledge Roche has abandoned plans to fight the legal battle with Amgen. Methoxy polyethylene glycol-epoetin beta continues to be used in clinical practice in EU countries without legal repercussions and the sales of the agent are unchanged.
Although not directly related to CKD, methoxy polyethylene glycol-epoetin beta has unfortunately found a place in sports doping: in the Olympics and in Tour de France.35,36
Future Directions
There are more than twenty on-going clinical studies comparing short term safety and efficacy of methoxy polyethylene glycol-epoetin beta.37–40 However, there is only one randomized study to our knowledge that has potential of giving critical safety and efficacy insight. This study expected to complete in September 2012 will compare methoxy polyethylene glycol-epoetin beta (ESA naïve or receiving ESA) with conventional ESA (darbepoietin or epoetin alpha or epoetin beta) estimates to enroll 2800 CKD (CKD-D and CKD-ND) from more than 140 centers worldwide except North America. The primary endpoint is time to composite of all cause mortality and non-fatal cardiovascular events. 41 Clinicians eager to switch to a longer acting ESA may want to wait until then.
Disclosures
The authors report no conflicts of interest.
