Abstract
The prevalence of osteoporosis is likely to rise with the increase in life expectancy of an ageing population. Current first line therapies for the treatment of osteoporosis are predominantly anti-resorptive. Teriparatide is a first in class, anabolic agent with a unique mechanism that results in increased bone formation. Daily subcutaneous injection for 6–24 months was effective in reducing vertebral and non-vertebral fracture rates, in improving bone mineral density (BMD) and in increasing bone formation rates in postmenopausal osteoporosis, with effects persisting following treatment cessation. Similar benefits on bone mass and bone formation were seen in men with osteoporosis and glucocorticoid induced osteoporosis. Beneficial effects on bone mass have been demonstrated in treatment naive subjects treated with teriparatide alone, sequentially with anti-resorptive therapy and concomitantly with some, but not all, anti-resorptive treatments due to an early blunting of the anabolic effect. Teriparatide is generally well tolerated. However, the high treatment cost and inconvenient mode of administration has limited it's use to patients with osteoporosis who have experienced an unsatisfactory response, who are intolerant to other osteoporosis therapies, or to patients at very high risk of fracture. Teriparatide treatment is currently restricted to a total lifetime treatment dose of 18 months of daily subcutaneous therapy due to concerns from animal studies suggesting an increased risk of osteosarcoma. More safety data may permit a longer duration of treatment in the future but will necessitate prolonged human studies. Teriparatide may serve a more prominent role in the treatment of older patients who continue to fracture despite low bone turnover or sustain side effects with anti-resorptive therapy.
Keywords
Introduction
Osteoporosis is characterised by the loss of bone mass or the presence of a fragility fracture. 1 The pathogenesis of osteoporosis focuses on an imbalance between osteoclastic bone removal and osteoblastic bone formation activity. Although fragility fracture is the hallmark of osteoporosis, diagnosing osteoporosis also involves assessment of bone mineral density with dual energy X-ray absorptiometry. 1 However, many individuals with osteoporosis are not diagnosed until fragility fractures occur. Fragility fractures are associated with significant disability, mortality and cost. 2
The prevalence of osteoporosis is increasing because of increased life expectancy and an ageing population. 2 In Australia, 75,000 fragility fractures occur annually with approximately 21,000 hip fractures. 3 The average cost of hospitalisation post-hip fracture is $23,000 per individual. 3 Twelve months after a hip fracture, 20% of patients die, 50% of patients experience permanent disability and 25% of patients require permanent nursing home care. 3 In 2001, the direct costs attributable to osteoporotic fractures in Australia were estimated at $1.9 billion. 4
Patients with previous fractures have a higher risk of sustaining subsequent fractures than the non-fracture population. 2 However, available therapies can reduce the risk of recurrent fractures by 30% to 60% within a year of starting treatment. 1
Current pharmacological options for treatment of osteoporosis include antiresorptive agents (eg, bisphosphonates, calcitonin and raloxifene) which reduce osteoclastic activity, strontium ranelate which reduces osteoclastic activity and may have some anabolic properties and teriparatide, the first pure anabolic agent, which stimulates bone turnover with a positive bone balance resulting in increased bone mass.2,5 Teriparatide (1–34 PTH–-parathyroid hormone fragment) has been approved in some countries including Australia and the US as an alternative treatment in patients who have a very high risk of fracture. This review focuses on published data on teriparatide and other 1–34 PTH analogues. The 1–84 PTH analogue is not considered in this review.
Mechanism of Action
Teriparatide is a recombinant human protein composed of the first 34 amino acids of human parathyroid hormone (PTH). This amino-terminal PTH fragment interacts with PTHR1 receptors expressed on bone and kidney cells in an identical fashion to full-length PTH. Teriparatide is a marketed pharmaceutical product produced by Lilly (Indianapolis, USA) and represents a first in class, bone anabolic agent.
Endogenous PTH has a pivotal role in calcium and phosphate metabolism. Any reduction in endogenous circulating calcium levels are detected by calcium sensing receptors expressed on the parathyroid glands and results in an increase in PTH secretion. 6 PTH stimulates osteoclastic activity, increases renal tubular calcium reabsorption but also causes renal phosphate wasting. Indirectly, via its effect on 1 alpha hydroxylase activity, the increase in 1,25 dihydroxyvitamin D mediated by PTH also culminates in an increase in intestinal calcium and phosphate absorption. 7 These effects all act to redress circulating calcium levels and account for the tight regulation of extracellular calcium homeostasis.
In contrast to the above scenario which occurs with continuous PTH secretion, alternative actions occur when PTH is administered intermittently in low dose. Following intermittent PTH release, PTH stimulates osteoblastic activity. The positive extracellular calcium balance induced by the effects of PTH on renal and intestinal calcium transport in this scenario provide a supply of mineral for utilisation by osteoblasts involved in the new bone formation process. Ultimately, intermittent PTH action also results in an increase in bone resorption but the delay in onset of this effect provides an “anabolic window” of opportunity where bone formation occurs exclusively. 8 Consequently, unlike other states where remodelling rates are increased, such as oestrogen deficiency, this temporal sequence of effects on the bone remodelling unit by intermittent PTH release result in a net increase in bone formation. Furthermore, this effect is confirmed by data showing the amount of new bone formed per remodelling unit is increased with teriparatide therapy as determined by the increase in osteon thickness with therapy.9,10
Effects on bone mass and bone quality are equally important. Microarchitectural changes can account for important effects on fracture risk. Consequently, therapies used to reduce fracture rates should preferably exert beneficial affects on bone mass as well as bone quality. Once daily exposure to teriparatide results in new bone formation on trabecular as well as cortical bone surfaces. In contrast, continuous PTH administration may stimulate bone resorption greater than bone formation resulting in net bone loss, especially at cortical sites with relatively less bone loss at trabecular bone surfaces. The effects of intermittent teriparatide treatment not only increase trabecular thickness but also increase trabecular connectivity as verified by microcomputer tomography of transiliac bone biopsies.11,12 Animal data also demonstrates increased remodelling of the endocortical surface of bone but overall increased cortical thickness due to increased periosteal bone formation. As a consequence, both trabecular and cortical bone microarchitecture improve with therapy.
Teriparatide effects are mediated via interaction with a specific G-protein coupled receptor with ligand binding inducing a cascade that activates cyclic AMP/protein kinase A and protein kinase C pathways. Multiple pathway activation accounts for the increase in osteoblast activity, a decrease in osteoblast apoptosis (programmed cellular death) and may result in conversion of bone-lining cells into osteoblast cells. 13 The precise cellular mechanisms that exert an anabolic effect in preference to an anti-resorptive effect are not known. Other anabolic factors may play a role in the osteoblastic effect. IGF-II expression has been found to be twice as high on the periosteal surfaces of bone biopsy samples from women treated with teriparatide compared to placebo treated women. 14 IGF-I may also be stimulated by PTH secretion. 15 This finding is relevant as low serum IGF-I and IGF-II levels are seen in hip fracture patients. These findings implicate impaired bone formation secondary to IGF deficiency as playing a pathogenic role in bone loss. 16 Basic fibroblast growth factor 2 (bFGF-2) is also up-regulated in teriparatide treated individuals. 17 Since bFGF-2 regulates the proliferation and differentiation of osteoblast progenitors, this cytokine could play an important role in the bone formative response to teriparatide therapy. 18 Finally, the osteocytic SOST gene may be transcriptionaly suppressed by PTH. As a result reductions in sclerostin, a potent inhibitor of bone formation could account for part of the anabolic response to PTH. 19
Metabolism
Whilst no metabolism or excretion studies have been conducted in humans, peripheral metabolism of teriparatide is believed to be initiated in the liver and PTH fragments are then excreted via the kidneys. It is not anticipated that teriparatide should accumulate within bone tissue which provides further differentiation from bisphosphonate metabolism and action. 20
Pharmacokinetic Profile
Data has been derived from phase I and phase III trials in men and women with osteoporosis receiving subcutaneous teriparatide 20 or 40 micrograms daily. 21 When teriparatide is administered as a daily subcutaneous injection it is quickly and almost completely absorbed with a peak serum concentration at 30 minutes after administration to healthy volunteers. Teriparatide has a half-life of approximately 1 hour. After approximately 3 hours, the concentration of teriparatide declines significantly and is no longer readily measurable. Whilst teriparatide is detectable in the circulation, endogenous PTH is undetectable and as the level of teriparatide declines, the level of endogenous PTH increases. 22
Following teriparatide 20 micrograms subcutaneous dose administration, serum calcium levels increase approximately 2 hours after dosing, reach a modest median increase of 0.2 mmol/L and start to decline 6 hours after administration. Serum calcium reaches a nadir at approximately 16 to 24 hours after subcutaneous administration. 23
Pharmacokinetics of single dose therapy was similar in patients with mild or moderate chronic kidney disease (creatinine clearance ≥30 mL/min) compared to healthy volunteers (creatinine clearance ≥90 mL/min). Within the age group of 31 years to 85 years, similar pharmacokinetics were identifiable so no dose adjustment is probably needed based on age alone. 24 No dose adjustments are suggested according to gender but circulating levels of teriparatide are 20%–30% lower in men compared with women. 20
Clinical Studies
Postmenopausal women with osteoporosis
Most randomised published clinical studies were conducted in postmenopausal women. The main outcome measures were fracture reduction with surrogate endpoints including the change in bone mineral density (BMD) and/or biochemical markers of bone remodelling.
Fracture prevention
Compared to placebo
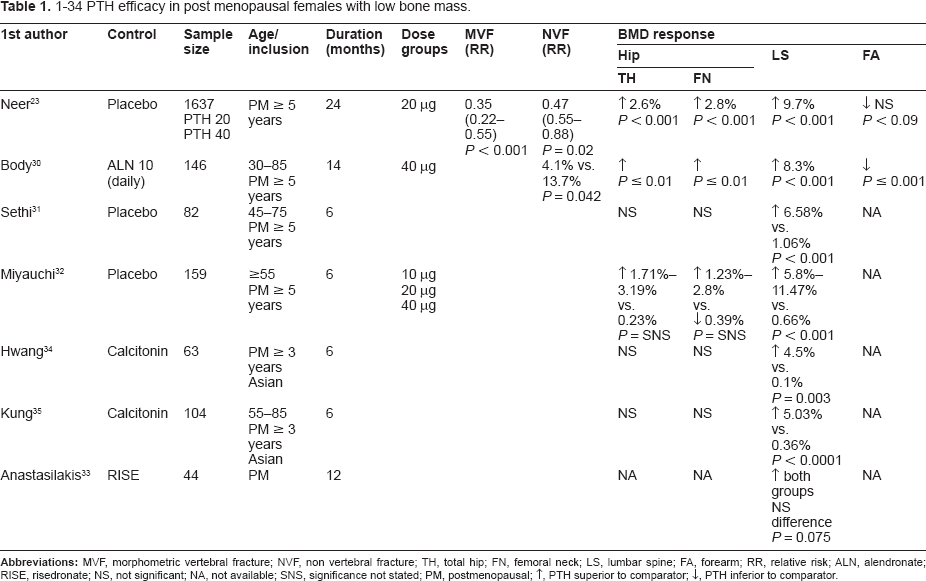
The Fracture Prevention Trial by Neer et al 23 is currently the only adequately powered study to address fracture reduction with teriparatide. A total of 1637 postmenopausal women were randomised to daily subcutaneous PTH at either 20 ug or 40 ug/day, or placebo. At the completion of the study at 24 months (median duration of treatment of 21 months), the rate of new morphometric vertebral fractures was lower with PTH 20 ug (relative risk (RR) 0.35; 95% C.I 0.22–0.55; P ≤ 0.001) and PTH 40 ug (RR 0.31; 95% C.I 0.19–0.50; P ≤ 0.001) compared to placebo. New fragility nonvertebral fractures were also lower with PTH 20 ug (RR 0.47; 95% C.I 0.25–0.88; P = 0.02) and PTH 40 ug (RR 0.46; 95% C.I 0.25–0.86; P = 0.01) compared to placebo. The onset of efficacy for nonvertebral fracture reduction was significant after 9–12 months of treatment.
In a follow up study of the Fracture Prevention Trial, Lindsay et al 25 reported sustained reduction in the risk of new vertebral fracture during an 18 month period after teriparatide cessation (RR 0.59; 95% C.I 0.42–0.85). The estimated number needed to treat to prevent 1 fracture over the total study duration was 8. Prince et al 26 reported persistent benefit in the rate of new non-vertebral fractures up to 30 months from study initiation (9% vs. 13%; HR 0.62; 95% C.I 0.41–0.93) in this population.
The benefit of teriparatide appears to be independent of the severity of osteoporosis, the presence of prevalent fracture, BMD, bone turnover markers, age or renal impairment in post menopausal females.27–29
Compared to bisphosphonates
Body et al 30 studied 146 postmenopausal women aged 30–85 years with lumbar spine (LS) or femoral neck (FN) BMD T ≤ −2.5. Patients were randomised to daily subcutaneous teriparatide 40 ug or daily oral alendronate 10 mg for 12 months. All non-vertebral fractures were radiologically confirmed but the degree of trauma was not ascertained. The teriparatide treatment group had a lower rate of non-vertebral fracture than the alendronate treatment arm (4.1% vs. 13.7%; P = 0.042).
Effects on BMD
Compared to placebo
The 24 month Fracture Prevention Study 23 in post-menopausal women with prevalent vertebral fracture demonstrated a significant dose-dependent increase in BMD of the spine and hip, but a reduction in radial BMD. PTH 20 ug resulted in a significant (P < 0.001) 9.7% increase in LS BMD, 2.8% increase in FN BMD, 2.6% increase in total hip (TH) BMD; and a non-significant 0.1% (P = 0.09) reduction in distal radius BMD and 2.1% (P = 0.09) reduction in shaft of radius BMD. PTH 40 ug resulted in a significant (P < 0.001) 13.7% increase in LS BMD, 5.1% increase in FN BMD, 3.6% increase in TH BMD and 3.2% decrease in shaft of radius BMD; and a non-significant (P = 0.74) 1.5% reduction in distal radius BMD.
Sethi et al 31 studied 82 postmenopausal osteoporotic women aged 45–75 years with BMD (LS, FN or TH) T scores ≤ −2.5. Patients were randomised to teriparatide 20 ug or placebo and treated for 6 months. At the end of the study period, patients treated with teriparatide had significantly higher percentage increase in LS BMD than placebo (6.58% vs. 1.06%, P = 0.001). In contrast, there were no differences in TH or FN BMD.
Miyauchi et al 32 studied 159 postmenopausal osteoporotic women aged ≥55 years. Patients were randomised to daily subcutaneous teriparatide (10 ug or 20 ug or 40 ug) or placebo and treated for 6 months. At study end point, LS BMD was significantly higher in all teriparatide treatment groups compared to placebo (P < 0.001).
Compared to bisphosphonates
Body et al 30 compared teriparatide with alendronate in osteoporotic postmenopausal women. The increased in LS BMD was significantly greater in the teriparatide group compared to the alendronate treated group. The differential effect on spinal BMD was evident within 3 months (2.7% higher; P < 0.001) and persisted until the end of the study at 12 months (8.3% higher; P < 0.001). FN and TH BMD were also higher with teriparatide compared to alendronate at 12 months (P ≤ 0.01). BMD at the one third distal radius was significantly lower with teriparatide treatment (P ≤ 0.001) when compared to baseline measurements and 12 months of alendronate treatment.
Anastasilakis et al 33 studied 44 postmenopausal osteoporotic women aged 65.1 ± 1.6 years. Patients were randomised to risedronate 35 mg weekly or subcutaneous teriparatide 20 ug daily and treated for 12 months. LS BMD increased significantly in both treatment groups at 12 months with no significant differences between treatments (P = 0.075).
Compared to other agents
Hwang et al 34 studied 63 postmenopausal Asian women with BMD T scores <-2 and radiologically proven vertebral or non-vertebral fracture. Patients were randomised to daily subcutaneous teriparatide 20 ug or daily subcutaneous calcitonin 100IU, and treated for 6 months. At study end point, LS BMD was significantly greater in the teriparatide treated group than the calcitonin treated group (4.5% increase vs. 0.1% increase; P = 0.003). There was no significant change in FN or TH BMD within and between the treatment groups.
Kung et al 35 studied 104 postmenopausal Asian women aged 55–85 years with at least one baseline vertebral or non-vertebral fracture. Patients were randomised to daily subcutaneous teriparatide 20 ug or daily subcutaneous calcitonin 100IU, and treated for 6 months. At study completion, LS BMD was significantly higher in the teriparatide treatment group than the calcitonin treatment group (5.03% increase vs. 0.36% increase; P < 0.0001). No significant change was observed in FN or TH BMD in either group.
Sequential and combination treatment
Loss of bone mass gained during teriparatide treatment is the principal concern following cessation of anabolic therapy. Teriparatide treatment is time restricted due to concerns regarding osteosarcoma risk. Studies suggest that the bone loss resulting after teriparatide cessation can be attenuated with anti-resorptive treatment. Patients who received at least 24 months treatment with a bisphosphonates during the 30 month post-teriparatide phase of the Fracture Prevention Trial demonstrated further increases in TH BMD (P < 0.05). 26 In contrast those who did not receive anti-resorptive treatment during the 30 month post-teriparatide treatment phase demonstrated reduction in TH and FN BMD that was not different from placebo (P > 0.05). Similar improvements in BMD were demonstrated in 2 studies with teriparatide followed by raloxifene compared to teriparatide followed by placebo.36,37
One year of teriparatide therapy increased vertebral BMD in both treatment naive and those pre-treated with antiresorptives in the EUROFORS trial (P > 0.001). 38 However, TH BMD only improved in treatment naive patients (P > 0.001) and this effect occurred regardless of the type of antiresorptives, ie, bisphosphonates or non bisphosphonates. 39 In contrast, a study by Ettinger et al 40 suggested that prior alendronate treatment prevented an increase in BMD in the first 6 months, while prior raloxifene treatment permitted an expected increase in TH BMD (-1.8% vs. +0.5%, P < 0.05) and LS BMD (+0.5% vs. +5.2%, P < 0.05).
At 1 year of combination therapy with teriparatide and annual zoledronic acid, LS BMD increased more than teriparatide treatment alone, with both treatments being superior to zoledronic acid alone therapy (7.5% vs. 7.0% vs. 4.4%; P < 0.001). However, although combination was superior at the TH BMD as well, zoledronic acid alone therapy was superior to teriparatide alone therapy at this site (2.3% vs. 2.2% vs. 1.1% (P < 0.05). 41
At 6 months, combination teriparatide plus raloxifene treatment increased LS, FN and TH BMD significantly from baseline and the increase in TH BMD was significantly greater than teriparatide only treatment (P = 0.04). 42 At 1 year, compared to hormone replacement therapy (HRT) alone, teriparatide 40 ug plus HRT resulted in greater increase in LS BMD (14% vs. 3%), TH BMD (5.2% vs. 1.6%) and FN BMD (5.2% vs. 2%) (P < 0.001). 43
Effects on remodelling markers: Bone turnover
The effect of teriparatide treatment on bone formation and resorption biochemical markers has been extensively studied. The mainformation markers studied include bone serum alkaline phosphatase (BSAP), osteocalcin (OC), carboxyterminal propeptide of type 1 procollagen (P1CP) and the amino terminal extension peptide of procollagen type 1 (P1NP). The main resorption markers studied include urinary deoxypyridinoline creatinine ratio (DPD), urinary type 1 collagen cross-linked C-telopeptide creatinine ratio (CTX) and urinary type 1 collagen N-telopeptide (NTX).
Compared to placebo
A study by Sethi et al 31 of postmenopausal osteoporotic women showed increased bone turnover markers in subjects treated with teriparatide 20 ug. Bone formation markers measured included BSAP and OC with both markers reaching zenith at 3 months and remaining higher than placebo at 6 months. A single bone resorption marker, urinary DPD, was higher than placebo at 3 and 6 months.
A study by Miyauchi et al 32 of Japanese postmenopausal osteoporotic women examined the effect of teriparatide on bone turnover markers. P1CP was increased and peaked at 4 weeks. Thereafter, at 6 months, in the 10 ug treatment group, P1CP decreased below baseline, the 20 ug treatment group returned to similar values to baseline, and the 40 ug treatment group remained higher than baseline. P1NP also peaked at 4 weeks in all treatment groups. At 6 months, P1NP returned to similar values to baseline in the 10 ug treatment group, whereas in the 20 ug and 40 ug treatment groups the values remained higher than baseline. BSAP in the 20 ug and 40 ug treatment groups remained higher than baseline at 6 months. Urinary CTX showed a peak increase at 3 months for the 40 ug treatment group and remained elevated at 6 months. The increased level of CTX in the 20 ug treatment group reached a maximum level at 6 months.
Compared to bisphosphonates
Body et al 30 compared teriparatide and alendronate treatment in postmenopausal osteoporotic women and demonstrated higher BSAP at 1 month, peak values at 6 months and persistent increase at 12 months (P ≤ 0.001 at all time points). Urinary NTX was increased at 1 month, remained higher at 12 months (P ≤ 0.001 at all time points) with teriparatide compared with alendronate treatment. As expected, bone resorption (urine NTX) (P < 0.001) and formation (serum BSAP)(P < 0.001) were suppressed in the alendronate treated group.
Anastasilakis et al 33 compared risedronate with teriparatide and showed a significant decrease in serum P1NP, CTX and total ALP in the risedronate treatment group, with corresponding increased levels in the teriparatide treatment group.
Compared to other agents
Hwang et al 34 examined changes in bone formation in postmenopausal osteoporotic Asian women treated with either teriparatide or calcitonin. BSAP and OC levels were significantly higher in teriparatide treatment groups compared to calcitonin treatment at 3 months (P < 0.05 for BSAP, P < 0.001 for OC) and 6 months (P < 0.001 for both).
Kung et al 35 studied postmenopausal osteoporotic Asian women treated with teriparatide and demonstrated a significant elevation in bone formation markers (BSAP and OC) above baseline within 3 months which was sustained at 6 months. There was no significant change in BSAP in calcitonin treatment but a significant reduction in OC was demonstrable at 6 months.
Recker et al 44 studied 80 postmenopausal women aged 45–90 years with LS, FN or TH BMD T ≤ −2.5. Patients were randomised to daily subcutaneous teriparatide 20 ug or strontium ranelate 2 g daily. P1NP levels were significantly higher than baseline by 1 month in the teriparatide group and continue to increase at 6 months, and were significantly higher than strontium ranelate at all time points (P < 0.001). In the strontium ranelate group, P1NP was lower than baseline at 3 and 6 months. BSAP increased gradually in the teriparatide group and was higher than the strontium ranelate treatment group at 6 months (P < 0.005). In the strontium treatment group, the levels of BSAP remained essentially unchanged from baseline. β-CTX increased gradually after 1 month in the teriparatide treatment group and was higher than the strontium ranelate group by 3 and 6 months (P < 0.001). In the strontium ranelate group, β-CTX level was lower than baseline at 1 and 3 months and returned towards baseline at 6 months.
Male osteoporosis
Effects on BMD
Compared to placebo
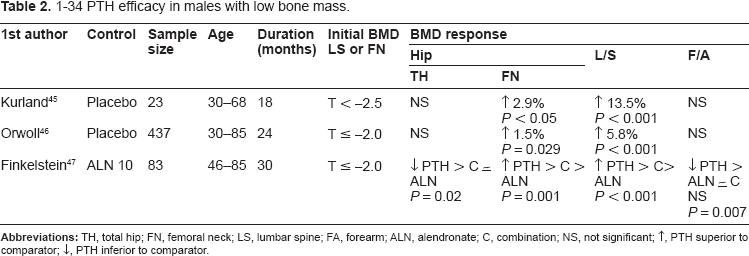
Kurland et al 45 conducted an 18 month placebo controlled trial of 23 men with idiopathic osteoporosis. Patients were aged 30–68 years with LS or FN BMD T scores <-2.5 or Z scores <-2, and randomised to daily subcutaneous PTH 400 IU or placebo. LS BMD increased linearly in the PTH treated group, and was 2% higher than placebo by 6 months (P < 0.05) and remained higher (13.5%) at 18 months (P < 0.005). FN BMD increased gradually in the PTH treated group, and was 2.9% higher than placebo at 18 months (P < 0.05). TH BMD did not change significantly. BMD at the 1/3 distal radius was lower at 12 months in the PTH treated group compared to placebo (P < 0.05), but by 18 months was not significantly different.
Orwoll et al 46 studied 437 men aged 30–85 years with LS, FN or TH BMD T scores ≤ −2. Patients were randomised to daily subcutaneous teriparatide (20 ug or 40 ug) or placebo for a pre-specified 24 months trial. However, the trial was stopped prematurely at 11 months due to reports of osteosarcoma in toxicology studies of rats administered with teriparatide. There was a dose dependant increase in LS and FN BMD. LS BMD was significantly higher in the teriparatide treated group compared to placebo within 3 months (P < 0.001) and remained higher at 12 months (placebo 0.52% higher than baseline, 20 ug teriparatide 5.87% higher than baseline, 40 ug teriparatide 9.03% higher than baseline; P < 0.001 for teriparatide 20 ug or 40 ug vs. placebo, and teriparatide 20 ug vs. 40 ug). FN BMD was also significantly higher in teriparatide treated group compared to placebo at 12 months (placebo 0.31% higher than baseline, 20 ug teriparatide 1.53% higher than baseline, 40 ug teriparatide 2.93% higher than baseline; P = 0.029 20 ug vs. placebo, P < 0.001 40 ug vs. placebo and P = 0.023 20 ug vs. 40 ug). TH BMD was significantly higher in the 40 ug teriparatide group compared to placebo (P < 0.001) at 12 months, but not for the 20 ug teriparatide group. Distal radius BMD was lower in all treatment groups at 12 months (placebo −0.15%, teriparatide 20 ug −0.46%, teriparatide 40 ug −0.56%; P = NS for all comparisons). Subgroup analysis showed the BMD response to teriparatide treatment was unaffected by age, BMI, baseline LS BMD, smoking or alcohol intake.
Compared to bisphosphonates or combination treatment
Finkelstein et al 47 studied 83 males aged 46–85 years with LS or FN BMD T scores ≤-2. Patients were randomised to daily subcutaneous PTH 40 ug (a different 1–34 analogue from teriparatide) or daily oral alendronate 10 mg or both, and followed for 30 months. Alendronate therapy was started at the baseline visit and continued for 30 months. PTH therapy commenced at the 6 month visit and continued for 24 months. At the study end point, LS BMD was higher in those treated with PTH, compared to alendronate alone or combination therapy (P < 0.001). Those treated with combination therapy had higher LS BMD than alendronate alone (P < 0.001). PTH therapy alone also resulted in higher FN BMD (P < 0.001 vs. alendronate; P = 0.001 vs. combination) and TH BMD (P = 0.005 vs. alendronate; P = 0.08 vs. combination). Combination therapy resulted in non-significantly higher FN and TH BMD compared to alendronate alone. Radial shaft BMD was slightly lower than baseline in the PTH group at 30 months, and lower than alendronate group (P = 0.002) and combination group (P = 0.009).
Effects on remodelling markers: Bone turnover
Compared to placebo
Kurland et al 45 studied men with idiopathic osteoporosis. All markers of bone turnover increased in the PTH treated group. The bone formation marker, P1CP, peaked at 6 months and decreased to baseline at 18 months (P = NS compared to placebo), BSAP peaked at 9 months and remained higher than placebo at 18 months (P < 0.05), and OC which peaked at 12 months and remained higher than placebo at 18 months (P < 0.005). Bone resorption marker, urinary DPD, peaked at 9 months, and remained higher than placebo at 18 months (P < 0.05) and urine NTX peaked at 12 months and remained higher than placebo at 18 months (P < 0.005). The highest correlation and therefore best predictor of LS BMD response to PTH therapy at 18 months was a combination of DPD at baseline and OC at 3 months.
Orwoll et al 46 studied men with low BMD treated with teriparatide (20 ug or 40 ug) and demonstrated a dose dependent increase in bone turnover markers compared to placebo. BSAP increased with both doses of teriparatide at 1 month, peaked at 6 months and remained higher than placebo at 12 months (P < 0.001). P1CP increased with both doses of teriparatide at 1 month (P < 0.001), however returned to baseline values at 12 months with no difference compared with placebo with the 40 ug dose but the 20 ug teriparatide dose reduced below placebo values at 12 months (P = 0.021). Urinary NTX/Cr was higher than placebo by 1 month, (20 ug: P = 0.04, 40 ug: P < 0.001), peaked at 3–6 months and remained higher than placebo at 12 months (P < 0.001 for both doses). Urinary DPD was higher than placebo by 1 month (20 ug: P = 0.021, 40 ug: P < 0.001), peaked at 6 months for both doses and remained higher than placebo at 12 months for both doses (P < 0.001).
Compared to bisphosphonates or combinations
Finkelstein et al 47 study of men with low BMD treated with PTH alone reported higher BSAP levels, which peaked at 12 months and remained elevated at 30 months compared to baseline. In the alendronate treated group, BSAP was reduced by 6 months and remained suppressed until the end of study period. The combination group of PTH and alendronate showed an initial reduction in BSAP at 6 months which then increased but did not reach the level of the PTH alone group. In 2006, Finkelstein et al 48 published further bone turnover data on 63 patients from the original study. Treatment with PTH alone resulted in elevated serum OC, P1NP and NTX, which peaked at 12 months and then declined gradually but remained higher than other treatment groups at 30 months. Treatment with alendronate alone resulted in reduced serum OC and P1NP by 6 months and serum NTX by 1 month which remained suppressed until 30 months. Treatment with alendronate and PTH resulted in serum OC, P1NP and NTX levels that were similar to alendronate alone in the first 6 months. After 6 months, when PTH therapy is instituted, serum OC and P1NP increased throughout the study period, whereas there was minimal NTX increase.
Glucocorticoid-induced osteoporosis
Limited data is available for glucocorticoid induced osteoporosis (GIO). The effect of teriparatide on BMD, bone turnover markers and fractures in patients with GIO compared to alendronate treatment was published by Saag et al. 49 This study included 428 men and women with GIO who had received prednisolone at a dose of 5 mg or equivalent per day for 3 months or more and were randomised in a double blind, 18 month study. Fewer radiographically identified vertebral fractures were reported at the end of the study in the teriparatide group compared to alendronate (0.6% vs. 6.1%; P = 0.004). Although both treatments improved LS BMD from baseline at 3, 6 and 12 months (all time points P < 0.001), teriparatide increased BMD 2-fold higher than alendronate at 18 months (8.0% vs. 3.9%; P < 0.001). The difference between treatment groups for LS BMD was significant from 6 months onwards (P < 0.001) and at the last measurement (7.2% vs. 4.4%; P < 0.001). TH BMD also improved with both treatments at 12 and 18 months. However, the increase in TH BMD was greater with teriparatide at 12 months (3.1% vs. 1.9%; P < 0.05) and 18 months (3.8% vs. 2.2%; P < 0.01) compared with alendronate.
Teriparatide improved bone formation markers (P1CP, P1NP, and BSAP) from baseline while all three markers decreased with alendronate. The increase in P1NP was significant at 1 month (P < 0.001), peaked at 6 months (69.8%) and remained elevated at 12 months (40%). In contrast P1NP decreased by 1 month and remained reduced with alendronate (P < 0.001 at all time points). Bone resorption marker (CTX) significantly increased with teriparatide at 1 month, peaked at 6 months (44.8%) and remained elevated at 12 months (10%). In contrast, with alendronate, CTX decreased from baseline within 1 month and remained reduced (55%–60% below baseline) during the 18 months of treatment (all time points P < 0.001).
Use of teriparatide in other conditions
Parathyroid hormone has been utilised in other areas where fracture risk may not be increased. Because of the unique mode of action, teriparatide could be associated with increased bone formation at skeletal sites where fracture does not occur commonly. This potential benefit has been explored in a small randomised trial of 40 patients suffering from severe periodontal disease and consequent alveolar bone loss. 50 Twenty subjects received 20 ug of subcutaneous PTH daily for 6 weeks with 1000 mg of calcium and 800 IU of cholecalciferol and compared with 20 subjects who received a matching placebo that was self administered by subcutaneous injection daily along with oral calcium and vitamin D. Subjects with BMD T scores less than −2.5 were excluded from the trial. All subjects then received periodontal surgical debridement. Periodontal bone improved in a linear and sustained fashion over the next 12 months by 29% in the teriparatide treated subjects compared with 3% in the placebo treated group according to radiographic imaging (P < 0.001). This effect resulted in improved clinical outcomes as determined by reductions in probing depth and increases in clinical attachment (P = 0.02 for both observations). Further data is, however, required before adopting this strategy routinely in the management of periodontal disease.
Several case reports published recently indicate that subcutaneous teriparatide may be associated with resolution of osteonecrosis of the jaw, a complication associated rarely with long term bisphosphonates use in osteoporosis.51–53 Randomised clinical trial data in this area is currently lacking.
Safety
Adverse effect compared to placebo
The largest published study by Neer et al 23 in females reported no significant differences in the number of deaths, hospitalisation, cardiovascular outcomes, nephrolithiasis or gout with teriparatide compared with placebo. The 40 ug teriparatide group reported more nausea (18% vs. 8%; P < 0.001) and headache (13% vs. 8%; P = 0.01) compared with placebo. The frequency of the latter symptoms were not significantly different in the 20 ug teriparatide treatment group compared to placebo. However, dizziness (9% vs. 6%; P = 0.05) and leg cramps (3% vs. 1%) were more common in the 20 ug teriparatide treatment group compared to placebo, but did not differ between the 40 ug teriparatide treatment group and placebo. Mild hypercalcemia (serum total calcium >2.6 mol/L) occurred more commonly in teriparatide treated subjects (placebo 2%, 20 ug PTH 11%, 40 ug PTH 28%). Mild or moderate hypercalcaemia (total calcium values <2.95 mmol/L) was reported in 95% of hypercalcemia episodes. One third of patients with hypercalcemia had persistent elevation on repeat testing within a few weeks. Women who did not suffer hypercalcemia during the first six months of treatment rarely developed hypercalcemia with longer duration treatment.
Findings were similar in a study by Miyauchi et al 32 which reported more frequent nausea, eczema, headache, falls, muscle spasm and increased uric acid with PTH therapy. Nausea and headache occurred more commonly in the 40 ug teriparatide group than any other group. There were no differences in the rate of gout, arthralgia or urinary calculus. Serum calcium was higher in all teriparatide groups 4 hours post injection, and remained higher in the 20 ug teriparatide group at 3 months and 40 ug teriparatide group at 6 months of treatment. The degree of calcium elevation was negligible and deemed not to be clinically significant. A third study by Sethi et al 31 did not report any significant difference in adverse events.
Data on adverse events is much more limited for males treated with teriparatide with smaller published studies. Orwoll et al 46 reported that nausea was significantly more common in the 40 ug teriparatide group compared to placebo. Serum calcium concentrations 4–6 hrs after injection were higher in the teriparatide group at all time points compared to placebo (P < 0.001). Serum calcium levels were above the upper reference limit in 6.2% of the 20 ug teriparatide treatment group (P = 0.003 vs. placebo) and 16.8% of 40 ug teriparatide treatment group (P < 0.001 vs. placebo). Most episodes of hypercalcemia were demonstrable in the first 28 weeks of the trial. Urinary calcium was not significantly different between treatment groups. Rates of nephrolithiasis were not different between treatment groups. Kurland et al 45 studied men with idiopathic osteoporosis and reported those treated with PTH had an expected reduction in endogenous serum PTH, but no change in serum calcium or 24 hr urinary calcium level.
No case of osteosarcoma, the most serious theoretical concern with teriparatide treatment, was reported in any of the human studies reviewed.
Adverse effect vs. active comparators
Body et al 30 noted the teriparatide treated group had a higher post-dose (4–6 hr) serum calcium than the alendronate treated group but subjects were asymptomatic. The teriparatide treated group also had a higher urinary calcium excretion; however this was only evident at the 1 month visit and not present at subsequent visits. Hwang et al 34 compared teriparatide and calcitonin and reported more hypercalcemia, dizziness, vasodilatation, and vomiting in the teriparatide treated group. Kung et al 35 compared teriparatide and calcitonin but reported no significant difference in adverse event, except higher calcium levels in the teriparatide treatment group.
Finkelstein et al 48 studied men and showed that hypercalcemia and hypercalciuria occurred more commonly in PTH only treatment compared with combination treated groups or alendronate only treatment. Headache, dizziness and joint pain occurred more commonly in the PTH and combination groups.
Efficacy
Teriparatide has been shown in randomised controlled trials to reduce vertebral and non-vertebral fracture risk, increase BMD at the lumbar spine and hip, and increase bone turnover markers. Effect on fracture and BMD are summarised in Table 1 (females) and Table 2 (males).
1–34 PTH efficacy in post menopausal females with low bone mass.
1–34 PTH efficacy in males with low bone mass.
Currently the best available evidence is for post-menopausal women, with demonstrated reduction in morphometric vertebral fracture and non-vertebral fracture. This benefit is sustained for 18 months and 30 months after teriparatide cessation for vertebral fracture and non-vertebral fracture respectively. However, this sustained benefit was dependent on the use of anti-resorptive agents after cessation of teriparatide to avoid rapid bone loss that eventuates after anabolic therapy cessation. Teriparatide was significantly more effective in reducing non-vertebral fracture rate than alendronate in the only available head to head trial with fracture data. At present there is no data available on fractures in men or on hip fractures outcome.
In postmenopausal women, teriparatide has been shown to be effective at improving LS BMD as early as 6 months and sustained for up to 24 months. Comparative studies have demonstrated superior efficacy over alendronate and calcitonin, but not risedronate. Teriparatide was also more effective than placebo and alendronate in improving TH BMD but not forearm BMD in published studies.
Studies in men suggest teriparatide alone is superior to alendronate alone or combination treatment in improving LS, FN or TH BMD. Although combination treatment was superior to alendronate in improving LS BMD, there was no difference in FN and TH BMD. This may suggest a blunting of the anabolic effect of teriparatide with combined alendronate treatment. This finding was supported by the EUROFORS studies which showed that although teriparatide improved LS BMD in treatment naïve and those pre-treated with antiresorptives, TH BMD only improved in treatment naïve patients. The study by Ettinger et al 40 suggested this blunting of the anabolic effect of PTH only occurred with prior alendronate but not prior raloxifene treatment. This finding was supported by studies comparing combination raloxifene and teriparatide 42 and combination HRT and teriparatide 43 which showed combination treatment was superior to either agent alone in improving LS and TH BMD at 6 months. This effect does not appear to be bisphosphonates related as the combination of teriparatide and zoledronic acid is superior to either agent alone in improving LS and TH BMD. 41
Teriparatide treatment is currently approved for a total lifetime treatment dose of 18 months of daily therapy. More safety data may permit a longer duration of treatment in the future but will necessitate prolonged human studies. The main concern following cessation of teriparatide is accelerated bone loss with reduction in BMD and consequent increased fracture risk. Patients who received anti-resorptive treatment post completion of teriparatide therapy maintain BMD compared to those who did not.
Teriparatide increases bone turnover and has a potent effect on both formation and resorption markers with a greater magnitude of effect on bone formation markers compared with placebo, bisphosphonates, raloxifene and strontium ranelate. Although teriparatide also increases bone resorption, the delay in the increase in bone resorption compared to the increase in bone formation provides an anabolic window of opportunity with demonstrable increases in LS and TH BMD. In contrast, bisphosphonates, calcitonin and raloxifene decrease bone remodelling with strontium ranelate having less potent effects on bone formation and resorption.
In GIO teriparatide appears to be superior to alendronate in improving BMD and reducing morphometric vertebral fractures.
Patient Preference
Anabolic therapy with teriparatide may be superior to combination or antiresorptive therapy alone in some settings but other factors may limit utility. If adherence and/or preference for therapy is poor then efficacy is equally diminished. There is only limited data on patient preference for teriparatide treatment. A single preference survey of 212 patients with osteoporosis (120 on bisphosphonates, 1 on rPTH, 80 treatment naive, and in the remainder treatment was not specified) found that patients preferred both weekly oral and annual IV bisphosphonates over daily subcutaneous rPTH even when informed that bisphosphonate therapy was inferior in fracture prevention to rPTH (38%, 43% and 7% respectively). The challenge with teriparatide treatment remains its mode and frequency of delivery and cost. 54
Place in Treatment
Teriparatide is the first anabolic agent available for the treatment of osteoporosis. Evidence is limited regarding comparison of efficacy to other available therapies. Teriparatide requires daily subcutaneous injection, manual dexterity and compliance issues that may be a challenge to patients. Teriparatide is an expensive treatment which may not be subsidised by governments/health organisations or subject to limited or restricted funding. Consequently, it is reserved as a second line treatment in patients who are unable to tolerate or in whom currently available treatments are ineffective or have failed. Teriparatide has a role in the treatment of patients with severe osteoporosis based on very low BMD or in those sustaining recurrent fragility fractures. It may also serve a more prominent role in the treatment of older patients who continue to fracture despite low bone turnover, are intolerant of antiresorptive therapy or in the treatment of GIO where it has demonstrated superiority to alendronate treatment.
Conclusion
Teriparatide and other 1–34 PTH analogues, represent the first available line of anabolic agents for the treatment of osteoporosis. The novel mode of action of teriparatide with its principal effect on bone formation places this agent in a unique position in the osteoporosis management algorithm. However, the limited evidence of efficacy in older populations and specifically on hip fracture reduction make cost-benefit analyses important when determining the benefit of this agent on morbidity and mortality rates associated with fractures in an ageing population. Teriparatide represents an expensive and inconvenient treatment so more evidence is needed before its role can be expanded to first line agent for the treatment of osteoporosis.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
