Abstract
Hypertension is the most important preventable cause of premature death in the developed countries, and the benefits of antihypertensive drugs regarding cardiovascular morbidity and mortality are well established. Nevertheless, adequate control is achieved only in the minority of patients. All official guidelines recommend blood pressure targets of 140/90 mmHg in every patient and even lower in high-risk groups, and support the use of two of more drugs as initial therapy in specific populations. Multiple drugs acting by different, but complementary, mechanisms of action are increasingly implemented in the treatment of hypertension and fixed-dose combinations are gaining popularity. Simplifying therapy is particularly important for elderly patients, who are more likely to have co-morbid conditions and to be taking multiple medications. The once-daily administration of a fixed dose enalapril/lercanidipine effectively reduces blood pressure in patients inadequately controlled by either component, is generally well tolerated and exhibits a favourable metabolic profile.
Hypertension is highly prevalent in both developing and developed countries and represents a major modifiable risk factor for cardiovascular morbidity and mortality. About a quarter of the world's adult population had the disease in 2000 and this proportion will increase to 30% by 2025. 1 The association between inadequate blood pressure control and worse cardiovascular outcome remains constant and strong among all age groups.2,3 Data from observational studies in individuals with uncomplicated hypertension suggest that, within the range of 115/75 mmHg to 185/115, the risk of vascular mortality approximately doubles for every 20 mmHg or 10 mmHg increase in systolic and diastolic blood pressure respectively. 4 When other cardiovascular risk factors, such as diabetes or chronic kidney disease are present, the risk is even higher.
Blood pressure lowering is the most important issue in the management of hypertension. Treatment with any regimen reduces the risk of major cardiovascular events and larger reductions in blood pressure produce larger reductions in risk. 5 Furthermore, in high risk populations early response to antihypertensive therapy is a critical determinant of outcome. In a post hoc analysis of data from the VALUE (Valsartan Antihypertensive Long-Term Use Evaluation) trial, patients whose hypertension responded immediately and those in whom target levels were attained within 6 months, demonstrated significant reductions in the risk of cardiovascular events, irrespective of the medication used. 6
Nevertheless, adequate blood pressure control is difficult to achieve and the co administration of two or more drugs is necessary in the majority of the patients. In the most recent US National Health Survey (2003-2004), only 37% of patients being treated for hypertension had blood pressure levels below 140/90 mmHg. 7 In the Losartan Intervention for Endpoint (LIFE) trial, >90% of the patients required more than one antihypertensive agent 8 and only one third of the participants in the Hypertension Optimal Treatment (HOT) trial reached target blood pressure with monotherapy. 9 Therefore, recent recommendations emphasize to consider initiating therapy with two drugs in patients with grade 2 hypertension or in those with high or very high total cardiovascular risk. In all these conditions greater reductions in blood pressure are pursued, because of the higher initial values or the lower target levels.10,11
Introduction to Lercanidipine and Enalapril
Antihypertensive drugs of different classes can be combined if they have different and complementary mechanisms of action, exhibit greater efficacy than the respective components and exert a favourable tolerance profile. This approach is more advantageous than the upward dose titration of one drug, where modest increases in blood pressure response are accompanied by a disproportional increase in adverse effects. 11 Furthermore, fixed dose combinations of the various agents in a single tablet for once daily administration are available. Simplifying therapy by reducing the number of pills leads to enhanced compliance and this is particularly important for elderly patients, who are more likely to have co-morbid conditions and to be taking multiple medications.
A pharmacological rationale underpins the use of fixed-dose combination of an angiotensin converting enzyme inhibitior (ACEI) and a long acting dihydropyridine calcium channel antagonist (DHP-CA). The DHP-CA is a potent vasodilator that activates the sympathetic nervous system and stimulates renin secretion, events, which can be buffered by the addition of an ACEI. Furthermore, it is known that the incidence of adverse effects increases dose dependently during calcium entry blockade and that ACE inhibitors have the capacity to preserve or even improve the tolerability profile of calcium blockers. Especially the incidence of peripheral edema can be considerably reduced, because of improved local hemodynamics (venodilation induced by the ACEI) and by using a lower dose of the calcium antagonist. In addition, other, mostly concentration–related adverse effects, such as constipation or headache, may occur less frequently with the lower dose of calcium channel blocker provided in the combination preparations. Lastly, calcium antagonists display a neutral profile regarding lipid metabolism and glucose tolerance and ACEI can improve insulin sensitivity and prevent or delay the onset of diabetes in hypertensive individuals.12–14
Enalapril and lercanidipine are well-established antihypertensive medications. Recently, their fixed dose combination has been approved for the treatment of hypertension in Germany. Although the experience is limited and large-scale comparative trials are lacking, the available results are promising. Other detailed overviews appear elsewhere.15,16
Pharmacology
Since limited data have been published about the fixed-dose combination, basic pharmacodynamic and pharmacokinetic aspects of their individual components will be presented in the following sections. Experimental data and available clinical studies will be included.
Chemistry
Lercanidipine hydrochloride
Lercanidipine is a dihydropyridine (DHP) derivative. It is a racemate due to the presence of a chiral carbon atom at position 4 of the 1,4-dihydropyridine ring.
The chemical name is 3,5-pyridinedicarboxylic acid, 1,4- dihydro-2, 6-dimethyl-4-(3-nitrophenyl)-2-[(3,3-diphenylpropyl)methylamino]-1,1 dimethylethyl methyl ester hydrochloride and the MW is 648.2 (free base: 611.7). Lercanidipine hydrochloride is a microcrystalline, odourless, citrine powder, readily soluble in chloroform and methanol, practically insoluble in water. Its chemical structure is shown in Figure 1.
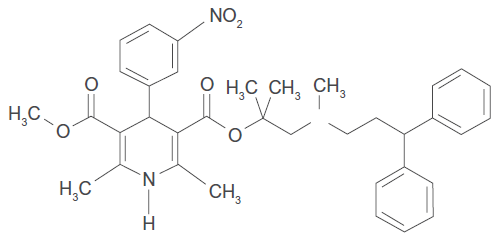
Lecarnidipne hydrochloride.
Enalapril maleate is the maleate salt of enalapril, the ethyl ester of a long-acting angiotensin converting enzyme inhibitor, enalaprilat. Enalapril maleate is chemically described as (S)-1-[N-[1-(ethoxycarbonyl)-3-phenylpropyl]-L-alanyl]-L-proline, (Z)-2-butenedioate salt (1:1). Its empirical formula is C20H28N2O5•C4H4O4, and its structural formula is shown in Figure 2. Enalapril maleate is a white to off-white, crystalline powder with a molecular weight of 492.53. It is sparingly soluble in water, soluble in ethanol, and freely soluble in methanol.
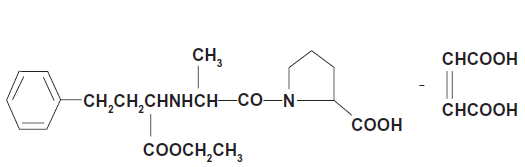
Enalapril maleate.
Lercanidipine
Mechanism of action
Three main classes of calcium antagonists are currently used: the benzodiazepines (diltiazem), the phenylalkylamines (verapamil), and the dihydropyridines. Despite their chemical diversity, they share the common property of blocking the transmembrane flow of calcium ions through voltage gated L-type calcium channels in the cell membrane. The L-type calcium channel was first isolated from cardiac muscle and was since then detected in vascular smooth muscle (arteriolar and venous) and a variety of nonvascular and noncontractile tissues. Blockade of L-type channels in vascular tissues results in the relaxation of vascular smooth muscle and in cardiac tissue results in a negative inotropic effect. 17
Lercanidipine belongs together with amlodipine and lacidipine to the third generation dihydropyridines, which are characterized by a slow onset, a long duration and a slow offset of action. The antihypertensive effect is primarily attributed to the S-enantiomer and results from peripheral vasodilation and decreased total peripheral resistance. Due to its high lipophilicity, lercanidipine is easily taken up and stored in the hydrophobic compartment of the lipid bilayer of the cell membrane, which enables a gradual onset, and long duration of action despite a relatively short plasma half-life. Numerous data from in vitro and ex vivo studies are available.18,19 In patients with mild to moderate hypertension blood pressure reductions assessed via ambulatory monitoring persisted over 24 hours after a single dose of lercanidipine 10 or 20 mg and a through to peak ratio of over 50% was measured. 20
Cardiovascular effects
Lercanidipine also exhibits some other important pharmacodynamic characteristics. It is highly selective for the vascular smooth muscle over other smooth muscle types, as has been corroborated in animal models. The relaxant potency in the rat aorta was 177-fold higher than in the rat bladder and 8-fold higher than in the rat colon. 21 The high vascular selectivity has been attributed to the higher density of the L-type channels in the vasculature. 22 In addition, at least in experimental settings, it has the lowest cardiodepressant activity among the DHP calcium blockers. Even in comparison with amlodipine, lercanidipine expressed a 4.4-fold lower negative inotropic action. 23 Much more pronounced differences have been observed with the other compounds. In the clinical setting, direct and indirect evidence has been obtained from comparative and non-comparative studies. In particular, the impact of the agent on heart rate, and sympathetic activation has been shown to be minimal. For example, in comparison to extended release nifedipine, the drug given at the usual therapeutic doses did not cause reflex tachycardia or other signs of sympathetic activation.24,25 In another open label, observational, non-comparative study, involving nearly 3000 patients at medium to high or very high cardiovascular risk (as defined by the ESH/ESC guidelines), lercanidipine, administered for 6 months, exhibited besides the good antihypertensive action a favourable tolerability profile, without significant hemodynamic or sympathetic adverse effects. 26
Organprotective actions (in vitro data and clinical studies)
Regarding the target organ damage, sound experimental data have been gathered. Menne et al recently elucidated novel molecular and cellular mechanisms of action, using transgenic rat models (of angiotensin II mediated organ damage) and endothelial and vascular smooth muscle cell cultures. Inhibition of PKC activation and reduced levels of the endogenous NO inhibitor asymmetric dimethyl arginine (ADMA) was observed. 27 In another animal model of hypertension lercanidipine reversed endothelial dysfunction through antioxidant effects and reduced matrix metalloproteinase 2 activation. 28 In vitro, antiatherogenic actions through effects on cholesterol accumulation and matrix metalloproteinase's secretion by macrophages have been described. 29 In clinical studies of patients with essential hypertension and /or diabetes, antioxidant and favourable vasodilatory effects have been demonstrated. Lercanidipine decreased oxygen-free radicals, 30 reduced significantly and to a similar extend with Losartan the levels of LDL oxidation 31 and exerted prolonged beneficial effects in the microcirculation. 32
There are ample experimental data about the nephroprotective properties of lercanidipine but the respective evidence in humans can be regarded as inconclusive. In spontaneously hypertensive rats it inhibited glomerular hypertrophy, induced similar degree of vasodilation in the afferent and efferent arteriole, reduced albuminuria and restricted inflammation. 33 In diabetic hypertensive patients with microalbuminuria, lercanidipine compared with ramipril lead to similar reductions in blood pressure and albumin excretion rate. 34 In patients with impaired kidney function and proteinuria, administration of lercanidipine resulted in optimal blood pressure control, reduction in proteinuria and preservation or slight improvement in creatinine clearance, but additive renoprotective effects that go beyond blood pressure reduction have yet to be demonstrated. 35 Nevertheless, it should be remembered that concerns have been raised about the safety of DHP calcium blockers when given as monotherapy in patients with significant proteinuria. 36
Finally, data published only as abstracts and not as full papers, indicate a favourable effect of lercanidipine in the regression of left ventricular hypertrophy in individuals with hypertension and a reduced incidence of stroke in spontaneously hypertensive rats reviewed in. 33
Enalapril
Mechanism of action and antihypertensive efficacy
Enalapril is an orally administered prodrug, which is deesterified in the liver to form the active ACE inhibitor enalaprilat. ACE is a peptidyl dipeptidase that catalyzes the conversion of angiotensin I to the vasoconstrictor substance angiotensin II. Inhibition of ACE results in decreased plasma angiotensin II, which leads to decrease vasopressor activity and decreased aldosteron secretion. Removal of the angiotensin II negative feedback on renin secretion increases plasma renin activity. The beneficial effects of enalapril in hypertension appear to result primarily from suppression of the renin-angiotensin-aldosterone system. Administration of enalapril in patients with hypertension results in a reduction of both supine and standing blood pressure. Although symptomatic postural hypertension is infrequent it might be anticipated in volume-depleted patients. 37 The antihypertensive effects are dose dependent at doses until 20 mg. 38 The onset of antihypertensive activity is seen at one hour after oral administration, with peak reduction of blood pressure achieved by 4 to 6 hours. 38 At recommended doses, antihypertensive effects have been maintained for at least 24 hours 39 and achievement of optimal blood pressure reduction may require several weeks of therapy. 37 In hemodynamic studies of patients with essential hypertension, blood pressure reduction was accompanied by a reduction in peripheral arterial resistance with an increase in cardiac output and little or no change in heart rate. 37
Organprotective actions
In addition, ACE inhibitors have well known renoprotective properties, at least but not only, by reducing glomerular capillary pressure. 40 Enalapril compared with placebo attenuated the decline in renal function and prevented the development of microalbuminuria in normotensive and normoalbuminuric individuals with type 2 diabetes mellitus. 41 Furthermore, recent data from trials in patients with hypertension and/or diabetes demonstrated pleiotropic effects of enalapril in microvascular remodelling, carotid artery intima-media thickness, platelet activation, coagulation factors and inflammatory markers of atherosclerosis and endothelial dysfunction.42–44 This is in accordance with older well-designed, multicenter, placebo control studies in patients with heart failure, where a significant reduction in morbidity and mortality was observed.45,46
Pharmacokinetic properties
The pharmacokinetic profile of the fixed–dose combination was examined only in one study, provided by the manufacturer, and was found to be similar of that of the individual components. 15 Therefore, the majority of the information presented in this section relates to studies performed in healthy volunteers or patients with essential hypertension who received either lercanidipine or enalapril alone. Most data are retrieved from previous excellent reviews.15,16,33,37,47,48
The main pharmacokinetic characteristics are summarized in Table 1.
Pharmacokinetic profile of enalaprilat and lercanidipine.

Pharmacokinetic profile of enalaprilat and lercanidipine.

Cmax, Time point of maximum plasma concentration; t1/2, serum half life.
Lercanidipine is rapidly and almost completely absorbed from the gastrointestinal tract. It undergoes extensive first-pass metabolism to largely inactive metabolites and has an absolute bioavailability of 10% in fed patients. Thus it should be taken before meals. Lercanidipine is highly liphophilic and exhibits a high plasma protein binding capacity (>98%), which can explain many of its characteristics: high volume of distribution, nonlinear pharmacokinetics and high membrane partition coefficient. Consequently, the drug provides a long lasting effect at receptor and membrane level allowing for once daily administration despite a relative short half-life time. On the contrary, the risk of accumulation and toxicity is greater if the elimination is impaired. Lercanidipine is metabolized by the cytochrome P450 3A4 and is excreted to a similar extend in the urine and faces. As with other DHP's, the potential for interactions with drugs that inhibit or induce cytochrome P450 exists and must be considered. For example, ingestion of grape fruit should be avoided and concomitant administration with cyclosporine is contraindicated. In a series of recently published studies involving patients older than 65 years, lercanidipine given at doses up to 20 mg exhibited a favourable antihypertensive efficacy and was well tolerated.26,49–52 Nevertheless, although the pharmacokinetics of the drug in elderly individuals or patients with mild to moderate renal or mild hepatic (Child-Pugh A) impairment are similar to that observed in the general population, caution is advised when increasing the dose to 20 mg. However, the drug is not recommended in patients with advanced renal (creatinine clearance <10 ml/min or dialysis) or hepatic insufficiency.
Enalapril
Enalapril is bioactivated in the liver to form enalaprilat, which does not undergo further metabolism and is excreted unchanged in the urine. The absolute bioavailability of oral enalapril as enalaprilat is 40% (not reduced by food) and the binding rate to plasma proteins is 60%. Enalaprilat is not absorbed orally and is only available for intravenous administration. After oral administration the peak serum concentrations of enalapril occur within about one hour. The respective concentrations of enalaprilat are achieved after 4-6 hours, which coincides with the maximum antihypertensive activity. Enalapril has a half-life of only 1.3 hours, but enalaprilat, because of tight binding to ACE, has a plasma half-life of about 11 hours. As mentioned, the primary route of elimination is the kidney and dose reduction is required in patients with renal impairment. Altered metabolism has been observed even in older patients with mild reduction in GFR (<80 ml/min). Consequently, drug accumulates in the elderly if they receive the standard dose, increasing the risk of adverse effects. In patients with advanced renal failure (mean GFR = 15 ml/min) a low dose of enalapril (1.25-5 mg) afforded the same degree of blood pressure control and renoprotection as a high dose (2.5-30 mg) with less unwanted effects. 53 Dose adjustment is not necessary in patients with hepatic dysfunction.
Efficacy Studies
The efficacy of lercanidipine and enalapril has been assessed in patients with hypertension and diabetes. Although fixed dose combination tablets are available, data from studies in which patients received the two components separately are also included. Unfortunately, apart from a few studies, most of the data are available from the manufacturer's prescribing information or have been published in abstract form. To our knowledge, no comparative trials have been conducted yet and the clinical relevance of the significant blood pressure reductions in terms of cardiovascular morbidity and mortality remains to be established.
Studies published as full papers
Puig et al recently demonstrated the additive effects of lercanidipine and enalapril in combination on systolic blood pressure in elderly patients with hypertension. 54 Seventy five patients aged 60-85 years with an office sitting systolic blood pressure between 160-179 mmHg, an office sitting diastolic blood pressure of <110 mmHg, and a mean daytime systolic blood pressure of ≥ 135 mmHg were enrolled. Individuals with severe hypertension or a history of cardio- or cerebrovascular disease were excluded. This was a randomized, double blind, placebo-controlled, four way crossover study. Every patient received the following four treatments in randomized order for 4 weeks each: lercanidipine 10 mg, enalapril 20 mg, lercanidipine 10 mg plus enalapril 20 mg and placebo. At the end of each treatment period office blood pressure was measured and a 24 h ABPM was performed. All treatments were superior in comparison to placebo. The response to combination therapy was significantly greater than that to either monotherapy, supporting a favourable interaction with a greater than additive effect on blood pressure. Particularly, the reductions in office systolic blood pressure in the combination group, the enalapril group and the lercanidipine group were 16.9 mmHg, 5.9 mmHg, and 5.0 mmHg respectively. Interestingly, the effect of the combination therapy on the ambulatory systolic blood pressure was no more than additive. Thus, the mean 24 hour systolic blood pressure was reduced by 17.9 mmHg in the combination group, by 12.9 mmHg in the enalapril and by 8.2 mmHg in the lercanidipine group (Fig. 3). Importantly, 45% of the patients receiving the two medications achieved a normal blood pressure (<140/90 mmHg) whereas the respective percentages in the enalapril and lercanidipine group were 18% and 19% (Table 4).
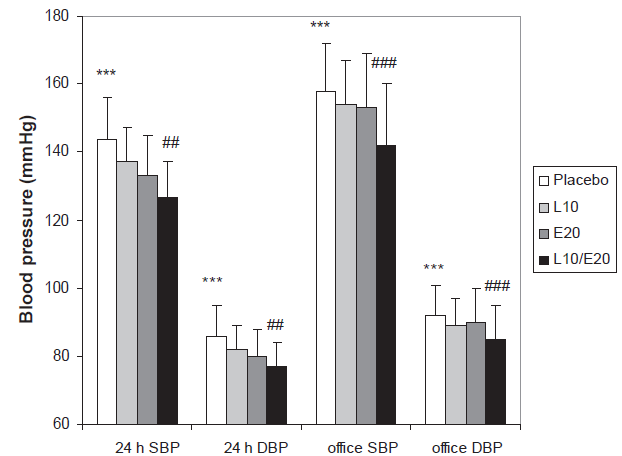
Results from a randomized, double blind four way crossover study in 62 elderly hypertensive patients. The mean 24 h systolic (24 h SBP) and diastolic (24 h DBP) from a ABPM measurement as well as the sitting office SBP and DBP are shown.
In an other study, Agrawal et al evaluated the efficacy and safety of lercanidipine versus hydrochlorothiazide as add-on therapy to enalapril in patients with diabetes mellitus and uncontrolled hypertension. 55 This multicenter, double blind, randomized trial included 174 patients aged 18-80 years. The patients were initially treated with enalapril 20 mg daily for 4 weeks. Those not adequately responding to enalapril monotherapy received for 20 weeks a combination therapy with lercanidipine 10 mg daily or hydrochlorothiazide 12.5 mg daily. The primary objective was to demonstrate the non-inferiority of lercanidipine compared to hydrochlorothiazide as add-on therapy to enalapril. At the end of the study lercanidipine decreased diastolic blood pressure by a mean of 9.3 mmHg and hydrochlorothiazide by a mean of 7.4 mmHg. The respective reductions in systolic blood pressure were 9.6 mmHg and 6 mmHg. 30.4% of the patients in the lercanidipine group versus 23.2% in the HCTZ group achieved a blood pressure below 130/85 mmHg (Table 4).
Thirty four patients with diabetes mellitus type II and a resting diastolic pressure above 90 mmHg, despite administration of 10 mg enalapril or equipotent doses of other ACE I (perindopril 4 mg in 6 cases and quinalapril 20 mg in 3 cases) were treated in an open label sequential comparison study with the same ACE inhibitor in combination with metoprolol 100 mg for 3 months, and subsequently with lercanidipine 10 mg for 3 more months. After 6 weeks on combination treatment the dose of metoprolol or lercanidipine was titrated up to 200 mg and 20 mg respectively, when the diastolic blood pressure was still greater than 90 mmHg. After 3 months on lercanidipine the mean arterial pressure fell by 6 mmHg compared to the point of withdrawal of the beta-blocker (p = 0.002). The respective reductions in systolic and diastolic blood pressure were 8 mmHg (p < 0.05) and 4 mmHg (non significant). Thus, in diabetic individuals replacement of metoprolol with lercanidipine did not cause rebound hypertension, as can be observed after withdrawal of the beta-blocker. Furthermore, the DHP calcium antagonist was shown to be a more potent second line antihypertensive agent than the beta-blocker in these patients. 56
Studies published in abstract form
The efficacy of lercanidipine as add on therapy has been evaluated in an uncontrolled study of 1562 patients with mild to moderate essential hypertension (ZANYCONTROL). Only the preliminary results are available in abstract form. The mean initial sitting office blood pressure was 160.4/94.1 mmHg. Treatment was started with lercanidipine (unfortunately the administered dose is not mentioned in the abstract) and if the blood pressure was not controlled after 1 month the patient received 20 mg of enalapril as add-on treatment. At the end of the study, which lasted 6 months 38% of the patients, received enalapril. The mean office blood pressure was 125.5/80.5 mmHg. 72% of the patients in the lercanidipine monotheray group and 61% in the combination group achieved optimal blood pressure (140/90 mmHg). 16
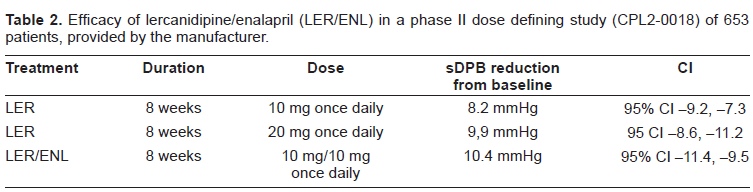
In a phase II, dose-fixing trial various combinations of lercanidipine (5, 10 or 20 mg), enalapril (5 or 10 mg) and placebo were administered in a total of 653 patients with uncomplicated mild to moderate essential hypertension. In two phase III trials, the fixed-dose combination of lercanidipine/enalapril was evaluated in hypertensive patients who where not responding sufficiently to lercanidipine or enalapril monotherapy. Details from these studies, which are provided by Recordati, the manufacturer of the medicament, and have been recently reviewed15,16 are summarized in Tables 2 and 3. The efficacy of the fixed dose combination of lercanidipine/enalapril was maintained over the long term in the 9 month non-comparative extension phase of each of the above mentioned phase III trials.15,16
Efficacy of lercanidipine/enalapril (LER/ENL) in a phase II dose defining study (CPL2-0018) of 653 patients, provided by the manufacturer.
Efficacy of lercanidipine/enalapril (LER/ENL) fixed–dose combination in phase III (CPL1-0018 and CPL1-0019) studies provided by the manufacturer.
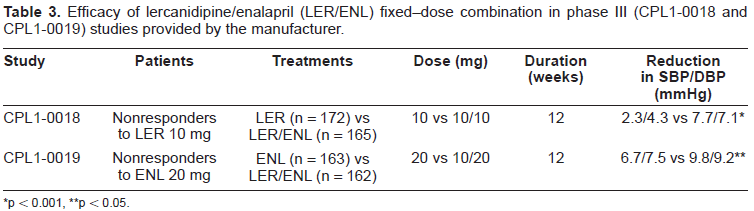
p < 0.001
p < 0.05.
The fixed dose combination of lercanidipine and enalapril is generally well tolerated in patients with mild to moderate hypertension and/or diabetes mellitus type II. The majority of patients developed adverse events that were either mild or moderate in severity and unrelated to treatment. Tolerability data are presented as a pooled safety analysis from the above mentioned phase II and III trials, their corresponding extension phases and the pharmacokinetic intervention trial.15,16
Data are also available from the published studies presented in the previous section.
According to the manufacturer, less than 5% of the patients stopped medication because of side effects. There were no differences between the combination therapy and the monotherapy groups. The most common treatment related adverse effects were cough, headache, dizziness, peripheral oedema and flushing. Cough was the most common cause of treatment discontinuation, was attributed to enalapril and occurred in nearly 5% of patients. Dizziness and headache, which occurred in 2.4% and 2.7% of patients respectively, were considered to be due to the blood lowering effect. Indeed, one patient in the phase I trial developed a true circulatory collapse. Therefore, caution is advised when using the drug in patients with reduced effective circulating volume. Only 1.5% of the patients developed peripheral edema, which is in contrast to the much higher rates reported in trials with other dihydropyridine calcium blockers. The low occurrence of edema can be explained by the peculiar pharmacodynamics of lercanidipine and its favourable interaction with enalapril as described previously. The overall incidence of another troublesome side effect of calcium antagonists, flushing, was also low, occurring in only 1.2% of patients. Notably, adverse effects specific to the ACE inhibitors such as angioedema and hyperkalemia were not reported. Finally, the fixed dose combination therapy exhibited a neutral metabolic profile since no patient developed new abnormalities in plasma levels of glucose or lipids.
The most common treatment related adverse effect reported in the study of Pugh et al was headache, which occurred more frequently with the placebo (8.5%) than with lercanidipine (2.9%), enalapril (5.7%), or combination therapy (4.2%). Discontinuation of therapy was necessary in four patients due to flushing and headache (combination group), fatigue, flushing and vertigo (combination group), dizziness (placebo group) and hypertensive crisis (placebo group). No clinically significant differences in heart rate or orthostatic hypotension were observed between the groups. Data on the incidence of peripheral edema or hyperkalaemia were not reported.
In both combination treatment groups as well as following enalapril monotherapy, the majority of patients presented adverse events that were either mild or moderate in severity and unrelated to treatment. In the study of Agraval et al treatment related adverse events concerned mostly heart rate and rhythm disorders (enalapril 1 event of arrhythmia, lercanidipine add-on 1 event of palpitation and tachycardia each, HCTZ add-on 1 event of arrhythmia and palpitation each). Overall, there were 4 treatment related adverse events in the lercanidipine (palpitation, tachycardia, nervousness and flush) and 3 events in the HCTZ add-on group (arrhythmia, palpitation, psychiatric disorder). In the lercanidipine group 2 adverse events (tachycardia and nervousness) observed in 1 patient induced a discontinuation of treatment.
As no study about drug interaction with the fixed dose combination has been published yet, the same precautions as for the individual components exist. For example, the co-administration with drugs that are strong inhibitors of the cytochrome CYP 3A4, such as cyclosporine and ketoconazole is contraindicated. 47 Finally the drug should not be given at any time point during pregnancy, because of the well-known toxic effects of ACE inhibitors to the fetus 57 and the rarity or non availability of data concerning lercanidipine.
Patient Focused Perspectives
Consistent control of blood pressure requires that patients follow medication and dietary regimens. However, antihypertensive therapy may have untoward side effects and result in little symptomatic relief, since hypertension often causes no symptoms. 58 Moreover, as has been shown in several intervention trials, an average of two to four antihypertensive agents are required to achieve effective blood pressure control to target levels. One must also consider that, especially in the elderly, the treatment regimens may become even more complex due to the co-existence of various comorbid conditions, such as diabetes, coronary heart disease and heart failure. 59 Thus, poor compliance to medication regimen contributes to the practice – outcome gap, in which clinical guidelines are implemented but expected benefits are not realized. 60 Even in clinical trials where patients receive special attention and are carefully selected, adherence rates of only 43 to 78 percent have been reported. 58 Finally, the higher frequency of adverse effects seen with higher doses of antihypertensive drugs explains the reluctance of primary care physicians to increase or modify the initially chosen medication despite the inadequate blood pressure control (therapeutic inertia). 61
There is little question that the use of fixed-dose combination antihypertensive therapy can improve patient compliance. A meta analysis of 9 studies comparing fixed-dose combinations versus free-drug components for the treatment of hypertension, diabetes, tuberculosis and HIV disease, demonstrated that the risk of non compliance to the medication regimen is reduced by 24%-26% with fixed dose combinations 60 In another study, hypertensive patients receiving once daily, single capsule fixed-dose combination of amlodipine–-benazepril showed significantly better adherence than patients receiving amlodipine and benazepril separately (80.8% vs 73.8%). 62 A recent survey objected to determine rates of compliance with antihypertensive therapy and total costs of care among elderly Medicaid recipients treated with a fixed-dose combination amlodipine besylate/benazepril versus a DHP calcium channel antagonist and an ACE inhibitor prescribed as separate agents. Compliance was significantly higher with fixed-dose versus free combination therapy (63.4% vs 49%; p < 0.001). The average total cost of care was also lower for patients receiving the fixed-dose combination (US$ 3179 vs US$ 5236). 63 Thus, fixed-dose combination antihypertensive products will find increased use in the coming years and physicians will embrace multi drug therapy to meet the currently advocated lower blood pressure goals. Unfortunately, regarding the fixed dose combination of enalapril/lercanidipine, direct evidence about compliance rates, healthcare costs or head to head comparisons with other therapeutic regimens are lacking. Nevertheless, the available data indicate that the once daily administration of a fixed-dose lercanidipine/enalapril 10 mg/10 mg or 10 mg/20 mg: a) improves blood pressure control in patients not responding to monotherapy with the separate components; and b) exerts a favourable tolerability profile. Treatment discontinuation due to serious adverse events was reported only in a substantial minority of the patients (5%).
Efficacy lercanidipine/enalapril (LER/ENL) in published studies.
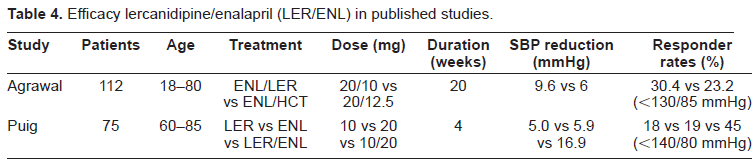
Efficacy lercanidipine/enalapril (LER/ENL) in published studies.

Treatment guidelines
Despite the evidence for a decrease in cardiovascular morbidity and mortality from normalizing blood pressure by several classes of antihypertensive drugs, the control of arterial hypertension remains disappointingly inadequate. As already mentioned, initial therapy for hypertension with a combination of drugs is recommended by both the seventh report of the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure and (JNC 7) and the European Society of Hypertension-European Society of Cardiology (ESH-ESC) guidelines for patients whose blood pressure are 20/10 mmHg or more above their treatment goals. Therefore combination therapies are gaining popularity. However, the treatment strategies vary. For instance, according to the ESH-ESC guidelines any of the major drug classes is suitable for therapy initiation and therefore combinations that do not include diuretics could be considered. Conversely, the JNC 7 guidelines advocate the use of diuretics as the basis of treatment, either alone or in combination with other agents. Nevertheless, both agree that clinicians should modify drug selection on the basis of patient characteristics and comorbid conditions. Elderly and African-American patients tend to be salt-sensitive and respond well to diuretics. Younger patients respond well to suppression of the RAS and an ACEI or ARB may be a good initial choice. ACE inhibitors are particularly helpful in patients with diabetes and microalbuminuria. Patients with heart failure can benefit from ACEI diuretics, β-blockers and ARB's. β-blockers and ACEI are good antihypertensive agents for patients with a history of myocardial infarction. Lastly, in patients with renal insufficiency and proteinuria ACEI can retard progression. Regarding treatment of hypertension three other important considerations should me made.
New-onset diabetes mellitus during antihypertensive therapy
First, the incidence of diabetes mellitus is alarmingly increasing. According to an analysis, the number of patients with diabetes is estimated to increase by 122% from 135 millions in 1995 to 300 millions in 2025. The worldwide prevalence of diabetes will also increase during the same period by 35%, from 4 to 5.4%. 64 Hypertension is often associated with impaired glucose tolerance, insulin resistance and obesity, and many patients develop diabetes, even when treated with placebo. Although new onset diabetes is well described with the use of thiazide diuretics, the concomitant use of RAS blockers is thought to be protective, so their combination appears appealing,14,65 but recent evidence advises for care. In a multicenter trial, changes in 2-h OGTT were assessed after the introduction of fixed dose combination therapy with tandolapril/verapamil or losartan/hydrochlorothiazide for reduction of blood pressure in patients with metabolic syndrome and impaired glucose tolerance. A significantly higher proportion of patients in the ACE inhibitor /diuretic group had higher HbA1c, insulin levels and 2-h OGTT values, consistent with a diagnosis of diabetes (11 vs 26%), within one year. These data support the notion that use of a thiazide diuretic at a commonly used dose worsens glucose tolerance even in the presence of a high-dose RAS blocker. 66 Furthermore, in a subgroup analysis of the ALLHAT trial, in participants with metabolic syndrome but without diabetes, at 4 years of follow up, the incidence of newly diagnosed diabetes was 12.6% for lisinopril 16.0% for amlodipine and 17,1% for chlorthalidone. For those without the metabolic syndrome, the respective rates of newly diagnosed diabetes were 4.7%, 4.2% and 7.7%. 67 Admittedly, although the metabolic effects of the various drugs have not resulted in differences in cardiovascular outcomes, it should be noticed that the complications of such drug induced diabetes may take several more years to become overt. 68 Thus, according to some investigators diuretics should not be considered as first line treatment in patients with uncomplicated hypertension. 69
Comparison of the various fixed-dose combinations of antihypertensive agents
Second, until recently most previous comparison trials of the various combination treatment regimens have failed to show significant differences regarding major outcomes, as long as equivalent decreases in blood pressure were achieved. 70 Even in the Anglo-Scandinavian Cardiac Outcomes Trial (ASCOT), the 23% reduction in the secondary end point of fatal and nonfatal stroke in the perindopril+amlodipine treatment group should be interpreted in the context of the greater average blood pressure level reduction (2.7 mmHg systolic and 1.9 mmHg diastolic), although the investigators content that the benefits seem to be somewhat greater than might be anticipated from the observed difference in blood pressure. 71 But for the first time, conclusive evidence is provided by a randomized, double blind controlled study. The “Avoiding Cardiovascular Events through Combination Therapy in Patients Living with Systolic Hypertension” (ACCOMPLISH) trial aimed at comparing the effects of two commonly used types of antihypertensive drug combinations (benazepril combined with either amlodipine or hydrochlorothiazide) on cardiovascular outcome in patients with high risk hypertension. The results were impressive and lead to the premature termination of the study after a mean follow up of 36 months. The mean systolic and diastolic blood pressures were 131.6/73.3 mmHg in the benazepril-amlodipine group and 132.5/74.4 in the benazepril-hydrochlorothiazid group. Importantly, blood pressure control, which was defined as a blood pressure of less than 140/90 mmHg, was attained in more than 70% of patients in both treatment groups. The primary outcome event (a composite of death from cardiovascular causes and cardiovascular events) occurred in 9.6% of patients in the benazepril-amlodipine group and in 11.8% of patients in the benazepril-hydrochlorothiazide group, representing an absolute risk reduction of 2.2% and a relative risk reduction of approximately 20% (Fig. 4). 72 Consequently, the evidence provided by the ACCOMPLISH trial, namely the excellent blood pressure control and the clear benefit regarding cardiovascular outcomes with the combination therapy of a calcium antagonist and an angiotensin converting enzyme inhibitor, gives further support to recommendations favouring the initiation of therapy with 2 or more drugs in patients with severe or complicated hypertension, and will considerably influence clinical practice. Nevertheless, these results should not diminish the value of treatment with the combination of an ACE inhibitor and a diuretic. In the recently published Hypertension in the Very Elderly Trial (HYVET) the combination of perindopril and indapamide lead to a significant reduction in mortality and morbidity. 2 Furthermore, in the blood pressure lowering part of the Action in Diabetes and Vascular disease (ADVANCE) trial those taking perindopril and indapamide had lower blood pressure (5, 6 mmHg systolic, 2, 2 mmHg diastolic) than did those on placebo, and a 9% reduction in the relative risk of major macrovascular or microvascular complications. 73
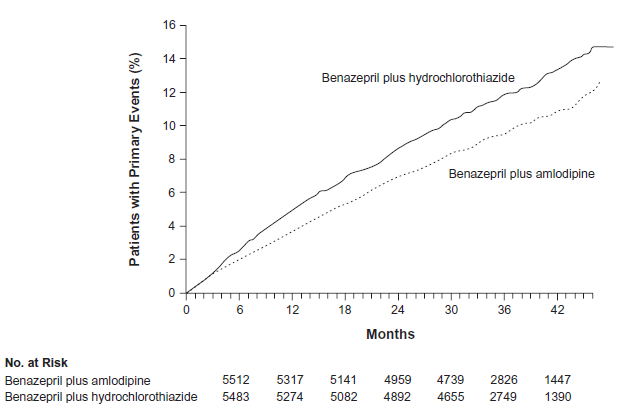
ACOOMPLISH Study. Caplan-Meier curves for time to first primary composite end point. There were 552 pts with events (9.6%) in the benazepril-hydrochlorothiazide group, as compared with 679 patients with events (11.8%) in the benazepril-amlodipine group (Adapted from Jamerson et al 72 with permission).
Thirdly, the available evidence does not support the hypothesis that ACE inhibitors and ARB have clinically meaningful differences in benefits or harms for individuals with essential hypertension. On the contrary, their combination do not seem to have advantages, as has been shown in the ONTARGET (Ongoing Telmisartan Alone and in Combination Ramipril Global Endpoint Trial) study. 74 Especially in the subgroup with impaired renal function, the combination therapy lead to more adverse events and a faster decline in glomerular filtration rate. 75
In conclusion, it should be again emphasized, that treatment with any commonly used regimen reduces the risk of major cardiovascular events and greater reductions in blood pressure produce greater reductions in risk. 5 Thus, although additional benefits may be provided by a specific drug or drug combination, the most important aspect of treatment is the reduction of blood pressure to goal levels. Additionally, in view of the diabetic epidemic, particular attention should be paid to patients with the metabolic syndrome and attempts should be made to minimize the occurrence of new onset diabetes mellitus during treatment of hypertension. The fixed dose combination of lercanidipine/enalapril could be a valuable option, since it offers the potential to: improve blood pressure goal attainment rates, reduce adverse events, increase compliance, not affect glucose and lipid metabolism and lower treatment costs.
Disclosure
The preparation of this review was not supported by any external founding. The author(s) declare that they have no competing interests.
