Abstract
Completing its initial phases of drug development in the mid 1990s as the one of the first fluoroquinolones that could be used with confidence to treat respiratory tract infections, levofloxacin went on to become one of the most widely prescribed antibiotics in the world. Available in both oral (po) and intravenous (IV) formulations and with characteristics of over 90% bioavailability, distribution into both extracellular and intracellular pulmonary compartments, highly predictable pharmacokinetics with over 90% of the drug being excreted unchanged in urine, and reliable activity against a broad spectrum of clinically important pathogens, levofloxacin has been used successfully to treat patients with a variety of serious infectious diseases as well as common infections most often treated outside of the hospital setting. Results of clinical trials involving patients with respiratory tract, urinary tract, and skin infections have consistently shown rates of clinical success and bacteriological eradication that were comparable to other widely used broad-spectrum agents. Regimens of levofloxacin, initially involving total daily doses of 250 mg to 500 mg, but more recently regimens involving 750 mg doses, have been shown to be safe and effective. Nearly a decade and a half of clinical experience has defined a safety and tolerability profile that permits data-driven assessment of the risks and benefits of using levofloxacin. As resistance to currently available fluoroquinolones has emerged, the clinical value of levofloxacin deserves continued evaluation. However, consistently high rates of susceptibility of clinically important bacteria, especially among those bacteria that commonly cause respiratory tract infections, such as Streptococcus pneumoniae and Haemophilus influenzae, suggest that this agent will continue to be a widely used well past the 20-year anniversary of its introduction into the antibacterial armamentarium.
Introduction
Levofloxacin is a member of the fluoroquinolone class of antibacterial agents with microbiological activity against clinically relevant bacteria that cause respiratory, skin and skin structure, and genitourinary tract infections (Table 1; personal communication, Dr. Alan Evangelista; hereafter cited as TRUST 2008). In the United States, levofloxacin is currently indicated for the treatment of respiratory tract infections, including acute bacterial exacerbations of chronic bronchitis, acute bacterial sinusitis, and community-acquired and nosocomial pneumonia; complicated and uncomplicated skin and skin structure infections; post-inhalational anthrax; and genitourinary infections including uncomplicated and complicated urinary tract infections, acute pyelonephritis, and chronic prostatitis. 1 Similar indications have been approved for levofloxacin in Europe (Tavanic®) and Asia (Cravit®). As the understanding of pharmacodynamics and the drug's safety and tolerability evolved, clinical trials have been conducted with several dose regimens. Initial work largely assessed regimens involving not more than a 500 mg daily dose: 250 mg dose regimens for patients with urinary tract infections and 500 mg dose regimens for respiratory tract infections. Higher dose regimens involving 750 mg doses were subsequently studied in patients with skin and skin structure infections 2 and in patients with nosocomial pneumonia. 3 Most recently, higher-dose 750 mg dose regimens given over a shorter course (5 days vs. 10 days) were studied in patients with respiratory tract infections, as the potential for this modification to favorably impact drug resistance became apparent. 4
Antimicrobial susceptibility from the TRUST Program: 2008 results (personal communication, Alan Evangelista, PhD).
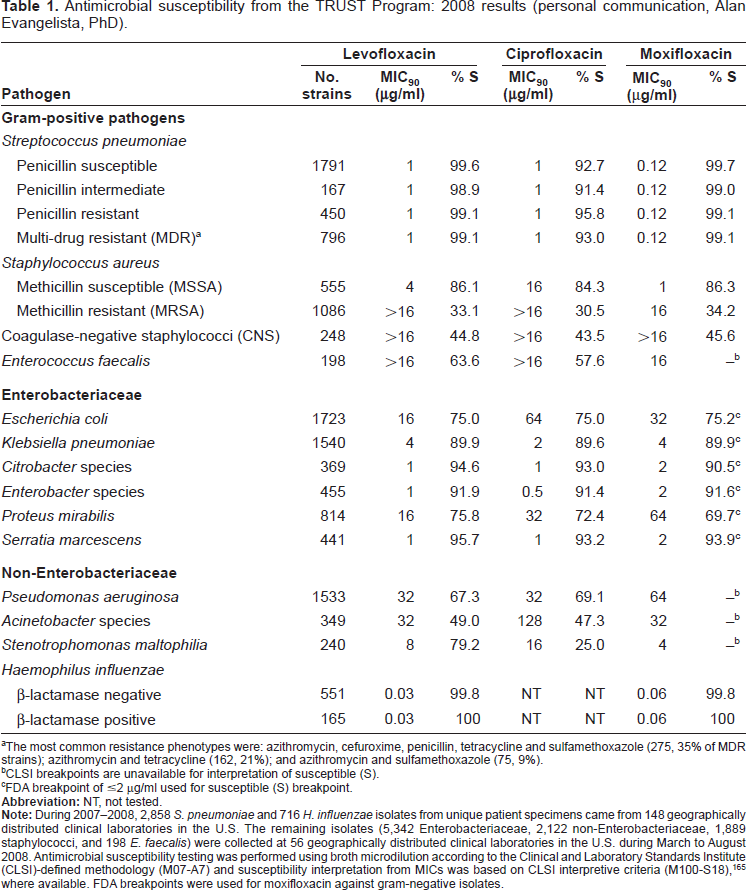
The most common resistance phenotypes were: azithromycin, cefuroxime, penicillin, tetracycline and sulfamethoxazole (275, 35% of MDR strains); azithromycin and tetracycline (162, 21%); and azithromycin and sulfamethoxazole (75, 9%).
CLSI breakpoints are unavailable for interpretation of susceptible (S).
FDA breakpoint of ≤2 μg/ml used for susceptible (S) breakpoint.
The primary focus of this review is on the utility of levofloxacin in respiratory and genitourinary tract infections, including relevant pharmacokinetic, pharmacodynamic, and in vitro data that support the clinical data in these indications. Discussion on current issues involving fluoroquinolone resistance among specific pathogens (including methicillin-resistant Staphylococcus aureus), the use of levofloxacin as part of a drug regimen in treating tuberculosis (an indication which has not been given regulatory approval) or in prophylaxis against organisms that could be used as weapons (Bacillus anthracis) is beyond the scope of this review.
This review was based on information collected from a Medline search conducted in October 2008 that was directed at identifying publications in English involving levofloxacin. The following key words were used: levofloxacin, antibiotic resistance, pharmacokinetics, pharmacodynamics, pneumonia, acute sinusitis, chronic bronchitis, urinary tract infection, pyelonephritis, chronic prostatitis, adverse events, seizure, tendonopathy, hyperglycemia, hypoglycemia, QT, hepatotoxicity, allergy, Clostridium difficile-associated diarrhea, peripheral neuropathy, and phototoxicity.
Mode of Action
Levofloxacin is the L-isomer of the racemic agent ofloxacin. 1 Fluoroquinolones interfere with bacterial DNA metabolism by inhibiting two bacterial enzymes—DNA gyrase and topoisomerase IV—which are critical to DNA replication, transcription, repair, and recombination. Their primary targets against Gram-positive and Gram-negative bacteria are bacterial topoisomerase IV and DNA gyrase, respectively. 5 The primary mechanism by which bacteria develop reduced susceptibility to fluoroquinolones is mutation in genes that code for DNA topoisomerase IV (i.e. par C and parE) and/or DNA gyrase (gyr A and gyr B).6–8 Less often, resistance results from mutation in efflux regulatory genes (mexR and nfxB), resulting in active drug efflux from the cytoplasm to the cell exterior.6,9
Pharmacokinetics and Pharmacodynamics of Levofloxacin
Pharmacokinetic profile of levofloxacin
The pharmacokinetic properties of levofloxacin in humans have been extensively studied and are reviewed in detail elsewhere.10–12 Pharmacokinetic parameters for levofloxacin 500 mg and 750 mg, following oral (po) or intravenous (IV) administration of single or multiple doses, are summarized in Table 2.13–17
Mean pharmacokinetic parameters of levofloxacin in healthy adult subjects.
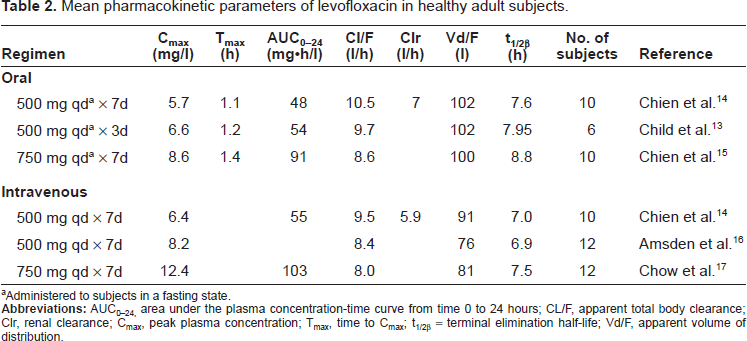
Administered to subjects in a fasting state.
There is rapid and virtually complete absorption following oral administration of levofloxacin (bioavailability ≥ 99%). 14 This absorption is not considered to be affected by food. 18 Linear pharmacokinetics have been observed with both oral and IV levofloxacin (e.g. maximum plasma concentration [Cmax] and area under the concentration-time curve [AUC] increase in a dose-proportional manner). Given its relatively long half-life (7-9 hours) and plasma concentration curve, concentrations achieved with once daily doses of levofloxacin (in patients with normal renal function) provide sufficient AUC to achieve AUC: MIC (minimal inhibitory concentration) ratios associated with clinical cure of a broad spectrum of bacteria.
Children, especially those younger than 5 years of age, have markedly different pharmacokinetics than adults. 19 Young children may eliminate levofloxacin nearly 2-fold more quickly than adults; therefore, extrapolating dose regimens from adults to children based on weight may result in drug exposure in children well below pharmacodynamic targets that have been associated with efficacy.
Systemic exposure with oral therapy is similar to that of IV therapy when identical levofloxacin doses are given (Table 2).15,17 Thus, patients can be treated with oral levofloxacin with the same dose regimen that they were receiving via the IV route. This has made transitioning from IV to oral therapy relatively easy, less prone to dosing errors, and done with considerable clinical confidence when a patient's condition improves to the extent that oral therapy can replace IV therapy.
Distribution into body tissues and fluids is extensive with levofloxacin (i.e. concentrations similar to, or substantially higher than, those observed in plasma). Relevant to the treatment of respiratory tract infections, levofloxacin concentrations in epithelial lining fluid, bronchial mucosa, and lung tissues were ~2- to 5-fold higher than those concurrently measured in plasma.20–24 Mean peak (at 4 hours) levofloxacin levels in alveolar macrophages ranged from 84 to 98 μg/ml in subjects given levofloxacin 500 mg orally or IV once daily for 5 days and from 82 to 105 μg/ml in those given 750 mg orally or IV once daily for 5 days.22,23 These levofloxacin concentrations in alveolar macrophages exceeded concurrently measured mean plasma concentrations by 9- to 19-fold. These findings in healthy volunteers indicate that levofloxacin penetrates well into the extracellular and intracellular compartments of the lung. Even higher intrapulmonary concentrations may be achievable in patients with lower respiratory tract infection. 25
Distribution of levofloxacin to other sites has also been documented, supporting its role in treating infections such as acute maxillary sinusitis, urinary tract infections, and chronic prostatitis. After a single 500 mg oral dose, the paranasal sinus mucosa to plasma ratio was 2.6 at the time of maximum plasma concentration (Tmax) in 15 patients with acute bacterial sinusitis. 26 In a related study, Ambrose et al. inserted an indwelling catheter into the maxillary sinus of 17 patients who received levofloxacin 750 mg once daily for 5 days for acute maxillary sinusitis. The median sinus to plasma penetration ratio was 1.1 based on sinus aspirate and blood samples collected at multiple time points on days 2 to 4. 27 Likely relevant to all infections, extensive accumulation of levofloxacin by phagocytes (polymorphonuclear [PMN] leukocyte/plasma Cmax ratio and PMN/plasma AUC ratio of 8) may promote drug delivery to extravascular sites of infection, 28 and may explain, at least in part, the effectiveness of levofloxacin against atypical pathogens.
Levofloxacin undergoes minimal metabolism and is primarily excreted unchanged (i.e. as active drug) in urine, achieving levels of active drug in urine that far exceed those in plasma. This excretion of active drug into urine suggests that levofloxacin would be well suited as a treatment of urinary tract infections (clinical data summarized later in this paper). In a study of 10 healthy subjects by Stein et al., mean peak/trough urine concentrations in samples collected from 0-1.5 hours (peak) and 12-24 hours (trough) post-dose were 210/31 mg/l, 347/92 mg/l, and 620/174 mg/l and mean urine AUC0-24s were 2216, 4351, and 7328 mg·h/l following single 250 mg, 500 mg, and 750 mg levofloxacin doses, respectively. By way of comparison, mean Cmax values in serum were 2.4, 5.1, and 7.6 mg/l for the respective doses. 29
A conservative view of penetration into (non-inflamed) prostatic tissue comes from a population-based pharmacokinetic analysis of data collected from 22 men who received levofloxacin 500 mg every 24 hours for the 2 pre-operative days and once on the day of prostatectomy (at either 0-0.5, 3.75-4.25, 7.5-8.5, or 22-24 hours prior to the procedure based on random assignment). 30 The drug penetration ratio (prostate to plasma) was 2.96 when calculated from the mean parameters and 4.1 from the mean of a 1,000 subject Monte Carlo simulation. More than 70% of patients had a penetration ratio exceeding 1. Even better penetration is anticipated for patients with inflammation associated with prostatitis.
Pharmacodynamic considerations
Pharmacodynamic targets have been derived in human studies, as well as in a number of supportive hypothesis-generating in vitro studies and animal infection models. 31 The use of pharmacodynamic principles should allow clinicians to select antibiotic dosages that maximize clinical efficacy of anti-infective agents and limit the development of resistance.32,33
Fluoroquinolones exhibit concentration-dependent (time independent) killing. Models that correlate clinical and microbiological responses with pharmacokinetic factors in plasma suggest that AUC to MIC (and in some studies, Cmax to MIC) is the pharmacodynamic parameter that best predicts the therapeutic efficacy of fluoroquinolones.34–37
Non-clinical and in vitro pharmacodynamic models have been used to identify drug exposure levels that are expected to be efficacious and minimize the emergence of resistance. This work has served as the basis for the continued refinement of dose regimens of levofloxacin, especially as they relate to the treatment of respiratory tract infections where large numbers of organisms in infected tissues may present the greatest risk for resistance selection. Early studies, using 500 mg dose exposure in an in vitro model, showed that this exposure would likely provide bactericidal activity against multidrug-resistant S. pneumoniae (i.e. resistant to penicillin, macrolide, clindamycin, trimethoprim-sulfamethoxazole, tetracycline, and second-generation cephalosporins) without re-growth over 48 hours. 38 Subsequent work, also in an in vitro model, showed that while 500 mg and 750 mg doses of levofloxacin were comparable with regard to providing bactericidal activity against S. pneumoniae strains that were intermediately resistant to ciprofloxacin, the 750 mg dose was needed to avoid re-growth of bacteria over time (Figure 1). 39 Consistent with this simple model that assesses lysis of bacteria in vitro, Jumbe et al. showed in a murine model of Pseudomonas aeruginosa infection, that high exposure to levofloxacin (fAUC0-24:MIC ≥ 110) was associated with prevention of in vivo amplification of preexisting mutant subpopulations of bacteria. 4 This threshold was ~2-fold higher than that required for 99% reduction of bacterial burden and therefore supported the concept that exceeding exposure associated with bactericidal activity may have the added benefit of reducing the emergence of resistant bacteria. Subsequent work31,40 supports that this concept is likely to be relevant in treating infections due to S. pneumoniae.
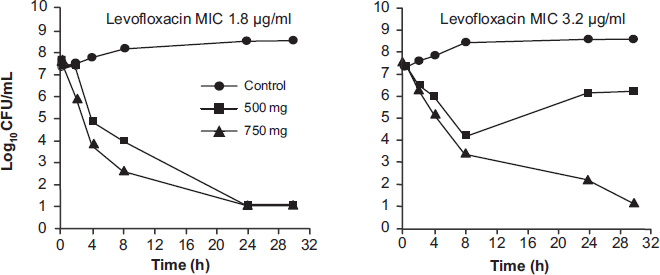
Bactericidal activity of levofloxacin against ciprofloxacin-nonsusceptible S. pneumoniae (reprinted with permission). 39
Taken together, this body of work suggests that levofloxacin exposures needed to inhibit the emergence of resistant organisms are higher than those associated with favorable clinical and microbiological outcomes. Although treatment of patients with dose regimens that provide exposure similar to that achieved with a 500 mg dose will be effective, higher drug exposure that is likely to be achieved with a 750 mg dosing regimen is predicted to be more effective in reducing the selection of resistance. It was with this background that clinical studies, aimed at assessing the efficacy and tolerability of these higher (750 mg) dose regimens over shorter periods of time (another variable) associated with lower selection of resistance, 4 were undertaken.
Efficacy in Clinical Trials
Respiratory tract infections
The use of levofloxacin as a treatment for patients with respiratory tract infections is supported by its excellent penetration to the site of these infections, as well as the drug's consistent activity against the leading causes of bacterial (both typical and atypical pathogens) infections involving the upper and lower respiratory tract. In vitro susceptibility surveillance of respiratory tract bacterial isolates over the past decade has documented that susceptibility of these pathogens to levofloxacin has remained relatively constant. Data from 2008 and the TRUST surveillance program in the U.S. showed the MIC90 of levofloxacin against 2,858 unique S. pneumoniae isolates collected from patient specimens is currently 1 μg/ml, resulting in >99% of isolates currently susceptible (Table 1). In the U.S. focused surveillance, the antimicrobial activity of levofloxacin is unaffected by source (respiratory tract or otherwise), geographic location, or age of patients from which the specimen was collected, resistance to other drug classes (β-lactam [penicillin], macrolide), or multi-drug resistance (MDR) phenotype.
Levofloxacin susceptibility rates for Haemophilus influenzae (>99%) and Moraxella catarrhalis (100%) also remain high in the U.S. Reports of fluoroquinolone resistance in Haemophilus species occurring in patients that have been hospitalized for long periods and/or treated with multiple courses of fluoroquinolones occurred in the late 1990s, but seemed to be confined to well-defined geographical locations.41–44 A report of a fatal infection due to a well characterized H. influenzae biotype II was described in Spain in 2003. 45 To date, however, resistance among H. influenzae has not appeared to have spread, and the percent susceptibility has been reported to be above 99% in most large surveillance studies.
Studies of levofloxacin in adult patients with community-acquired pneumonia.
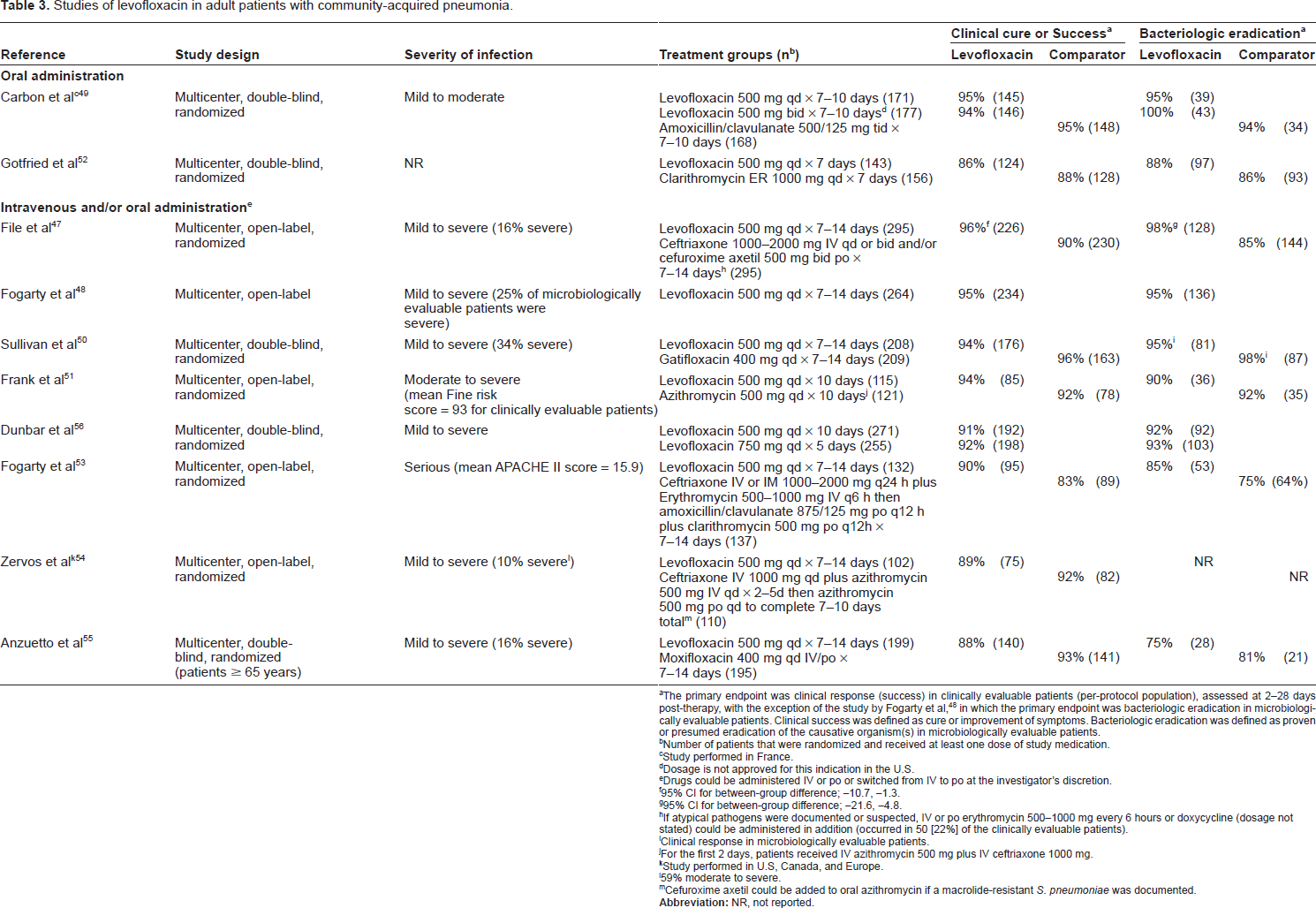
The primary endpoint was clinical response (success) in clinically evaluable patients (per-protocol population), assessed at 2-28 days post-therapy, with the exception of the study by Fogarty et al, 48 in which the primary endpoint was bacteriologic eradication in microbiologically evaluable patients. Clinical success was defined as cure or improvement of symptoms. Bacteriologic eradication was defined as proven or presumed eradication of the causative organism(s) in microbiologically evaluable patients.
Number of patients that were randomized and received at least one dose of study medication.
Study performed in France.
Dosage is not approved for this indication in the U.S.
Drugs could be administered IV or po or switched from IV to po at the investigator's discretion.
95% CI for between-group difference; −10.7, −1.3.
95% CI for between-group difference; −21.6, −4.8.
If atypical pathogens were documented or suspected, IV or po erythromycin 500-1000 mg every 6 hours or doxycycline (dosage not stated) could be administered in addition (occurred in 50 [22%] of the clinically evaluable patients).
Clinical response in microbiologically evaluable patients.
For the first 2 days, patients received IV azithromycin 500 mg plus IV ceftriaxone 1000 mg.
Study performed in U.S, Canada, and Europe.
59% moderate to severe.
Cefuroxime axetil could be added to oral azithromycin if a macrolide-resistant S. pneumoniae was documented.
Clinical efficacy of levofloxacin in patients with acute bacterial sinusitis.
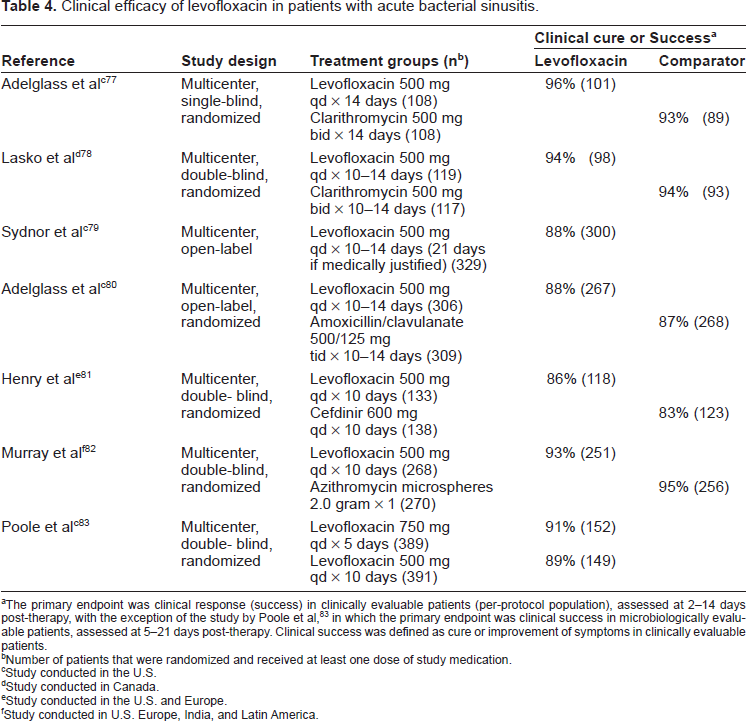
The primary endpoint was clinical response (success) in clinically evaluable patients (per-protocol population), assessed at 2-14 days post-therapy, with the exception of the study by Poole et al, 83 in which the primary endpoint was clinical success in microbiologically evaluable patients, assessed at 5-21 days post-therapy. Clinical success was defined as cure or improvement of symptoms in clinically evaluable patients.
Number of patients that were randomized and received at least one dose of study medication.
Study conducted in the U.S.
Study conducted in Canada.
Study conducted in the U.S. and Europe.
Study conducted in U.S. Europe, India, and Latin America.
Bacteriologic eradication a by pathogen in patients with acute bacterial sinusitis.83
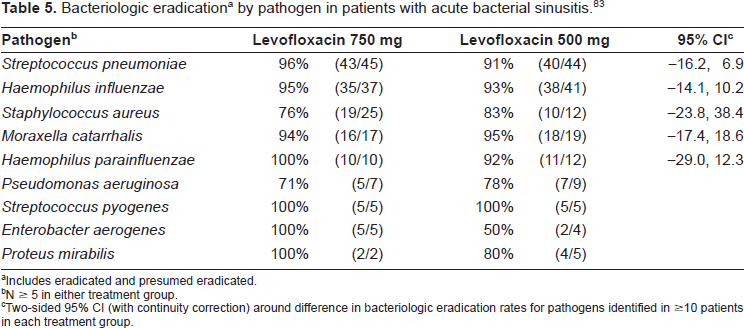
Includes eradicated and presumed eradicated.
N ≥ 5 in either treatment group.
Two-sided 95% CI (with continuity correction) around difference in bacteriologic eradication rates for pathogens identified in ≥ 10 patients in each treatment group.
Efficacy of levofloxacin in patients with acute exacerbations of chronic bronchitis.
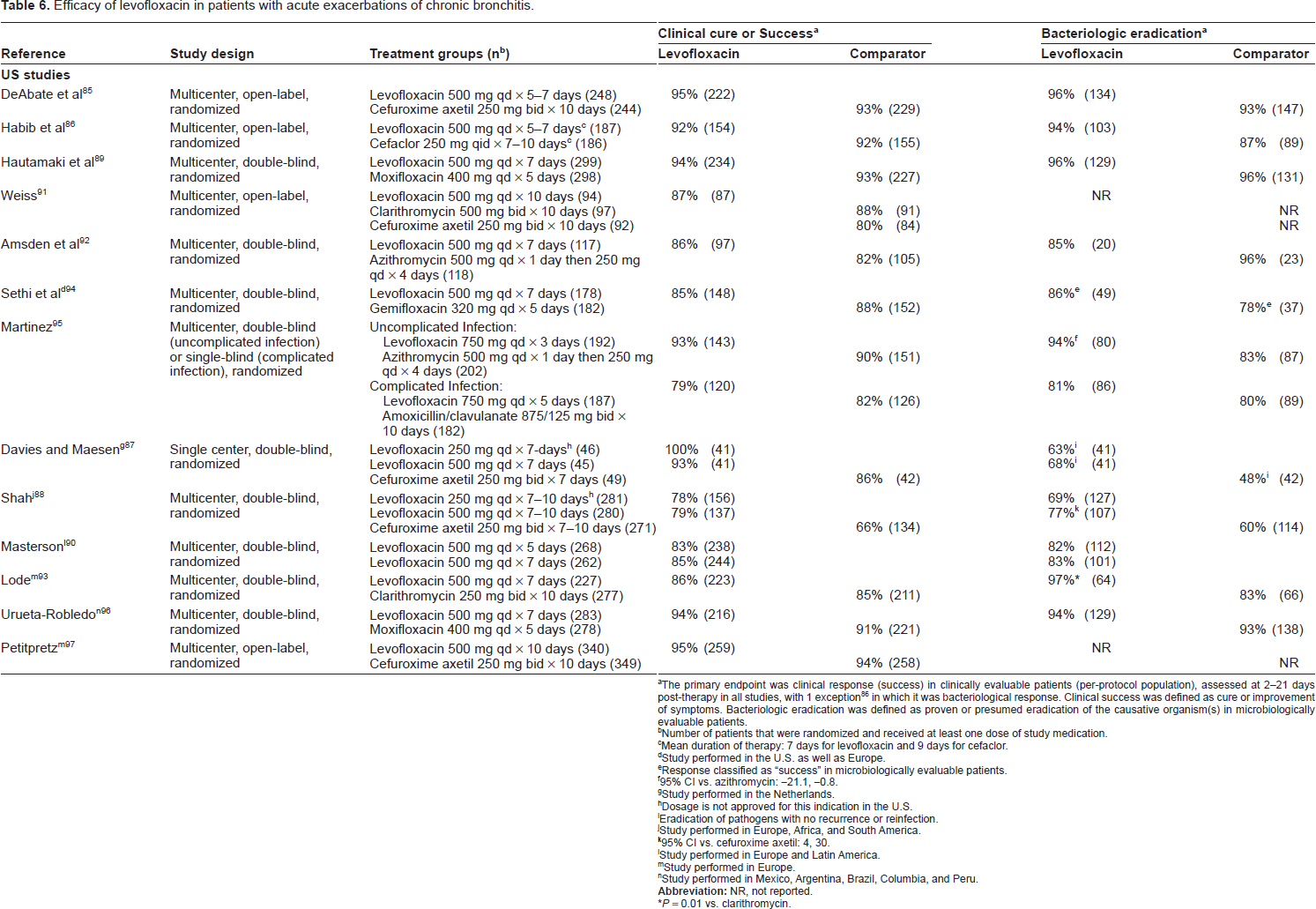
The primary endpoint was clinical response (success) in clinically evaluable patients (per-protocol population), assessed at 2-21 days post-therapy in all studies, with 1 exception 86 in which it was bacteriological response. Clinical success was defined as cure or improvement of symptoms. Bacteriologic eradication was defined as proven or presumed eradication of the causative organism(s) in microbiologically evaluable patients.
Number of patients that were randomized and received at least one dose of study medication.
Mean duration of therapy: 7 days for levofloxacin and 9 days for cefaclor.
Study performed in the U.S. as well as Europe.
Response classified as “success” in microbiologically evaluable patients.
95% CI vs. azithromycin: −21.1, −0.8.
Study performed in the Netherlands.
Dosage is not approved for this indication in the U.S.
Eradication of pathogens with no recurrence or reinfection.
Study performed in Europe, Africa, and South America.
95% CI vs. cefuroxime axetil: 4, 30.
Study performed in Europe and Latin America.
Study performed in Europe.
Study performed in Mexico, Argentina, Brazil, Columbia, and Peru.
P = 0.01 vs. clarithromycin.
Community-acquired pneumonia (CAP)
There is a large evidence base for the use of levofloxacin in the treatment of community-acquired pneumonia, leading experts to recommend it as first-line treatment for patients with comorbidities (e.g. diabetes, chronic lung disease), who have used an antimicrobial agent within the previous 3 months, have other risk factors for drug-resistant pneumococcal infection, or are being treated in an inpatient setting, either on the general ward or in an intensive care unit. 46 Levofloxacin has been evaluated in numerous clinical studies in patients with mild-to-severe CAP, initially administered at a daily dose of 500 mg for 7-14 days (Table 3).47–55 Recently, based on the pharmacokinetic and pharmacodynamic principles discussed above, it has been studied at a daily dose of 750 mg for 5 days (Table 3). 56 In these studies, levofloxacin was shown to be at least non-inferior, and in some trials superior, to the comparator regimen, including β-lactams (i.e. amoxicillin/clavulanate, cefuroxime axetil, ceftriaxone), macrolides/azalides (clarithromycin, azithromycin), and fluoroquinolones (gatifloxacin) based on clinical and microbiological endpoints. In a head-to-head comparison of the 500 mg and 750 mg daily doses of levofloxacin, the 2 regimens were shown comparable (clinical success and bacteriological eradication rates ≥91%). 56
Post hoc analyses of the data collected in these trials have provided additional information regarding outcomes in levofloxacin-treated patients. An analysis focused on pneumococcal bacteremia, 57 which involved 9 trials and 108 levofloxacin-treated patients, showed that the clinical success occurred in 98 (91%). This included success in 5 of 6 patients infected with penicillin-resistant isolates and 6 of 6 infected with macrolide-resistant isolates. The <1% mortality rate was lower than expected based on historical experience.
Response rates for levofloxacin were evaluated in patients with severe infections (Table 3). Fogarty et al. determined a bacteriologic eradication rate of 94% for patients with mild-to-moderate infections and 97% for patients with severe infections (25% of the total, including bacteremia, hypotension in the absence of volume depletion, altered mental status, baseline respiratory rate ≥30/minute, or intubation/mechanical ventilation). 48 In another study published by the same author, monotherapy with levofloxacin was comparable to the combination of a β-lactam and macrolide in patients with serious CAP (mean APACHE II score = 15.9) who required hospitalization or had remarkable derangements in vital signs. 53 In a study by Querol-Ribelles et al. of patients with CAP requiring hospitalization, there was a 60% reduced likelihood of death (OR = 0.39) with levofloxacin, as compared to ceftriaxone plus clarithromycin, after adjustments for infection severity. 58
The high overall bacterial eradication rate of levofloxacin in patients with CAP is reflected in high eradication rates (generally >90%) for the most common infecting pathogens. By way of example, in the largest study, all strains of H. influenzae, S. pneumoniae (from sputum and blood cultures), M. catarrhalis, and S. aureus isolated from microbiologically evaluable patients were eradicated with levofloxacin. 47 In this experience, levofloxacin-treatment was shown to have better eradication rates compared to ceftriaxone and/or cefuroxime axetil treatment in patients with infections caused by H. influenzae (100% vs. 79%; 95% confidence interval around difference −39.2, −2.5). Furthermore, the utility of levofloxacin for treatment of CAP is underscored by its efficacy against antibiotic-resistant pathogens. In CAP caused by P. aeruginosa, the bacteriologic eradication and clinical success rates were 79% and 90%, respectively, across levofloxacin studies. 59 Consistent with expectations based on in vitro susceptibility testing (Table 1), high microbiological and clinical response rates (97%) have been observed in levofloxacin-treated patients with pneumonia caused by macrolide-resistant pneumococci. 60 These findings may be important to consider as the prevalence of isolates resistant to macrolides appears to increase (PROTEKT study).61–63
Efficacy observed with levofloxacin in a large (n > 1,700), “real-world” population of patients with a pneumonia diagnosis based on clinical and radiological findings have been reported to be associated with a 96% bacteriological eradication rate and 94% clinical success rate. 64 A similar clinical cure rate (94%; 382/405) was reported in an open label, comparative trial involving children (6 months to 16 years) given a levofloxacin dose regimen of 10 mg/kg twice daily for children less than 5 years and 10 mg/kg once daily (to a maximum dose of 500 mg once daily) for children 5 years or older. Levofloxacin is not approved for use in children with CAP, largely because of considerations of risk/benefit related to the well-described effects of fluoroquinolones on cartilage of juvenile laboratory animals and the observations made regarding differences in the incidence of reported musculoskeletal adverse disorders observed in a large, prospective, comparative clinical experience. 65
Based, in part, on the results of the clinical study of levofloxacin 750 mg administered for 5 days (summarized below), current treatment guidelines recommend that patients with CAP be treated for a minimum of 5 days, and until they are afebrile for 48 to 72 hours and have no more than 1 CAP-associated sign of clinical instability. 46 In this context, short-course treatment, when clinically feasible, is preferred based on improved compliance, fewer adverse effects, and the theoretical consideration that these short-course regimens may be less likely to select for drug resistance. 66 In a multicenter, double-blind study in which subjects were randomized to treatment by study center and severity of infection (Pneumonia Severity Index [PSI] score), Dunbar et al. 56 showed 750 mg, 5-day treatment with levofloxacin to be comparable to traditional (500 mg), 10-day treatment with the fluoroquinolone in patients with CAP, ranging from mild to severe (i.e. PSI score > 70, Fine class IV–V). The clinical success rate, which was assessed 7 to 14 days following the completion of treatment, was 92% (of 198 clinically evaluable patients) for the 5-day treatment group and 91% (of 192) for the 10-day treatment group (95% CI −7.0, 4.4); response rates were similar across severity of illness strata. In multivariate analysis adjusting for baseline severity of infection, clinical success rates in patients aged 65 years and older were also comparable for the 5- and 10-day groups. 67 Consistent with the clinical results, bacteriologic eradication rates were 93% and 92% for the respective groups. 56
Although overall clinical response rates were high and comparable between treatment groups in the 5- day to 10-day study of levofloxacin, patients in the short-course group had earlier defervescence, as evidenced by a higher proportion of afebrile patients by day 3 (P = 0.006 vs. 500 mg, 10-day group) as well as trends suggesting more rapid resolution of sputum purulence (P = 0.059) and shortness of breath (P = 0.132), especially among the more severely ill CAP patients (P < 0.01 for fever and purulent sputum production). 68 This led to an earlier switch from parenteral to oral treatment—after an average of 2.7 of the 5 days and 3.0 of the 10 days. 68 It is noteworthy that symptoms of levofloxacin-treated patients continued to improve over the course of the study, 69 since it has been shown that the majority of CAP patients remain symptomatic for several weeks after completion of antibiotic therapy. 70 In a multivariate logistic regression model of data from a large observational study (n = 535), treatment with levofloxacin (relative risk [RR] 1.7, P = 0.010), younger age, absence of asthma, and absence of chronic obstructive pulmonary disease (COPD) were identified as significant, independent predictors of complete symptom resolution in patients with CAP. 70
Levofloxacin has been shown to be an effective treatment for atypical bacterial causes of CAP. Experience across 6 clinical studies (1,997 total patients) provided data for 75 patients with infection caused by Legionella, as confirmed by seroconversion, positive urinary antigen test result, or isolation from a pretreatment sputum culture. 71 Co-infection with Mycoplasma pneumoniae (25% of patients) and Chlamydophila (formerly Chlamydia) pneumoniae (17%) was established by seroconversion. The clinical success rate was 93%, and none of the patients had a documented microbiological relapse (evaluated 3-5 weeks post-treatment). Others have also reported favorable outcomes in patients with Legionella pneumonia who were treated with levofloxacin, including significantly faster time to clinical stability (defined by multiple vital sign thresholds, normalization of mental status, and ability to eat) (3 vs. 5 days, P = 0.002), 72 and shorter length of hospital stay (mean of ~5 fewer days), as compared to macrolides.72,73 Adding to these findings was a subgroup analysis of the Dunbar et al. study described above, 56 in which the impact of short-course treatment was assessed. 74 High response rates were observed in CAP patients infected with an atypical pathogen who were treated with levofloxacin 750 mg for 5 days: 96% of 66 patients vs. 97% of 57 patients treated for 10 days. Clinical response was sustained (≥98% for each regimen) at long-term follow-up (~30 days post-treatment).
Hospital-acquired/nosocomial pneumonia
Current treatment guidelines recommend levofloxacin, among selected other agents, as monotherapy in patients with early-onset infection and without risk factors for multi-drug resistant pathogens, the presence of which should prompt combination therapy (levofloxacin plus another antipseudomonal agent [cephalosporin, carbapenem, or β-lactam/β-lactamase inhibitor], and vancomycin or linezolid if MRSA is suspected). 75 Given concerns about evolving antibiotic resistance patterns in the hospital setting, the future value of all antimicrobial agents, including levofloxacin, for treating serious infections caused by resistant Gram-negative organisms must be carefully considered.
A multicenter, open-label study has been conducted in which 438 adult patients with nosocomial pneumonia were randomized to levofloxacin 750 mg IV for a minimum of 24 hours then po or imipenem/cilastatin 500 mg to 1000 mg IV every 6 to 8 hours, followed by oral ciprofloxacin 750 mg every 12 hours for 7 to 15 days. 3 Adjunctive therapy was initiated in patients with documented or suspected P. aeruginosa (i.e. ceftazidime or another non-carbapenem β-lactam added to levofloxacin and aminoglycoside added to the comparator) or MRSA infection (vancomycin). At study entry, 71% of patients were receiving mechanical ventilation (ventilator-associated pneumonia [VAP] results reported separately by Shorr et al. 76 and summarized below), and mean APACHE II score was 14.9. Mean length of hospital stay prior to the diagnosis of hospital-acquired pneumonia (HAP) was 9.5 days and 8.2 days for patients randomized to levofloxacin and the comparator regimen, respectively. In patients evaluable for microbiologic efficacy, clinical success was achieved in 58% (54/93) of patients who received levofloxacin and 61% (57/94) of patients who received the comparator regimen (95% CI, −12.0 to 17.2) and bacteriologic eradication was achieved in 67% (62/93) and 61% (57/94) of patients in the levofloxacin and comparator groups, respectively (95% CI, −20.3 to 8.3). Clinical response rates for the respective treatment groups were 65% and 41% for patients with confirmed P. aeruginosa infection (n = 34) and 62% and 79% in patients with confirmed S. aureus infection (n = 40).
The study population included 222 patients with VAP, with half of each randomized to levofloxacin and imipenem/cilastatin. 76 The treatment groups were similar with respect to patients’ age (mean = 52.9 years), severity of illness (APACHE II score = 15.0), and duration of mechanical ventilation before the onset of VAP (mean = 8.8 days). IV therapy was administered, on average, 1 day less before being switched to oral therapy for patients who received levofloxacin (8.4 vs. 9.4 days for the comparator, P = 0.07). In the intention-to-treat population, clinical success was achieved in 59% and 63% of patients treated with levofloxacin, and imipenem-cilastatin, respectively.
Acute bacterial sinusitis
Treatment guidelines for acute bacterial sinusitis recommend a respiratory fluoroquinolone (e.g. levofloxacin) as first-line treatment of patients with mild infection who have received an antibiotic in the most recent 4-6 week period and for those with illness of moderate severity. 66 Levofloxacin has been evaluated in 7 published studies of patients with acute bacterial sinusitis (Table 4).77–83 Clinical success rates determined 2-14 days post-treatment ranged from 86% to 96% in clinically evaluable patients treated with levofloxacin 500 mg po once daily for 10-14 days. In the comparative studies, levofloxacin was similar to clarithromycin, amoxicillin/clavulanate, cefdinir, or azithromycin (investigational formulation) based on clinical response. Relapse rates were low (≤8%).77,80 Bacteriological outcome was assessed in 1 study in which there was a 92% (among 138 levofloxacin-treated patients) overall pathogen eradication rate, including eradication rates of 100%, 97%, 94%, and 93% for S. pneumoniae, H. influenzae, S. aureus, and M. catarrhalis, respectively. 79
Levofloxacin administered at a high dose over a shorter duration of treatment has also been studied in patients with acute bacterial sinusitis. In a multicenter, double-blind, randomized study, Poole et al. 83 compared traditional (500 mg), 10-day treatment to high-dose (750 mg)-short-course, 5-day treatment with levofloxacin in culture-positive patients with clinical and radiologic evidence of acute bacterial sinusitis. Clinical success at day 17-24 was observed in 89% (of 149) and 91% (of 152) of microbiologically evaluable patients (the primary analysis population) in the 10-day and 5-day treatment groups, respectively (95% CI −10.0, 4.2). Bacteriologic eradication rates exceeded 90% in both treatment groups for the most common pathogens causing acute sinusitis (Table 5). 83 At the long-term follow-up visit (day 35-45), the rates were 95% and 94% in the respective treatment groups. 83
Support for rapid killing with the 750 mg dose in acute bacterial sinusitis was demonstrated in analyses showing that pathogens (isolated from sinus aspirate samples obtained from an indwelling sinus catheter) were rapidly eradicated after 1 to at most 3 days. 84 Consistent with this observation, Ambrose et al. reported that the median time to maxillary sinus sterilization was 1 day following the initiation of levofloxacin 750 mg dosing. 27
Acute bacterial exacerbation of chronic bronchitis (ABECB)
In studies of acute bacterial exacerbation of chronic bronchitis, levofloxacin, at a daily dose of either 500 mg or 750 mg, was shown to have clinical and microbiological response rates similar to those with a β-lactam, macrolide/azalide, or another fluoroquinolone (Table 6).85–97 In some studies, the eradication rate was higher with levofloxacin than the comparator agent in patients infected with H. influenzae or S. pneumoniae.86,88 Across the studies, no additional clinical benefit was observed with 7-10 days treatment beyond that seen with 5 days89,90,92,94,96 and response with the shorter treatment course was not affected by patient age, frequency of exacerbations, or comorbidities. 90
Treatment with levofloxacin has been shown to reduce need for hospitalizing chronic bronchitis patients during treatment of their acute bacterial exacerbation (P = 0.02 vs. standard therapy comprised of clarithromycin, cefuroxime, or amoxicillin/clavulanate) 98 and prolong the post-treatment infection-free interval.93,97
In a study of short-course treatment of acute bacterial exacerbation in patients with chronic bronchitis, after stratifying patients by severity of underlying disease, Martinez et al. randomized uncomplicated patients to levofloxacin 750 mg once daily for 3 days or azithromycin once daily for 5 days and complicated patients to levofloxacin 750 mg once for 5 days or amoxicillin/clavulanate 875/125 mg twice daily for 10 days. 95 The clinical response rates for the study antibiotics within each disease severity sub-group were similar to one another (Table 6). For microbiologically confirmed cases, clinical status was improved to a greater degree after 3 days of levofloxacin as compared to 5 days of azithromycin (96% versus 87% clinical success rates, respectively, 95% CI −17.6, −0.1), and the bacteriological eradication rate was significantly higher (94% vs. 83%, respectively; 95% CI −21.2, −0.8). Eradication rates with levofloxacin exceeded 90% for all isolated pathogens, the most common being H. influenzae, S. pneumoniae, H. parainfluenzae, and M. catarrhalis. Complicated patients treated with levofloxacin had earlier resolution of their symptoms (cough, sputum purulence, increased sputum production, each P < 0.05) than those treated with amoxicillin/clavulanate. 99 Early response with levofloxacin (92% at day 4) has also been observed by others. 92
Genitourinary tract infections
Urinary tract infection is the most common indication for which fluoroquinolones are prescribed. 100 Experts recommend their use as first-line agents for treating complicated urinary tract infections as well as chronic bacterial prostatitis because of their reliable activity against most of the expected uropathogens (Table 1) and their ability to achieve high concentrations in urine and urogenital tissues. In general, fluoroquinolones have emerged as preferred agents because several of the more commonly prescribed antibiotics used to treat these infections (e.g. trimethoprim-sulfamethoxazole and amoxicillin) have become substantially less active against the bacteria that cause these infections. 101
There are concerns, however, about increasing rates of fluoroquinolone resistance in Gramnegative pathogens including Escherichia coli and P. aeruginosa. Resistance rates in E. coli vary according to the source of the pathogen, with rates currently less than 2% and 10% in isolates from patients with acute pyelonephritis or complicated urinary tract infection, respectively. 102 Rates of trimethoprim-sulfamethoxazole and ampicillin resistance in urinary isolates of E. coli are currently 26% and 46%, respectively, with multi-drug resistant phenotypes increasing (TRUST 2008) and no new, potentially effective oral antibacterials for treating community-acquired urinary tract infections on the horizon. Therefore, it is imperative that strategies are implemented that may preserve the fluoroquinolone class.
Levofloxacin was initially studied at dose of 250 mg for complicated urinary tract infection and acute pyelonephritis.103,104 Even at this lower dose, high urine levels of levofloxacin were achieved, resulting in high uropathogen eradication rates: 99% (n = 93) of E. coli, 93% of Klebsiella pneumoniae (n = 28), 100% of Proteus mirabilis (n = 10), 86% of P. aeruginosa (n = 7), and 67% of Streptococcus faecalis (n = 6). 103
Given the increase in rates of resistance, a more recent study in complicated urinary tract infection or acute pyelonephritis evaluated a 5-day course of levofloxacin (750 mg IV or po once daily) to a 10-day course of ciprofloxacin (400 mg IV and or 500 mg po twice daily) 102 and demonstrated, for the first time, the ability to treat these complicated infections with shorter courses of therapy (Table 7).102–107 Post-treatment blood cultures were negative for all 10 levofloxacin-treated patients with E. coli urosepsis. Eradication of resistant pathogens (based on in vitro susceptibility testing) was observed, albeit at lower rates than for susceptible isolates. In a sub-group of 35 patients with complicated acute pyelonephritis (characterized by obstruction, retention, renal calculi, diabetes, immunocompromised status, presence of catheter, neurogenic bladder, ileal loop, or recent invasive procedure), overall pathogen eradication rates were 95% and 100% for levofloxacin- and ciprofloxacin-treated patients, respectively. 105
Studies of levofloxacin in patients with genitourinary tract infections.
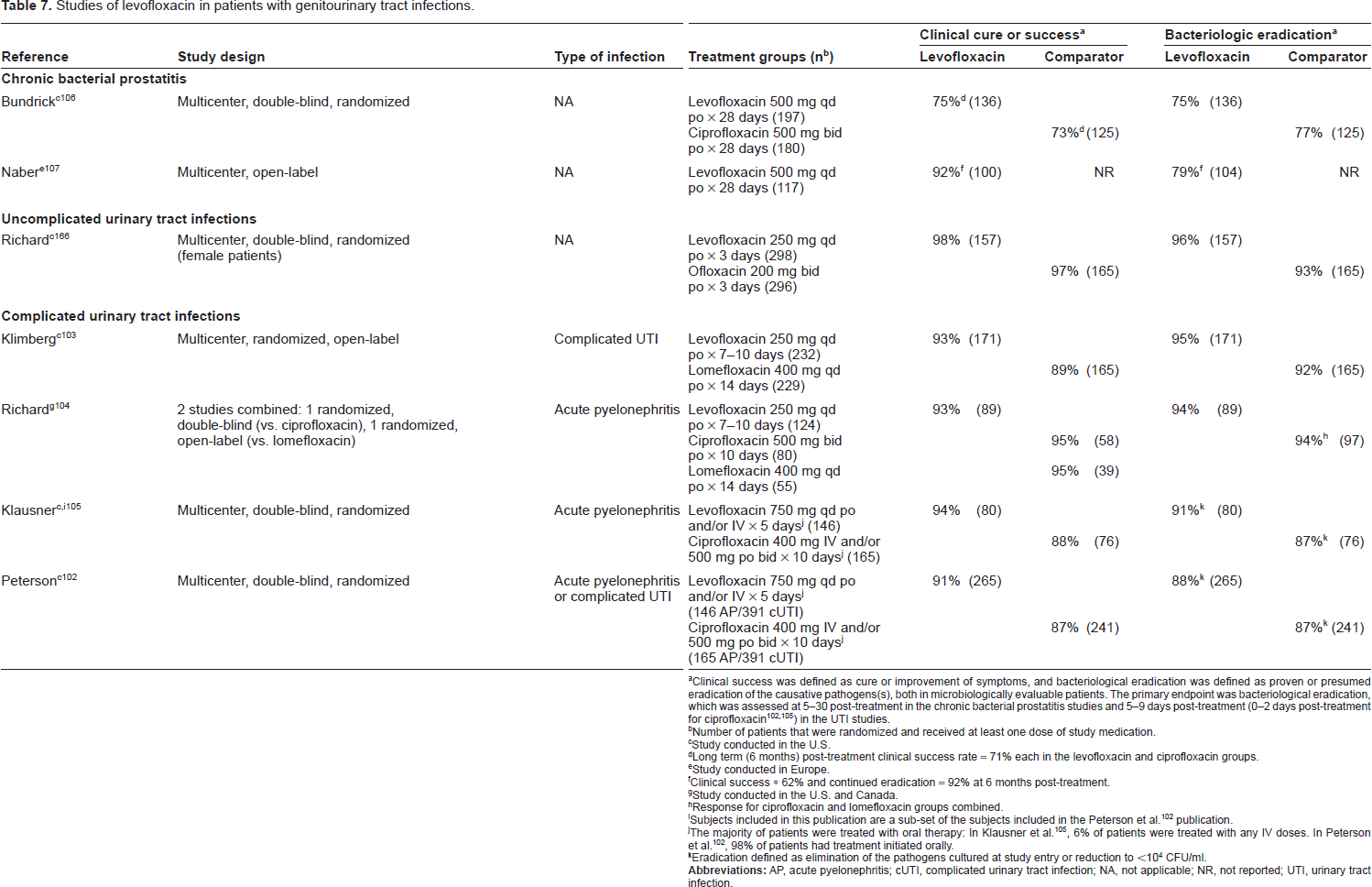
Clinical success was defined as cure or improvement of symptoms, and bacteriological eradication was defined as proven or presumed eradication of the causative pathogens(s), both in microbiologically evaluable patients. The primary endpoint was bacteriological eradication, which was assessed at 5-30 post-treatment in the chronic bacterial prostatitis studies and 5-9 days post-treatment (0-2 days post-treatment for ciprofloxacin102,105) in the UTI studies.
Number of patients that were randomized and received at least one dose of study medication.
Study conducted in the U.S.
Long term (6 months) post-treatment clinical success rate = 71% each in the levofloxacin and ciprofloxacin groups.
Study conducted in Europe.
Clinical success * 62% and continued eradication = 92% at 6 months post-treatment.
Study conducted in the U.S. and Canada.
Response for ciprofloxacin and lomefloxacin groups combined.
Subjects included in this publication are a sub-set of the subjects included in the Peterson et al. 102 publication.
The majority of patients were treated with oral therapy: In Klausner et al. 105 , 6% of patients were treated with any IV doses. In Peterson et al. 102 , 98% of patients had treatment initiated orally.
Eradication defined as elimination of the pathogens cultured at study entry or reduction to <10 4 CFU/ml.
Levofloxacin, at a daily dose of 500 mg, has been shown effective for treatment of chronic bacterial prostatitis based on clinical and microbiological outcomes (Table 7).106,107 In a study conducted by Bundrick et al. resolution or improvement of symptoms and pathogen eradication, assessed 5-18 days following the completion of treatment, were accomplished in approximately 3 of every 4 men, and clinical response was sustained, as confirmed by an evaluation made 6 months later. 106 Eradication rates with levofloxacin for the most frequently isolated pathogens were: 93% (n = 15) for E. coli, 90% for coagulase-negative staphylococci (n = 10), 83% for Staphylococcus epidermidis (n = 24) and Streptococcus mitis (n = 12), 78% for Streptococcus agalactiae (n = 18), 74% for Staphylococcus haemolyticus, and 72% for Enterococcus faecalis (n = 54). These findings are supported by a recently conducted open-label study. 107
Safety and Tolerability
Levofloxacin has a well-defined safety profile that is based on clinical trial experience as well as clinical use of the drug for over 12 years. The foundation for the safety experience with levofloxacin was laid during clinical trials (>7,000 patients) in which adverse events, laboratory testing, vital signs, and electrocardiograms (ECG) were evaluated. Across clinical trials and dosages of levofloxacin, the most frequently reported adverse events (without regard to whether they were related to treatment or not) involved the gastrointestinal (GI) tract (nausea [7% of 7,537 patients], diarrhea [5%], constipation [3%], abdominal pain, vomiting, and dyspepsia (each 2%]). 1 In the clinical studies reviewed in this paper (Tables 3–7), levofloxacin was at least as well tolerated as the non-quinolone comparators, including amoxicillin/clavulanate, imipenem/cilastatin, clarithromycin, azithromycin, ceftriaxone, cefuroxime axetil, cefdinir, and cefaclor. Of note, the incidence of GI events is similar among the fluoroquinolones and other systemic antimicrobial agents. 108
There is no evidence from levofloxacin clinical trials that the incidence of any specific adverse event is related to dose (i.e. 500 mg vs. 750 mg). 109 And, using logistic regression analyses, Preston et al. could not identify a relationship between exposure to levofloxacin (i.e. peak and trough concentrations, AUC) and incidence of treatment-emergent adverse events involving the GI tract, skin, or central nervous system (CNS) (including psychiatric disturbances). 110
Safety data for levofloxacin collected during post-marketing surveillance studies confirm the findings from the clinical studies based on type and frequency of commonly reported adverse events. 111 However, the fluoroquinolones are associated with less frequently reported, but potentially serious adverse events, some leading to restriction or discontinuation of selected agents within this class. These events include seizures, tendonitis, allergic reactions, disturbances in glucose metabolism, hepatotoxicity, peripheral neuropathy, Clostridium difficile-associated diarrhea and prolongation of the QT interval. 1 The prevalence of these reactions can differ among agents within the class; however, all currently marketed fluoroquinolones include warnings about these events.
CNS effects
Fluoroquinolones have proconvulsant properties, which depend on both their dosage and chemical structure. 112 Structural differences among the agents at position 7 of the quinolone nucleus affect their interaction with γ-aminobutyric acid (GABA) receptors in the CNS. Those with an unsubstituted piperazine (e.g. ciprofloxacin) or unsubstituted pyrrolidine moiety (tosufloxacin, clinafloxacin) are more likely to cause seizures via displacement of GABA or competition with GABA binding to receptors, leading to CNS stimulation. 113 Among the fluoroquinolones, levofloxacin has been shown to be one of the least epileptogenic, perhaps due to its weak binding affinity to the GABA receptor in the CNS. 114 The low predicted potential of levofloxacin for neurotoxicity has been confirmed by low incidence of convulsions and other CNS adverse events during treatment of humans.1,115
Tendonopathy
Arthropathy and tendon disorders have been reported in patients being treated with, or following treatment with, fluoroquinolones;65,116–120 estimates of the prevalence of these events vary. In a case-control study, the event rate of Achilles tendonitis and rupture associated with fluoroquinolones was 3.2/1000 patient treatment-years. 117 Patients older than 60 years and persons taking corticosteroids may be at increased risk of tendonopathy,117,118 Sporadic case reports also suggest that transplant patients may also be at higher risk. A case-controlled study aimed at identifying factors associated with Achilles tendon rupture with fluroquinolones 119 reported an odds ratio (adjusted for age, gender, obesity, skin and soft tissue infection, oral steroid use, injected steroid use, other non-fluoroquinolone antibiotic, arthritis, diabetes, and trauma) associated with rupture for fluoroquinolone use in the preceding 90 days to be 1.2 (95% CI 0.8-1.8). This did not appear to differ substantially from the odds ratio observed for treatment with azithromycin (1.2; 95% CI 0.9-1.6) but appeared to be considerably lower than the odds ratio observed for patients given two or more corticosteroid injections (2.4; 95% CI 1.4-4.1). In this study, the adjusted odds ratio found for levofloxacin was 0.6 (95% CI 0.3-1.4).
The mechanisms of fluoroquinolone-associated effects on connective tissue are not completely understood. The reasons for the effects on tendons and the effects on the cartilage of juvenile animals, which has led to concern about use of this class of drugs in children, may be similar.121–123 One possible mechanism involves chelation of magnesium in cartilage, resulting in impaired integrin function and disturbance of chondrocyte-extracellular matrix interaction followed by formation of reactive oxygen species, apoptosis, and tissue damage. 124
A boxed warning regarding the risk of tendonopathy was added to the labels of all fluoroquinolones in 2008. 125
Regarding effects in children, an increased incidence of musculoskeletal disorders (i.e. arthralgia, and less frequently, arthritis, tendonopathy, and gait abnormality) was observed in 1,340 patients treated with levofloxacin (6 months to 16 years of age), compared to 893 controls treated with a non-fluoroquinolone: 2.1% vs. 0.9% of patients in the respective treatment groups reported musculoskeletal disorders during the first 60 days following the first dose of antibiotic (P = 0.038, 2-sided Fisher's exact test), as did 3.4% and 1.8%, respectively (P = 0.025) during a 1-year follow-up period.1,65 Most of these musculoskeletal adverse events were characterized as being mild, and none severe, and all were self-limited, resolving without sequelae after a median of 7 days in levofloxacin-treated patients and 9 days in non-fluoroquinolone-treated patients.
Glucose homeostasis alterations
Alterations in glucose metabolism have been reported, albeit infrequently, during treatment with fluoroquinolones. Like the insulin secretagogues, they have insulinotrophic effects, caused by direct inhibition of pancreatic β-cell potassium channel activity (KATP), leading to hypoglycemia. 126 The mechanism(s) by which they cause hyperglycemia is less clear. Several research groups have evaluated the incidence of dysglycemia in patients being treated with fluoroquinolones. In a retrospective review of > 17,000 hospitalized patients, Mohr et al. documented poor glucose control (i.e. blood glucose >200 mg/dl or <50 mg/dl) within 72 hours of dosing in 0.93%, 1.01%, and 0% of inpatients treated with levofloxacin, gatifloxacin, and ciprofloxacin, respectively. 127 Concomitant sulfonylurea treatment was determined to be the only independent risk factor for fluoroquinolone-associated hypoglycemia. The relative risk of hypoglycemia (<51 mg/dl) with fluoroquinolones was assessed by Graumlich et al. in a nested case-control study of 7,287 inpatients that received levofloxacin or gatifloxacin. 128 After adjustments for significant predictors (e.g. concomitant antihyperglycemic drugs, renal failure, sepsis syndrome), the risk of hypoglycemia was 2.8 times higher after gatifloxacin than levofloxacin therapy. When studied in an outpatient setting, the prevalence of dysglycemia (~40% hyperglycemia) was 2.6% in patients treated with oral levofloxacin. 129 After adjustments for confounding factors, treatment with levofloxacin did not increase the risk of developing a dysglycemic event, whereas lack of dosage adjustment for impaired renal function (OR 10.3), presence of diabetes (OR 17.1), and treatment with insulin (OR 5.3) or sulfonylureas (OR 3.6) did. Hypoglycemia during treatment with levofloxacin has been reported in patients also taking oral antihyperglycemic agents, notably sulfonylureas,130,131 diabetics being treated with insulin, 132 as well as in patients without diabetes. 133
QTc prolongation
Delay in cardiac repolarization, as measured by prolongation of the QTc interval on ECG and, in rare cases, torsade de pointes, a potentially life-threatening arrhythmia, have been reported with fluoroquinolones. The conduction delay and dysrhythmias are related to fluoroquinolone blockade of the rapid-acting portion of the delayed rectifier potassium channel, which is controlled by the human ether-a-go-go-related gene (HERG). Agents within the class have different antagonistic potencies for the HERG channel: Sparfloxacin and grepafloxacin have the most potent, moxifloxacin has intermediate, and levofloxacin and ciprofloxacin have the least potent inhibitory effects, 134 likely explaining different incidences of conduction delays and ventricular tachyarrhythmias reported with the fluoroquinolones. These findings were consistent with results from a double-blind, 4-period, 4-sequence crossover study in which 47 healthy volunteers were given single (supra-therapeutic) doses of placebo, levofloxacin 1000 mg, moxifloxacin 800 mg, and ciprofloxacin 1500 mg, with ECGs recorded 24 hours before and after dosing. 135 In this study, a single 800 mg dose of moxifloxacin was shown to have a greater effect on prolongation of QTc than either levofloxacin or ciprofloxacin as measured by change from baseline QTc (Bazett) at 1 hour following dose or by incidence of >30 or >60 msec change on QTc (Bazett) above baseline over a 24-hour period after treatment. Using a different design for measuring effects on QT intervals, an open-label, randomized, crossover study of multiple doses showed that levofloxacin (500 mg qd × 7 days) had no effect on QTc, whereas QTc was prolonged by 6 and 11 μsec (each P < 0.05) relative to baseline and from the 2-hour measurements, respectively, after 7 days of moxifloxacin. 136
Hepatotoxicity
The extremely low risk of liver injury with levofloxacin, as predicted based on mechanistic studies,137,138 has been confirmed in a large clinical experience in humans. Abnormal liver function, characterized by elevations in transaminase and/or alkaline phosphatase levels, was observed during routine chemistry testing in <1% of more than 7,000 patients treated with levofloxacin in clinical trials. No evidence of serious drug-related hepatotoxicity with levofloxacin was detected in clinical trials. 1 Severe liver injuries (e.g. hepatitis, necrosis, hepatic failure) in patients treated with levofloxacin occurred at a rate of less than 1 per million in worldwide and U.S. post-marketing surveillance;139,140 and have been reported rarely in the medical literature;141–144 most cases are not associated with hypersensitivity.
Allergic reactions
Allergic reactions, including both immediate- and delayed-type hypersensitivity reactions, are reported with fluoroquinolones, but occurring at a rate less than with β-lactams. 145 Immediate hypersensitivity reactions (e.g. urticaria, angioedema, anaphylaxis) appear to be primarily mediated by IgE, whereas delayed-type events (e.g. cutaneous reactions) are mediated by T-cells. 146 There appears to be substantial cross-reactivity among the fluoroquinolones.146,147
The incidence of serious allergic reactions during antibacterial use was recently assessed by Johannes et al in a large, U.S. health insurance claims database (~10 million patients, July 2000 to June 2004). 148 Events of anaphylaxis, other allergic drug reactions, or cardiopulmonary resuscitation occurring in an emergency department or inpatient setting within 2 weeks after outpatient dispensing were identified in cohorts exposed to levofloxacin and other agents. The investigators documented a 0.1% incidence of anaphylaxis attributed to levofloxacin based on review of medical records by a healthcare professional. In a related study, according to estimates from a national, stratified probability sample, the annual rate of emergency department visits due to mild allergic reactions (e.g. dermatitis, drug eruption, erythema, pruritus, rash) was 2.8 per 10,000 (0.03%) outpatient prescription visits for fluoroquinolones (i.e. ambulatory care visits during which the agent was prescribed). 149
Salvo et al. evaluated allergic reactions associated with oral drug use in a large database of spontaneous adverse drug reaction reports. 150 An independent panel of experts inspected all reports and identified “cases” based on: reports described as anaphylactic shock or anaphylactoid reaction; events with onset consistent with allergic reaction involving at least 2 systems/organs from skin and appendages, respiratory, gastrointestinal, CNS, and cardiovascular; or, cutaneous reactions with an onset consistent with an allergic origin. Anti-infective agents (reporting odds ratio [ROR] as compared to non-allergic reports = 2.92) and musculoskeletal agents (plus aspirin) (ROR 1.65) were the only drug classes significantly associated with allergic reactions, accounting for 70% of the total cases. Levofloxacin (ROR 2.0) was among the frequently prescribed anti-infective agents with the lowest risk of allergic reactions.
Clostridium difficile diarrhea
Clostridium difficile is a spore-forming, toxin-producing gram-positive rod that is responsible for 15%-25% of antibiotic-associated cases of diarrhea. 151 Treatment with antibacterial agents alters the balance in normal colonic flora. After disruption of microflora within the colon, some antibiotic-treated patients (depending on immune status, number and severity of underlying disease, age, and other host factors) become infected with C. difficile. Exotoxins (A and/or B) produced by pathogenic strains of C. difficile contribute to the development of a broad spectrum of associated adverse clinical effects, ranging from mild diarrhea to life-threatening colitis. Strains producing excessive amounts of toxin are often refractory to antimicrobial treatment (metronidazole, oral vancomycin) and increase morbidity and mortality among affected patients, some ultimately requiring colectomy.151–153
There is accumulating data suggesting that fluoroquinolones are an important risk factor for C. difficile-associated diarrhea. 154 During the development of levofloxacin, the incidence of pseudomembranous/C. difficile colitis was approximately 0.06%. 1
Peripheral neuropathy
Sensory or sensorimotor axonal polyneuropathy affecting both small and large axons has been reported in a minority of patients during treatment with fluoroquinolones, resulting in paresthesias, hypoesthesias, dysesthesias, and weakness. 1 Peripheral nervous system effects may be severe and long lasting. 155
Photosensitivity/phototoxicity
During treatment with a fluoroquinolone, patients may experience photosensitivity or phototoxicity, manifested by an exaggerated sunburn reaction in locations exposed to sunlight or artificial ultraviolet light. Exposure to light produces reactive oxygen species that trigger prostaglandin release, inflammation, and tissue damage. 156 Risk of phototoxicity is related to chemical structure, being highest for agents with a halogen substituent at position 8 of the quinolone nucleus (e.g. lomefloxacin, sparfloxacin). 157 Using a mouse model, Hayashi et al. observed the lowest phototoxic potential for levofloxacin, ciprofloxacin, and norfloxacin among a series of 10 fluoroquinolones tested. 158 In phase 2 and 3 clinical trials, the incidence of phototoxicity was 0.03% (1/3,460) among levofloxacin-treated patients. 116
Patient Focused Perspectives
Levofloxacin has been shown to improve patient satisfaction and reduce not only the time from parenteral to oral (switch) treatment, but also the length of hospital stay (as previously described) and cost of care in studies of pneumonia. In lower-risk pneumonia patients (i.e. PSI risk class II or III, score ≤ 90) treated with levofloxacin po or IV/po, comparable successful outcomes were achieved and more of the patients treated on an outpatient basis expressed satisfaction with their overall care (91% vs. 79% of inpatients). 159 When the decision is made to hospitalize CAP patients for treatment, Marrie and coworkers found that those treated with levofloxacin based on practice guidelines (vs. conventional management) required 1.7 fewer days (P = 0.01) of parenteral therapy, 1.7 fewer inpatient (bed) days (P = 0.04), 160 and fewer inpatient medical resources, resulting in cost savings of between $457 and $994 per patient. 161 In a study of 495 CAP patients admitted to 1 of 6 community hospitals for treatment, Makos et al. documented significantly faster switch to oral therapy (1.2 fewer IV days, P < 0.01) and quicker discharge (0.8 days, P < 0.01) with levofloxacin 750 mg daily compared to ceftriaxone 1000 mg plus azithromycin 500 mg daily. 162 An economic analysis of data from the study by File et al. 47 came to the same conclusion—treatment with levofloxacin resulted in a shorter length of stay as well as lower hospitalization costs compared to ceftriaxone 163 and lower total outpatient costs compared to cefuroxime axetil. 164
Conclusions
The development of levofloxacin as an important addition to the fluoroquinolone class occurred during a period when advances were being made in the fields of pharmacodynamics, molecular biology, pharmacoepidemiology, toxicology, drug resistance, and health economics, which impacted the process of antibiotic drug research and development. The pharmacokinetics and antimicrobial activity of the drug stimulated initial interest in exploring its clinical value for treating patients with urinary tract and respiratory tract infections. Results of phase 2 and 3 clinical trials, which formed the basis for our early understanding of its safety and efficacy, suggested that levofloxacin would have broad clinical utility. Results of this clinical work supported continued study of the drug in treatment of serious skin infections, nosocomial pneumonia, and prostatitis. Accumulating clinical experience over the past two decades has led to the choice of levofloxacin, in some instances as preference over, and in other instances as an alternative to, established β-lactam and macrolide therapies.
Lessons learned during the last two decades predict that two factors—drug resistance and safety—are likely to shape the future utility of levofloxacin. Resistance to antimicrobials appears to reflect an unrelenting force of nature that may ultimately render even the most potent antibacterial useless over time. The appearance of fluoroquinolone resistance over a broad spectrum of organisms has occurred since these agents were introduced into clinical practice. Given the prominent role that the pneumococcus plays in respiratory tract infections, the appearance of levofloxacin-resistant strains in the 1990s was considered to foreshadow an end to the drug's clinical value for pneumonia. However, a clear rise in the prevalence of levofloxacin-resistant S. pneumoniae was not subsequently observed. Likewise, there have been no changes in the prevalence of resistance among other important bacterial causes of community-acquired respiratory tract infections (e.g. H. influenzae, M. catarrhalis, Legionella pneumophila, M. pneumoniae). In contrast, resistance to fluoroquinolones in general, and to levofloxacin specifically, has continued to rise in Enterobacteriaciae. Although this pattern suggests that future use of levofloxacin may be best directed toward treating respiratory tract infections, rather than urinary tract infections, it may be that strategies aimed at slowing this development of resistance could prolong levofloxacin's value in treating infections caused by Gram-negative pathogens. A higher dose (750 mg), short-course (3-5 days) levofloxacin regimen, which exceeds pharmacodynamic targets associated with effective bactericidal activity at the site of infection, not only provides effective and safe therapy but also may minimize the selection of resistance.
In the last two decades as we have continued to see the challenge of drug resistance defined, we have also witnessed remarkable growth in our understanding of the biology of, and our ability to recognize, drug-related adverse effects, especially among fluoroquinolones. Experiences that led to removal or restricted availability of several promising fluoroquinolones (e.g. sparfloxacin, lomefloxacin, trovafloxacin, gatifloxacin) taught us that even large clinical trials may not predict the complete safety and tolerability profile of new drugs. However, during this period vigilant post-marketing assessment of other agents belonging to the class (e.g. ciprofloxacin, levofloxacin) improved our understanding of this profile during use in a large and diverse patient population and, in doing so, better informed us about the risks of their use. This experience, which now includes use in hundreds of millions of patients, indicates that levofloxacin is one of the best-tolerated and safest antimicrobial agents currently available. Based on this large clinical experience, prescribing information for levofloxacin and all other fluoroquinolones includes warnings about infrequently reported, but potentially serious adverse events (e.g. seizures, tendonitis, allergic reactions, disturbances in glucose metabolism, QT prolongation).
Levofloxacin proved to be an important addition to the list of antibacterials that was still rapidly growing in the early 1990s. As the challenges of antibacterial drug development have slowed the approval of new antibacterial agents, it is likely that clinicians will need to continue to rely on and learn from agents like levofloxacin, with its well-characterized antimicrobial activity and safety profile, to best treat patients in the future.
Disclosure
The author is an employee of J & J pharmaceuticals Research and Development.
Footnotes
Acknowledgements
The author would like to acknowledge Sandra Norris, PharmD of Norris Communications Group for her writing and editorial assistance on this manuscript. The author would also like to thank Janet A. Peterson, PhD, Susan C. Nicholson, MD, and Behin Yektashenas, PharmD of Ortho-McNeil Janssen Scientific Affairs, LLC for their assistance with the preparation of this manuscript.
