Abstract
Once used only as third-line therapy in the management of chronic pain states, methadone is now being used as first- and second-line therapy. Most risks and the stigma associated with methadone use have been known for years. Only over the past decade or so have the unique pharmacokinetic-pharmacodynamic properties and methods for conversion from other opioids to methadone been established. Pertinent English-language literature was obtained from MEDLINE/PUBMED and EMBASE searches (1966-June 2009). This paper provides an overview of the cardiotoxicity of oral methadone, with an emphasis on its use as an analgesic. Cardiotoxicity during its use in the maintenance of opioid addiction has also been reviewed due to the wealth of epidemiologic, risk factor, and correlative analytic data contained therein. A series of recommendations are provided to improve the cardiac safety profile of oral methadone used for analgesia. In addition, there is a discussion of settings and patient types which may impact upon these recommendations.
Keywords
Introduction
Methadone, an opioid structurally related to propoxyphene and fentanyl and its congeners, was a product of German research just prior to World War II. It was planned to be a replacement for morphine and its congeners which were unavailable to the Axis powers under wartime conditions. Until prolonged-release formulations of other opioids became available, methadone was widely-used as an analgesic due to its long terminal disposition half-life (t 1/2). In addition, it was the drug-of-choice in opioid maintenance programs for opioid addicts which emerged in the late 1960's-early 1970's, again due to its long t 1/2.1,2
However, its clinical application was hampered by its unpredictable pharmacodynamics in combination with its long t 1/2 and drug-drug interactions. Moreover, from the patient perspective, there was the stigma of its use, legitimate though it was, in maintenance therapy of opioid addicts. Recently, methadone has been undergoing a renaissance in the management of pain, especially as a second-line agent via opioid-to-opioid switching (also called opioid rotation) when other opioids fail or are not tolerated. Methadone can be safely used when the prescriber understands its complex pharmacokinetic-pharmacodynamic relationships and the mechanisms of its drug-drug interactions, both of these being the products of extensive research from the 1980's to present. There is a reasonable volume of clinical data for the drug in the treatment of both malignant and non-malignant pain.1,2 Table 1 illustrates the potential advantages and disadvantages of methadone as an analgesic.
Potential advantages and disadvantages of methadone as an analgesic.

One of the challenges of using methadone (and its congeners) that remains is the unique (among the opioids) cardiotoxic adverse effects. In 2001-2002, the first case reports appeared of delayed ventricular repolarization (manifested as prolongation of the QTc interval on the electrocardiogram [ECG]) leading, in some cases, to torsade des pointes (TdP) (also called polymorphous ventricular tachycardia), a potentially lethal arrhythmia. Most of the initial reports involved the methadone congener, 1-alpha-acetylmethadol (LAAM), a very long-acting agent especially suited to maintenance of opioid addiction.
LAAM was removed from the European market in 2001 due to these case reports of its association with TdP. 3 In the US, the Food and Drug Administration (FDA) required the addition of a “black box” warning to the drug labeling in 2001. The labeling was modified to mandate ECG monitoring before and periodically during LAAM therapy. This warning led to a precipitous drop in use of the drug since most opioid maintenance programs did not want the expense or bother of an ECG monitoring program. In response to this drop in utilization, the manufacturer (Roxane Laboratories, Ridgefield, CT) made a business decision to discontinue the marketing of LAAM in the US in 2003. It should be remembered that LAAM is still FDA-approved. 4
The goal of this paper is to review, in detail, the cardiotoxicity potential of methadone and its congeners, with an emphasis on oral methadone used for analgesia. Recommendations will be provided to maximize the safety of oral methadone used for analgesia. Recommendations for the use of parenteral methadone in inpatient palliative care and the use of methadone in opioid maintenance programs have recently been published.5,6 Articles were identified by Medline/Pubmed and EMBASE literature searches conducted from 1966 to June 2009 using the following words and their combinations: methadone, 1-alpha-acetylmethadol, LAAM, pain, chronic pain, cancer pain, QTc, arrhythmia, bradycardia. Additional articles were identified in the bibliographies of retrieved articles.
Potential Confounders to Consider
When considering whether or not methadone is causally-linked to cardiotoxicity, as much information as possible must be obtained regarding potential confounders:
The effects of concurrent drugs (in terms of direct electrophysiologic effects and indirect effects mediated by drug-drug interactions, leading to elevated serum methadone concentrations) and
The prevalences of signs, symptoms, and electropathophysiology in the populations targeted for use of methadone (e.g. patients undergoing palliative care, IV drug abusers [both human immunodeficiency virus [HIV]-positive and -negative]).
The association of antiarrhythmics with prolongation of the QTc interval with subsequent development of TdP, ventricular fibrillation, and death, is well-known. This is especially the case with class I agents (quinidine [and related quinine], procainamide, N-acetyl-procainamide, disopyramide, propafenone, flecainide, and mexiletine), class II agents (sotalol), and class III agents (amiodarone, dofetilide, ibutilide, quinidine [and related quinine], and sotalol). 7
The number of non-antiarrhythmics associated with TdP is quite large, as illustrated in a review conducted using articles published between 1966 and 2000. One hundred and eighty-nine cases were identified in 129 articles (7 cases did not provide gender data, 56 did not provide actual QTc interval data, and 83 did not provide actual QT interval data). The age range was neonate to 95 years old. The nine most frequently associated drugs were haloperidol (13.2%), erythromycin (11.6%), pentamidine (9.8%), thioridazine (6.9%), probucol (4.2%), clarithromycin (4.2%), ketanserin (3.7%), terodiline (3.7%), and halofantrine (3.2%). Some of the preceding agents have been withdrawn due to their adverse electrophysiologic effects. The percentages of cases with QTc intervals exceeding 500 ms and 500 or more ms were 89.5 and 91.7%, respectively. The percentages of cases with QTc intervals in excess of 500 ms were 75.5% with non-antiarrhythmics compared with 80.2% for antiarrhythmic drugs (the latter figure being from historical data). The percentage of cases of TdP occurring in females was 67.2%, similar to the 70% figure for antiarrhythmics (the latter figure being from historical data). The TdP prevalence in females ranged, in the nine agents listed above, from 33.3% (pentamidine) to 100% (probuchol). Only two agents of the nine had a prevalence in females of less than 50%. 8
Of all of the opioids, methadone has been the most extensively evaluated with regard to pharmacokinetic drug-drug interaction potential. The first reason for this is the importance of specific cytochrome P450 (CYP450) isozymes in its metabolism. The second reason is the use of this agent as a maintenance therapy in opioid drug abusers (a high-risk group for blood-borne viral pathogens), leading to extensive evaluation of its interaction potential with antiretrovirals. The third reason is the low hepatic extraction characteristic of methadone, which enhances the risk of enzyme induction and inhibition significantly altering drug clearance and terminal disposition half-life.
Methadone alters the pharmacokinetics of other drugs to a limited extent. It may increase plasma/ serum concentrations of nifedipine, tricyclic antidepressants, β-blockers, phenothiazines, haloperidol, zidovudine, and risperidone.9,10 It may also decrease plasma concentrations of didanosine and stavudine.9,10 Table 2 illustrates the effects of other drugs on methadone plasma/serum concentrations. Reductions in drug concentration, which are usually due to the stimulation of methadone metabolism, can produce a loss of analgesia and potentially a drug withdrawal reaction. Increases in drug concentration, which are usually due to inhibition of methadone metabolism, can produce enhanced drug toxicity. 2 Despite a moderate-to-high degree of plasma protein binding and the ability of basic drugs to displace methadone from its binding sites on alpha1 acid glycoprotein (AAG), there are no data suggesting that these protein binding displacement interactions are clinically relevant.10,11
Drugs altering methadone pharmacokinetics.
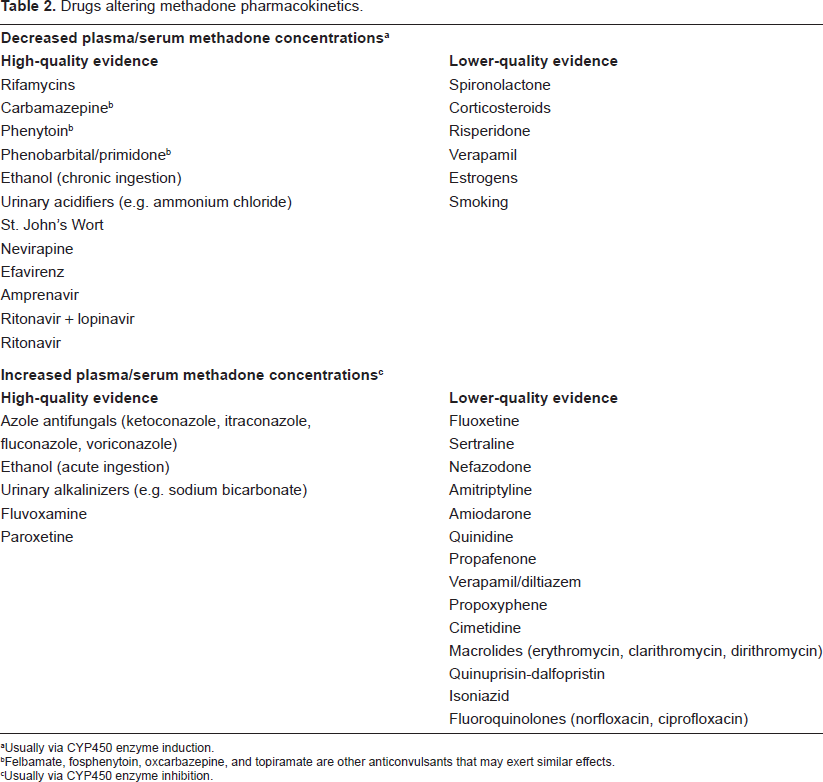
Usually via CYP450 enzyme induction.
Felbamate, fosphenytoin, oxcarbazepine, and topiramate are other anticonvulsants that may exert similar effects.
Usually via CYP450 enzyme inhibition.
Another potential confounder is cocaine, a common co-drug-of-abuse in opioid abusers. This is because cocaine suppresses the delayed rectifier potassium ion channels in cardiac myocytes (HERG current where HERG = human ether-a-go-go gene) like many other torsadogenic drugs, including methadone. 12 This suggests that the combination of methadone and cocaine may escalate TdP risk substantially. 13 However, there are currently no published data regarding the electrophysiological consequences of combination exposure.
Lofexidine is an alpha-2 adrenoceptor agonist used in opioid detoxification and to help transition from methadone to buprenorphine maintenance therapy.14,15 This agent, like clonidine, is an imidazoline alpha-2 adrenoceptor agonist which also possesses analgesic properties and theoretically may be coadministered with opioids for the management of pain. Lofexidine has been implicated in QTc interval prolongation and hypotension, the latter effect with the combination of lofexidine and methadone.16,17 The electrocardiographic effects of coadministered lofexidine and methadone were assessed in 14 opioid addicts who were stabilized on methadone maintenance therapy at a dosage of 80 mg daily. After randomization to lofexidine or placebo once daily, lofexidine was started at 0.2 mg daily with dosage escalation in 0.2 mg increments weekly until a dosage of 1.6 mg daily was attained by week 8. Multiple ECGs were obtained (before methadone, after methadone stabilization, and during and after lofexidine coadministration). Heart rate significantly decreased after the addition of lofexidine 0.4 mg daily (mean ± SD, -8.0 ± 7.3 beats/min, p = 0.0006). No significant effect of methadone monotherapy on QTc interval (vs. baseline) was noted. However, when lofexidine was added, the maximum increment in QTc interval (21.9 ± 40.8 ms) was significant (p = 0.018) as was the maximum decrement in heart rate (-9.6 ± 5.8 beats/min, p < 0.0001). In addition, significant increases were noted in PR interval (p = 0.026) and QRS interval (p = 0.002). Three female participants had clinically-important QTc interval prolongation (defined as an increase >40 ms) after the addition of lofexidine 0.4 mg daily (increments were 45, 65, and 124 ms). Thus, preliminary data have established the potential for lofexidine-methadone coadministration to induce significant QTc interval prolongation. 18
When evaluating baseline electropathophysiology in target patient populations, it is important to look for patterns consistent with sudden cardiac death as this pattern is consistent with TdP acutely degenerating into ventricular fibrillation and death, i.e. the most important pattern ascribed to methadone cardiotoxicity. In those papers describing morbidity/mortality in methadone recipients, it is also important to examine patterns in order to see whether or not they are consistent with this arrhythmogenesis hypothesis.
An epidemiological trial published in 1972 presented the mortality findings in opioid addicts in New York City from 1949 through 1970, focusing on the demographic parameters of age, race, and cause of death. Of interest, 80% of mortality was due to immediate acute reactions with 20% being due to long-term complications of drug abuse. During much of this time period, the rates of homicidal violence as a cause of death were low. Some proportion of these immediate acute reactions could have been due to drug-induced arrhythmogenic events. 19
In a literature search conducted up to May 2000, 502 deaths due to methadone were found. Of the 502 subjects, 348 (69%) had not been enrolled in an opioid maintenance program at the time of death, implicating diverted methadone as the etiology. The remaining 31% had been enrolled in an opioid maintenance program. Over one-half (51%) of deaths in enrollees in methadone maintenance programs occurred during the initial dose-finding phase of the program (days 1-28). Death during this time period could be consistent with an arrhythmogenic origin, at least in part. Later deaths in enrollees were felt to be due to both diverted methadone (including IV formulations) and methadone from maintenance program supplies. 20
Data were presented regarding a series of 10 known heroin addicts who died shortly after enrollment in a methadone maintenance program. Seven subjects were male and subjects ranged from 18 to 36 years old. Upon autopsy, five cases demonstrated no life-threatening anatomical abnormalities while the other five demonstrated bronchopneumonia and chronic persistent hepatitis. In all ten victims, methadone blood concentrations were consistent with those previously reported to be associated with death (mean 0.71 mg/L, range 0.30-2.52 mg/L). Other drugs were found in 6 subjects (benzodiazepines in 3, ethanol in 2, and morphine and propoxyphene in 1 each). In 4 of these 6 subjects, these other drugs may have contributed to death. In all subjects, methadone had been started between 2 and 6 days before death, with the mean dose at death being 60 mg daily. The timing of these deaths is consistent with a possible arrhythmogenic origin, with arrhythmias being precipitated by initiation at excessive methadone dosages and/or hepatic disease reducing drug clearance, causing serum concentrations to rise to toxic levels. 21
One ECG study was conducted in 95 asymptomatic individuals who were either enrolled in a methadone maintenance program and yet were still sporadically abusing multiple other drugs, including ethanol (group MDA) or were applicants to a methadone maintenance program, had not yet initiated methadone therapy, and used IV heroin within 24 hours before performance of the ECG (group DA). All group MDA subjects either had barbiturates in their screening urine samples and/or a history of illicit drug ingestion within 3 days before performance of the ECG. There were 41 and 54 subjects in groups MDA and DA, respectively. In addition, a control group matched for ethnic background and age was recruited. Exclusion criteria minimized the risk of confounding comorbidities. In the MDA group, the following ECG abnormalities were noted: prolonged QTc interval (34%), prominent U waves and bradyarrhythmias (each 31%), abnormal T waves (29%), premature systoles (7%), and first-degree heart block, intra-atrial conduction defect, and abnormal left axis deviation (each 4%). In the DA group, the following ECG abnormalities were noted: prolonged QTc interval (31%), abnormal T waves (16%), prominent U waves, bradyarrhythmias, and incomplete right bundle branch block (each 11%), and intra-atrial conduction defect (3%). It should be noted that quinine was found in the blood of 26% of subjects in this group. This was not unexpected due to the popularity of quinine as a diluent for illicit opioids. It should also be noted that class I antiarrhythmics like quinine prolong the QTc interval, predisposing to TdP. 7 In the control group, 11% of subjects exhibited sinus bradycardia. Twenty-two subjects had an ECG repeated at least 4 months after enrollment in a methadone maintenance program (18 had repeatedly negative urine screenings for drugs-of-abuse except methadone). In 13 of these 22 subjects, a prolonged QTc interval had been noted at baseline. In 7 of these 13 subjects (54%), QTc interval prolongation was not noted on the repeat ECG. In 4 subjects who continued to abuse multiple drugs, including ethanol, QTc interval prolongation was noted on the repeat ECG but not on the baseline ECG.22,23
All sudden death cases considered to be due to illegal substances or misuse of legal substances are reported to central authorities in France. Sudden death cases from 1994 through 1998 were evaluated in recipients of methadone and buprenorphine. The numbers of patients receiving each drug throughout France were estimated by dividing the quantity sold in the country by the mean estimated daily dose. It was estimated that 1.4-fold more sudden deaths occurred in buprenorphine than in methadone recipients and that 14-fold more patients received buprenorphine than methadone during this time period. Hence, the annual sudden death rate in methadone recipients was ≥3-fold greater than that due to buprenorphine. Methadone/buprenorphine sudden death rates (per exposed individual) were 0.0125/0.0017, 0.0025/0.0001, 0.0030/0.00001, and 0.0007/0.0002 in 1994-1995, 1995-1996, 1996-1997, and 1997-1998, respectively. In fact, had all buprenorphine recipients in France been required to take methadone instead, the estimated number of deaths from 1994 through 1998 would have been 288, rather than the observed 46. 24
An Australian epidemiological study utilized the Western Australian Data Linkage System (WADLS) to evaluate mortality in a cohort entering methadone maintenance therapy between January 2001 and December 2002. The study cohort consisted of 553 individuals, 59 percent being male (mean 32 years old) and 36 percent being female (mean 30 years old, p = 0.029). There were 2 deaths in the cohort during the first 14 days after entering therapy (crude mortality rate = 94.5 deaths/1,000 person-years) but both deaths were considered to be due to respiratory depression. Among the 13 deaths during the remainder of the study period (crude mortality rate = 5.66 deaths/1,000 person-years), none were felt to be cardiovascular in nature. 25
Over a four-year period, all patients experiencing sudden cardiac death and undergoing investigation by the medical examiner in the metropolitan Portland, OR area were consecutively evaluated in an epidemiologic trial. Case subjects were those with therapeutic blood concentrations of methadone (< 1 mg/L) while control subjects were those in whom no methadone was found in the blood. Patients with recreational drug use or drug overdose (including methadone overdose where the blood concentration > 1 mg/L) were excluded. Investigators looked for cardiac pathology consistent with sudden death (coronary artery disease [CAD], severe left ventricular hypertrophy [LVH], hypertrophic cardiomyopathy [HCM], anomalies, myocarditis, and dilated cardiomyopathy). A total of 22 sudden cardiac deaths occurred with therapeutic methadone blood concentrations (0.48 ± 0.22 mg/L, range 0.1-0.9 mg/L). In these individuals, 68 percent were male and their mean ± SD age was 37 ± 10 years old. These were compared to 106 consecutive sudden death cases without evidence of methadone being present. In these individuals, 69 percent were male and their mean ± SD age was 42 ± 13 years old. The most common indication for methadone use was pain control (55%). Among the group of 22 methadone recipients, sudden cardiac death-associated cardiac abnormalities (all CAD) were found in only 23 percent (N = 5), with no clear-cut cause of sudden cardiac death in the remaining 77 percent (N = 17). 26
Among the 106 non-methadone recipients, sudden cardiac death-associated cardiac abnormalities (CAD, severe LVH/HCM, anomalies, myocarditis, and dilated cardiomyopathy) were noted in 60 percent (N = 64, p = 0.002). The significantly lower prevalence of cardiac disease in the case (methadone) group implicates methadone, even at therapeutic concentrations, as a likely cause of sudden death. 26
Lastly, Florida Department of Law Enforcement data were analyzed to examine trends in death releated to or caused by methadone and heroin from 2001 to 2006. Medical examiners used toxicology reports to determine whether deaths were drug-related (i.e. drugs were present in the decedents) or drug-caused (i.e. drug toxicity reached lethal levels). In 2001, the heroin-caused death rate (1.65/100,000) was slightly higher than that due to methadone (1.09/100,000). However, a steady decline occurred thereafter in heroin-caused deaths (β = -0.986, r 2 = 0.972, p < 0.001) while a steady rise occurred in methadone-caused deaths (β = 0.991, r 2 = 0.982, p < 0.001). By 2006, the methadone-caused mortality rate was almost 12-fold greater than that due to heroin (4.11 vs. 0.35/100,000, respectively). In 2001, the heroin-related and methadone-related death rates were similar (2.00 vs. 2.17/100,000, respectively). Again, a steady decline occurred thereafter in heroin-related deaths (β = -0.985, r 2 = 0.971, p < 0.001) while a steady rise occurred in methadone-related deaths (β = 0.985, r 2 = 0.970, p < 0.001). By 2006, the methadone-related mortality rate was over 13-fold greater than that due to heroin (5.87 vs. 0.45/100,000, respectively). 27
Extending these observations to the drug abuser population also seropositive for HIV, there are several potential risk factors for prolongation of QTc interval besides methadone. These include HIV-associated dilated cardiomyopathy (wherein there is fibrous degeneration of the conduction system), HIV-associated coronary artery disease (due to highly-active antiretroviral therapy [HAART]-associated metabolic syndrome), direct electrophysiologic effects of drugs (antiretroviral drugs and drugs used to prevent opportunistic infections), and uncorrected electrolyte abnormalities (due to malnutrition and/or chronic diarrhea). 28
Recently, the results of a study of 300 patients undergoing palliative care who were not in a terminal state, were published. None of these patients were receiving methadone. Of these 300 patients, 47 (16%) had prolonged QTc interval values but only 2 of these 47 (4%) had QTc interval values exceeding 500 ms. In comparing the group of subjects with versus those without prolonged QTc intervals, the group with prolonged QTc intervals had an increased male:female ratio (p = 0.005), increased prevalence of cardiac disease (p = 0.05), reduced serum potassium concentrations (p = 0.005), reduced serum calcium concentrations (p = 0.002), reduced serum creatinine concentrations (p = 0.01), increased serum alkaline phosphatase concentrations (p = 0.003) and increased serum total bilirubin concentrations (p = 0.009). Weak but significant correlations existed between QTc interval and serum potassium concentration (r = -0.22, p < 0.01), serum calcium concentration (r = -0.16, p < 0.01), serum BUN concentration (r = -0.15, p < 0.05), serum creatinine concentration (r = -0.19, p < 0.01) and serum total bilirubin concentration (r = +0.18, p < 0.01). In summary, prolonged QTc intervals are relatively common in palliative care recipients but severely increased QTc intervals (defined as QTc intervals >500 ms) are rare. 29
In summary, available data suggest that the target populations for methadone use have some finite risk of exhibiting prolonged QTc intervals independent of methadone use. The data regarding the finite risk of TdP and subsequent arrhythmias in these populations are less clear-cut. In addition, many drugs that could be used as co-therapies with methadone as well as the common co-drug-of-abuse cocaine exhibit similar electrophysiological effects, potentially leading to a much enhanced risk of TdP during concurrent use. In observational studies evaluating mortality in methadone recipients, the patterns of death are frequently quite consistent with an arrhythmogenic origin, i.e. they are consistent with an immediate acute event and/or occur very early in the course of methadone therapy.
Mechanisms of Cardiotoxicity
Several hypotheses have arisen to explain the two major forms of cardiotoxicity caused by methadone and its congeners: bradycardia and, more important, ventricular arrhythmias.
Bradycardia
One study has evaluated whether LAAM and its congeners exhibit anti-cholinesterase activity that could explain induction of bradycardia. Moieties evaluated included LAAM and its two major metabolites (1-alpha-acetylnormethadol or N-LAAM and 1-alpha-acetyldinormethadol or DN-LAAM) and methadone. LAAM and its two metabolites (10-4 M) did exert significant anticholinesterase activity in guinea pig heart homogenate, in guinea pig plasma, against purified acetylcholinesterase, and against purified butyrlcholinesterase (all p < 0.05 from control). 30
The activities of all of these moieties were significantly greater in plasma compared with heart homogenate and against purified butyrlcholinesterase compared with purified acetylcholinesterase (all p < 0.05 from control). LAAM and methadone were compared for their effects on cholinesterase activities in heart homogenate and plasma. At 10-4 M, LAAM/methadone reduced heart homogenate activities to 82.11 ± 4.26/91.22 ± 5.12% of control and plasma activities to 43.43 ± 4.37/53.80 ± 9.90% of control (mean ± SEM, all p < 0.05 from control). Again, both agents were more active against plasma enzyme activity compared with heart homogenate enzyme activity. Unfortunately, the effects of LAAM and methadone were not compared statistically. In isolated guinea pig atria studies, LAAM at 5 × 10-6 M significantly potentiated the effects of bethanecol (55.96 ± 5.75% decrease in heart rate) compared with the effect of bethanecol alone (19.78 ± 2.12% decrease, p < 0.05). 30
In contrast, these moieties did not reduce heart homogenate cholinesterase activity at the low concentrations (5 × 10-6 M) which produce bradycardia ex vivo which can be blocked completely by atropine. It appears that cholinesterase inhibition does not contribute to the bradycardia induced by low concentrations of LAAM, methadone, and related compounds but may contribute to bradycardia induced by high concentrations. 30
Other investigators have wondered whether or not the bradycardia induced by methadone might be due to calcium channel blockade, considering the structural similarities of verapamil and methadone. In the mouse hot-plate test, using intraperitoneal and intracerebroventricular dosing routes, verapamil did not potentiate methadone analgesia, thus arguing for a similar mechanism of action of the two agents. Methadone 0.1 mM depressed barium-induced bovine adrenal catecholamine release and KCl-induced contractions of guinea pig ileum, both being calcium-dependent processes. Depression of the latter was a concentration-dependent phenomenon. In addition, calcium administration could reverse the opioid-induced inhibition in both tissues: 3 and 5 mM in the former and 7 mM in the latter. Lastly, in the spinal vagotomized rat model, methadone (2.5 mg/kg) produced hypotension similar to that induced by verapamil (0.5 mg/kg) while 2.5, 5, and 7.5 mg/kg doses of both drugs produced similar heart rate (bradycardia) responses. 31
Impaired contractility
Another area of interest has been the cardiac muscle contractile response when exposed to opioids. One study evaluated the effects of six opioids (methadone, morphine, meperidine, fentanyl, pentazocine, and LAAM) at bath concentrations of 10-8 to10-4 M in the presence/absence of naloxone using an isolated cat right ventricular papillary muscle preparation. Three parameters were of interest: maximum developed tension (T), maximum rate of tension development (dT/dt), and time to peak tension (Tpk). No major change occurred in any parameter with exposure to methadone and LAAM bath concentrations of 10-8 to 10-6 M. 32
At 10-5 M, methadone was associated with a small but significant decrease (-3%) in mean T (p < 0.05) while LAAM produced significant decreases in mean T (-9%) and mean dT/dt (-11%) and an increase in mean Tpk (+2%) (p < 0.001, p < 0.001, and p < 0.05, respectively). At 10-4 M, methadone was associated with significant decreases in mean T (-25%), mean dT/dt (-23%), and mean Tpk (-2%) (p < 0.001, p < 0.001, and p < 0.05, respectively). At the same concentration, LAAM was associated with significant decreases in mean T (-48%), mean dT/dt (-49%), and mean Tpk (-4%) (all p < 0.001). Pretreatment of the muscle preparation with naloxone (10-4 M) did not prevent this reduction in contractile performance. Following drug washout, contractile performance improved (for methadone where T and dT/dt returned to 89 and 90% of control, respectively) or did not (LAAM). Isoproterenol (0.8 × 10-7 M) elicited a positive inotropic response whether administered in the presence of 10-4 M methadone or LAAM or following their withdrawal. It thus appears that high opioid concentrations exerted a direct myocardial depressant effect which was not prevented by naloxone and, thus, this effect was not mediated by opioid receptor agonism. The effect was felt to be due to a nonspecific local anesthesic effect on cardiac muscle. 32 Of interest, a case of bi-ventricular heart failure has recently been reported in a 37 year old male initiated on methadone maintenance therapy at an inappropriately high dose (60 mg daily). The QTc interval was within normal limits. 33
Prolongation of QTc interval
Most drugs which prolong the QTc interval on the ECG, thus predisposing to TdP and then ventricular fibrillation and death, do so by interacting with voltage-dependent HERG potassium channels. For example, LAAM inhibited these channels in a dose-dependent fashion, with little/no blockade at concentrations of 100 nM or less, 24 ± 3% blockade at 0.3 μM, 60 ± 3% blockade at 3 μM, and 96 ± 3% blockade at 10 μM (IC50 of 3 μM).34,35 After drug washout, 75% of the blockade was eliminated over 5 minutes. 36 The major metabolite N-LAAM also inhibited HERG with an IC50 estimated at 12 μM 35 In contrast, LAAM had little or no effect on other potassium channels such as the Kv LQT1/min K and Kv 4.3 cardiac channels. 35
Methadone blocks HERG channels as well, with an order of potency of S-isomer being greater than the R, S-racemate, the latter being greater than the R-isomer (respective IC50 values of 12, 19, and 29 μM, respectively, at room temperature and 2, 3, and 7 μM, respectively, at 37 °C). 37 The two major metabolites of methadone (EDDP, EMDP) are extremely weak as HERG channel blockers. 37 This blockade occurs very rapidly, with steady-state inhibition being achieved in less than 1 second at the IC50 concentration of 10 μM. 36
As the commercial US formulation for IV methadone contains the preservative chlorobutanol, it is important to assess the effects of each agent separately and together in order to gauge the risks of QTc interval prolongation with IV methadone. Chlorobutanol (3 mM) alone caused an approximate 40% fall in HERG currents while the combination of 3 mM chlorobutanol plus varying methadone concentrations led to a clear, dose-dependent fall in HERG currents, demonstrating that both agents were capable of blocking HERG channels. Using permutations of multiple concentrations of each agent plus isobologram analysis, a synergistic interaction was demonstrated. For example, a 50% reduction in HERG currents was noted when both agents were present at only one-third of their IC50 concentrations. 38
In the isolated perfused rabbit heart preparation, perfusion with methadone 1-3 μM produced a 10-12% increase in QT interval duration (p = NS). When assessing absolute QT interval values, only with perfusion at the 30 μM concentration was a significant result compared with baseline obtained (p < 0.05). When assessing percentage change in QT interval from baseline, results with 10 μM (21 ± 5% increase, p < 0.05) and 30 μM (44 ± 12% increase, p < 0.001) concentrations were significant. Trends to dose-dependency of this phenomenon were seen. 39
The effects of LAAM and methadone on ECG parameters have been evaluated in one study utilizing patients addicted to opioids who were induced and maintained on methadone for 30 days and then LAAM for 30 days. Of 123 subjects enrolled (77 being male, mean 41.5 years old), 55 (33 being male) completed entire study period. In males and females, mean baseline QTc interval values were 411.4 and 427.3 ms, respectively. Methadone increased the QTc interval by means of 7.6% and 7.9% in males and females, respectively. Corresponding additional QTc interval increases of 1.55% and 3.51% occurred during LAAM administration. The proportions of subjects exceeding threshold QTc interval values of 450 ms (males) and 470 ms (females) during at least 1 study timepoint were 69.7% and 72.7%, respectively. No bradycardia or atrial or ventricular arrhythmias occurred at any point during the study. 40
In another study, ECG recordings were performed within a randomized controlled trial comparing the efficacy and tolerability of LAAM and methadone as opioid maintenance therapies. Recordings were done twice (during the run-in period when all patients were receiving methadone and 24 weeks after randomization into methadone or LAAM therapy groups). Complete ECG datasets were available for 53 patients (LAAM, N = 31; methadone, N = 22). During the 24-week randomized phase, the mean ± SD methadone group dose was 69 ± 28 mg/day and LAAM doses were 90 ± 32 mg on Monday and Wednesday (equivalent to 69 ± 25 mg methadone) and 110 ± 40 mg on Friday (equivalent to 69 ± 25 mg methadone). No clinical cardiac events were reported. After 24 weeks, LAAM recipients experienced a significant increase in mean ± SD QTc interval from a baseline of 409 ± 22 ms to 418 ± 28 ms (p = 0.046). Methadone did not produce a significant change in QTc interval. No significant changes in QTc interval dispersion were seen after either drug. More patients with borderline (431-450 ms in males, 451-470 ms in females) or prolonged QTc intervals were observed in LAAM recipients (N = 7) compared with methadone recipients (N = 1) although this difference was not significant. It should be noted that pre-randomization, there were 3 LAAM recipients and 2 methadone recipients with abnormal QTc intervals. Increases in QTc interval between 30 and 60 ms and greater than 60 ms were seen in two patients each during LAAM therapy and an increase between 30 and 60 ms was seen in one patient during methadone therapy (p = NS). The lack of significant change in the methadone group was not unexpected since the baseline condition for comparative purposes occurred during intake of methadone in a similar dosage (72 ± 26 mg/day) compared with the drug condition of methadone during the randomized phase (69 ± 28 mg/day). The study design did not allow a comparison of the electrophysiologic effects of the two opioids. 41
In addition, the electrophysiologic effects of methadone and buprenorphine have been evaluated in patients receiving opioid maintenance therapy with methadone (N = 35, 23 males, 35 ± 8 years old) or buprenorphine (N = 19, 11 males, 34 ± 6 years old) and non-dependent control subjects (N = 17, 9 males, 31 ± 8 years old). Methadone and buprenorphine recipients, having been on a stable drug dose for a minimum of four weeks, were taking maintenance doses of 69 ± 29 mg/day and 11 ± 5 mg/day, respectively. Mean ± SD QTc intervals did not differ between the three groups (methadone, 407 ± 23 ms; buprenorphine, 407 ± 18 ms; controls 397 ± 21 ms). No correlation was evident between methadone dose and QTc interval (p = 0.25). However, the QTc interval occurring with methadone doses of 60 mg/day or greater (405 ± 29 ms) was significantly greater than that occurring with doses of less than 60 mg/day (381 ± 27 ms, p = 0.02). Two methadone patients and one control subject (all males) had prolonged QTc intervals (defined in males as QTc interval >430 ms). The three groups significantly differed with respect to U wave frequencies. U waves were noted in 46%, 0%, and 13% of methadone, buprenorphine, and control subjects, respectively (odds ratio for methadone vs. other two groups pooled = 7.8). Lastly, methadone patients manifesting U waves were on a significantly higher dose than those methadone patients without U waves (89 ± 29 mg/day vs. 60 ± 24 mg/day, p = 0.004). 42
Although several other opioids are capable of blocking HERG currents, albeit in a much weaker fashion than do LAAM and methadone, the clinical relevance of HERG blockade can be assessed by dividing the IC50 value by the peak serum concentration value. For other opioids, the resulting values exceed 400 or 500, indicating a wide margin for cardiac safety. For methadone and LAAM, respective values of 2.7 and 2.2 indicate a much narrower margin for cardiac safety. 36
Brugada-like syndrome
Another diagnosis compatible with sudden cardiac death induced by methadone is a Brugada-like syndrome. Brugada syndrome is a heterogenous genetic disease predisposing to life-threatening ventricular tachycardia and sudden cardiac death, with about 1/4 of cases being due to a loss of function mutations in the cardiac sodium channel SCNSA gene. It is usually identified by a characteristic Brugada-type ECG (ST elevation of coved type in precordial leads V1-V3), although affected individuals may have normal ECG findings. Inducers of a Brugada-like ECG include febrile states, cocaine and methadone, sodium channel blockers (e.g. propafenone, procainamide, flecainide, bupivacaine, lidocaine, and tricyclic antidepressants), propofol, and electrolyte imbalances. In a recent multicenter observational trial of patients presenting with typical Brugada-like ECGs during an acute medical event (N = 47), 16, 26, and 5 cases were due to febrile episodes, drug-related, and due to electrolyte imbalances, respectively. Fifty-one percent had malignant arrhythmias, with 38% developing (aborted) sudden cardiac death, and 6% each experiencing ventricular tachycardia and syncope. One of the victims of sudden cardiac death was a methadone recipient. 43
Clinical Data
Review of FDA Medwatch reports involving methadone from 1969 through October 2002 revealed some interesting findings. Of the 5503 AEs, 43 (0.78%) involved TdP and 16 (0.29%) involved QTc interval prolongation. Doses were available in 42 (71%) events. In 10/42 (29%) events, doses were within the recommended range for opioid maintenance (i.e. 60-100 mg/day). The mean ± SD dose was 410 ± 349 mg/day (median 345 mg/day) (range 29-1680 mg/day). The route of administration was provided in 25 (42%) events, with 3 (12%) being by the IV route and 22 (88%) being by the oral route. Risk factors previously associated with the TdP risk of other drugs (e.g. female sex [in 59%], interacting medication [in 24%], hypokalemia [in 24%], hypomagnesemia [in 10%], and structural heart disease [in 5%]) were found in 44 (75%) cases. In 14 (24%) cases, subjects were taking at least one other drug associated with prolonged QTc intervals and/or TdP while in 10 (17%) cases, subjects were taking at least one other drug associated with inhibition of methadone metabolism. Almost one-half of the events resulted in a need to hospitalize the subject or resulted in prolonged hospitalization (28 cases [47%]). In 5 cases (8%), the subject died. 44
Adults
The majority of case report and case series data documenting methadone-associated prolongation of the QTc interval and TdP have involved the adult patient population using the drug for analgesic purposes (N = 174, Table 3).38,45–57 A minority of clinical data have involved the adult patient population undergoing methadone maintenance therapy (N = 46).34,58–81
Case reports/series and retrospective series/prospective trials of methadone-associated QTc interval prolongation and torsade des pointes/ventricular fibrillation in patients receiving the drug for analgesia.
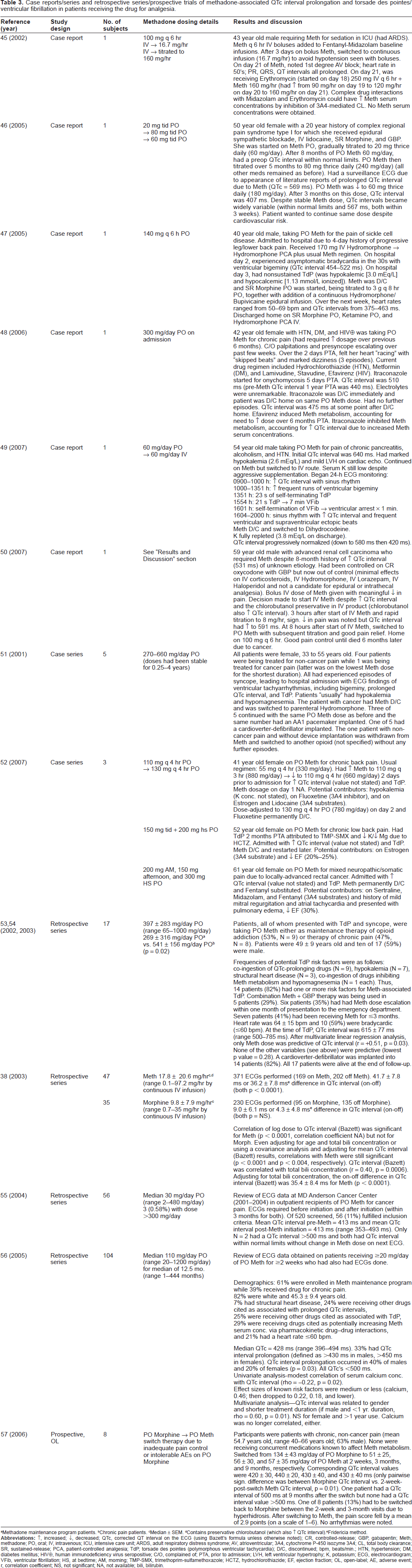
Methadone maintenance program patients.
Chronic pain patients.
Median ± SEM.
Contains preservative chlorobutanol (which also ↑ QTc interval).
Fridericia method.
However, the majority of study data providing the epidemiology of methadone-associated QTc interval prolongation and applied correlational and regression analyses have been obtained in patients undergoing methadone maintenance therapy for opioid addiction (Table 4).37,82–91 The prevalence of QTc interval prolongation varied widely from study to study, depending upon the definitions utilized. For example, when prolongation was defined as a QTc interval >440 ms (upper limit of normal), 450-499 ms, ≥460 ms, ≥500 ms, and >500 ms, the corresponding prevalence values were 29% (28% males, 32% females), 14%, 30%, 16%, and 0-2.2%. When prolongation was defined as a QTc interval equal to 450-470 ms (female) or 430-450 ms (male), >450 ms (female) or >430 ms (male), >470 ms (female) or >450 ms (male), and >480 ms (female) or >470 ms (male), the corresponding prevalence values were 30%, 17%, 8-20%, and 2.4% (Table 3).37,82–91
Synopsis of QTc interval effects of methadone in subjects undergoing methadone maintenance therapy of opioid addiction.

Vs. reference values for persons of same age and sex.
Bazett correction.
Fridericia correction.
PT level = surrogate of liver function. meth dose had weakest effect of the 4. serum K × 3A4 inhib use interaction: p < 0.001. serum K × PT interaction: p = 0.002.
Could not distinguish R- from S-isomer as too highly correlated with each other.
When racemate serum conc. >600 ng/mL (prolonged QTc interval: >450 ms [M], >470 ms [F]).
When racemate serum conc. ≤600 ng/mL.
% with prolonged QTc intervals.
% with borderline QTc intervals (borderline QTc interval: 430-450 ms [M], 450-470 ms [F]).
QTc interval >450 ms (M), >470 ms (F).
QTc interval >440 ms (M), >450 ms (F).
Significant correlations existed between QTc interval and methadone serum concentrations although correlation coefficients (r) were modest at best: 0.22 (for trough racemate concentration), 0.24 (for trough R-isomer concentration), 0.31 (for trough racemate concentration), 0.25 (for peak racemate concentration), 0.37 (for trough concentration), and 0.32 (for peak concentration). Significant correlations existed between QTc interval and methadone doses although, again, r values were quite modest: 0.53 (male), 0.35 (female), 0.20, 0.19 (QTc interval at trough), and 0.22 (QTc interval at peak).37,82–91
Univariate regression analyses identified several parameters significantly correlated with an increase in the QTc interval: decreasing serum potassium and calcium concentrations, use of comedications, increasing methadone dose and trough racemate serum concentrations, and positive slow metabolizer status for CYP450 isozyme 2B6. Univariate risk factors predicting an increased prevalence of prolonged QTc intervals in a population included use of methadone and ethanol abuse (Table 4).37,82–91
Multivariate regression analyses identified the following parameters as significantly correlated with an increase in QTc interval: decreasing serum potassium and calcium concentrations; increasing methadone dose, trough racemate serum concentration, and prothrombin time; use of comedications; HIV seropositivity; male sex; use of CYP450 isozyme 3A4 inhibitor therapy; and positive slow metabolizer status for CYP450 isozyme 2B6 (Table 4).37,82–91
Overall, the magnitude and prevalence of prolonged QTc intervals did not differ between male and female methadone recipients. Outcome data were available in two papers. In one paper, the proportion exhibiting syncope rose as methadone dose rose, although these data were not quantitatively analyzed. By logistic regression analysis, the odds for reporting any syncope rose by 1.2-fold for each 50 mg increment in the methadone dose. 83 In the other paper, TdP was reported in 6 methadone patients (3.6%) and ventricular bigeminy in 3 methadone patients (1.8%). In 2 patients, ventricular bigeminy preceded TdP. 86 Bradycardia did not appear to be a problem in methadone recipients. In one paper, heart rate rose by 5 ± 13 beats/minute at the six month visit (p < 0.001) and by 5 ± 12 beats/minute at the 12 month visit (p = 0.001) in methadone recipients. 90
There is no a priori reason to expect that these electrophysiological responses seen in patients undergoing methadone maintenance therapy of opioid addiction would not also be seen in patients receiving the drug for analgesia. The doses utilized in both conditions are similar in degrees of variability and range. Many of the risk factors for prolonged QTc interval with methadone can occur in both patient populations: electrolyte abnormalities; use of comedications with direct QTc interval-prolonging effects or indirect potentiating effects through inhibition of methadone metabolism, leading to elevated serum drug concentrations; methadone dose escalation; and hepatic impairment, leading to inhibition of drug metabolism. The two risk factors likely to be unequally distributed between the two populations are concurrent cocaine use and HIV seropositivity which would be expected to more commonly occur in the opioid addict population. However, the importance of HIV seropositivity as a risk factor compared to the aforementioned risk factors is unclear. Overall, the electrophysiological effects of methadone, as noted in Table 4, are likely to be applicable to methadone used for analgesia as well.
Pediatrics
There are only two published case reports documenting methadone cardiotoxicity in pediatrics (bradycardia, prolonged QTc interval): one in a neonate and one in an infant.92,93 Reviews of methadone poisoning in children mention only peripheral vasodilation (predisposing to orthostatic hypotension), occasional sinus bradycardia, and cerebrovascular vasodilation (due to decreased sensitivity of the respiratory center to CO2, leading to an increased pCO2) which can increase intracranial pressure. These are the only cardiovascular effects of methadone poisoning mentioned for children. No arrhythmias such as TdP are mentioned. 94
Whether or not methadone cardiotoxicity exists in utero due to maternal ingestion is not known. Of interest, the fetal effects of split dosing (the usual schedule for analgesic) compared with once daily dosing (the usual schedule for dosing during maintenance of opioid addiction) have been assessed recently. Split dosing has significantly less negative impact on fetal neurobehavior than once daily dosing. 95 Some parameters wherein split dosing was found to be the safer schedule included fetal heart rate (p < 0.05) and heart rate variability (p < 0.001) at both peak and trough maternal methadone concentrations. 95 The clinical importance of these findings in terms of methadone-associated arrhythmias is unknown at present.
Responses to Methadone Cardiotoxicity
The responses to methadone cardiotoxicity are dependent upon the manifestations thereof. Responses will differ for asymptomatic QTc interval prolongation compared with symptomatic QTc interval prolongation (TdP and its sequelae). After emergency arrhythmia management, the next response to symptomatic QTc interval prolongation should be an attempt to discontinue the drug and substitute an opioid devoid of cardiotoxicity risk. In the context of methadone maintenance therapy of opioid addiction, buprenorphine, naltrexone, or a combination of the two agents are potentially useful alternatives.81,96 Even slow-release oral morphine has been a potentially useful alternative. 97 In the case of methadone use for analgesia, this may be more complicated since methadone is frequently used in subjects with pain states poorly responsive to more frequently-prescribed opioids such as morphine, oxycodone, or hydromorphone or in subjects who tolerate these agents poorly. In such subjects, a change in opioid may not be desirable. Implantation of a cardioverter-defibrillator and continuation of methadone therapy may be the only reasonable option. However, device implantation should always be considered only after a switch from methadone to another opioid is thoughtfully considered and ruled out. These devices are expensive alternatives and patients will continue to be symptomatic when devices fire in order to terminate TdP/ventricular tachycardia/ventricular fibrillation.
In a series of 8 patients receiving methadone maintenance therapy who presented with prolonged QTc intervals and symptomatic TdP, 5 and 3 patients were implanted with dual-chamber and single-chamber devices, respectively. Two patients did not continue methadone therapy post-procedure. One patient had a periprocedural complication (lead perforation with tamponade, requiring pericardiocentesis). One had a postprocedural complication (presumed device infection followed by explantation and implantation of a new device after completion of antimicrobial therapy). Over a mean 27-month follow-up period, one patient died of unknown cause(s) and three patients had nine interrogation-proved device firings leading to 11 emergency room visits and eight hospitalizations. In these three patients, two exhibited prolonged QTc intervals during device therapy (mean ± SD, 581 ± 53 ms) and one had hypokalemia and a recently-escalated methadone dose as predisposing factors. In two patients, the only predisposing factor was continued methadone therapy. 98 A case could have been made not to perform device implanation in the two patients where the presumed precipitant (methadone) had been discontinued. In the other six patients, details regarding the absolute need for continued methadone maintenance versus a switch to (an)other opioid(s) were unavailable. Results of this series demonstrated the very real benefit of life prolongation but also the potential for complex and expensive complications of device implantation.
An example where a case could be made not to pursue device implantation in response to continued need for methadone despite arrhythmia risk is in those individuals with advanced terminal disease undergoing palliative therapy. In this circumstance, the benefits of continued methadone therapy may be perceived as greater than the risks of side effects, including arrhythmias, and device effects and complications.
Another potential response to symptomatic QTc interval prolongation is methadone dose reduction in an attempt to find an effective dose which is below the threshold for induction of arrhythmias. However, seldom is this successful and the end result is usually a subtherapeutic dose where the arrhythmias are reduced in frequency/severity or eliminated but the analgesic efficacy is seriously compromised.
Recommendations for Improving the Cardiac Safety of Oral Methadone
Before discussing recommendations to improve upon the cardiac safety of oral methadone, it would be useful to discuss the current state of knowledge of health professionals about the cardiotoxicity of methadone. This has been the subject of only one study. In an attempt to discern the extent of knowledge of the adverse electrophysiologic effects of methadone, a national mail survey was conducted in 2006 of physicians licensed as medical directors of accredited US opioid maintenance programs. The response rate was 66% (692/1049). Seventy-five and 71% of respondents correctly identified the QTc interval-prolongation and TdP-induction risks of LAAM, respectively. However, only 41 and 24% correctly identified the QTc interval-prolongation and TdP-induction risks of methadone, respectively. Respondents were better at correctly identifying the lack of association of buprenorphine with QTc interval prolongation (52%) and induction of TdP (58%). Although programs with a larger census (p < 0.001 for trend) and those in academic settings (p = 0.08) predicted greater awareness of the adverse electrophysiological effects of methadone, even with these characteristics, awareness rates did not exceed 54%. 99 It thus appears that substantial efforts are required to educate healthcare personnel with regard to the adverse electrophysiologic risks of methadone and steps to take to reduce these risks.
The first US guideline to be published, on parenteral methadone in analgesia and palliative care, was developed from a roundtable discussion held on March 3, 2007 in New York City. 5 The following recommendations from that discussion are also potentially applicable to recipients of oral methadone for analgesia. It was recommended to discuss openly with the patient, family, and healthcare proxy, the risk of QTc interval prolongation (and, therefore, TdP and sudden cardiac death) so that an informed decision could be made. It was felt that the risk was small and that close ECG monitoring could reduce that risk. Consideration of burden versus benefit was paramount in treating patients in pain. In those with a life-threatening illness, the potential benefit of controlling otherwise refractory pain may far outweigh the risks, even when ECG monitoring for arrhythmias was impractical. The following recommendations for ECG monitoring were offered:
at baseline, prior to initiation
at 24 hours after initiation
at 4 days after initiation (i.e. at steady-state)
when the dose was significantly escalated (note: no definition of “significantly” was provided) and
when there was a change in patient condition or therapy which further increased arrhythmia risk (i.e. electrolyte imbalance, CHF, or drug-drug interactions potentially leading to increased serum methadone concentrations).
It was suggested that electrolyte monitoring may be necessary in high-risk patients. In response to observation of a prolongation of the QTc interval, it was recommended to rule out the presence of additional causes. For individuals with a predisposition to or high risk of QTc interval prolongation, it was recommended that a preservative-free parenteral methadone product should be available (requires sterile compounding by pharmacy). However, there were notable omissions in the recommendations. First, no recommendations were made regarding recipients of oral methadone in the inpatient palliative care setting or oral/parenteral methadone in the outpatient palliative care setting. Second, no recommendations were provided regarding ECG monitoring in outpatients receiving methadone. Third, no specific recommendations were provided regarding methadone dosage adjustment based on the observed QTc interval.
The second US guideline to be published, on methadone use for opioid maintenance therapy and analgesia, was developed by an expert panel holding meetings in May 2003, July 2007, December 2007, and July 2008. 6 Although methadone use as an analgesic was one target of the panel, the Center for Substance Abuse Treatment of the Substance Abuse and Mental Health Services Administration convened the meetings and the majority of participants were involved only in opioid maintenance therapy. Five recommendations from the expert panel were potentially applicable to recipients of oral methadone for analgesia:
to inform patients of arrhythmia risk when prescribing methadone
to inquire regarding histories of structural heart disease, arrhythmias, and syncope
to obtain a pretreatment ECG to measure the QTc interval and obtain follow-up ECGs within 30 days then annually. additional ECGs were recommended if the daily methadone dose exceeded 100 mg or patients had unexplained syncope or seizures
if the QTc interval >450 but <500 ms, to address the benefit vs. risk ratio with patients and increase the monitoring frequency. if the QTc interval ≥500 ms, to consider stopping the drug or reducing its dose, eliminating any other contributors to arrhythmia risk, or changing to an alternative therapy
to be aware of drug-drug interactions between methadone and other drugs that possess QTc interval-prolonging properties or inhibit methadone metabolism.
Unlike the first guideline, this one was independent of the route of methadone administration. It also mentioned specific QTc interval ranges and the recommended responses to each.
Other organizations have provided less structured recommendations than the preceding two. The Medicines and Healthcare Products Regulatory Agency in the United Kingdom recommended ECG monitoring when the daily methadone dose exceeded 100 mg but did not provide a detailed monitoring approach. 100 The UK Department of Health guidance on clinical management of drug abuse and dependence incorporated this approach as well and recommended that patients be informed of the reasons(s) for monitoring if ECG screening was being considered. 101 The Methadone Maintenance Guidelines of the Ontario (Canada) College of Physicians was more specific and recommended ECG monitoring when daily methadone doses exceeded 150 mg and repeating the ECG when the daily dose approached 180 to 200 mg. If the QTc interval exceeded 470 ms, the recommendation was to reduce the daily dose and refer the patient to a cardiologist. 102
It should be remembered that there are no data proving the value of serial ECG monitoring in reducing the adverse cardiovascular outcomes of methadone therapy. At present, ECG monitoring is recommended solely on the basis of expert opinion that it is a reasonable approach.
From the data reviewed in this paper, it would appear that the safety of patients receiving oral methadone therapy for analgesia can be improved through institution of an ECG monitoring program. A selective amalgamation of the recommendations cited in the two published US guidelines5,6 plus additional ones appear to be reasonable for recipients of oral methadone for analgesia.
Recommendations for safe use of oral methadone as an analgesic
Electrocardiographic screening must utilize the standard 12-lead ECG since QTc prolongation can be missed if measured on single- or three-lead ECGs. 5 Since it is dependent on heart rate, the QT interval must be corrected (designated QTc) using an appropriate formula. Most frequently, the Bazett formula is used (QTc = QT interval in ms divided by the square root of the preceding RR interval [in seconds]). 103 Although this formula is likely to overcorrect in the setting of high heart rates, it is a reasonable method for screening purposes as long as patients remain supine for approximately 5 minutes before performing the ECG. Ideally, ECG screening should be performed at the time of attainment of peak serum methadone concentrations (i.e. 2 hrs. after dosing). There is controversy regarding manual versus automated interpretation of the QTc interval. Since a higher proportion of patients receiving methadone for analgesia are likely to have structural heart disease, manual interpretation is preferred. In addition, the same lead should be used each time.
Inform patients, families, and healthcare proxies regarding the risks of QTc interval prolongation (and, hence, the risks of TdP and sudden cardiac death) to allow an informed choice to be made.
Solicit histories of structural heart disease, arrhythmias, and syncope from patients and/or families. Look for other risk factors for prolonged QTc intervals: abnormal serum electrolyte concentrations and use of comedications that also prolong the QTc interval or impair methadone metabolism, leading to elevated methadone serum concentrations, hepatic impairment (esp. if severe [Child-Pugh class C]).
Consider the burden versus the benefit of using oral methadone in patients with severe pain, even in the presence of asymptomatic/symptomatic QTc prolongation. In those with a life-threatening illness or terminal illness, the potential benefit of controlling otherwise refractory pain may far outweigh the arrhythmia risks, even when ECG monitoring is impractical and/or implantation of a cardioverter-defibrillator is not deemed to be feasible. The choice by the patient/family to pursue methadone therapy even without ECG monitoring and/or implantation of a cardioverter-defibrillator should be respected by the clinician and a detailed note to this effect should be entered into the medical record.
Obtain ECGs at the following times: baseline (before initiation of oral methadone), 4-7 days after initiation (i.e. at steady-state) and annually. additional ECGs are recommended 4-7 days after dosage increments or when patients have unexplained syncope or seizures, or when there are changes in condition or therapy which increase arrhythmia risk (e.g. electrolyte imbalances, hepatic impairment, addition of drugs with QTc interval-prolonging properties or the ability to impair the metabolism of methadone).
Obtaining an ECG only after attainment of a particular daily dosage (see previous guidelines)5,6 or when a dosage increment exceeds a certain quantity of drug (see previous guidelines)5,6 is not appropriate. Frequently, patients can be maintained on much lower doses of oral methadone when used for analgesia as opposed to use in the maintenance of opioid addiction. In addition, the dosage increment for analgesia is usually calculated as a percentage of the current daily dose (usual range is 25-50%+, depending on pain severity) as opposed to fixed mg increments as used in the maintenance of opioid addiction. Thus, incremental methadone doses can vary over a wide range on a population basis. No evidence exists to support arbitrary daily doses or increments as the basis for performing follow-up ECGs. At present, this should be addressed on an individual patient basis. Changes in condition or therapy which potentially augment arrhythmia risk should be aggressively managed if consistent with the plan of care for the patient.
Follow serum electrolyte concentrations on an individualized basis.
In terms of the arrhythmia risk with specific QTc intervals in the asymptomatic patient, only for QTc intervals exceeding 500 ms is there general agreement that intervals in this range are potentially dangerous. In this situation, consideration should be given to substituting (rotating to) another opioid, reducing the methadone dose and augmenting analgesia with adjuvant agent(s), and/or discontinuing other agents potentially prolonging the QTc interval and/or elevating methadone serum concentrations.
Residence in certain settings such as long-term institutional care (assisted living, skilled care, palliative care) may make ECG monitoring quite challenging. Such monitoring may not be available in these settings, necessitating transport of the patient to locations (e.g. clinics, emergency departments, etc.) where 12-lead ECGs can be performed. This may have negative logistical, financial, and even clinical consequences. As mentioned previously, individuals undergoing hospice or palliative care frequently exhibit potential benefits from methadone use that far exceed the risks of cardiotoxicity, even if serial ECG monitoring proves impractical.
With recommendations now being advanced to screen potential methadone recipients with pretherapy and multiple on-therapy ECGs, it appears that LAAM and methadone will soon have similar safety monitoring requirements. This may bode well for a return of LAAM to the marketplace, at least in the US where it is still approved by the FDA. In fact, in terms of opioid maintenance therapy, in many ways, LAAM is a superior opioid. In clinical trials, LAAM has been non-inferior and, in many reports, superior to methadone and buprenorphine in suppressing illicit opioid use.104–109 The adherence burden is lower with LAAM due to its twice or thrice weekly dosing compared with the daily dosing of the other two agents. Having LAAM available may, in fact, reduce TdP risk by reducing the number of patients needing very high methadone doses to “stay clean”. These individuals can often be managed with much lower doses of LAAM which have a lower intrinsic electrophysiologic risk. 110 In summary, with the large cardiovascular “safety advantage” of methadone (i.e. methadone's presumed lack of effect on the QTc interval and, hence, risk of TdP and ventricular fibrillation) having been dispelled, the question now is when/whether Roxane Laboratories or another pharmaceutical manufacturer will bring LAAM back? 4
Conclusion
Once used as an “opioid of last resort” for analgesia, methadone is currently enjoying a renaissance in its use as an analgesic. As a result of its use today as a second-line (and, in some cases, first-line) opioid analgesic, formerly infrequent toxicities such as cardiotoxicity are becoming more prevalent. This paper has presented an overview of the epidemiology, mechanisms, and risk factors for methadone-associated cardiotoxicity (predominantly QTc interval prolongation, torsade des pointes and ventricular fibrillation). Of necessity, data from both the chronic pain and opioid addict populations have been reviewed in order to provide as complete an overview as possible. Recommendations have been made for the safe use of oral methadone for analgesia in all healthcare settings. Hopefully, utilization of these recommendations will reduce the frequency of cases of cardiotoxicity associated with the use of oral methadone.
Disclosure
The author reports no conflicts of interest.
