Abstract
Tigecycline is a first-in-class glycylcycline, broad-spectrum, intravenous antibacterial developed to overcome the two major mechanisms of tetracycline resistance (ribosomal protection and efflux). The drug has been approved in US for community-acquired bacterial pneumonia in adults. In vitro, tigecycline had good activity against a range of Gram-positive, Gram-negative and atypical community-acquired respiratory tract pathogens implicated in community-acquired pneumonia (CAP), including community-acquired Staphylococcus aureus, penicillin-resistant Streptococus pneumoniae and multidrug-resistant Enterobacteriaceae. Nonetheless, tigecycline shows in vitro low activity against against P. aeruginosa. Tigecycline provides high intrapulmonary concentrations that exceed the MIC90 of most of these respiratory pathogens. The combined results of two well designed, phase III studies demonstrated that tigecycline 100 mg initially, followed by 50 mg every 12 hours for 7-14 days was not inferior to recommended dosages of levofloxacin in the treatment of hospitalized patients with CAP. Clinical cure rates were 89.7% versus 86.3% in the clinically evaluable population and 81.0% versus 79.7% in the clinical modified intent-to-treat population. Tigecycline represents an appropriate choice for empirical monotherapy in the treatment of CAP, mainly in patients with risk factors for infections due to resistant bacteria.
Introduction
Community-acquired pneumonia (CAP) remains one of the most serious infections affecting outpatients, accounting for significant morbidity and mortality.1,2 In the United States, it is the sixth leading cause of death. In fact, it is estimated that roughly 5.6 million cases of CAP occur every year in this country and at least 1 million of them require hospitalization.2,3 Although most guidelines agree in that antimicrobial therapy should be initially tailored according to either the severity of the infection or the presence of co-morbidity and the local epidemiology, a great variability may be noted among the different countries in the selection of the primary choice in the antimicrobial agents, even for the cases considered as at a low-risk class.4–10 This fact may be due to the many microbial causes of CAP and specialties involved, as well as the different health-care systems effecting the availability or cost of antibiotics. 11
Tigecycline is a new glycylcycline antimicrobial that is active in vitro against a variety of gram-positive and gram-negative organisms, including multidrug-resistant (MDR) pathogens such as methicillin-resistant Staphylococcus aureus (MRSA), extended spectrum β-lactamases (ESBL)-producing Enterobacteriaceae, and MDR-Acinetobacter spp. 12 The FDA approved tigecycline for the treatment of complicated intraabdominal infections, and complicated skin and skin structure infections. 12 On March 20, 2009 the FDA approved a new indication for tigecycline intravenous infusion allowing its use for the treatment of community-acquired bacterial pneumonia (CABP) caused by Streptococcus pneumoniae (penicillin-susceptible isolates), including cases with concurrent bacteremia, Haemophilus influenzae (beta-lactamase negative isolates), and Legionella pneumophila. 13
Notwithstanding this, tigecycline's pharmacological and microbiological profiles encourage physicians’ use of the drug in other infections caused by resistant pathogens featuring limited therapeutics options (e.g. ventilator-associated pneumonia, VAP) 14 or in special situations within the approved indications (e.g. severe intrabdominal infections) “off label” indications-. 15
The aim of the present review is to evaluate the clinical evidence regarding the use of tigecycline for the treatment of CAP and analyze the potential role of tigecycline in this group of patients.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
The tetracycline class of antimicrobials inhibits bacterial growth by binding to the bacterial 30S ribosomal subunit, which blocks entry of amino-acyl transfer RNA molecules into the A site of the ribosome. The elongation of peptide chains is prevented because there is no more incorporation of amino acid residues into the peptide chains. This results in overall inhibition of protein synthesis. Tetracyclines are, for the most part, bacteriostatic. 16
Tigecycline is the first member of the glycylcyclines, a novel group of antimicrobial agents. These agents retain a central four-ring carbocyclic skeleton of the tetracycline class that is crucial for antimicrobial activity. Modifications include substitution of an N-alkyl-glycylamido group on the D ring at the 9th position that confers a broader spectrum of activity; this modification gives this antibiotic the ability to evade tetracycline resistance mechanisms. Tigecycline has a 9-t-butyl-glycylamido side chain on the central skeleton. It has a molecular weight of 585.65 Daltons and its chemical formula is C29H39N5O8. 17
Active efflux of drugs from inside the bacterial cell and ribosomal protection are the two main mechanisms of bacterial resistance to tetracyclines.18,19 The spread of resistance to tetracyclines by bacteria occurs through the acquisition of specific resistance genes, which are found in plasmids, conjugative transposons and integrons. 16 Tigecycline most likely overcomes these tetracycline resistance mechanisms due to steric hindrance by a large substituent at position 9. 20
Tigecycline is available only as an intravenous formulation and is administered twice daily as a one-hour infusion. Given at the standard clinical dose of 100 mg followed by 50 mg q12 h, tigecycline produced a Cmax was 0.85-1 mg/L and a relatively low mean steady-state serum concentrations of 0.403 mg/L and 0.633 mg/L.21,22 The half-life (t1/2) in humans is relatively long, ranging from 37 to 67 h in healthy volunteers, probably because of its large volume of distribution. Studies with 14 C in rats have confirmed that tigecycline is distributed extensively to tissues, including lung, skin, liver, heart, and bone. The concentrations of tigecycline in the skin and lungs in rats were approximately three-fold to fourfold higher than those in plasma.21,22
The mean clearance ranged from 0.2 to 0.3 L/h/kg, with ~8%-11% of the drug excreted in the urine as unchanged drug over 48 h. The major route of elimination appears to be through feces, via biliary excretion. Glucuronide conjugates of tigecycline, its epimer, and an N-acetyl-9-aminominocycline were the major metabolites found, in low concentrations, in feces, urine, and serum.21,22
The pharmacokinetic profile of tigecycline is not significantly modified by renal impairment, hemodialysis, or mild hepatic impairment. Moderate (Child Pugh B) and severe (Child Pugh C) hepatic impairment resulted in a 25% and 55% reduction in systemic clearance, respectively.21,22
Regarding tigecycline, in an experimental neutropenic mouse thigh infection model, the time above the MIC (multiplied by a factor varying from 0.5 to 4) was the best predictor of effectiveness of this agent, compared with the Cmax or AUC indices, against E. coli and Klebsiella pneumoniae, while the AUC and the time above the MIC were the best predictors of effectiveness of tigecycline against S. pneumoniae. 12 However, due to prolonged post-antibiotic effect against most pathogens, along with extensive volume of distribution, and prolonged elimination t1/2 in humans, the AUC/MIC ratio has been proposed as the most important pharmacodynamic index of tigecycline. 23
The AUC0-12 of tigecycline in epithelial lining fluid (ELF) of healthy volunteers was 32% higher than the AUC0-12 value in serum. One would expect higher AUC values with pneumonia. On the other hand, tigecycline concentrations in alveolar macrophages of healthy volunteers were 78-fold higher than in those found in serum. 32 Tigecycline concentrations in neutrophils have also been 20- to 30-fold higher than extracellular concentrations of the drug (Fig. 1). Tigecycline provides high intracellular concentrations in other tissues (such as lung and bowel) that exceed serum concentrations by 2- to 8-fold.21,22 While well-controlled clinical data are required to fully assess the viability of tigecycline as a therapeutic modality for pneumonia, Conte et al 24 reported that the Cmax/MIC90’ AUC/MIC90, time/MIC90 and extended serum and intrapulmonary half-lives of this compound were favorable for the treatment of tigecycline-susceptible respiratory pathogens. However, Koomanachai et al have studied in a murine model that the in vivo bactericidal activity of tigecycline against various A. baumannii (MIC 0.25-1.0 mg/L) causing pneumonia. These studies revealed that AUC/MIC exposures of 2.17 and 8.78 were required to produce 1 and 2 log kill, respectively. Given these confounding issues, extrapolation of our current dataset to man suggests that tigecycline doses of up to 200 mg/day may be required to provide adequate exposure for A. baumannii. 25 In concordance with Koomanachai, Burkhardt et al 26 have found that indicate that attainable extracellular ELF concentrations of tigecycline in patients with ventilator-associated pneumonia are absolutely insufficient to reliably eradicate extracellular bacteria such as S. pneumoniae, S. aureus, Escherichia coli or Klebsiella pneumoniae, based on the agent's current MIC profile, whereas the mean concentrations in alveolar cells exceed the MIC90 of all atypical bacteria through the entire dosing interval. Therefore, the current dosage of 50 mg tigecycline twice daily is probably underdosed for the treatment of pneumonia caused by typical, extracellular-acting bacteria.
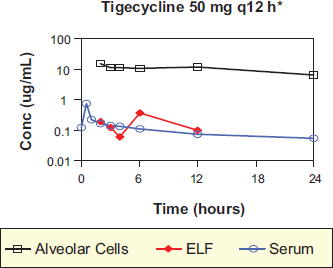
Tigecycline concentrations in serum, epithelial lining fluid (ELF) and alveolar cells at the time of serum Cmax concentrations.
However, high cure rates for tigecycline patients were observed in two observational studies of patients with nosocomial pneumonia (~70%) using the usual dose approved (100 mg loading dose and 50 mg q12 h).27,28
Surveillance studies provide invaluable information in the tracking of antimicrobial susceptibility both locally and globally. The Tigecycline Evaluation and Surveillance Trial (TEST) is a global surveillance study initiated in 2004 to assess the in vitro activity of tigecycline and comparator antimicrobials against key gram-negative and gram-positive organisms, including nosocomial MDR-pathogens.
According to these data, tigecycline exhibits antibacterial activity against a wide spectrum of aerobic and anaerobic bacteria which produce health care-associated pneumonia (HCAP) and CAP, including MDR-pathogens (Table 1).
Etiologic findings among pneumonia patients (%) and tigecycline in vitro activity.
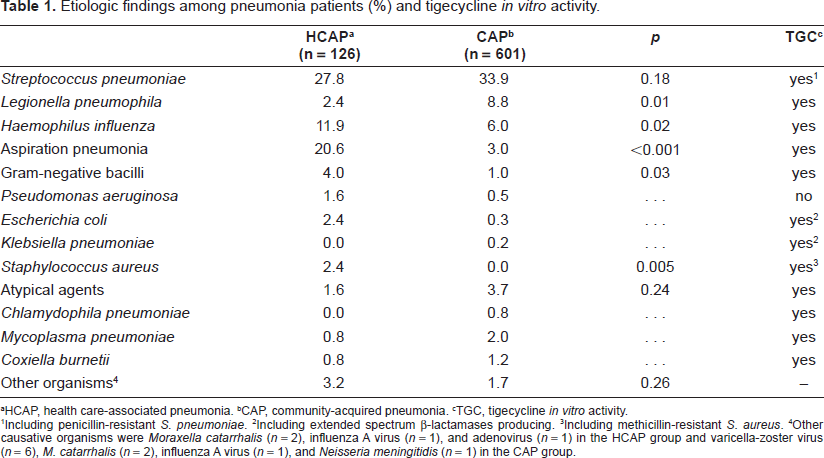
HCAP, health care-associated pneumonia.
CAP, community-acquired pneumonia.
TGC, tigecycline in vitro activity.
Including penicillin-resistant S. pneumoniae.
Including extended spectrum β-lactamases producing.
Including methicillin-resistant S. aureus.
Other causative organisms were Moraxella catarrhalis (n = 2), influenza A virus (n = 1), and adenovirus (n = 1) in the HCAP group and varicella-zoster virus (n = 6), M. catarrhalis (n = 2), influenza A virus (n = 1), and Neisseria meningitidis (n = 1) in the CAP group.
As with common aerobic gram-positive pathogens, including MRSA, vancomycin-intermediate S. aureus, vancomycin-resistant enterococi (VRE), and penicillin-resistant Streptococcus pneumoniae, MIC90 values range from 0.12 to 0.5 μg/mL. 29 All these isolated would be considered susceptible to tigecycline at the FDA susceptibility breakpoints of 0.25 and 0.5 μg/mL for streptococci and staphylococci, respectively. 30 In addition, both tetracycline-susceptible and resistant strains have exhibited almost identical MIC values for tigecycline.
Gales et al 31 have published that against S. pneumoniae, tigecycline, gatifloxacin, quinupristin/dalfopristin, vancomycin, teicoplanin, and linezolid showed a susceptibility rate of 100.0%. Four S. pneumoniae isolates were not susceptible to ceftriaxone when non-meningitis breakpoints were applied. Tigecycline (MIC50, 0.12 μg/mL) was as potent as teicoplanin (MIC50, 0.12 μg/mL) and two-fold more potent than ceftriaxone (MIC50, 0.25 μg/mL), gatifloxacin (MIC50, 0.25 μg/mL), and vancomycin (MIC50, 0.25 μg/mL) against S. pneumoniae. Tigecycline was also highly active against S. pneumoniae, including isolates resistant to penicillin and/or tetracycline and/or erythromycin. In the same study nearly 18.0% and 93.0% of the H. Influenzae and M. catarrhalis strains analized were β-lactamase, producing respectively. Haemophilus influenzae (MIC50/90, 0.25 μg/mL) and M. catarrhalis (MIC50/90, ≤0.12/0.5 μg/mL) isolates were very susceptible to tigecycline (100.0% susceptible), including β-lactamase producing isolates.
Community-acquired MRSA (CA-MRSA) isolates are usually recovered from complicated skin and skin structure infections. Which are associated with younger patients, sport team participation, recent incarceration, military recruits, snorting/smoking illegal drugs, lower comorbidity index, more frequent visits to bars, raves, and/or clubs. On a total of 1989 CA-MRSA (94.7% Panton-Valentine leukocidin positive), tigecycline susceptibility rate was 98.2% (MIC90 0.5 μg/mL). 32
Tigecycline is active against most gram-negative bacilli (not P. aeruginosa, Proteus spp., or Providentia spp. and many strains of Morganella morganii). Studies of these isolates suggest that these strains have constitutively overexpressed multidrug efflux pump systems (e.g. MexXY and AcrAB) for which tigecycline is a substrate. 33 More than 90% of strains of Pseudomonas aeruginosa have MIC values of ≥4 mg/mL and would be considered resistant to tigecycline. 30
For Enterobacteriaceae, tigecycline's MIC90 values for tetracycline-susceptible strains have ranged from 0.25 to 1 mg/mL. 29 Most studies of large numbers of isolates have shown that >95% of all Enterobacteriaceae are susceptible to tigecycline at the FDA susceptibility breakpoint of 2 μg/mL. Strains of Escherichia coli and Klebsiella species with and without ESBLs have shown very similar MIC values and susceptibility rates to tigecycline. Similar susceptibility profiles were seen for both E. aerogenes and E. cloacae. Against these microorganisms, susceptibility rates of >90.0% were seen for tigecycline. 29
Tigecycline is highly active against carbapenemase-producing Enterobacteriaceae isolates. Castanheira et al have published that all serine or metallo-β-lactamase-producing Enterobacteriaceae isolates (n = 104) were inhibited at the tigecycline susceptibility breakpoint approved by the FDA (<2 μg/mL). This compound was the most active antimicrobial tested against this collection of multidrug-resistant strains (MIC50, 0.5 μg/mL; MIC90, 2 μg/mL).
Tigecycline is active against Acinetobacter spp. including strains that are not susceptible to available antibiotics (MIC90 1-2 μg/mL). 29 Arroyo et al have published that tigecycline shows a very good activity against 46 MDR-Acinetobacter spp (carbapenems and colistin included), with an MIC90 of 1 μg/mL. 34
Tigecycline is active against most anerobic bacteria, including Clostridium difficile, Fusobacterium species, Prevotella species, Poryphymonas species, and the Bacteroides fragilis group. 30 The FDA susceptibility breakpoint to tigecycline for anerobes is 4 μg/mL, which fits the MIC distribution of anerobic organisms in the clinical trials.
Tigecycline has potentially useful antimicrobial activity but may not always be effective as empiric monotherapy (i.e. infections where P. aeruginosa is suspected or when high serum levels of antibiotic is required). Petersen et al have determined the interaction of tigecycline with other antimicrobial agents against a variety of bacterial isolates using the broth microdilution chequerboard method. 35 The combination of tigecycline and another antibiotic demonstrated either synergy (24%) or no interaction (76%) against the panel of gram-negative bacteria; antagonism was not observed for any combination with tigecycline, against any of the strains tested. A higher percentage of synergistic combinations with tigecycline were observed with amikacin (56%), ampicillin/sulbactam (33%), piperacillin/tazobactam (50%) and rifampicin (33%). Combinations of tigecycline with amikacin also showed synergy for 40%-100% of Enterobacter spp., Klebsiella pneumoniae, Proteus spp. and Stenotrophomonas maltophilia isolates.
With the gram-positive isolates, rifampicin displayed a synergistic effect with tigecycline for 66% of the isolates tested. The majority of these strains showing synergy were VRE and penicillin-resistant Streptococcus pneumoniae. Mercier et al have found that the combination of tigecycline and gentamicin gave enhanced activity against the three Staphylococcus aureus isolates. 36 At the same time, Entenza et al 37 have found that synergy occurred when tigecycline was combined with rifampicin against 64%-100% of Enterococcus spp., Streptococcus pneumoniae, Enterobacter spp. and Brucella melitensis isolates.
The main conclusion of the available data is that the interaction of tigecycline with other antimicrobials produced primarily an indifferent response and very rarely showed antagonism. Although synergy detected by in vitro studies, is an encouraging outcome suggesting that tigecycline may prove to be effective in combination therapy in particular situations (i.e. in potentially problematic MDR species).
Clinical Efficacy
Tigecycline has been compared with levofloxacin for the treatment of patients hospitalized for CAP, requiring intravenous antibiotics, in two multicenter, randomized, double-blind clinical trials. 38 Tigecycline was administered intravenously as a 100 mg loading dose followed by 50 mg twice daily. Levofloxacin was administered intravenously as a 500 mg dose every 24 hours, while in one of the two trials it could alternatively be administered at a dose of 500 mg twice daily, at the investigators’ discretion. Additionally, in one of the two studies, therapy could be switched to oral levofloxacin after at least 3 days of intravenous therapy in patients allocated to both treatment arms who satisfied certain criteria of response. The total duration of study treatments was between 7 and 14 days in both trials. At the test-of-cure evaluation (within 7 to 23 days after the completion of therapy), cure rates in the tigecycline treatment arms were not inferior compared to the levofloxacin treatment arms, regarding both the clinically evaluable (574 patients; 89.7% compared to 86.3%, respectively), and the modified intention-to-treat population (797 patients; 81.0% compared to 79.7%, respectively). A similar proportion of deaths were noted in both treatment arms (2.6 and 2.8%, respectively). 38
Clinical cure was achieved in 20/22 tigecycline patients (90.9%; 95% CI, 70.8-98.9) and 13/18 levofloxacin patients (72.2%; 95% CI, 46.5-90.3) with S. pneumoniae bacteremia in the microbiologically evaluable population. 38
For the common respiratory pathogens S. pneumoniae and Haemophilus spp., the clinical cure by baseline pathogen at test of cure assessment was 85/91 (93.4%; 95% CI, 86.2-97.5) and 14/17 (82.4%; 95% CI, 56.6-96.2) tigecycline patients and 90/99 (90.9%; 95% CI, 83.4-95.8) and 13/16 (81.3%; 95% CI, 54.4-96) levofloxacin patients, respectively. The clinical cure for the atypical bacteria, M. pneumoniae, C. pneumoniae, and L. pneumophila, was 37/39 (94.9%; 95% CI, 82.7-99.4), 18/19 (94.7%; 95% CI, 74.0-99.9), and 10/10 (100%; 95% CI, 69.2-100.0) for tigecycline and 44/48 (91.7%; 95% CI, 80.0-97.7), 26/27 (96.3%; 95% CI, 81.0-99.9), and 6/6 (100%; 95% CI, 54.1-100) for levofloxacin patients. Only 9 penicillin-resistant S. pneumoniae were isolated, and there was no significant difference in the clinical efficacy between the 2 treatment arms.
A considerable proportion of the included patients may not have been that severely-ill to require hospital admission and intravenous antimicrobial therapy. It was the major concerns that led the European Medicines Agency (EMEA) to recommend the refusal of the change to the marketing authorization of tigecycline in this indication. 39 Specifically, only approximately 20%, and 8% of the total modified intention-to-treat patient population was classified as having a Pneumonia Severity Index (PSI) of at least IV, and a CURB-65 score of at least 3, respectively.38,40 Nevertheless, the tigecycline cure rates did not differ in clinically evaluable patients with a CURB-65 score of at least 3 compared to levofloxacin. 38
In summary, these results presented here indicate that tigecycline is an efficacious and well-tolerated monotherapy, with comparable efficacy to levofloxacin for the treatment of hospitalized patients with CAP. Tigecycline was effective in eradicating the pathogens associated with CAP, including atypical bacteria.
Safety
Tigecycline was associated with significantly higher rates of drug-related adverse events compared to levofloxacin (47.9% versus 37.4%, respectively), mainly consisting of nausea (20.8% versus 6.6%, respectively), and vomiting (13.2% versus 3.3%, respectively). In contrast, elevated serum glutamic pyruvic transaminase (SGPT)/alanine aminotransferase (ALT) and serum glutamic oxaloacetic transaminase (SGOT)/ aspartate aminotransferase (AST) levels occurred at significantly higher rates (P < 0.01) for levofloxacin patients compared with tigecycline patients. There was no significant difference in reports of diarrhea between the 2 treatment arms (7.7% tigecycline and 5.5% levofloxacin). However, the rates of serious adverse events and of discontinuation of study medications due to adverse events did not significantly differ between the compared treatment arms (tigecycline 6.1% vs. levofloxacin 8.1%).
There were no significant differences between treatment groups with respect to the frequency of severe adverse events and deaths (9.9% and 10.9%; and 2.8% and 2.6% in the tigecycline and levofloxacin group respectively). 38 None of the deaths were considered related to study medication.
Patient Therapy
Based on the Type of Pneumonia
Currently accepted classifications of pneumonia include community-acquired pneumonia (CAP), hospital-acquired pneumonia (HAP), VAP, and healthcare-associated pneumonia (HCAP) (Table 2a).41,42
Classifications of pneumonia.
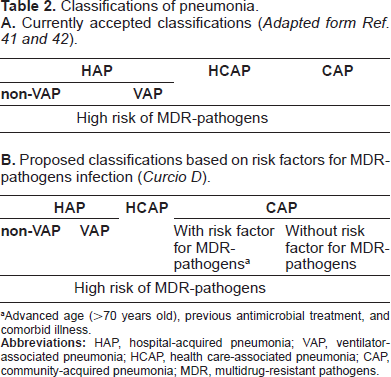
Advanced age (>70 years old), previous antimicrobial treatment, and comorbid illness.
HCAP is a category of nosocomial pneumonia defined by the 2005 American Thoracic Society/Infectious Diseases Society of America (ATS/IDSA) guidelines to include any patient who has been hospitalized in an acute care hospital for 2 or more days within the past 90 days; residents of a nursing home or long-term care facility; recipients of recent intravenous antibiotic therapy, chemotherapy, or wound care within the past 30 days; or patients who have attended a hospital or hemodialysis clinic. 42
In general, patients who develop HCAP are more similar to hospitalized patients than true community patients in that they have a greater burden of co-morbidities (i.e. cancer, chronic kidney disease, etc.)41,42 and in terms of clinical manifestations, HCAP can be graded between CAP and HAP, but it is more similar to HAP.43,44
An important characteristic of HCAP is that the pathogens are often MDR bacteria (MRSA, Pseudomonas aeruginosa, and other gram-negative bacilli). 42 Therefore, the initial treatment of HCAP should be similar to that of HAP and VAP, which also differentiates it from CAP. 45 Recognition of HCAP is particularly important for clinicians working in first-response areas such as emergency departments so that appropriate initial antimicrobial therapy is not delayed. Several studies have demonstrated that delaying the delivery of appropriate antimicrobial therapy in patients with HAP, VAP, and HCAP results in excess mortality.41,45
However the definitions of HAP, CAP, and HCAP have varied among different large-scale studies and the classification schemes are inherently imprecise because patient groups overlap in the different categories and the distinction between HCAP and CAP has never been totally clear. 46
The accepted classification of patients with pneumonia into those with CAP and those with HAP is very useful in the younger and middle age groups, but it is not appropriate for the elderly population. 47 The list of diseases that have been cited as risk factors for pneumonia in the elderly include all of the common diseases in this age group, such as chronic obstructive pulmonary disease, congestive heart failure, ischemic heart disease, diabetes mellitus, chronic renal insufficiency, neurological disease, malignancy and malnutrition and although they are more frequent in patients with HCAP,47,48 they can also be observed in those with CAP.49–52 In the large retrospective series of 4543 patients with culture-positive pneumonia Kollef and coworkers compared the demographic and clinical characteristics of patients with CAP, HCAP, and HAP and described that age and the prevalence of the main comorbidities of HCAP were intermediate between CAP and HAP. 41 Carratala et al 53 did not find significant differences between patients with HCAP (n = 126) and CAP (n = 601) in several co-morbidities such as chronic renal failure (p = 0.13), chronic liver disease (p = 0.60), autoimmune disease (p = 0.82), smoking (p = 0.45) and heavy drinking (0.77).
In conclusion, there are several studies suggesting that age and comorbidities play an important role in disease morbidity also causes HCAP but in the CAP.
Related with the etiologic agents, Grossman et al assessed the occurrence of culture-positive CAP and HCAP at a single center. 54 Among the 639 patients hospitalized with pneumonia, HCAP was more common than CAP (67.4% vs. 32.6%). The most common pathogens identified overall included MRSA (24.6%), Streptococcus pneumoniae (20.3%), Pseudomonas aeruginosa (18.8%), methicillin-sensitive Staphylococcus aureus (13.8%), and Haemophilus influenzae (8.5%). Patients with HCAP were statistically more likely to be infected with MRSA, Pseudomonas aeruginosa, and other gramnegative bacilli compared with patients with CAP.
Kollef et al 41 found important differences between the microbiology of CAP and HCAP S. aureus was the dominant pathogen, identified in 25.5% of CAP patients and 46.7% of HCAP patients (p < 0.01). MRSA was identified in 8.9% of CAP patients and 26.5% of HCAP patients (p < 0.01). Other frequently identified gram-positive pathogens were streptococcus nongroup (13.4% versus 7.8%, respectively; p < 0.01) and Streptococcus pneumoniae (16.6% versus 5.5%, respectively; p < 0.01). Rates of S. aureus and MRSA did not differ among patients with HCAP, HAP, and VAP, suggesting etiologic similarities among these three pneumonia categories. 5 The gram-negative pathogens Pseudomonas spp. (25.3% versus 17.1%, p < 0.01) and Acinetobacter sp. (2.6% versus 1.6%, p < 0.05) were significantly more common in patients with HCAP than CAP. 41
Even CAP caused by MRSA and or MDR-gram-negative bacteria, such as Klebsiella pneumoniae, is uncommon, there are certain situations in which these pathogens must be considered.
Arancibia et al 55 studied 559 patients with CAP to assess risk factors for infection with gram-negative bacteria, including Pseudomonas aeruginosa. 12 Bacterial etiology was identified in 309 (55%) of the patients. Sixty patients (11%) had CAP due to gram-negative bacteria. P. aeruginosa [39/60 (65%)] and Escherichia coli [12/60 (20%)] were the most common gram-negative pathogens. Multivariate analysis demonstrated that probable aspiration [OR 2.4, 95% CI 1.1 to 5.2), previous hospital admission (OR 5.1, 95% CI 2.6 to 10.0), previous antimicrobial treatment (OR 1.9, 9% CI 1.0 to 3.7), and pulmonary comorbidities (OR 2.8, 95% CI 1.5 to 5.5) were significant risk factors for gram-negative pneumonia.
In that sense, Pop-Vicas et al 56 performed a case control study over a 6-year period evaluating risk factors for patients who harbored MDR-Gram-negative bacilli on admission to the hospital. 13 Of 464 MDR-gram-negative pathogens isolates 12%, 35%, and 53% of isolates were resistant to five, four, or three antimicrobial groups, respectively. Three variables were significantly associated with the presence of MDR gram-negative pathogens infection: age ≥65 (OR 2.8, 95% CI 1.1 to 7.4), prior exposure to antibiotics ≥ 14 days (OR 8.7, 95% CI 2.5 to 30), and prior residence in a long-term care facility (OR 3.5, 95% CI 1.3 to 9.4). Patients with chronic skin ulcers and patients with a history of recurrent urinary tract infection also often harbored MDR-gram-negative pathogens (100% and 93%, respectively). The risk associated with these conditions can best be explained by the likelihood that these patients were more likely to have received prior repetitive courses of antibiotics. Prior usage of fluoroquinolones was a risk factor for fluoroquinolone-resistant enterobacteriaceae (OR 1.08, CI 95% 1.04 to 1.11).
In a study focusing on pneumonia in the elderly, El Solh et al 57 performed a prospective cohort study in patients over the age of 75 with severe pneumonia requiring intensive care unit (ICU) admission. 21 They focused on risk factors for resistant pathogens. Of the 104 patients included in this study, 55% came from the community and 45% were admitted from a nursing home. This study suggests that elderly nursing home patients, as compared with elderly community-based patients, have a higher incidence of S. aureus pneumonia, including MRSA, but have similar risks for MDR-Gram-negative bacilli (14% in the community group vs. 15% in the nursing home group). In other publications the same authors have found by multivariate analysis in older patients (>70 years old) with severe pneumonia that prior antibiotic exposure was the single most important risk factor for infection with with MDR-pathogens (OR 24.5, CI 95% 3.3 to 118, p < 0.001).
In summary, it is well-established that the presence of co-morbidities and MDR-pathogens are more common among patients with HCAP, the patients with CAP can have infections by MDR-bacteria too (i.e. MRSA, gram-negative bacilli, Pseudomonas aeruginosa) that's why is necessary to consider several risk factors in these groups of patients in order to improve the empirical antibiotic treatment (i.e. advanced age, comorbid illness, and prior exposure to antibiotics)58,59 (Table 2b).
Tigecycline would be an option in patients with CAP with risk factors for infections due to MDR-pathogens in combination with an anti-pseudomonal antibiotic if P.aeruginosa is suspected.
Based on the Characteristic of the Patient
Elderly Patients
As were described previously, the older patients have an increased risk to suffer CAP by MDR-resistant pathogens.
Another important characteristic of the etiology of CAP in the elderly is the relatively high rate of CAP caused by more than one pathogen. More than one pathogen is involved in a third of the patients with CAP, 60 and despite its therapeutic importance, remains undiagnosed in most. 61
The relative prevalence of Legionella spp. among the etiologies of CAP in the elderly ranges in various series between 0 and 15%.60,62 The importance of Chlamydia pneumoniae as a CAP etiology has become clearer over recent years. It is found in 6 to 26% of elderly patients hospitalized for CAP.44,50,60,63 To date, the data collected on this etiology are too limited to reach definite conclusions, but it is becoming clearer that this pathogen, like Legionella spp., has strong geographic variability and is very dependent on the intensity of the etiological work-up and the diagnostic methods used. Mycoplasma pneumoniae is either rarely alluded to in studies of CAP in the elderly, or referred to as an unimportant etiological factor. However, it is impossible to ignore the reports of a significant proportion of patients with M. pneumoniae pneumonia being elderly.60,64
Another etiological factor that has not been mentioned in most series on CAP in the elderly is aspiration pneumonia. 48
Although all cases of pneumonia suppose a previous “micro-aspiration” with oropharyngeal content into the larynx and lower respiratory tract, this item is specially referred to that evidential aspiration with both oropharyngeal and gastric contents occurred in ambulatory patients, i.e. the aspiration pneumonia preceded by a chemical injury (the Mendelson's syndrome), or a large-volume aspiration following to an obstruction with a foreign body or fluids.
The risk factors are the impaired consciousness due to alcohol, drugs, or hepatic failure; 65 beside, it has been largely accepted that disorders in swallowing and in cough reflex are a considerable source of this kind of pneumonia in the elderly but the hypothesis that poor oral hygiene could be linked to respiratory pathogen colonization has only recently aroused great interest. In severe aspiration pneumonia, 20% of organisms implicated are anerobic and 80% aerobic, most of which are gram-negative Enterobacteriaceae. Poor oral hygiene increases subsequent risk of pneumonia: dental plaque may act as a reservoir for pathogenic organisms implicated in CAP or HCAP.66,67
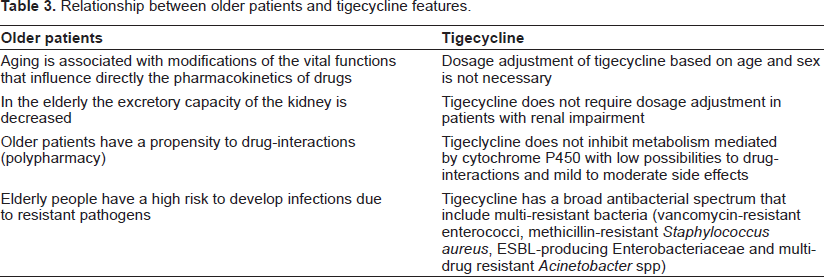
The treatment of complicated infections in critically ill older patients implicate, in most cases, the use of antibiotics such as vancomycin, carbapenems, third- or fourth-generation cephalosporins, piperacillin tazobactam, and aminoglycosides that require dosage adjustment related to renal function to reduce the risk for adverse events and medication interactions. 68 In this context, there are several reasons to consider the use of tigecycline in elderly people (Table 3):
Neither severe renal impairment nor hemodialysis significantly modifies its pharmacokinetic profile,
Does not require dosage adjustment in older people, nor does it inhibit the metabolism of the drugs mediated by cytochrome P450, reducing the risk of medication interaction, and
Has a broad antibacterial spectrum that includes multiresistant bacteria and anerobes. 68
Relationship between older patients and tigecycline features.
Nonetheless, tigecycline shows in vitro low activity against P. aeruginosa, therefore, in patients with risk factors, an anti-pseudomonal antibiotic should be added.
Patients with Risk Factor for Community-Acquired MRSA (CA-MRSA)
The spectrum of disease caused by CA-MRSA occurs worldwide and primarily encompasses skin and soft-tissue infections,69–72 but deep-seated infections such as pyomyositis, osteomyelitis, septic arthritis, and severe infections such as necrotizing pneumonia and bacteremia have also been reported. 73
Staphylococcus aureus has long been recognized as a cause, albeit an infrequent one, of CAP. Estimated to represent 1%-10% of CAP and 20%-50% of HAP, 2-5 it had been uncommon in healthy children and adults from high-income countries except when seen in the post-influenza setting.42,74
In 1999, four pediatric deaths were reported due to CA-MRSA necrotizing pneumonia; these infections were caused by strains that differed from typical nosocomial strains in their antibiotic susceptibility patterns and pulsed field gel electrophoresis (PFGE) characteristics. 75 Since then, many case reports and series have been described.75–82 However, the overall incidence of CA-MRSA pneumonia remains unknown.
CA-MRSA pneumonia generally affects young and previously healthy patients,75–83 Clinical presentation is usually that of a severe pneumonia with high fever, hypotension, and hemoptysis followed by rapid progression to septic shock and requirement for ventilatory support. More than a quarter of patients with CA-MRSA pneumonia are reported to have multilobar infiltrates and/or cavitation in imaging studies.75,84–89 Substantial morbidity and mortality have thus been reported due to CA-MRSA pneumonia, but mortality varies widely.87,89,90 However, despite reporting bias, many reports from the USA and Europe have noted mortalities greater than 50%.75,84,85
The epidemiology of CA-MRSA has not been fully elucidated; however, several important risk factors for the development of CA-MRSA have been described in the literature (Table 4).
Risk factors for community-acquired Staphylococcus aureus.

The use of vancomycin or linezolid has been recommended for empirical treatment of CAP in cases in which CA-MRSA is a consideration. 86 However, reported treatment failures for MRSA infections with MICs of vancomycin in both the susceptible and non-susceptible range remain an important concern.91–93 Whether optimization of vancomycin pharmacokinetic parameters will improve outcomes for patients with MRSA pneumonia is not clear, 94 but the American Thoracic Society/Infectious Disease Society of America guidelines for pneumonia recommend aiming for vancomycin trough concentrations of 15-20 mg/mL 95 Responses in patients with MRSA bacteremia, for example, have been found to be better among those with isolates that have vancomycin MICs of at least 0·5 μg/mL compared with those with 1-2 μg/mL 96 Slightly contrasting with this observation, patients with MRSA pneumonia who attained vancomycin serum trough concentrations four times the MIC had improved responses at 72 h but not at the end of therapy. 97
Even though, there are no studies which have measured the relevance of the vancomycin MIC creep in CA-MRSA, Mendes et al have published that the vancomycin MICQ0 in 1989 isolates of CA-MRSA recovered from patients presenting to North American was 1 μg/mL. In this study, tigecycline showed excellent in vitro activity against this pathogens and its activity was comparable with those other highly active parenteral agents, such as the glycopeptides, streptogramin combinations, and oxazolidinones. 32
The lack of data on the efficacy of tigecycline for MRSA pneumonia limit the use of this drug, however, its excellent antimicrobial activity and high intrapulmonary concentration represent two attractive features to use this drug in patients with CAP caused by CA-MRSA.
Patients with Penicillin Allergy
The CAP management algorithm in the guidelines recommends alternative antibiotic choices for patients with a penicillin allergy. To ensure the most appropriate alternative is prescribed, a detailed history of penicillin reaction is needed. 98
Maxell et al 98 have published that in total of 691 CAP, 79 patients (11%) had documented penicillin allergy. Of these, 17 were documented to have immediate hypersensitivity, 55 had documented non-immediate hypersensitivity, and seven patients had assumed non-immediate hypersensitivity due to nonspecific allergy documentation.
The standard teaching is that patients who have had possible penicillin anaphylaxis should not be treated with cephalosporins. There is evidence that, among patients with a history of penicillin allergy, the rate of allergic reaction to any other antibiotic is 3 times the rate among control subjects. 98 In the general population, the risk of serious allergic reactions to cephalosporins appears to be <0.02%; 99 the risk is lowest for third-generation cephalosporins (possibly because free drug competes with bound drug for antibodies to the side chain). 99 Therefore, even if patients with a history of penicillin allergy have twice as great a risk of having a serious reaction to cephalosporins that do control subjects, this risk may be lower than the risk that they will have a serious reaction to any alternative antibiotic. 100 Furthermore, the results of penicillin skin testing do not predict cephalosporin allergy which again suggests that there is limited cross-reactivity. 100
On the basis of the structure of the drugs, cross-reactivity between penicillin and carbapenems would be expected. A retrospective study of 63 febrile neutropenic bone marrow transplant recipients who had a history of penicillin allergy and received imipenem-cilastatin revealed 1 definite, 3 probable, and 2 possible allergic reactions. 100 To our knowledge, there are no published data on allergy to meropenem in patients who are allergic to imipenem or to penicillins. The cross-reactivity rate between cephalosporins and carbapenems is unknown but is probably quite low, because most reactions to cephalosporins involve side chains rather than the β-lactam ring.
The main issue related with the true penicillin allergy in CAP has been observed in patients who need combination therapy.
It should be noted that most of recommendations of combination therapy in severe CAP are based upon either descriptive works (including some case-control studies) or consensus of experts. In fact, as far as we know, prospective, randomized trials specifically designed to ascertain the safety and efficacy of the different regimens for the treatment of CAP in patients admitted to ICU remains rare.4–10 The “atypical” bacteria have been reported to a wide range of frequencies among these patients. Moreover, besides the lethal potential of Legionella species,101–103 one study suggested that the incidence of this pathogen in severe CAP is being replaced by others “atypical” bacteria, such as M. pneumoniae and C. pneumoniae. 104
Thus, in view of the high mortality rate of severe CAP, the inclusion of an antibiotic proving active against “atypical” bacteria beside the β-lactams, such as fluoroquinolones or macrolides, in the initial therapy appears to be mandatory.4–10
The CAP Australian guidelines recommend that all severe CAP patients receive combination therapy 98 with two exceptions: moxifloxacin/gatifloxacin is recommended for patients with immediate hypersensitivity to penicillin. Torres et al 105 have published that in patients with severe CAP (Pneumonia Severity Index V) moxifloxacin (n = 169) was noninferior to treatment with ceftriaxone plus levofloxacin combination therapy (n = 167). Clinical cure rates at test of cure 84.8% and 86.8% respectively.
At the same time, it is possible to consider using tigecycline like monotherapy in patients with allergy to the β-lactams that require combination treatment? Only 2 patients with PSI V were included in the tigecycline arm of the CAP clinical trial, 38 that's why, the data are insufficient to support this recommendation.
However, tigecycline includes in its spectrum all the microrganisms which are covered by the combination β-lactams plus macrolides or fluoroquinolones with an excellent pharmacodynamic profile in pulmonary tissue. 24
A special concern is the possibility of bacteremia in severe CAP patients and the low serum concentration which tigecycline achieve at the usual dose. As it was mentioned previously, in the clinical trial of CAP there was insignificant difference in the clinical cure between tigecycline and levofloxacin patients with S. pneumoniae bacteremia. The high concentration of tigecycline in the source of infections (lung) and the low MICQ0 of the S. pneumoniae to tigecycline could be the reasons for this findings. 38
Conclusions
Tigecycline represents an appropriate choice for empirical monotherapy in the treatment of CAP. No dosing adjustments are needed in patients with renal dysfunction or mild-to-moderate hepatic dysfunction. In addition, tigecycline is not metabolized by, nor does it inhibit or induce, cytochrome P450. As a pragmatic approach to antimicrobial agent stewardship, there are a number of factors that favor the use of a single broad-spectrum agent over combination therapy for initial treatment when the infecting pathogens are not known in a seriously ill patient, including the ease of administration, the ability to use one agent as opposed to 2 or more drugs, and a reduced likelihood of drug interactions. 12
I propose an algorithm rational to prescribe tigecycline in CAP (Fig. 3); based on its microbiological and phamacological profile, knowing that the evidence derived from the clinical trials are not adequate to prove all this conclusion. Nonetheless, tigecycline is not active against against P. aeruginosa, which is a significant pathogen, not only in VAP, HCAP, but also in CAP in patients with specific risk factors.
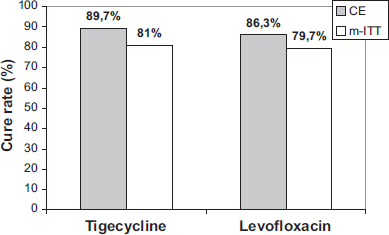
Clinical cure rates in community-acquired pneumonia phase 3 studies of tigecycline compared with levofloxacin.
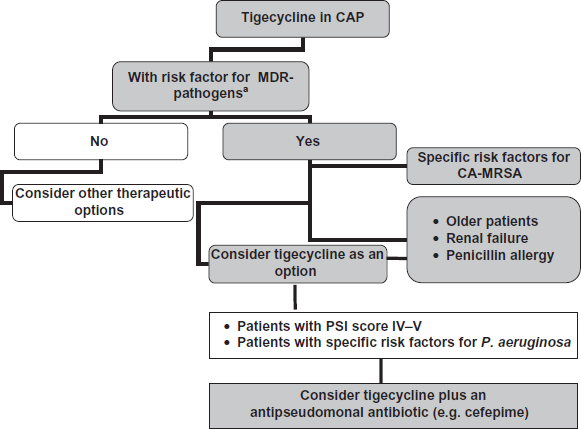
Algorithm rational to prescribe tigecycline in Community-Acquired Pneumonia (CAP).
In sumary, tigecycline would be an appropriate consideration for CAP when there are medical concerns for MDR, an elevated severity score mandating inpatient care, and care paradigm requires intravenous antibiotics.
Only controlled new clinical trials will provide sufficient evidence to support approval for different clinical scenarios (i.e. tigecycline in severe CAP).
Transparency Declarations
DC is speaker of Wyeth Laboratories Argentina for Tygacil®.
Disclosure
The author reports no conflicts of interest.
