Abstract
Chronic stable angina pectoris results from a fixed coronary arterial obstruction causing an imbalance between myocardial oxygen supply and demand. Current therapy aims to reduce cardiovascular events (vasculoprotective) thereby improving survival, and/or relieve ischemic symptoms (antianginal) thereby improving the quality of life. Vasculoprotective therapy consists of lifestyle modification, antiplatelet agents, lipid lowering therapy and angiotensin-converting enzyme (ACE) inhibitors. Conventional antianginal therapy for patients with chronic stable angina consists of beta-blockers, calcium channel blockers and nitrates, with surgical or percutaneous revascularization serving an adjunctive role. Despite the investigation of multiple novel therapies and medications over the past 25 years, arguably the most significant contribution to antianginal therapy during that time involved the recent introduction of ranolazine. Ranolazine acts via a distinctive pathway, inhibiting the late sodium current of the action potential in ischemic myocytes. Multiple studies have demonstrated that ranolazine significantly reduces anginal symptoms and improves exercise performance in patients with chronic stable angina but does not reduce mortality. Ranolazine does not affect either heart rate or blood pressure, a unique property among the current antianginal agents. Despite its QT prolongation, ranolazine has a proven safety profile and is not proarrhythmic. In fact, in a recent large randomized trial, ranolazine reduced the incidence of supraventricular tachycardia, ventricular tachycardia, new-onset atrial fibrillation and bradycardic events. Ranolazine may confer some additional benefits such as a reduction in HbA1c levels and improved left ventricular diastolic function. Ranolazine is now approved for use in chronic stable angina. Current guidelines recommend beta-blockers as the first line antianginal agent due to the proven mortality reduction. However, for patients with bradycardia or hypotension, ranolazine may be considered as initial antianginal therapy.
Introduction
Approximately 1 in 3 American adults has 1 or more forms of cardiovascular disease, and over 9 million have angina pectoris. 1 Chronic angina pectoris represents a frequent and significant problem for both the patient and the clinician. Although revascularization has proven to reduce myocardial infarction (MI) and death in patients with acute coronary syndromes (ACS), the same is not true for chronic stable angina. 2 Therapy for chronic angina currently is based predominantly on medical therapy, with revascularization serving an adjunctive role. Therapy for stable coronary artery disease (CAD) has two major focuses. The first is a vasculoprotective role, aimed to retard atherosclerotic disease progression, thereby reducing future cardiovascular events and death and improving the quantity of life. The second is an anti-ischemic (antianginal) role focused on improving the ischemic imbalance, thereby reducing the severity and frequency of anginal symptoms and improving the quality of life.
Chronic stable angina is the result of an imbalance between myocardial oxygen supply and demand in the setting of a fixed atherosclerotic coronary plaque. In patients with chronic stable angina, the median frequency of anginal episodes is 2 per week, with increased angina frequency (p < 0.0001) associated with a decreased overall feeling of wellbeing. 3 The majority of medical therapy aimed at reducing anginal symptoms is based on reducing myocardial oxygen demand by decreasing the heart rate and blood pressure, while revascularization therapy (percutaneous and surgical) is aimed at improving myocardial oxygen supply. Unfortunately, novel therapies in the field have been lacking until recently. Nitrates were first introduced in 1867, beta-blockers in 1962, coronary artery bypass graft surgery (CABG) in 1967, percutaneous transluminal coronary angioplasty in 1977 and calcium channel blockers in the early 1980s. Nearly 25 years passed before a novel antianginal therapy was available for managing these patients. Ranolazine was approved for use in January 2006 by the U.S. FDA for patients with chronic stable angina. In contrast to previous antianginal medications, ranolazine works via a unique mechanism to reduce anginal symptoms and improve exercise capacity without any effect on either the heart rate or blood pressure. 4
Vasculoprotective Therapy
Lifestyle
In addition to medical therapy, exercise, dietary modification, and smoking cessation are essential elements in managing patients with coronary artery disease. Exercise training has been proven to improve myocardial perfusion, exercise performance, endothelial function and to raise the anginal threshold in symptomatic patients with CAD. 5 Additionally, in a study of patients with stable coronary artery disease, exercise training significantly improved event-free survival at lower costs when compared to percutaneous coronary intervention (PCI). 6 Current guidelines for patients with chronic angina recommend 30-60 minutes of moderate-intensity aerobic activity 7 days a week. 7
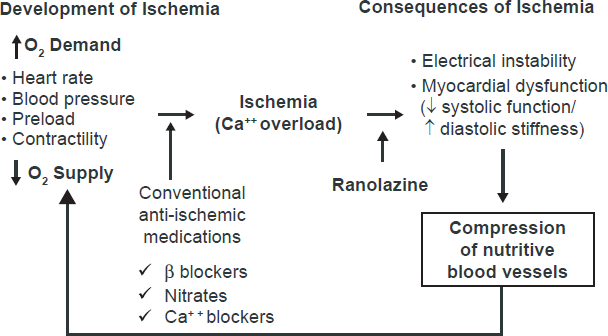
Sites of action of anti-ischemia medication. Reproduced with permission from stone ph. ranolazine: new paradigm for management of myocardial ischemia, myocardial dysfunction, and arrhythmias. cardiol clin. 2008;26:605.
Randomized controlled trials studying dietary effects in patients with stable angina are limited. However, the Lyon Diet Heart Study concluded that institution of a Mediterranean diet after a first myocardial infarction significantly reduced cardiovascular outcomes such as death and nonfatal myocardial infarction. 8 Further evidence supporting the benefits of intensive lifestyle modification derives from a variety of studies using a multifactorial approach. The Stanford Coronary Risk Intervention Project (SCRIP) demonstrated a reduced progression of angiographic coronary arterial stenosis and decreased hospitalizations for clinical cardiac events using a multifaceted lifestyle risk reduction model. 9 More recently, a study of patients with stable coronary artery disease who subscribed to a low-fat diet, engaged in moderate exercise and practiced stress management showed a significant reduction in anginal episodes, improved quality of life and improved exercise capacity. 10 The benefits of smoking cessation in patients with coronary artery disease have long been understood and should be encouraged in all patients. 11
Antiplatelet therapy
Aspirin, at a dose of 50-325 mg per day, has been proven to reduce morbidity and mortality in a variety cardiovascular disease conditions. A meta-analysis involving over 9,000 patients with stable cardiovascular disease found a significant 21% reduction in the risk of cardiovascular events, a 26% reduction in non-fatal MI, a 25% reduction in stroke and a 13% reduction in all-cause mortality, with a slightly increased risk of bleeding. 12 The treatment effect was similar for both low (50-100 mg daily) and high dosing (300 mg daily) regimens, with higher doses increasing the risk of bleeding. Although clopidogrel, a thienopyridine derivative, has been shown to reduce the composite endpoint of stroke, MI and vascular death in patients with atherosclerotic vascular disease over aspirin monotherapy the benefit was largely driven by patients with peripheral arterial disease. 13 The CHARISMA (Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management, and Avoidance) trial included a subgroup of patients with documented CAD and angina and found only a possible benefit of the addition of clopidogrel over aspirin monotherapy with an increased bleeding risk. 14 Based on these findings, aspirin has been recommended as first line antiplatelet therapy for patients with stable coronary disease.
Lipid lowering therapy
Cholesterol reduction via medical therapy and/or lifestyle modification has clear benefit in patients with CAD. The extent of data exploring the merits of HMG Co-A reductase inhibitors (statins) is broad and will be only briefly reviewed. A meta-analysis of over 90,056 patients in 14 randomized trials demonstrated the potent effect of statins in reducing death and cardiovascular events in a LDL dependent fashion. 15 The Adult Treatment Panel III (ATP III) of the National Cholesterol Education Program issued a report based on clinical trial data, recommending the treatment goal of low-density lipoprotein cholesterol (LDL-C) < 70 mg/dL for high risk patients. 16 This recommendation was later supported by a meta-analysis of over 27,000 patients showing a 16% odds reduction in coronary death, MI or any cardiovascular event with intensive vs. moderate statin therapy. 17 Several of the studies were in higher risk patients with acute coronary syndromes. The TNT (Treating to New Targets) trial concluded, however, that even patients with stable coronary disease derived a 22% relative risk reduction in major cardiovascular events with high dose statin therapy compared with low dose statin therapy. 18 Secondary analysis of the TNT trial concluded that additional benefit with intensive statin therapy may be achieved in patients 65 years or older, with an acceptable safety profile. 19
Angiotensin Converting Enzyme inhibitors (ACE inhibitors)
Current ACC/AHA guidelines recommend that ACE inhibitors be used in all patients with stable coronary disease and left ventricular (LV) systolic dysfunction (ejection fraction (EF) < 40%), hypertension, diabetes or chronic kidney disease in the absence of contraindications. 7 For patients with stable coronary artery disease and preserved LV systolic function, however, clinical trial data have been somewhat conflicting. The EUROPA trial (European Trial on Reduction of Cardiac Events with Perindopril in Stable Coronary Artery Disease) and HOPE trial (Heart Outcomes Prevention Evaluation study) reported reduced morbidity and mortality among patients with coronary disease and preserved LV systolic function, but the PEACE trial (Prevention of Events with Angiotensin Converting Enzyme Inhibition Trial) did not support these findings, possibly attributable to a relatively low risk study population in the latter study. 20 A meta-analysis of randomized controlled trials of ACE inhibitors in coronary artery disease with preserved LV systolic function, which included these trials, found a modest but significant reduction in cardiovascular mortality, nonfatal MI, all-cause mortality and revascularization rates. 21 Based on these results, updated guidelines conclude it is reasonable to use ACE inhibitors in patients with stable coronary disease and preserved LV systolic function.
Similar recommendations relate to the use of angiotensin-receptor blockers (ARBs) based on two large recently published clinical trials. The ONTARGET trial (Ongoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial) and the TRANSCEND trial (Telmisartan Randomised Assessment Study in ACE Intolerant Subjects with Cardiovascular Disease) demonstrated that the ARB telmisartan provided similar reductions in cardiovascular death, myocardial infarction, stroke or hospitalization for heart failure and was equally tolerated in patients with vascular disease or high risk diabetes without heart failure when compared with ACE inhibitors.22,23
Antianginal Therapy
Beta-blockers
Beta-blockers provide their antianginal effect by reducing the heart rate, blood pressure and myocardial contractility thereby reducing myocardial oxygen consumption and improving diastolic perfusion time. Although they have proven mortality benefit in patients with a prior history of myocardial infarction,24,25 the same is not necessarily true for patients with chronic stable angina. Beta-blockers, along with calcium channel blockers and nitrates, have been proven to decrease the frequency of anginal episodes, increase exercise duration and prolong the time to ST-segment depression on exercise treadmill testing. 26 A large meta-analysis, however, concluded that beta blockers provide slightly greater reductions in angina frequency with less adverse events compared with calcium channel blockers. 27 Too few trials compared nitrates with either beta blockers or calcium channel blockers from which to draw any significant conclusions. Given the beta-blocker mortality reduction in patients with prior MI, acute coronary syndrome and LV dysfunction, current guidelines recommend beta-blockers as first line agents for those with chronic stable angina in the absence of contraindications. 7
It was previously believed the side effects and contraindications to beta-blockers precluded broad use of these agents in patients with stable CAD. Absolute contraindications include severe bradycardia, advanced atrioventricular (AV) block, decompensated congestive heart failure and severe reactive airway disease. Potential side effects include reduced exercise capacity, fatigue and sexual dysfunction, with the latter two being significantly less common than previously thought. 28
Calcium channel blockers
Calcium channel blockers reduce angina by dilating systemic and coronary arteries, decreasing myocardial oxygen demand and increasing coronary arterial blood flow. Clinical trials using short-acting formulations of calcium channel blockers have shown increased rates of cardiac events and mortality in patients with hypertension and acute coronary syndromes respectively. 27 Subsequent studies and reviews of long-acting formulations of calcium channel blockers such as the ALLHAT trial (Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial), 29 the ACTION trial (A Coronary Disease Trial Investigating Outcome with Nifedipine GITS) 30 and the Blood Pressure Lowering Treatment Trialists’ Collaboration 31 indicate that the long-acting formulations reduce the need for coronary interventions without increased morbidity or mortality rates in patients with stable CAD. When beta-blockers are contraindicated or unable to control anginal symptoms, either long-acting dihydropyridine or non-dihydropyridine calcium channel blockers should be instituted. Adverse effects include bradycardia, heart block, dizziness, edema, headache and constipation.
Nitrates
By dilating systemic and coronary arteries as well as systemic veins, nitrates cause a decrease in left ventricular preload and afterload, subsequently decreasing myocardial oxygen demand and hence ischemia. Long-acting nitrates have been shown to decrease anginal episodes and increase exercise duration. 20 Chronic administration of long-acting nitrates is associated with development of tolerance, characterized by a decrease in hemodynamic and antianginal efficacy or an escalation of dose to achieve a similar benefit, which likely occurs through a variety proposed mechanisms.32,33 Tolerance can be avoided by maintaining at least a 12 hour nitrate free period every 24 hours, preferably at night when myocardial oxygen demands are typically lowest. Nitrates are available in a variety of formulations (oral, ointment or transdermal patch) including short acting sublingual and spray dosing which can be used for acute symptom relief within 5-10 minutes. Nitrate use within 24 hours of the phosphodiesterase inhibitors type 5 (sildenafil, vardenafil, and tadalafil) is contraindicated as life-threatening hypotension may result.
Revascularization (Percutaneous or coronary artery bypass graft surgery)
Current guidelines recommend that coronary artery bypass graft surgery should be reserved for patients with stable angina who have either left main disease, multivessel disease or left ventricular systolic dysfunction with diabetes. Controversy has existed surrounding the possible benefits of percutaneous coronary intervention (PCI) over medical therapy for patients with stable CAD. Meta-analyses comparing these two treatment strategies have yielded conflicting results.34–36 The reasons for these discrepancies are unknown but could be related to the diversity of patient populations, variability in the use and intensification of medical therapy, and evolution of interventional techniques.
More recently, the COURAGE (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation) trial randomized 2,287 patients with objective evidence of ischemia and significant coronary artery disease to optimal medical therapy with or without PCI. 37 Unlike many of the previous trials, the medical therapy utilized in the trial was optimal by today's recommended guidelines for patients with CAD. The study concluded that there was no difference between the two groups in death, non-fatal MI or other major cardiovascular events during a median follow-up period of 4.6 years. PCI reduced the frequency of angina in the first year compared with medical therapy, but there was no difference at 5 years. Even though there was a high cross-over from the medical therapy alone group to the medical therapy with PCI group and drug eluting stents were used in less than 3% of cases, the COURAGE trial suggests that PCI should be reserved for patients with stable coronary disease who are symptomatic despite optimal medical therapy. Nonetheless, cross-over was more likely in patients with multiple perfusion defects in radionuclide stress testing. 38
Transmyocardial revascularization
Transmyocardial revascularization (TMR) uses either a CO2 or holouim: YAG laser to create channels in the ischemic myocardium to relieve angina. This technique was first proposed in 1965 based on a model of the reptilian heart, but has yet to become a mainstay of antianginal therapy. The mechanism by which TMR works is poorly understood. Initial thoughts were that it provides better myocardial perfusion via these direct channels. Subsequent studies, however, have concluded that these channels fill up and occlude with necrotic debris shortly after their generation. 39 Currently hypotheses support laser-induced angiogenesis and denervation of the myocardium. 40 Although clinical trials have shown benefit of TMR in relieving angina and reducing cardiac events, 41 they have been limited by lack of a sham-controlled arm. Therefore, much of the benefit of TMR is thought to be placebo. 26 A similar technique, called percutaneous laser revascularization (PMR), has been developed but clinical trials supporting its benefit are also limited. Current guidelines from the Society of Thoracic Surgeons state that TMR may be an acceptable form of therapy for selected patients with stable coronary disease and refractory angina. 40
Enhanced External Counterpulsation (EECP)
The technique of EECP therapy consists of ECG-gated rapid sequential compression of bilateral lower extremities in diastole and decompression in systole, similar to that of an intraaortic balloon pump. A typical course consists of 35 treatments administered 1 hour daily for 7 weeks. The first randomized, blinded, controlled clinical trial to study EECP, the MUST-EECP trial (Multicenter Study of Enhanced External Counterpulsation), enrolled 139 patients with angina, documented CAD and positive exercise treadmill test (ETT). 42 EECP therapy was well tolerated, significantly reduced angina and extended time to exercise-induced ischemia compared to the sham EECP group. Subsequent clinical trials have supported these findings and concluded that EECP is a safe, beneficial, low-cost and noninvasive treatment for patients with stable angina.43,44 The mechanisms by which EECP provides benefit may involve improvement in endothelial function, promotion of collateralization, enhancement of ventricular function, improvement in oxygen consumption (VO2), regression of atherosclerosis and peripheral training effects similar to exercise.
Spinal Cord Stimulation (SCS)
SCS involves placing a stimulating electrode in the dorsal epidural space at the level of C7-T1 with a pacemaker sized generator located in the abdominal area. By receiving 1 hour of stimulation a day, with the ability to self-deliver extra stimulation for breakthrough pain, it is thought that SCS can provide analgesia for patients with chronic angina pectoris refractory to medical or interventional therapy. There have been several small clinical trials investigating SCS in chronic angina, none with a sham-controlled arm, which have suggested SCS may increase exercise capacity and reduce anginal episodes. The ACC/AHA guidelines on managing chronic angina conclude that although these trials appear promising, there is a paucity of data from which to recommend SCS for managing chronic angina. 11
Novel Medical Agents
Experimental therapy
Several pharmacologic agents have been tested in experimental trials with variable success in treating stable angina. Trimetazidine modulates cardiac metabolism by inhibiting the enzyme involved in β-oxidation of free fatty acids (abnormally utilized in the setting of ischemia), thereby causing a shift toward the more energy-efficient glucose oxidation. Clinical trials have shown that it can produce significant improvements in exercise stress tests and the symptoms of angina without any hemodynamic effects.45–47 Additionally, recent trials using trimetazidine in patients with ischemic and non-ischemic cardiomyopathy have shown significant improvement in LV function compared to conventional therapy alone.48,49 Trimetazidine is well tolerated and its benefits are seen when used as monotherapy or in combination with conventional antianginal agents.
Nicorandil, a nicotinamide ester, causes venous and arterial vasodilatation by its dual mechanism of action: 1) opening ATP-sensitive potassium channels and 2) possessing a nitrate moiety. Thus, nicorandil increases coronary blood flow while reducing preload and afterload. Although limited clinical trial data exist, the IONA (Impact of Nicorandil in Angina) study enrolled over 5,000 patients with stable CAD and found nicorandil, in addition to standard antianginal therapy, significantly reduced the primary end point (coronary death, MI or hospitalization for angina) compared with placebo. 50 Ivabradine, an inhibitor of the If channel in the sinus node, causes bradycardia in the absence of any negative inotropic effects. In randomized, double-blind, placebo controlled trials, ivabradine has been shown to reduce the incidence of coronary artery disease outcomes (in those with heart rates ≥70 bpm) 51 and improve exercise test parameters (in a dose dependent fashion). 52 Fasudil inhibits the enzyme rho kinase, an intracellular signaling molecule involved in vascular smooth muscle contraction. In a double-blind, placebo-controlled, randomized trial of 84 patients with stable angina, fasudil significantly improved time to ≥1 mm ST-segment depression compared with placebo, but had no significant effect on Canadian Cardiovascular Society (CCS) class, time to angina, frequency of angina or nitroglycerin use. 53 None of these drugs are approved in the U.S. for treatment of chronic stable angina.
Sibutramine, a serotonin and norepinephrine transporter blocker is approved for use in the U.S. for weight loss management. Clinical trial data suggests that it can improve endothelial function in patients with stable CAD, 54 thereby serving as adjunctive therapy to optimize outcomes. It is not clear, however, whether the endothelial benefit was attributable to the medication itself or the associated weight loss. A final experimental therapy still early in investigation is Ad5FGF-4, an angiogenic gene therapy agent. Preclinical studies suggest that it may induce angiogenesis and serve a unique role in treating patients with chronic stable angina. Despite clinical trial data showing no effect in men, there was a significant improvement for women in total ETT duration, time to 1 mm ST-segment depression, time to angina and CCS class. 55 Each of these experimental agents provides novel mechanisms of antianginal therapy but are not approved for use in the U.S. for the treatment of chronic stable angina.
Ranolazine
Ranolazine is a piperazine derivative that was patented in 1986 and approved for use in chronic stable angina in January 2006. It is available in intravenous and oral forms, with only the latter approved for clinical use. Unlike traditional antianginal therapies, which aim to reduce myocardial oxygen demand (i.e. heart rate, afterload, myocardial contractility and preload), ranolazine has been proven to reduce ischemic symptoms and improve exercise capacity without any clinically significant alterations in heart rate or blood pressure.56,57 Ranolazine improves the ischemic imbalance via its novel downstream metabolic and cellular effects.
Mechanism of action
Originally, it was thought that ranolazine exerted its therapeutic efficacy by partially inhibiting fatty acid oxidation, causing preferential use of the more energy efficient glucose oxidation.56, 58,59 However, this appears only to occur at serum levels well above the therapeutic values (>10 μmol/L). 60 Subsequent research concluded that ranolazine likely provides its cardioprotective effect by altering sodium shifts and intracellular calcium. 61 By preferential inhibition of the late sodium current (late INa) at serum levels proven efficacious in clinical trials, ranolazine attenuates the electrical and mechanical consequences of ischemia. 62
Under physiologically normal conditions, electrical stimulation of cardiac myocytes causes rapid opening of membrane sodium channels (peak INa) and inward flux of sodium ions. Rapid depolarization results, representing Phase 0 of the action potential. 63 These sodium channels rapidly inactivate and remain closed during the plateau phase (phase 2) of the action potential. As a result of depolarization, transmembrane ion fluxes continue, leading to intracellular release of calcium from the sarcoplasmic reticulum. Intracellular calcium then interacts with actin and myosin, leading to myocardial contraction.
Various pathological conditions, including ischemia, left ventricular hypertrophy and heart failure, can lead to abnormal late INa.63,64 The increased intracellular accumulation of sodium ions causes reverse mode transport of calcium to the intracellular space via the Na+/Ca2+ exchanger, leading to intracellular calcium overload and sustained myocardial contraction. Electrical instability, arrhythmia and mechanical dysfunction (reduced contractility and increased diastolic wall tension) result from excess intracellular calcium accumulation. 65 Increased diastolic wall tension causes vascular compression and further reduction of coronary blood flow to the ischemic territory at the same time of increased metabolic demand, leading to a vicious circle of further myocardial ischemia and eventual necrosis. Ranolazine has been proven to attenuate the abnormalities of ventricular repolarization and contractile function in a variety of pathologic models via inhibition of the late INa and its subsequent effects.62,66–69 However, in healthy nonischemic hearts, nonfailing myocytes, where the contribution of late INa is minimal, ranolazine does not have any measurable effect at therapeutic concentrations. 70 Ranolazine, by targeting a unique downstream component of the ischemic cascade only in pathological states, provides a novel therapeutic option in the treatment of chronic angina and possibly arrhythmias.
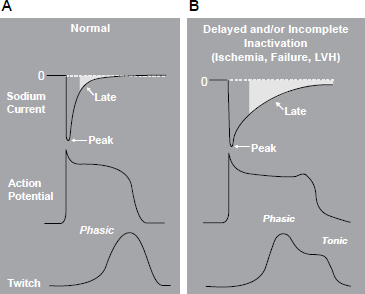
Relationship between late Na+ current and ventricular action potential and contraction. A) Normal conditions. B) Delayed or incomplete inactivation in the setting of ischemia, heart failure, or left ventricular hypertrophy. Reproduced with permission from Belardinelli L, Antzelevitch C, Fraser H. inhibition of late (sustained/persistent) sodium current: a potential drug target to reduce intracellular sodium-dependent calcium overload and its detrimental effects on cardiomyocyte function. Eur Heart J. 2004;6:14;. Permission received from European Heart Journal Supplements, Oxford Press and author Belardinelli L.
Pharmacokinetics, metabolism and drug-drug interactions
Ranolazine is available for clinical use in the oral form as film-coated extended-release 500 mg tablets. The intravenous formulation is only available for experimental use and the immediate-release oral ranolazine is no longer used. Oral bioavailability ranges from 35% to 50%, with food having no significant impact. The time to maximal plasma concentration (Cmax) is typically 4-6 hours, with an elimination half-life of approximately 7 hours and steady state achieved within 3 days of multiple dosing (500 mg-1000 mg twice daily). 71 The peak-trough ratio at steady state ranges from 1.8-2.2. Ranolazine plasma concentrations that are therapeutically effective for chronic angina based on clinical trials ranged from 2-6 μmol/L. 70
Ranolazine is metabolized primarily by the cytochrome P450 (CYP) 3A4-mediated pathway. Additional pathways for metabolism include the CYP2D6 (10%-15%), glucuronidation (<5%) and renal excretion of unchanged ranolazine (5%). 71 Four metabolites (CVT-2738, CVT-4786, CVT-2514 and CVT-2512) occur at concentrations of >10% the parent compound at steady state concentrations, but their potential pharmacologic activity is unknown. In patients with moderate liver dysfunction (Child-Pugh grade B), significant elevations of plasma ranolazine levels are noted, whereas mild liver dysfunction (Child-Pugh grade A) has no effect. 72 Similarly, moderate to severe renal impairment increases ranolazine steady state levels by 1.7 to 2.0 fold respectively. 73
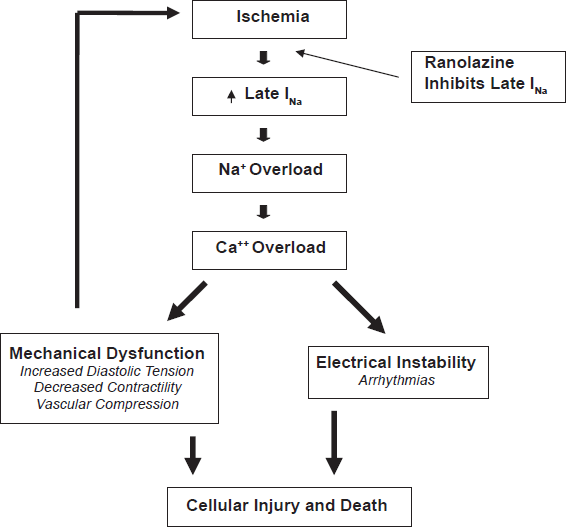
Pathophysiology of ischemia and mechanism of action of ranolazine.
Due to its metabolism via the CYP3A4 pathway, drug-drug interactions are a significant concern. Ketoconazole, a potent inhibitor of CYP3A4, has been shown to increase plasma ranolazine levels between 2.5 and 4.5 fold. Moderate inhibitors, such as diltiazem, verapamil, macrolide antibiotics, protease inhibitors and grapefruit juice increase ranolazine levels 2 fold.71,74 Ranolazine increases plasma levels of simvastatin roughly 2 fold and digoxin 1.5 fold, with neither affecting ranolazine levels. Drug labeling indicates that ranolazine is contraindicated with moderate and potent inhibitors of CYP3A4. P-glycoprotein inhibitors, such as cyclosporine, increase absorption of ranolazine and should be used with caution.
Clinical trials: Stable angina
The initial clinical trial of ranolazine for chronic angina pectoris compared immediate release ranolazine (400 mg three times daily) with atenolol (100 mg daily) and placebo in a randomized, doubleblind, 3-period, crossover study of 158 patients. Ranolazine compared to placebo produced significant improvements in total exercise duration, time to angina onset and time to ST-segment depression without any significant effect on heart rate or blood pressure. Compared with atenolol therapy, ranolazine therapy resulted in significantly longer total exercise duration by a mean of 21 seconds. 75 However, all subsequent studies have used the extended-release formulation, on which FDA approval of ranolazine was largely based.
The MARISA trial (Monotherapy Assessment of Ranolazine in Stable Angina) was 4 week, doubleblind, four-period crossover study of extended-release ranolazine (500 mg, 1000 mg, 1500 mg) versus placebo in 191 patients with at least 3 months of exertional angina and angina-limited ETT with documented ST-segment depression. 56 Significant exclusion criteria included NYHA class III-IV congestive heart failure, acute coronary syndrome or revascularization within the previous 2 months, corrected QT interval (QTc) > 500 ms or any medication know to prolong the QTc. All other antianginal therapy was discontinued except sublingual nitroglycerin as needed. Exercise testing was performed at the end of each treatment during both peak (4 hours after dosing) and trough (12 hours after dosing), with the primary efficacy end point being total exercise duration at trough. Secondary endpoints included time to onset of angina and time to 1 mm ST-segment depression at trough and the same three ETT end points at peak. All three ranolazine doses significantly increased exercise duration at trough compared with placebo (p < 0.005) in a dose dependent fashion, with negligible effects on heart rate and blood pressure. Statistically significant improvements were also noted in times to 1 mm ST-segment depression and times to onset of angina (all p < 0.005). Ranolazine was generally well tolerated, with fewer than 8% of patients discontinuing therapy because of adverse effects, and almost three-quarters of the withdrawals occurring with 1500 mg dose.
The CARISA trial (Combination Assessment of Ranolazine in Stable Angina) enrolled 823 patients with symptomatic chronic angina who were already on standard doses of antianginal therapy (atenolol 50 mg/day, diltiazem 180 mg/day or amlodipine 5 mg/day). Patients were randomized in a 3-group parallel, double-blind, placebo-controlled fashion to ranolazine 750 mg, ranolazine 1000 mg or placebo twice daily. 57 Patient selection and exclusion criteria where otherwise similar to those enrolled in MARISA. Treadmill exercise 12 hours (trough) and 4 hours (peak) after dosing was assessed after 2, 6 (trough only), and 12 weeks of treatment. The primary endpoint, exercise duration at trough, was improved in both ranolazine groups (pooled) compared with placebo (115.6 vs. 91.7 seconds, p = 0.01), again without any clinically meaningful changes in blood pressure or heart rate. The effects were sustained throughout the entire study suggesting that no significant tolerance developed. Similar improvements were seen with time to angina onset and time to ST-segment depression, most pronounced at peak. Ranolazine also significantly reduced angina attacks and nitroglycerin use by about 1 per week compared with placebo. The mean QTc increased 6.1 ms for the 750 mg and 9.2 ms for the 1000 mg ranolazine groups over placebo. The CARISA study demonstrated that ranolazine provides additional antianginal and anti-ischemic efficacy in patients with severe chronic angina who are symptomatic despite standard doses of antianginal therapy and may have additional non-anginal benefit in diabetic patients.
Although post-hoc analysis of CARISA did not show any significant difference in exercise improvement or angina control between diabetic and non-diabetic patients, it produced an interesting glycometabolic finding. Ranolazine 750 mg and 1000 mg reduced HbA1c vs. placebo by 0.48% (p = 0.008) and 0.70% (p = 0.0002) respectively. 76 This finding was recently supported by an exploratory analysis of the MERLIN trial, discussed below, which found ranolazine compared with placebo significantly reduced HbA1c at 4 months (5.9% vs. 6.2%; p < 0.001), was more likely to achieve an HbA1c < 7% at 4 months in diabetics (59% vs. 49%; p < 0.001) and in patients with diabetes reduced overall HbA1c from 7.5 to 6.9 (change from baseline -0.64; p < 0.001). 77 Although the mechanism for this benefit is unclear, it warrants further investigation to explore its potential for additive glycemic control in diabetic patients.
The ERICA trial (Efficacy of Ranolazine in Chronic Angina) sought to answer the question whether ranolazine improves anginal control in patients with stable coronary disease symptomatic despite maximal traditional antianginal therapy. 78 A total of 565 patients with stable coronary artery disease and ≥3 anginal attacks per week despite maximal amlodipine 10 mg/day were randomized to 1,000 mg ranolazine or placebo twice daily for 6 weeks. Compared with placebo, ranolazine significantly reduced the frequency of angina episodes (2.88 ± 0.19 on ranolazine vs. 3.31 ± 0.22 on placebo; p = 0.028) and nitroglycerin consumption (2.03 ± 0.20 on ranolazine vs. 2.68 ± 0.22; p = 0.014). Subgroup analysis showed that patients with more frequent angina (>4.5 episodes/week) appeared to derive greater treatment benefit with ranolazine. Based on the findings of ERICA, ranolazine was approved by the FDA for use in chronic stable angina.
Clinical trial: Acute coronary syndrome
The MERLIN-TIMI 36 trial (Metabolic Efficiency with Ranolazine for Less Ischemia in Non-ST-Elevation Acute Coronary Syndrome) sought to determine the efficacy and safety of ranolazine in a higher risk population of patients with CAD. 79 A total of 6,560 patients with either unstable angina or non-ST-elevation myocardial infarction (NSTEMI) were randomized to ranolazine (initiated intravenously followed by oral ranolazine 1,000 mg twice daily) vs. placebo within 48 hours of ischemic symptom onset in a double-blind, placebo-controlled fashion with therapy continued for a median of 348 days. Although there was no significant difference between ranolazine and placebo in the primary efficacy endpoint of combined rate of cardiovascular death, MI or recurrent ischemia compared with placebo (21.8% vs. 23.5% respectively, HR 0.92, 95% CI 0.83-1.02, p = 0.11), analysis of the individual elements showed a significant reduction of recurrent ischemia with ranolazine compared to placebo (13.9% vs. 16.1%, HR 0.87, 95% CI 0.76-0.99, p = 0.03). Consistent with the results from CARISA and ERICA, long-term treatment with ranolazine also significantly improved anginal control in several prespecified exploratory end points: worsening angina by ≥1 CCS class requiring intensification of therapy, increase in or addition of antianginal therapy and angina frequency.
Additional findings of the MERLIN trial, some of which will be discussed later, included a reduction in ventricular and atrial arrhythmias, incremental benefit in those with elevated BNP as well as women and a safety profile consistent with previous studies. In fact, based on a post hoc analysis, women had a significant reduction of the primary endpoint with ranolazine compared with placebo (n 2291; HR, 0.83; 95% CI, 0.70-0.99), largely driven by a 29% relative reduction in ischemia. 79
A recently published subgroup analysis of 3,565 patients with prior chronic angina in the MERLIN trial, lends further support to previous smaller studies that ranolazine significantly improves angina and exercise performance in patients with stable CAD. 80 However, ranolazine did not reduce the rate of ischemia detected on continuous ECG during the acute coronary syndrome hospitalization, which is an independent predictor of death. 81 The findings in MERLIN confirm that ranolazine is an effective and safe antianginal therapy in both stable angina and a higher risk CAD group, but does not afford a reduction in major cardiovascular events and all-cause death in NSTEMI ACS patients despite its antiarrhythmic properties.
Reproduced with permission from Nash DT. Ranolazine for chronic stable angina. Lancet. 2008;372:1335-41.
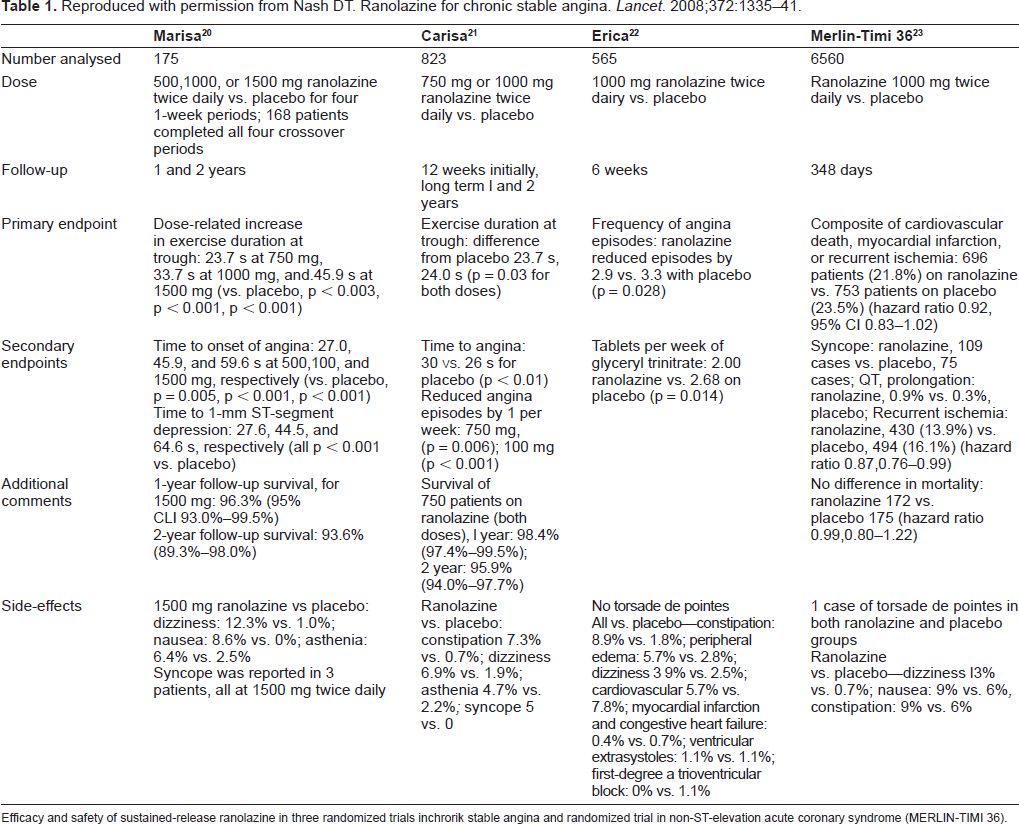
Efficacy and safety of sustained-release ranolazine in three randomized trials inchrorik stable angina and randomized trial in non-ST-elevation acute coronary syndrome (MERLIN-TIMI 36).
Electrophysiological properties
Ranolazine has two major ion channel effects: inhibition of the late INa which shortens the action potential duration (APD) and inhibition of the delayed potassium rectifier (IKr) which lengthens the APD. 66 The extent of action potential alteration ultimately depends on the relative concentrations and inhibition of the INa and IKr in addition to the cellular state of the myocyte. Lengthening of the APD in association with early afterdepolarizations (EADs), delayed afterdepolarizations (DADs), triggered activity and increased transmural dispersion of repolarization are the substrates for ventricular arrhythmias, including Torsades de Pointes (TdP) and atrial arrhythmias such as atrial fibrillation. 82
Despite ranolazine prolonging the QTc interval on average by 2.4 ms, 83 experimental and clinical trials to date have not demonstrated any increased risk for arrhythmias. In fact, multiple preclinical trials have demonstrated that ranolazine actually shortens the APD, reduces EADs, DADs, triggered activity and transmural depolarizations, suggesting a possible antiarrhythmic effect of ranolazine.66,67,69,82,84 This may in part be explained by the fact that in the ventricular midmyocardium (M cells) and Purkinje fibers, INa predominates and the APD is shortened with ranolazine. Based on this evidence, some recent experimental models have demonstrated that ranolazine can suppress atrial and ventricular arrhythmias such as atrial fibrillation and TdP.85,86
Consistent with this preclinical data, the MERLIN-TIMI 36 trial showed a reduction in both ventricular and atrial arrhythmias with ranolazine for the first time in a human clinical trial. 87 Continuous ECG (Holter) recording was performed for the first 7 days after randomization to ranolazine vs. placebo in these patients with non-ST-elevation ACS. Treatment with ranolazine compared with placebo resulted in fewer episodes of ventricular tachycardia (VT) ≥ 8 beats (166 [5.3%] vs. 265 [8.3%]; p < 0.001), supraventricular tachycardia (1413 [44.7%] vs. 1752 [55.0%]; p < 0.0010, new-onset atrial fibrillation (55 [1.7%] vs. 75 [2.4%]; p = 0.08) and pauses ≥3 seconds (97 [3.1%] vs. 136 [4.3%]; p = 0.01). Although no difference was found in the occurrence of sustained VT (monomorphic or polymorphic), the frequency with which these occurred in the study was likely too low to reach any conclusions. Therefore, ranolazine appears to be antiarrhythmic despite its minimal prolongation of the QTc; however, more trials are needed to support this conclusion considering that patients on current therapy with potentially QT prolonging medications were excluded from MERLIN.
Safety and tolerability
Based on clinical trial data in patients both with stable angina and with higher risk non-ST-elevation ACS, ranolazine is well tolerated with an acceptable safety profile. In the MARISA trial, adverse events were reported in 14.5%, 15.5%, 20.6% and 33.2% of patients on placebo and ranolazine 500 mg, 1000 mg and 1500 mg respectively, which led to 1000 mg twice daily being the recommended maximal dose. 56 Syncope occurred in 3 patients, all of whom were on the 1500 mg dose regimen. In the CARISA trial, there were 5 reported cases of syncope, all in patients on the 1000 mg twice daily dose. 57 Four of the 5 patients were on diltiazem and all 5 patients were on an ACE inhibitor, both of which are know to raise plasma concentrations of ranolazine. These events occurred in the absence of any clinically meaningful differences in blood pressure or heart rate. In the ERICA trial there were no reported cases of syncope. Ranolazine was started at 500 mg twice daily for 1 week and titrated to a maximum of 1000 mg twice daily, suggesting that subscribing to a judicious use of ranolazine based on symptom relief with a slow titration and monitoring for side effects and possible drug-drug interactions can negate this side effect. 70 In review of these trials, the most common adverse effects reported include dizziness, constipation, nausea, asthenia, syncope, headache and abdominal pain.
The ROLE (Ranolazine Open Label Experience) trial enrolled 746 patients with severe functional impairment from angina (as assessed by the Duke Treadmill Score (DTS), mean -14.4) who had completed either the MARISA or CARISA trials in an open-label extension program. 83 Ranolazine could be titrated to optimal doses ranging from 500 mg to 1000 mg twice daily based on symptoms and patients were followed for a mean of 2.8 years. After two years of therapy, 72 patients (9.7%) discontinued ranolazine due to adverse events, 46 patients (6.0%) due to elective withdrawal and 34 patients (4.6%) due to death. Among 6 factors evaluated, only age ≥64 years predicted higher withdrawal rates. The most common adverse events were dizziness (11.8%) and constipation (10.9%). There were no discontinuations for QTc prolongation and no reports of TdP. Annual mortality was 2.8% compared with >5% as predicted by the DTS. Moreover, the MERLIN trial demonstrated that even in a high risk population with non-ST-elevation ACS, ranolazine was safe. At 1 year, no difference in cardiovascular death or total mortality was observed with ranolazine compared with placebo. 79
Rate of tachyarrhythmias detected on cECG monitoring after non-ST-segment elevation Ml.
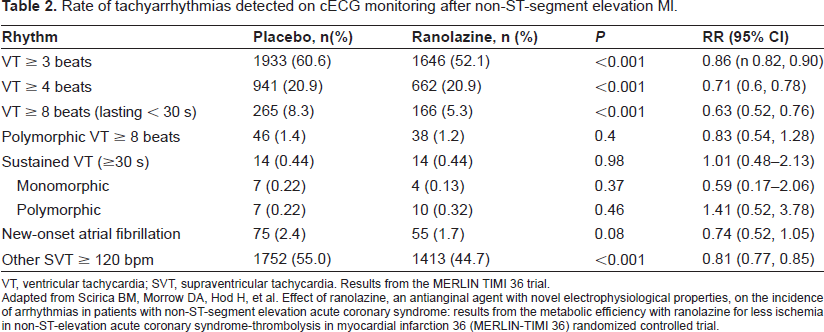
VT, ventricular tachycardia; SVT, supraventricular tachycardia. Results from the MERLIN TIMI 36 trial.
Adapted from Scirica BM, Morrow DA, Hod H, et al. Effect of ranolazine, an antianginal agent with novel electrophysiological properties, on the incidence of arrhythmias in patients with non-ST-segment elevation acute coronary syndrome: results from the metabolic efficiency with ranolazine for less ischemia in non-ST-elevation acute coronary syndrome-thrombolysis in myocardial infarction 36 (MERLIN-TIMI 36) randomized controlled trial.
To further evaluate the safety of ranolazine, several specified populations have been investigated. A review of the previous clinical trials prior to MERLIN suggested that women showed less improvement than men in exercise testing but similar improvements in angina frequency and nitroglycerin consumption with equal safety. 88 In contrast, subgroup analysis from MERLIN found the effect of ranolazine on the primary end point was significant among women (n = 2291; HR, 0.83; 95% CI, 0.70-0.99), driven by a 29% relative reduction in recurrent ischemia with ranolazine (p = 0.002). 79 These results are encouraging but have to be validated in future clinical trials.
Despite the elderly population being more likely to have adverse events and discontinue therapy, their incidence of serious adverse events is not increased with ranolazine.83,89 Post-hoc analysis from the CARISA trial showed that diabetics were equally as likely as non-diabetics to derive benefit without any increased risk of adverse events with ranolazine. 76 Finally, QTc prolongation has been described in numerous clinical trials but has not been associated with any proarrhythmic effects. In fact, based on the MERLIN trial, ranolazine may actually be antiarrhythmic. Ranolazine is a safe and well tolerated antianginal agent when prescribed appropriately for use in high risk patients with CAD, including women, elderly and diabetics.
Future applications
Ranolazine may be useful to treat heart failure through its unique ability to maintain intracellular sodium and calcium homeostasis in pathological conditions. Under conditions of myocardial ischemia, left ventricular hypertrophy and oxidative stress, abnormal late INa causes intracellular calcium overload, prolonged myocardial contraction and abnormal left ventricular diastolic function. 90 Several experimental models have demonstrated improvement in diastolic wall tension, left ventricular end-diastolic pressure and left ventricular filling with ranolazine.59,69,91,92 These effects were demonstrated without any negative inotropic effects, suggesting a possible new therapy for diastolic heart failure. Beyond its diastolic improvements, ranolazine at therapeutic concentrations has also been shown to improve systolic function.59,93 This benefit is additive to concurrent therapy with beta-blockers and ACE inhibitors and occurs without any increase in VO2.94–96 Much of this work is based on animal models, leading to the conclusion that further investigation involving human clinical trials has to be undertaken to confirm these findings.
Another potential application of ranolazine recently explored after publication of the MERLIN trial rests in its antiarrhythmic effects. Despite prolonging the QTc, the MERLIN trial showed that ranolazine significantly reduced episodes of supraventricular and ventricular arrhythmias in patients with ischemia. 79 Consistent with these results, in vitro models have shown that ranolazine can mitigate the proarrhythmic milieu in animal hearts and even terminate lethal rhythms such as Torsades de Pointes.85, 97 More recently, a targeted human model was explored in patients with Type-3 long QT-syndrome. LQT3-δKPQ, is associated with a sustained inward sodium current during membrane depolarization and a substantial increase in lethal cardiac events. In this high-risk population, an 8-hour intravenous ranolazine infusion shortened a prolonged QTc by 26 ± 3 ms (p < 0.0001) and improved diastolic relaxation. 98 The growing body of evidence supporting ranolazine's potential use as an antiarrhythmic was tested in a published case report, showing resolution of a malignant ventricular tachycardia with ranolazine 1,000 mg twice daily in a patient with alcohol cardiomyopathy refractory to aggressive conventional therapy for VT. 99
Ranolazine's antiarrhythmic properties, however, do not appear limited to the ventricle. Within the atria, ranolazine has been shown to inhibit the rapidly activating IKr as well as use-dependent inhibition of peak INa. In doing so, ranolazine leads to prolongation of the effective refractory period, intra-pulmonary vein conduction block and suppression of EAD and DAD-mediated triggered activity, which are thought to be inciting events in the generation of atrial fibrillation. 84 Although limited by small numbers and isolated trials, ranolazine has been effective in terminating atrial fibrillation as well as preventing its initiation in both animal and human studies.86,100
Conclusion
Chronic stable angina is an exceedingly prevalent condition, only expected to increase as trends in obesity and diabetes continue to worsen. Although recent strides in vasculoprotective therapy for stable angina have been impressive, little has been added to antianginal therapy over the past 25 years until ranolazine. Conventional therapy with beta blockers, calcium channel blockers and nitrates remain the cornerstone of antianginal therapy. Because of the mortality benefit of beta-blockade, current guidelines recommend beta-blockers as initial therapy in patients with CAD in the absence of contraindications. Based on revised labeling, however, ranolazine is also approved as initial antianginal therapy. Given that ranolazine does not affect the heart rate, blood pressure or bronchial reactivity, it may well be the preferred agent for CAD patients with bradycardia, AV block, hypotension or reactive airway disease while calcium channel blockers should remain the preferred second agent after beta-blockers for patients with hypertension. Ranolazine is well tolerated with a proven safety profile and its minimal QTc prolongation is without clinical sequelae. In fact, ranolazine has several intriguing antiarrhythmic and metabolic properties which may lead to future applications beyond its proven antianginal benefit.
Disclosure
Commitment: Research Grants/Contracts/Trial Steering Committee/Trial Adjudication Committee Organization: Pfizer, Merck, NHLBI, Gilead Sciences (formerly CV Therapeutics), Abbott, Sanofi-Aventis, Eli Lilly
Commitment: Consultantship
Organisation: Women's Advisory Board, Gilead Sciences (formerly CV Therapeutics); Cardiovascular Advisory Board, Leadership Council for Improving Cardiovascular Care (LCIC) Executive Committee, Schering-Plough; AstraZeneca; Abbott; Merck; Pfizer, Boston Scientific, Medtronic Women's CV Health Advisory Panel; Genzyme
