Abstract
Mycophenolic acid inhibits an enzyme, inosine monophosphate dehydrogenase (IMPDH), blocking purine synthesis of lymphocytes and therefore functioning as an effective immunosuppressive agent in transplantation. Currently, there are two available forms of mycophenolic acid (MPA) available; mycophenolate mofetil (MMF) and enteric-coated, delayed-release mycophenolate sodium (EC-MPS). Both products are approved for prophylaxis of organ rejection in renal transplant recipients. The use of MPA may be associated with adverse gastrointestinal effects which can lead to a reduction of the dose or discontinuation of therapy. Enteric-coated MPS was developed to reduce the upper gastrointestinal side effects due to its delayed release in the small intestines. Similar systemic MPA exposure is provided by oral administration of MMF 1000 mg daily and EC-MPS 720 mg, which contain near equimolar MPA content. Clinical trials in renal transplant recipients have demonstrated that EC-MPS is therapeutically equivalent to MMF when used at the time of transplantation and when used for conversion for gastrointestinal complications. The available literature regarding the incidence and severity of gastrointestinal adverse effects and the impact on quality of life remains controversial. Prospective, randomized trials of the available MPA formulations are warranted to further explore the gastrointestinal adverse effect profiles.
Introduction
Mycophenolic acid (MPA) is well established as an immunosuppressive agent for use in renal transplantation patients. Currently there are two forms of MPA available on the market, mycophenolate mofetil (MMF, CellCept®, Roche Laboratories, Nutley, New Jersey, USA) and mycophenolate sodium (EC-MPS, Myfortic®, Novartis Pharmaceuticals, East Hanover, New Jersey, USA). The use of MPA may be associated with adverse gastrointestinal effects which can lead to a reduction of the dose or discontinuation of therapy. Enteric-coated, delayed-release MPS was developed to reduce upper gastrointestinal side effects and as an alternate treatment option in patients who cannot tolerate MMF. This review article analyzes available data published to date regarding MPA preparations and compares the efficacy and safety, including the incidence of gastrointestinal side effects, of MMF to EC-MPS.
Mechanism of Action
Mycophenolic acid exhibits an immunosuppressive effect by non-competitively inhibiting inosine monophosphate dehydrogenase (IMPDH), which inhibits lymphocyte purine synthesis. Inhibition of IMDPH causes inhibition of de novo guanosine nucleotide synthesis, therefore exhibiting a cytostatic effect on T and B lymphocytes. 1 Inosine monophosphate dehydrogenase is the rate limiting step in converting inosine monophosphate (IMP) to guanosine monophosphate (GMP), an important intermediate in the synthesis of lymphocyte DNA, RNA, proteins, and glycoproteins. T and B lymphocytes cannot synthesize GMP sufficiently, which is unlike other types of cells, so the cytostatic effect on lymphocytes is greater than on other types of cells. 1 By exerting this cytostatic effect on T and B lymphocytes, mycophenolic acid inhibits the production of antibodies and prevents rejection.
Pharmacokinetics
Mycophenolate mofetil (MMF) is available as 250 mg capsules, 500 mg tablets, a 200 mg/ml suspension, or intravenously (Table 1). Mycophenolate sodium (EC-MPS) is only administered orally and available as 180 and 360 mg tablets. 2 Mycophenolate mofetil and EC-MPS should be taken on an empty stomach, and should not be crushed or split, however, MMF can be taken with food, but EC-MPS has to be administered on an empty stomach. Both formulations undergo hydrolysis to form the active compound mycophenolic acid (MPA, Fig. 1). The kinetics of the two formulations differ with regard to the speed with which the drug is released from the formulation. The peak plasma concentrations of MPA occur within 1-1.5 hours after oral administration of MMF and between 1.5 and 2.5 hours after administration of EC-MPS.1,2 This difference is largely due to the fact that EC-MPS is enteric-coated and insoluble in the acidic pH of the stomach, while it is highly soluble in the neutral pH of the intestine. Similar systemic MPA exposure is provided by oral administration of MMF 1000 mg daily and EC-MPS 720 mg, which contain near equimolar MPA content. 3 Mycophenolic acid is highly, 97%-99%, bound to albumin. Therefore, the free fraction of MPA may be altered in patients with hyperbilirubinemia, severe renal impairment or hypoalbuminemia.4,5
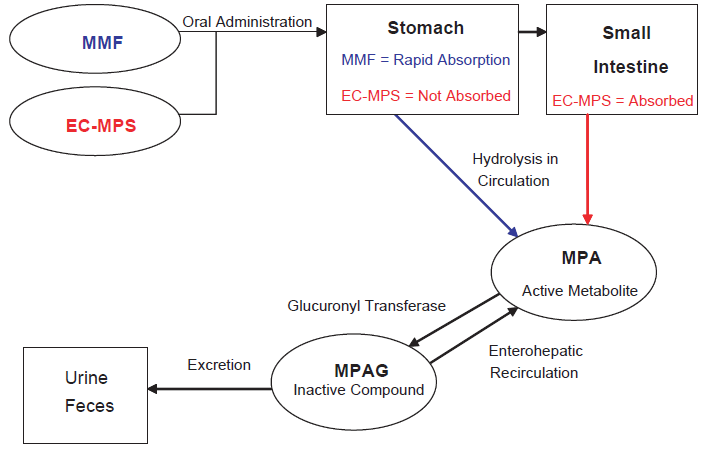
Metabolic pathway of MPA.
Comparison of mycophenolate mofetil and enteric-coated mycophenolate sodium.
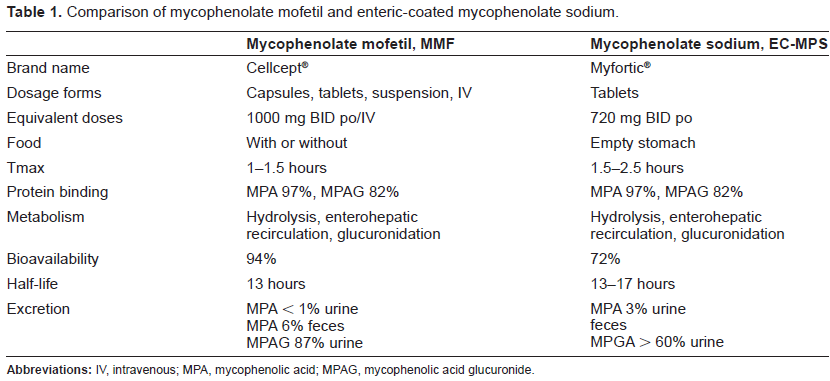
In the liver, MPA is conjugated by glucuronyl transferase to form mycophenolic acid glucuronide (MPAG), the inactive metabolite. 1 Mycophenolic acid also has a minor acyl glucuronide metabolite with similar pharmacologic activity to that of MPA. Mycophenolic acid and MPAG are excreted in bile, urine, and feces. The mean elimination half-life of MPA is 17.9 hours following oral administration of MMF and 13-17 hours following oral administration of EC-MPS.1,2 In stable renal recipients, total MPA exposure negatively associates with renal function, through accumulation of both MPA and MPAG in patients with moderately reduced renal allograft function. This is in contrast to severe graft dysfunction, where MPA clearance is higher due to increased free fraction of MPA. 6
Drug Monitoring
The clinical utility of MPA monitoring remains controversial and it is further complicated by a delayed release, enteric coated product (EC-MPS). Some authors have reported that MPA AUC correlates with rejection,7–10 while other data reveals that MPA concentrations are not correlated with effect, but rather the dose is related to renal transplant recipients’ outcomes. 11 Pharmacokinetic data has shown that 1000 mg of MMF and 720 mg of EC-MPS deliver similar systemic exposure of MPA using estimated through 12 hour MPA AUCs (area under the time concentration curve), 3 which are unrealistic in the clinic setting because of multiple blood draws. Furthermore, although AUCs are similar between MMF and EC-MPS, the tmax is delayed in the EC-MPS product, leading to the possibility that single point concentration monitoring or abbreviated AUCs may not accurately predict full 12 hour AUCs.12,13 Currently, evidence does not exist to recommend therapeutic drug monitoring of EC-MPS. In fact one study demonstrates that trough levels may be 30% higher with EC-MPS when compared to MMF, while AUCs are similar. 14 Further study of EC-MPS pharmacokinetics is needed to determine a clinically useful tool to help predict EC-MPS efficacy and toxicity.
Adverse Events
The most common adverse events associated with the use of MMF and EC-MPS, including nausea, vomiting, diarrhea, and leucopenia, are typically dose-related.1,2 Other adverse events, reported in greater than 20% of patients, may include pain, fever, headache, infection, asthenia, chest pain, sepsis, anemia, thrombocytopenia, leukocytosis, hypertension, hypotension, tachycardia, edema, anorexia, dyspnea, tremor, and insomnia.1,2
Drug Interactions
Several factors may influence mycophenolic acid levels including concomitant administration of immunosuppressive agents. Cyclosporine may lower MPA plasma concentration through inhibition of enterohepatic recirculation, while tacrolimus does not have this effect. 15 Likewise, higher MPA exposure was reported in patients receiving MMF and sirolimus as compared to those receiving concurrent cyclosporine.16,17 Thus it may be important to closely monitor a patient when changing immunosuppressive regimens. Neither MMF nor EC-MPS have been studied with concomitant azathioprine, but it is not recommended to use in combination since both drugs inhibit purine metabolism.
Mycophenolic acid, the active form of both drugs, competes with acyclovir and ganciclovir for tubular secretion, therefore increasing the concentration of both drugs in the body and increasing the potential for hematological or gastrointestinal toxicity. Patients taking concomitant therapy should be monitored closely.
Antacids containing magnesium and aluminum hydroxide and calcium free phosphate binders can decrease the absorption of both MMF and EC-MPS when administered at the same time. It is recommended that if patients need to take these medications, they should be separated by at least 2-4 hours of administration of either drug.
Cholestyramine and other drugs that bind bile acid resins should be avoided completely with MMF and EC-MPS. These drugs reduce MPA exposure by interrupting enterohepatic recirculation of MPAG.
When studied with oral contraceptives, both MMF and EC-MPS showed much interpatient variability. Additional birth control methods should be considered.
Pivotal Clinical Trials
There are two major clinical trials that are published comparing the safety and efficacy of MMF to EC-MPS18,19 (Table 2). The trials showed that EC-MPS is therapeutically equivalent to MMF, and that both drugs have a similar incidence and severity of side effects. The evidence from these trials was the basis of market approval of EC-MPS.
The first pivotal trial, a phase III, international, randomized, double-blinded, double-dummy, multicenter, 12 month, parallel group trial, compared the incidence of gastrointestinal adverse events and neutropenia at three months of treatment and evaluated whether maintenance renal transplant patients receiving MMF could be converted to EC-MPS. 18 Patients that were included in the study were male and female, age 18-75 that had undergone primary or secondary deceased donor or living donor kidney transplantation, already on established immunosuppressive therapy of MMF and cyclosporine with or without corticosteroids, and were at least six months post transplant. The primary endpoint was the incidence and severity of gastrointestinal adverse events at three months and neutropenia within the first three months of study drug administration. The intent-to-treat trial enrolled 163 into the MMF group and 159 patients in the EC-MPS group.
The incidence and severity of gastrointestinal adverse events at three months was not statistically significant between groups (MMF 20.9% vs. EC-MPS 26.4%). Incidence of upper gastrointestinal adverse events, defined by occurrence of nausea, dyspepsia, abdominal upper pain, gastroesophageal reflux disease, esophageal reflux, gastritis, and anorexia, was similar between both groups (MMF 13.5% vs. EC-MPS 13.2%). Non-upper gastrointestinal adverse events occurred in 12.9% of MMF-treated patients and 18.2% of EC-MPS- treated patients. Secondary measures of gastrointestinal adverse events at six and nine months also showed a similar incidence between drugs and a statistically significant difference was not shown. The occurrence of neutropenia, defined as a low absolute neutrophil count of <1500 cells/mm3 within the first 3 months of treatment was not statistically different between groups (3.1% MMF and 0.6% EC-MPS; 95% CI: (-6.74, +0.80)). These results remained unchanged throughout the remainder of the study.
Summary of pivotal trials.
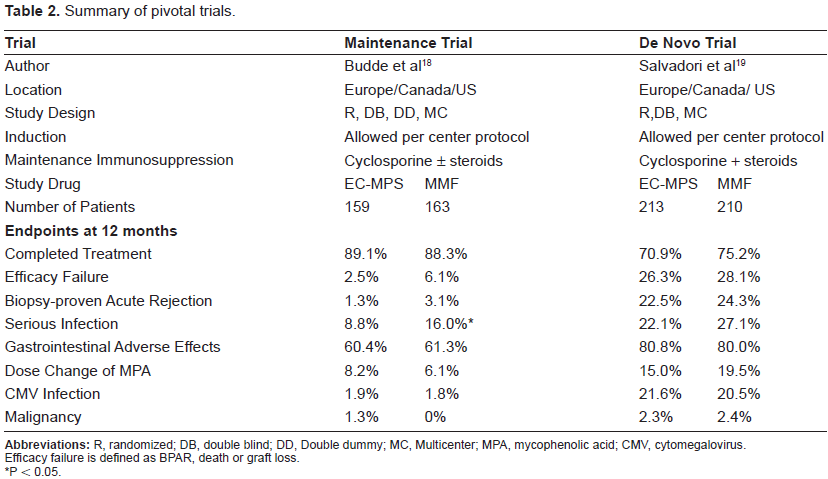
P < 0.05.
Both MMF and EC-MPS were shown to be equally efficacious without statistically significant differences in graft loss or death. The authors concluded that patients on immunosuppressive therapy for renal transplant can safely be converted from MMF to EC-MPS with all safety and efficacy measures being comparable between groups.
The second pivotal trial evaluated MMF versus EC-MPS in de novo renal transplant recipients. This 12-month, parallel group, randomized, double-blind, international trial involving 423 patients attempted to demonstrate the therapeutic equivalence of MMF and EC-MPS and to compare their safety profiles. 19 Male and female patients aged 18-75 who were recipients of a first deceased donor, living-unrelated, or human leukocyte antigen (HLA) mismatched living-related donor kidney transplant were eligible to be included in the study. The primary efficacy measure was treatment failure, defined as the incidence of biopsy-proven acute rejection (BPAR), graft loss, death, or loss to follow up, within 6 months of the start of treatment. The safety analysis included all randomized patients who received at least one dose of study medication and had at least one safety assessment.
The trial had a sufficient sample size for claiming clinical equivalence with power set at 85% and alpha set to 0.05. There was no statistical difference found between the MMF and EC-MPS groups regarding the incidence of efficacy failure at 6 months, 25.8% and 26.2% (95% CI: -8.7, +8.0), or 12 months, 28.6% and 28.1% (95% CI: -8.0, +9.1). These results indicate clinical equivalence between the two study treatments. The overall incidence of adverse events and the safety profiles of both treatment drugs were similar. The majority of adverse events were mild or moderate and the incidence of severe adverse events, including infection, were similar between the two groups. A similar proportion of patients, 80.0% in the MMF group and 80.8% in the EC-MPS group, experienced a gastrointestinal related adverse event over the 12 month study period.
The authors concluded that EC-MPS was therapeutically equivalent to MMF and that both study drugs had a comparable safety profile. The trial was not powered to statistically detect a difference in the gastrointestinal tolerability between the two treatments and no difference in the incidence of gastrointestinal adverse events was observed.
Post-marketing Studies
There are several other clinical trials that have been published since EC-MPS was approved that have attempt to explore the gastrointestinal profiles of the two formulations of MPA.20–28 The Progris study demonstrated a significant reduction in gastrointestinal-related symptom burden and improved patient functioning and well being within 4-6 weeks after conversion from MMF to EC-MPS in patients experiencing mild to moderate gastrointestinal complications. 20 A similar study conducted in India demonstrated an improvement in gastrointestinal complications with conversion to EC-MPS. 21 Additionally, a longer, 3 month multicenter, prospective study of conversion from MMF to EC-MPS for mild to moderate gastrointestinal symptoms (myTIME) resulted in a significant improvement in gastrointestinal symptom rating scale (GSRS) regardless of calcineurin inhibitor, ethnicity or presence of diabetes. 22 The main limitation of this trial is that it did not include a control group. To address this limitation an ongoing multicenter, double-blind, double-dummy, 4 week trial (myGAIN) of patients with mild, moderate, or severe gastrointestinal complaints related to MMF will evaluate equimolar conversion to EC-MPS. Gastrointestinal symptom rating scale subscales, gastrointestinal quality of life index (GIQLI), and gastrointestinal symptomatology are being evaluated.
Other post-marketing trials have explored the use of MMF and EC-MPS in de novo renal transplant recipients.23–28 Pelletier and colleagues recently published a retrospective comparison of 217 patients that were prescribed either MMF or EC-MPS in combination with sirolimus. 23 In the trial, MMF-sirolimus treated patients required more MPA drug manipulations when compared to EC-MPA-sirolimus treated patients. Interestingly, patients that had drug manipulations also had a higher incidence of acute rejection and graft loss. 23 Likewise another, a retrospective, sequential cohort analysis of de novo renal and pancreas transplants (n = 198) resulted in more adverse event driven drug manipulations in the MMF versus EC-MPS group. 24 In contrast, several authors have not reported a difference in gastrointestinal related adverse effects between MMF and EC-MPS.24–27
Conclusion
Mycophenolate mofetil and mycophenolate sodium appear to have comparable efficacy and safety profiles (Table 1 and 2). Pivotal trials in renal transplant recipients have demonstrated that EC-MPS is therapeutically equivalent to MMF when used at the time of transplantation and when used for conversion for gastrointestinal complications. The post-marketing trials comparing MMF and EC-MPS are controversial. Some have shown a benefit in gastrointestinal rating subscales. At this time, it is not known whether or not EC-MPS will alleviate gastrointestinal adverse events. It is possible that gastrointestinal events are multifactoral (infectious etiology, related to gastroparesis or other concomitant medications) and EC-MPS may offer benefit to specific populations. If a patient fails MMF because of the gastrointestinal side effects, then the patient may benefit if switched to EC-MPS. Also, if the patient is predisposed to gastrointestinal disorders, then EC-MPS may be a better initial choice for the patient.
An economic cost analysis comparing the available formulations has not been performed to date. With the upcoming release of generic MMF, patients may see a substantial cost savings from using this drug over EC-MPS. Prospective, randomized trials fully evaluating the adverse effects and economic aspects of the available MPA formulations are warranted.
Disclosure
The authors report no conflicts of interest.
