Abstract
Didanosine (ddl) has been used to treat HIV infection, in combination with other anti-retroviral drugs, for over 15 years. However, the use of the original formulation of ddI was limited by serious gastro-intestinal adverse effects, which were mainly attributable to the buffer used to protect ddI from the effect of gastric pH. Didanosine enteric-coated capsule (ddI-EC), a more recently introduced formulation, is less likely to cause gastrointestinal intolerance and its absorption might not be compromised by food intake. In this review we discuss efficacy of ddI-EC-containing anti-retroviral combinations (cART) both in naïve and previously treated patients. Because of its favorable resistance profile, ddI-EC has been shown to be potentially efficacious in rescuing patients in virological failure, even if they have nucleoside reverse transcriptase inhibitors (NRTI)-associated resistance mutations. However, ddI has been shown as a potent inducer of mitochondrial dysfunction. Peripheral neuropathy, severe hyperlactatemia and pancreatitis have all been described in patients exposed to ddI. Close monitoring of patients on ddI-EC-containing cART and low threshold for treatment modifications are required to prevent major complications. In the context of once daily cART, ddI-EC is a valid option, particularly when other agents are not available, or when other medical conditions preclude the use of drugs such as tenofovir or abacavir. The role of ddI-EC in second line cART may be even more important in resource-limited settings where additional options are lacking.
Introduction
Didanosine (ddl) is a member of the first drug family licensed for the treatment of HIV infection, the nucleoside analogue reverse transcriptase inhibitors (NRTI). It is a purine (adenine) analogue which was licensed in 1991 for the treatment of HIV infection. However, the use of ddI was mainly limited by gastrointestinal intolerance and some serious adverse effects. Didanosine is unstable in acid solutions and therefore, its original formulation was buffered to protect the active drug from gastric pH. Gastrointestinal intolerance consistently reported in ddI-treated patients was mainly attributable to the buffer present in the old presentation of ddI and has been significantly reduced when the introduction of the enteric-coated formulation of ddI (ddI-EC). As pharmacological information and pre-clinical data on ddI-EC have been extensively reviewed recently, 1 this review will focus on clinical aspects of ddI use, particularly efficacy in anti-retroviral therapy (ART)-naïve and experienced patients, safety issues, resistance profile and clinically relevant drug-drug interactions with ddI.
Studies included in this review were identified by searching PubMed, the Cochrane Clinical Trials Register and the ClinicalTrials.gov website. The terms used were ddI, didanosine, HIV and cohort or randomized trial. In addition, abstracts of relevant specialized conferences and papers cited on the selected studies were also searched. We limited our search to studies on adults.
Clinical Efficacy
Once daily (OD) combination ART (cART) regimens have been shown to be at least as effective as combinations requiring more frequent doses. This has been mainly attributed to better adherence to OD regimens than cART needing twice daily dosing. 2 Didanosine was approved for once daily administration from licensing and has been extensively investigated as a component of OD combinations both in naïve and experienced patients. However, its dietary restrictions and the imposed separation of dosing times from other concomitant medication tended to make regimens involving ddI complex and difficult for patients. 3 However, recently published studies have shown that administration of ddI-EC with food may have little impact on its anti-viral efficacy. In an open label clinical trial about 100 ART-naïve patients were started on once daily ddI-EC with lamivudine (3TC) and efavirenz (EFV) taken on either an empty stomach or with food according to patients’ preferences. In this study 33 of the 44 patients (75%) who took cART without and 36 of the 47 patients (76.5%) who had their anti-retrovirals with food had HIV-RNA < 50 copies/ml at week 48. Furthermore, virological failure was reported in 2 patients in the fasting group (4.5%) and 4 patients (8.5%) in the non-fasting group during the study period (p = 0.3). Finally, the mean plasma ddI concentration measured in 15 patients from the fasting group (0.27 g/ml; standard deviation–-SD- 0.8) was no different from the level measured in 17 patents from the non-fasting group (0.22 g/ml; SD 0.11). 4 Similarly, a small, randomized, open label clinical trial in which ART-naïve patients were started on ddI-EC monotherapy for 28 days either with empty stomach or with a fat-rich meal (about 350 kcal) failed to show any difference in the trough plasma concentration of ddI. The mean ddI level in the fasting group was 0.0234 mg/l whereas it was 0.0227 in the other group (p = 0.96) at day 28. 5
In an open-label study (GESIDA-3903) comparing EFV with either ddI-EC/3TC OD or zidovudine (AZT)/3TC twice daily, both regimens were administered with food. The study included 369 ART-naïve participants and 70% of those randomized to the ddI-EC arm and 63% of the patients on the AZT arm had HIV-1 RNA levels < 50 copies/ml at week 48 respectively, in an intention-to-treat (ITT) analysis. In this GESIDA study ddI-EC-based combination was not inferior when compared to the AZT-based regimen. In addition, this study also showed no difference between the arms regarding immunologic response or clinical disease progression. 6
Based on previous pharmacokinetic studies, the manufacturer's recommendation is to take ddI-EC on an empty stomach. 7 However, from the data summarized above it seems unlikely that taking ddI-EC with food compromise its anti-viral efficacy, although larger studies would be needed to confirm this. If ddI-EC could be prescribed without dietary restrictions it would significantly increase its acceptability to patients.
Efficacy in naïve patients
In a systematic review including 64 randomized clinical trials and 15 observational cohort analyses (accounting for over 20,000 participants) Carr and Amin have found that exposure to ddI with either 3TC or emtricitabine (FTC) was independently associated with a higher rate for treatment success. Virological success was defined as the proportion of patients with undetectable viral load by intention to treat analysis (ITT) in first line ART. 8 In an observational study of 163 ART-naïve patients who were started on ddI-EC, 3TC and EFV 62.9% had HIV-RNA < 50 copies/ml by week 48 with a mean increase in CD4 cell count of 199 cell/ml (96-288). The authors reported virological failure in 7 patients (4.1%) during the study period and only two of the 18 participants who discontinued study treatment because of adverse events had events attributable to ddI-EC (i.e. peripheral neuropathy and gastro-intestinal intolerance). 9 The same study group has recently reported data on longer follow-up showing that by the end of month 36 about 42% of the study population (70 individuals) remained on the initial cART with undetectable HIV viral load with no further cases of severe adverse drug reactions (ADR) attributable to ddI. 10
However, in naïve patients not all ddI-containing ART combinations have shown the same level of efficacy. A three-arm open-label, randomized clinical trial (N = 180) comparing ddI/abacavir (ABC)/stavudine (d4T) against AZT/3TC/saquinavir (SQV) and ritonavir (Rtv) or AZT/3TC with nelfinavir (NFV) or nevirapine (NVP), showed a significant lower virological efficacy in the group of patients receiving triple NRTI therapy compared with the other arms. Patients on the triple NRTI combination also developed more ADR, including peripheral neuropathy and symptomatic hyperlactatemia. 11 However, other triple NRTI combinations such as AZT/3TC/ABC have also been shown to be inferior to protease inhibitors (PI) or non-nucleoside analogue reverse transcriptase inhibitors (NNRTI)-containing cART. 12 In addition, the combination of ddI with tenofovir (TDF) is associated with early virological failure and paradoxical decrease in CD4 lymphocyte count.13,14 In a small pilot intensification study 36 patients were randomized to receive TDF/ddI/EFV with or without lopinavir/ritonavir (LPV/rtv) for the first 12 weeks and then followed up to week 48. There was a high rate of virological failure overall and all 6 patients who developed virological failure had EFV-related mutations. Nevertheless, 4 of these failing patients expressed L74V and 2 also had K65R mutations which have been associated with ddI resistance. 14 When co-administrated with TDF the levels of ddI increase both in plasma and intracellularly, 15 leading to an increased risk of ddI-induced toxicities. In a retrospective observational study including over 250 patients of whom 60% were started on high dose (400 mg/day) and the rest on low dose (250 mg/day) of ddI, the authors found that those patients on a high dose ddI were three times more likely to stop therapy because of ddI-induced ADRs than patients on reduced doses of ddI (Odds ratio–-OR -3.0; 95% CI 1.30-7.09). 16 It has therefore been suggested that the dose of ddI should be reduced when co-administration with TDF is unavoidable.
A Chinese open label study where 200 ART-naïve patients were randomized to AZT/ddI, d4T/3TC or AZT/3TC with NVP showed that 40% of those randomized to ddI-based combination achieved HIV-RNA < 50 copies/m whereas about 69% in the other two study groups achieved undetectability by week 52. In addition, all isolates from 17 of the 26 patients who presented virological failure after week 12 showed NNRTI-related mutations while eight of them also showed NRTI-associated mutations, including L74V and K65R. 17
Another open label trial performed in Botswana where over 600 ART-naïve HIV-1C-infected patients were randomized to AZT/ddI, AZT/3TC or d4T/3TC with either EFV or NVP showed that although there was no difference between the study arms regarding overall survival, 11% of the patients randomized to AZT/ddI-based cART showed virological failure and genotypic resistance mutations by week 96 compared to 2% in the other group (p = 0.002). 18 However, none of the patients who developed virological failure while receiving AZT/ddI-based cART developed any of the reverse transcriptase mutations classically associated with ddI resistance.
The data summarized above suggest that efficacy of ddI in first line cART depends on the other components of the regimen. Virological failure in ddI-based cART is often related to non-ddI selected resistance mutations.
Efficacy in experienced patients
In the ALIZE trial 355 patients who had HIV-RNA < 400 copies/ml for at least six months while on a PI-based cART were randomized to either continuing the regimen they were receiving or to switch to once daily ddI-EC,/FTC/EFV. By week 48 the OD combination was shown to be non-inferior than the PI-based combinations as in an ITT analysis, 90.5% and 87% of the patients randomized to OD treatment and 87.6% and 79% of those in the PI-based therapy group had HIV-RNA < 400 and 50 copies/ml respectively. 19 In addition, a sub-analysis of the ALIZE study showed that switching from AZT-based cART to ddI-based combination was associated with a modest increase on hemoglobin (Hb) levels by week 48 (+0.73 and -0.37 g/dl in the OD and the continuation arms respectively). 20 Although the clinical implications of such a change in the Hb level might be questionable, anemia has been consistently found to be associated with poor outcome in patients with HIV infection. For instance, a recently published systematic review including 10 observational studies with over 48,000 person-years of follow up has shown that even mild anemia (Hb 11-13 and 10-12 mg/dl for male ad female patients respectively) at baseline was independently associated with mortality (Hazard ratio–-HR -1.42; 95% CI 1.17-1.73) and progression to AIDS (HR 1.6; 95% CI 1.37-1.86) in patients starting first line ART. 21
Although concomitant exposure to ddI and TDF has been associated with both a poorer outcome when compared to other NRTI backbone and a higher incidence of ADRs, a non-randomized simplification study with ddI/TDF has been published. A total 169 well suppressed patients (HIV-RNA < 50 copies/ml for at least 6 months) while on PI or NNRTI-based cART were allocated to either continuing their current regimen (N = 84) or to switch to OD ddI/TDF/NVP (N = 85) based on patients’ preferences. The study showed, in an ITT analysis, that 76% of the OD group and 86% of those continuing their cART had HIV-RNA < 50 copies/ml by week 48 (p = 0.176) suggesting that ddI/TDF/NVP combination was non-inferior than twice daily cART in experienced patients. 22 In this study, slightly more than half of the patients included in the continuation cART arm were also taking ddI in combination with either 3TC (42%) or d4T (14%). Interestingly, patients started on ddI/TDF-based cART showed a significant reduction in their CD4 cell count over the study period (mean 95 cell/ml; 95% CI 45-115) whereas no significant change was noted in those patients who continued their previous cART (p = 0.021).
Because of its favorable resistance profile and its suggested activity against virus strains harboring resistance mutations to other NRTIs, particularly thymidine analogues mutations (TAM), efficacy of ddI has been investigated in the context of patients with failing cART. In a small clinical trial, the RESTART study, 21 patients with at least 8 weeks exposure to NRTIs and who had virological failure were randomized to adding ddI and d4T, with or without hydroxyurea, to their failing cART for 12 weeks. Addition of ddI/d4T was associated with a significant reduction in HIV-RNA levels with no additional effect attributable to hydroxyurea. Baseline resistance tests failed to predict response to ddI and d4T. 23 Similarly, in the Jaguar trial, 168 patients on cART with HIV-RNA ranging between 1,000 and 100,000 copies/ml were randomized to receive either ddI-EC or placebo added to their failing regimen for a period of four weeks. By the end of the study period 31 and 11% of those randomized to ddI achieved HIV-RNA < 400 and 50 copies/ml respectively whereas in the placebo group, only 6% showed HIV-RNA levels below 400 copies/ml and none achieved viral loads below 50 copies/ml (p < 0.001 and <0.01 respectively). The median reduction in HIV-RNA in the ddI group was -0.56 log10 whereas the variation in HIV-RNA levels in the placebo group went in the opposite direction (0.007 log10) (p < 0.001). interestingly, previous exposure to ddI did not affect the results. 24 Bearing in mind the limitations of the last two studies, even in those previously exposed to it, ddI appears to retain at least partial antiviral activity and may therefore be useful in rescuing experienced patients from virological failure.
The Jaguar study also reported data on baseline resistance test results in most of patients. Only 14 patients (10 randomized to the ddI arm) had L74V or K65R (2 in the ddI group) mutations whereas the frequency of other NRTI-associated mutations (NAM) such as M184V/I was almost universal (about 91%). The investigators found significant anti-viral activity associated with ddI in patients with up to 5 NAMs. The median reduction on HIV-RNA level was -0.45 log10 copies/ml in those patients with up to 5 NAMs randomized to the ddI group whereas those randomized to placebo showed a median increase on their HIV-RNA level of 0.07 log10 copies/ml (p = 0.047). 24 Although the magnitude of the association found is not clear, as the range of the change in HIV-RNA was not reported, the study shows that ddI can maintain some anti-viral effect even in very experienced patients harboring a significant number NAMs, at least in the short term. Therefore, available information suggests that ddI-EC-based cART is effective in controlling viral replication in patients with undetectable HIV-RNA in peripheral blood but also has been shown to be potentially efficacious in rescuing patients in virological failure, even if they have NRTI-associated resistance mutations.
Safety Issues
Most ADRs associated with NRTI exposure, and particularly to dideoxynucleosides (zalcitabine–-ddC-, d4T and ddI), have been attributed to mitochondrial dysfunction. Some NRTIs can induce impairment of mitochondrial function primarily by inhibiting polymerase γ which eventually leads to mitochondrial DNA (mtDNA) depletion and energy deprivation in the affected cell. 25 Several in vitro and in vivo studies have demonstrated that dideoxynucleosides tend to be the strongest inducers of polymerase γ inhibition among NRTIs.26,27 In general, ADRs attributable to mitochondrial function impairment appear to be more frequently seen in patients with advanced HIV-induced immunosuppression.28–30
In addition, it has been suggested that dideoxynucleoside-induced mitochondrial dysfunction is a consequence of a cumulative effect. That would imply that the longer the exposure the higher the incidence of such complications.31,32 However, recent evidence suggests that this may not be the case. In a recently published analysis of the Delta trial data, using a flexible parametrical survival model, it was demonstrated that the incidence rate of peripheral neuropathy rose over the first 90 days of exposure to either AZT/ddI or AZT/ddC and showed a sharp decline immediately after. 28 In addition, a large case-control study on risk factors for NRTI-induced lactic acidosis has shown that the duration of exposure to ddI in cases was significantly shorter than in controls. There was a significant reduction in the risk of developing lactic acidosis over time. 29 Similarly, a cohort study found that the risk of developing peripheral neuropathy was higher during the first year of exposure to either ddI or d4T. 33 This pattern of early onset of ddI-induced adverse events in patients on first line ART has also been reported for pancreatitis. An early singe-centre cohort study found that among patients started on ddI monotherapy the incidence of pancreatitis peaked in the first six months of exposure showing a sharp decline thereafter. 34 It therefore appears that for dideoxynucleosides, and specifically ddI, mitochondrial dysfunction is related to individual susceptibility rather than a cumulative effect in all patients. Individuals with borderline mitochondrial function might be at higher risk of developing adverse events when exposed to dideoxyucleosides.
Recent studies have shown that some mitochondrial genetic polymorphisms might lead to a higher risk of developing mitochondrial dysfunction. Among patients of white European ethnicity included in the ACTG 384 study, mtDNA haplogroup T was independently associated with peripheral neuropathy. This association was stronger among those patients exposed to ddI/d4T, when compared to those started on AZT/3TC-based combinations (OR 5.4; 95% CI 1.4-25.1). 35 Sub-haplogroup L1c was independently associated with the likelihood of developing peripheral neuropathy (OR 3.68; 95% CI 1.13-11.95) when the analysis was limited to patients with black African ethnic background. 36 These data are consistent with individuals expressing certain DNA polymorphisms, either mitochondrial or nuclear, being at higher risk of developing dideoxynucleoside-induced mitochondrial dysfunction.
Peripheral neuropathy
A prospective cohort study including about 150 patients has shown that individuals ever exposed to ddI had a significantly higher risk of developing symptomatic peripheral neuropathy (OR 3.21; 98% CI 1.56-6.60) than patients who have never been exposed to this drug. 37 However, as with other dideoxynucleoside-induced mitochondrial toxicities, peripheral neuropathy is more frequently seen when patients are exposed simultaneously to ddI and d4T. A cohort study has shown that the incidence rate of peripheral neuropathy in patients ever exposed to ddI was 6.8 per 1000 persons-year (95% CI 4.3-9.2) whereas the rate among those exposed to ddI/d4T simultaneously was 17.5 events per 1000 persons-year (95% CI 10.0-28.2). The incidence rate was even higher in those patients exposed to ddI/d4T/hydroxyurea simultaneously (28.6 per 1000 persons-year; 95% CI 17.0-45.1). After adjusting for age and CD4 cell count, patients exposed to ddI/d4T simultaneously had a significantly higher risk of developing peripheral neuropathy (HR 3.5; 95% CI 1.81-6.77) than those receiving only ddI. 38
Although the prevalence of peripheral neuropathy has decreased since the introduction of HAART, this remains the most common neurological complication seen in HIV-infected patients. A cohort analysis, comparing the incidence of peripheral neuropathy before and after the introduction of HAART combinations, showed that in the pre-HAART era exposure to ddI, high dose d4T (40 mg twice daily) and ddC for up-to a year was associated with an increased incidence of peripheral neuropathy. The risk decreased significantly with longer exposure to dideoxynucleosides, particularly in the case of ddI. 33 Interestingly, according to the data presented in this study, during the HAART era exposure to dideoxynucleosides was not independently associated with peripheral neuropathy. Furthermore, other studies have also shown that exposure to HAART, even if including dideoxynucleosides, reduces the risk of developing peripheral neuropathy. 39 Therefore, a critical factor in reducing the incidence of clinically evident peripheral neuropathy is controlling HIV replication and restoring immune function. In a large cohort study including over 1900 patients with a median persons-year follow-up of 3.1 years (IQR 1.1-6.4) it was shown that the incidence of peripheral neuropathy in patients starting ART with CD4 cell counts between 200-349 cell/mm3 (3.37 per 100 PY) and >350 cell/mm3 (2.95 per 100 PY) was significantly lower than the incidence rate calculated for those who started ART with CD4 cell counts <200 cell/mm (4.96 per 100 PY) (p = 0.016 and <0.001 respectively). Although exposure to ddI was associated with an increased risk of developing peripheral neuropathy (HR 1.34; 95% 1.04-1.73), other factors were also independently associated with this complication including exposure to d4T (HR 2.16; 95% CI 1.64-2.86), older age (HR 1.39 per 10 years increment; 95% CI 1.23-1.58) and baseline CD4 cell count (HR 1.54; 95% CI 1.22-1.96). 40 Other factors such as alcohol abuse and diabetes mellitus as well as markers of HIV disease progression have also been found to be independently associated with peripheral neuropathy in HIV-infected patients30,41 and might certainly affect the impact of dideoxynucleosides on peripheral nerve function.
Kelleher et al published a combined analysis of the data generated in four early randomised clinical trials using ddI. Low baseline CD4 cell count was the only factor independently associated with the development of peripheral neuropathy. There was no difference between patients exposed to either 500 mg or 750 mg/day of ddI or any association with cumulative ddI exposure. 42 In another retrospective cohort analysis including over 500 patients followed by a median of 19 months (IQR 12 to 31) there was no difference on worsening of peripheral neuropathy in patients exposed to dideoxynucleosides compared to those not exposed to these drugs (OR 0.94; 95% CI 0.84-1.07), suggesting that most patients receiving dideoxynucleoside-based combinations might be able to continue their treatment without any increased risk of worsening of neuropatic symptoms. 43
It has been suggested that exposure to dideoxynucleosides, and particularly ddI, might unmask sub-clinical peripheral nerve dysfunction. 44 Other medical conditions likely to cause peripheral neuropathy such as diabetes mellitus or alcohol abuse may predispose to the toxic effect of neuro-toxic drugs such as dideoxynucleosides. Therefore, patients with other known risk factors for peripheral neuropathy should not been exposed to ddI-EC, if other treatment options are available.
Pancreatitis
During 33,742 person-years follow-up in EuroSIDA cohort there were 43 episodes of pancreatitis, representing an incidence rate of 1.27 events per 1000 person-years (95% CI 0.89-1.66). When limiting the analysis to those patients exposed to ddI and d4T for less than two years the incidence rate was 1.73 events per 1000 person-years (95% CI 0.83-3.19) while it was 0.78 per 1000 person-years (95% CI 0.16-2.29) in those patients exposed to that combination for longer than two years. The incidence rate of pancreatic events in patients exposed to ddI but not to d4T for less than two years was 1.24 per 1000 person-years (95% CI 0.64-2.16) and 1.09 per 1000 person-years (95% CI 0.40-2.308) with longer exposures. In this EuroSIDA report, the multivariate model presented failed to show any association between ART and the likelihood of developing pancreatitis. Furthermore, there was some evidence suggesting a reduction in the risk of developing pancreatitis by 4% per year of exposure to ddI and d4T (RR 0.96; 95% CI 0.72-1.27). The effect of ddI exposure in the absence of concomitant d4T showed a similar pattern (RR 0.92; 95% CI 0.72-1.19). 45
However, combined analyses of the data generated in a number of ACTG studies before and after 1999 had reported incidence rates (6.1 and 8.5 per 1000 person-years respectively) higher than the incidence reported in EuroSIDA. Furthermore, the analysis of 20 ACTG studies performed before 1999 showed that the incidence rate of pancreatitis among patients exposed to ddI seems to be dose dependent and tends to be higher when ddI is co-administered with d4T. 46 A smaller study from a Serbian cohort has reported an incidence rate of acute pancreatitis of 0.03 episodes per 100 person-years in 445.6 person-years of follow-up. In this study the patients exposed to ddI and d4T simultaneously were three times more likely to develop acute pancreatitis than those patients treated with ddI but not d4T (RR 3.13; 95% CI 1.43-12.56). 47
Although the differences in the incidence of pancreatic events seen across the published studies might be at least partially explained by the different definitions used for pancreatitis, one cannot exclude the potential effect of concomitant medication and other causes of pancreatic events. A recent retrospective cohort study found that patients who developed at least one pancreatic event were more likely to have chronic viral hepatitis, billiary disorders, alcohol or recreational drug abuse and hypertriglyceridemia than patients who never developed any pancreatic events. In addition, patients who developed pancreatitis in this Italian cohort study had a longer history of HIV infection and more AIDS defining events. 48
Lactic acidosis
A large, multi-national case-control study on risk factors for severe hyperlactatemia or lactic acidosis including over 100 cases has shown that after adjusting for age, gender and current CD4 cell count, patients exposed to ddI-containing cART were five times more likely (OR 5.24; 95% CI 4.49-87.91) than those patients on AZT/3TC-based cART to develop hyperlactatemia. Patients exposed to ddI and d4T simultaneously were at even higher risk of developing severe hyperlactatemia (OR 19.87; 95% CI 4.49-87.91) when compared with those receiving AZT/3TC, 29 but as referred to above, the magnitude of the association between ddI exposure and severe hyperlactatemia decreased over time. The adjusted odds ratio for exposure to ddI for less than 12 months was 9.26 (95% CI 2.47-34.67) whereas it was 5.01 (95% CI 1.02-24.56) and 2.28 (95% CI 0.57-9.10) for exposures of 12-24 months and > 24 months respectively. 29 These data support the hypothesis of individual susceptibility to the dideoxynucleoside effect on mitochondrial function.
Previous studies have also shown an association between exposure to ddI and the likelihood of developing severe hyperlactatemia, although as with other mitochondrial toxicities, the magnitude of the association tend to be higher when used in association with d4T.29,49 Finally, female patients appear to be at higher risk than male patients and therefore the drug manufacturer has issued a warning letter to drug-prescribers highlighting that the combination of ddI and d4T must be avoided in female patients, particularly during pregnancy as cases of fatal lactic acidosis have been reported. 7
Monitoring blood lactate levels and having a low threshold for stopping exposure to dideoxynucleosides in patients developing symptoms suggestive of hyperlactatemia may avoid serious complications and fatality. Point of care lactate readers have been used to monitor capillary blood lactate levels in patients on dideoxynucleoside-based ART in some resource-limited countries. Although not fully validated yet, this monitoring strategy might be a valid alternative to prevent severe hyperlactatemia.
Peripheral lipoatrophy
Body shape changes, and particularly changes on the distribution of peripheral adipose tissue, is one of the ART-induced complications more feared by HIV-infected patients. The etiology of peripheral lipoatrophy may be multi-factorial but it has been shown that the loss of peripheral fat is, at least in part, a consequence of mitochondrial dysfunction. The content of mtDNA in adipose tissue from patients exposed to either d4T or ddI is depleted when compared to patients not on NRTIs. 50 In addition, data from some randomized clinical trials have shown that simultaneous exposure to d4T and ddI was more likely to be associated with peripheral fat loss than exposure to AZT/3TC. In a study of 150 ART-naïve patients were randomized to start treatment with either AZT/3TC or ddI/d4T with EFV, NFV or both, patients exposed to ddI/d4T had a median change in limb fat (assessed by DEXA scan) of -32.5% from base-line to week 144. There was no significant loss of limb fat among patients randomized to AZT/3TC-based cART. 51 However, there is no evidence of an association between ddI and lipoatrophy, in the absence of concurrent d4T exposure. A small study where patients were randomized to start first line ART with ddI and either d4T or 3TC, showed that patients allocated to ddI/d4T lost a median of 1.8 Kg (-26%) of limb fat from baseline to week 96, whereas patients allocated to ddI/3TC-based cART actually gained between 0.4 and 1.6 Kg during the study period, depending on the third drug included on their cART. 52 In addition, a trial comparing FTC and d4T, both in combination with ddI and efavirenz, reported a significant difference between groups in the incidence of lipoatrophy. After 60 weeks of follow up, 0.4% of the patients receiving ddI/FTC and 6% of those randomized to ddI/d4T had developed investigator-defined lipoatrophy. 53 Therefore, the available evidence suggest that, although ddI is a strong inducer of mtDNA depletion in adipose tissue, exposure to ddI in the absence of concomitant d4T is unlikely to be associated with peripheral lipoatrophy.
Cardiovascular disease
Large observational studies have recently reported an association between some ARV drugs or drug families and the likelihood of developing cardiovascular events. The DAD study reported an association between recent exposure (within the previous six months) to both ABC and ddI and an excess risk for myocardial infarction. After adjusting for classical cardiovascular risk factors, exposure to ddI increased the relative risk for myocardial infarction in about 50%. 54 Further analyses from the DAD study have confirmed the association seen between recent exposure to ddI and myocardial infarction (RR = 1.41; 95% CI 1.09-1.82) in 74,407 person/years of follow-up. The risk for cardiovascular events was not related to any change in blood lipid profile and tends to disappear soon after the exposure to ddI is terminated. 55
However, the joint analysis of the DAD and SMART studies failed to demonstrate any significant association between ddI exposure and cardiovascular disease but confirmed the previously mentioned association between ABC exposure and cardiovascular events. 54 A case-control study nested from the French Hospital database cohort recently confirmed the association between recent exposure to ABC (shorter than one year) and myocardial infraction. This study which included 286 cases of myocardial infraction and 865 controls failed to find any association between case status (i.e. myocardial infraction) and ddI exposure. 56
These data have important limitations, some of them related to the design of observational and case-control studies. Therefore, more research is needed to elucidate the potential role of ddI in cardiovascular disease, which remains uncertain. It is important to recognize that the magnitude of the effect attributable to NRTI exposure is still modest compared with the impact of other factors classically associated with cardiovascular disease. Interventions such as smoking cessation would have a much higher impact on the overall cardiovascular risk of patients. Nevertheless, given the lack certainty, switching any patient away from ddI or ABC might not be justified if the overall cardiovascular risk assessment is lower than perhaps 20%-30% of developing a cardiovascular event within the next 10 years according to the Framingham equation. It may also be reasonable to avoid starting any patient on ddI-containing cART if the overall cardiovascular risk is high if other treatment options are still available.
Drug-interactions
There are some online tools where potential interactions between anti-retrovirals and other drugs can be checked. The site www.hiv-druginteractions.org hosted by the University of Liverpool is one of these options where clinicians can check for pharmacological interactions. It is advisable to check for interactions in every patient who is about to start on ART or when adjustments are going to be performed on the combination the patient is receiving. Table 1 shows some of the most clinically relevant drug interactions with ddI.
Clinically significant drug interactions reported with ddI.
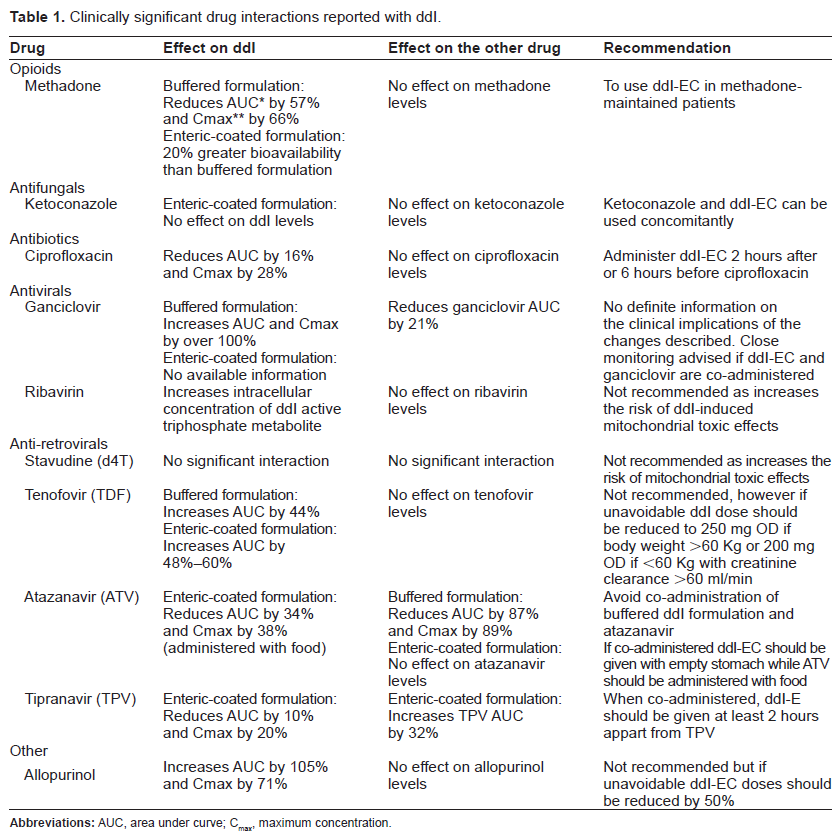
It would be important, however to make a few remarks on a number of interactions which might be clinically important in some settings. Hepatitis C and HIV co-infected patients represent one of the clinical challenges where interactions with ddI are critically important. Ribavirin, used to treat HCV infection, has been shown to increase the intracellular concentration of the tri-phosphorylated ddI leading to severe adverse drug reactions such as pancreatitis and lactic acidosis. 57 The effect of ribavirin on ddI metabolism appears to be independent of concomitant exposure to d4T which is not associated with any increased risk of clinically relevant mitochondrial toxicity in patients receiving ribavirin-based HCV therapy. 58 As recommended on product labels of both ddI and ribavirin patients on ddI-based cART should adjust their regimen before starting anti-HCV therapy.
Co-administration of ddI with allopurinol is also not recommended as this drug increases ddI AUC by more than 100%. 59 Finally, for the reasons discussed above, a reduction on the dose of ddI is recommended if co-administered with TDF. However, given the observed poor immunological response when ddI and TDF are used in combination, 13 these two drugs should not be used in combination when other options are available.
Resistance Profile
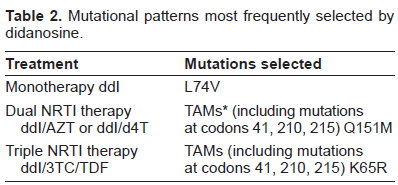
Interpretation of ddI-associated resistance mutations and prediction of clinical response to ddI can be difficult as the ddI resistance profile has not been fully defined. 60 As mentioned above, the Jaguar study has shown a significantly anti-viral activity associated with ddI in patients with up to 5 NAM mutations showing that ddI can maintain its anti-viral effect even in very experienced patients harboring a significant number NRTI-associated mutations at least in the short term. 19 Table 2 shows the resistance mutations more frequently selected by ddI and ddI-containing cART.
It has been extensively shown that the M184V mutation is associated with high level resistance to 3TC and FTC. Some in vitro studies have suggested that M184V might also reduce sensitivity to ddI,61,62 while other studies suggest that M184V might increase response to ddI. 63 In a sub-analysis of the ACTG 364 (N = 104), the effect of baseline TAM and NAM on virological failure was assessed in NRTI-experienced patients randomized to start either NFV, EFV or both at the time of switching their NRTI-backbone to either ddI/d4T or d4T/3TC, depending on their previous NRTI-backbone. At baseline the median number of TAMs was 3 (range 0-6) whereas 92 patients (88%) had M184V/I and 4 patients had L74V. The analysis of time to virological suppression (HIV-RNA < 50 copies/ml) showed a trend towards more effective viral replication control in those patients started on ddI-containing combinations (p = 0.094; HR = 1.8) when compared to those patients who continued on 3TC. In addition, the study showed that the likelihood of developing virological failure was about two fold higher in patients continuing 3TC-based cART compared to those switched to ddI. 64 In the sub-analysis of the ACTG 364 study none of the four patients started on ddI-based cART who had L74V at baseline developed virological failure during the study period.
The only two mutations in the transcriptase gene listed as associated with resistance to ddI on the International AIDS Society-USA database are L74V and K65R. 65 It has been also shown that K65R, besides reducing significantly the virus susceptibility to a number of NRTI, including ddI, also impairs viral replication capacity. 66 However, as with L74R, it is unusual to find this mutation in patients failing on ddI-based cART. Concomitant use of K65R-inducing NRTI, including ddI and TDF, has been shown as associated with a higher risk of developing this mutation. 67
Including data from over 1400 drug-experienced patients who had a resistance test performed at the time of starting a ddI-based cART Assoumou et al have recently reported a significant degree of discordance between different algorithms used to predict ddI failure. At baseline, the prevalence of L74V and K65R was 4% and 2% respectively. However, by week 24, 56% of the patients had failed to treatment. None of the algorithms accurately predicted the six month response to ddI. 60
Conclusion
Once daily cART regimens are often preferred by patients and have been shown to be non-inferior compared to twice daily regimes. In that context ddI-EC is a valid option, particularly when other agents are not available, or when other medical conditions preclude the use of drugs such as TDF or ABC. The enteric coated formulation of ddI is generally well tolerated and dietary restrictions might be less critical than previously described. The efficacy of ddI-EC-containing cART has been shown in both naïve and experienced patients. Its capacity of controlling viral replication in the presence of NRTI-selected resistance mutations suggests that it also has role in second-line cART, particularly in resource-limited settings.
Mutational patterns most frequently selected by didanosine.
Close monitoring for ddI-induced ADRs is essential to prevent any major complication, particularly those related to mitochondrial dysfunction. Further studies are needed to elucidate the role of ddI exposure in the overall risk of cardiovascular disease and peripheral lipoatrophy. Similarly, it is important to identify markers of a predisposition to major mitochondrial impairment if a genetic basis for the susceptibility to this complication is established.
Disclosure
The author reports no conflicts of interest.
