Abstract
The cornerstone in treatment of psychosis is antipsychotic drugs. Treatment options have increased over the years; newer antipsychotic drugs with a proposed increased efficacy regarding negative and cognitive symptoms, but also a shift in side-effects from neurological side-effects to metabolic side-effects have arisen as the new challenge. The basis of successful pharmacological treatment is a fundamental understanding of the mechanisms of action, the desired effects and side-effects of antipsychotic drugs, a good relationship with the patient and a thorough monitoring of the patient before and during treatment. The clinically relevant aspects of antipsychotic drug treatment are reviewed; mechanism of antipsychotic drug action, clinical considerations in treatment, switching antipsychotic drugs, polypharmacy, safety and patient preference.
Introduction
The basis of successful pharmacological treatment is a fundamental understanding of the mechanisms of action, the desired effects and side-effects of antipsychotic drugs, a good relationship with the patient and a thorough monitoring of the patient before and during treatment. The treatment of psychotic symptoms should not solely be based on psychotropics, but also include psychosocial and psychotherapeutic interventions. 1
The introduction of chlorpromazine by Laborit, Delay and Deniker in 1952 changed the treatment of psychosis dramatically,2,3 and initiated the development of several new antipsychotic drugs, all with a dopaminergic receptor antagonism in common. 4 A promising new class of antipsychotic drugs appeared in the 1990s with a proposed increased efficacy regarding negative and cognitive symptoms, but also a shift in side-effects from neurological side-effects to metabolic side-effects as the new challenge.5–7 After more than ten years with atypical antipsychotic drugs, the gloss is wearing off and studies now suggest the clinical differences are small and the side-effects troublesome. 8
The authors hope that this clinical overview will throw light on the different antipsychotic drugs, desired effects, side-effects, safety concerns and the importance of patient preferences, and by that improve the treatment of patients with psychosis.
Mechanism of Action
Dopamine hypothesis
The dopamine hypothesis attributes disturbed dopamine neurotransmission as an explanation for symptoms in schizophrenia. 9 Patients with schizophrenia have an increased activity in the mesolimbic dopamine receptor systems which is believed to be responsible for the psychotic or positive symptoms, e.g. hallucinations and delusions. 4 The level of dopamine in the prefrontal cortex is lower compared to healthy controls, probably causing negative and cognitive symptoms. 9 The reduced striatal dopamine receptor activity, as a result of treatment with dopamine D2 blocking agents, causes extra pyramidal side-effects (EPS). 9 The antagonistic dopaminergic effect on the pituitary gland causes hyperprolactinemia.
Since the advent of chlorpromazine, the dopaminergic system has had a core role in our understanding of how antipsychotic drugs work. 10 Newer neuroimaging binding studies show that blocking the dopamine D2 receptors more than 60%-70% corresponds to the antipsychotic effect whereas blocking more than 75% increases the risk of EPS.11,12 This theory is questioned by the fact that clozapine, the antipsychotic drug with the highest efficacy,5,13 only has a dopamine D2 receptor blockage of 40% in clinical relevant dosages 11 which suggests that antipsychotic effect is more than pure dopamine D2 receptor antagonism.
The glutamate hypothesis
Glutamate functions as the main excitatory neurotransmitter in the brain, and is involved in many functions e.g. cognition and perception. 14 The glutamate receptors are divided into ionotropic and metabotropic receptors consisting of several subunits, 15 where the ionotropic N-methyl-D-aspartate (NMDA) receptors is of special interest in schizophrenia research and treatment. 16
The glutamate hypothesis presumes hypofunction of the glutamatergic system causing symptoms of schizophrenia, 16 partly by increasing dopaminergic tone in the mesolimbic system secondary as a result of the aforementioned glutamatergic hypofunction. 16 NMDA receptors in particulary have been implicated in the pathogenesis, but their role is not fully elucidated, although trials with e.g. ketamine and phencyclicine (PCP) that antagonize the NMDA receptor has been shown to induce symptoms mimicking the positive and negative symptoms of schizophrenia in healthy volunteers, and worsen symptoms in patients with schizophrenia. 15 Data on brain morphology postmortem and NMDA receptors has been gathered, but no clear conclusions has been drawn. 15 There has also been focus on genes coding for the NMDA receptor, both in studies of animals with knock-out genes but also in humans with gene-mapping; 16 but again no clear conclusions can be drawn. 16
The use of glutamate NMDA receptor agonists in combination with dopamine blocking antipsychotic drugs have only been used in trials, and no compounds have reached the market yet. 17 The trials have generally been small, on treatment refractory patients and have not been conclusive; 17 further research is still needed.
This review will focus on drugs marketed and used for treating patients with psychosis and experimental drugs will not be discussed further.
Types of antipsychotic drugs
Antipsychotics are usually divided into typical/first generation, or atypical/second generation antipsychotics. 18 No clear terminology or definitions exist. In this overview the terms typical and atypical antipsychotic drugs will be used.
What makes atypical antipsychotic drugs atypical is not clearly defined, but a low tendency to produce EPS and a low frequency of sustained increased levels of prolactin are often used in the definition.19–21 The atypical antipsychotic drugs are listed in Figure 1.
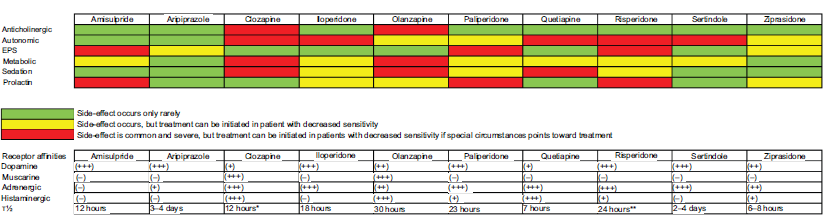
EPS was a common side-effect when treating psychotic symptoms in patients with typical antipsychotic drugs. 22 This side-effect was not seen as frequent with atypical antipsychotic drugs, 23 due to either a more selective mesolimbic dopaminergic blockade,24,25 a powerful serotonergic antagonism on the 5-HT2A receptor 26 a 5-HT1A agonism, or a rapid dissociation from the dopamine D2 receptor.7,19
The atypical antipsychotic drugs gave hope for better compliance due to fewer side-effects, 18 but time has shown that atypical antipsychotic drugs do not improve compliance compared to typical antipsychotic drugs,27–31 and although EPS is no longer the most troublesome side-effect,22,23 other side-effects have arisen, e.g. weight gain, dyslipidemia and decreased insulin sensitivity.18,32–34
All antipsychotic drugs have the dopamine D2 receptor blockade in common,4,35 but variations in other receptor affinities differentiate the side-effects of antipsychotic drugs. A short description of clinically relevant receptors will follow.
Receptors and Antipsychotic Drugs
Dopamine receptors
The dopaminergic system consists of five receptors named D1 to D5 divided into two families, the D1-like receptor family consists of the D1 and D5 receptors, and the D2-like receptor family consists of the D2 to D4 receptors.9,36 As described earlier, the dopamine D2 receptor is essential in the pathophysiology of psychotic disorders. The effects of the remaining receptors are not fully elucidated, e.g. the D1 and D4 receptors, are probably involved in the mechanism of cognitive symptoms in patients with schizophrenia.9,37
Serotonin receptors
The serotonergic (5-HT) receptor system is also involved in the mechanism of action of antipsychotic drugs, as most atypical antipsychotic drugs have an antagonistic effect on the 5-HT2A receptor and a partial agonistic effect on the 5-HT1A receptor. 26 This mechanism is probably responsible for an increase in nigrostriatal and prefrontal cortex release of dopamine resulting in lower levels of EPS and perhaps fewer cognitive side effects, respectively. 26 The D2/5-HT2A ratio seems to be more important for an anti-EPS profile than the absolute 5-HT2A affinity. 26 The 5-HT2C receptor has been associated with increased appetite, especially in combination with affinity for the histaminergic H1 receptor26,38,39 as described later. A genetic variation on the 5-HT2C receptor could also be involved in the pathogenesis of antipsychotic drug induced weight gain. 40
Adrenergic receptors
Animal studies have shown that a central α-1 receptor antagonism has a stabilizing effect on dopamine release in the mesocorticolimbic part of the brain, which is the proposed mechanism behind antipsychotic effect.41,42 The central α-1 receptor antagonism is not only believed to be involved in the mechanism of antipsychotic effect but also in sedation, 41 which is evident in patients treated with, e.g. clozapine.41,42
The α-2 receptor antagonistic effect of many antipsychotic drugs is not fully understood, but the combination of α-2 receptor antagonism and dopamine D2 antagonism has been proposed as mediating antipsychotic effect at lower D2 occupancies. 41
The peripheral effects of adrenoreceptor blockade are discussed in the safety paragraph of this overview.
Muscarinic receptors
The cholinergic system consists of nicotinergic and muscarinergic receptors but antipsychotic drugs mostly affect the muscarine system. The muscarinic receptor system consists of 5 receptors named M1 to M5. 43 A muscarinic antagonism, or anticholinergic effect, is especially seen in the older high dose/low potency antipsychotic drugs, e.g. chlorpromazine, but also seen to a lesser extent in some of the atypical drugs, e.g. olanzapine and clozapine. 43 The anticholinergic effect reduces EPS through the M4 receptor antagonism, but increases the risk of confusion, seizures, constipation, urinary retention, sinus tachycardia and cognitive deficits.43–46 The muscarinic receptor system modulates the dopaminergic system when used in the treatment of EPS, through M4 antagonism that increases the dopaminergic load in the nigrostriatal area. 43 Muscarinic agonism has been proposed as mediating decreased dopaminergic tone in the mesolimbic area, and thereby decreasing psychotic symptoms. 43
The antagonistic effect of some antipsychotic drugs on the muscarinic M3 receptor has been implicated in the development of diabetes by a direct inhibitory effect of the pancreatic β-cells insulin release.38,47
The nicotinergic receptor agonistic drugs have shown promise in the treatment of negative and cognitive symptoms in patients with schizophrenia. 48
Histamine receptors
The histaminergic receptor system consists of four receptors labeled H1 to H4 with the H4 receptor primarily located peripherally. 49 Both the atypical and typical antipsychotic drugs exert antagonistic properties on the histamine receptors, especially, olanzapine, quetiapine and clozapine. 50 The histaminergic receptor antagonism on the H1 receptor is involved in the changed feeding patterns, increased appetite and decreased satiety, mediating the antipsychotic drug induced weight gain.33,49,50 Sedation and decreased arousal are also linked to the H1 receptor and, moreover, sedation decreases cognitive functioning. As a consequence, histamine receptor agonists are targets for future cognitive enhancing drugs.49,51
Treatment with Antipsychotics
Efficacy vs. effectiveness
Two concepts are important when discussing the effects of antipsychotic drugs: Efficacy and effectiveness. Efficacy is defined as “the ability to produce the desired beneficial effect in expert hands and under ideal circumstances” and is derived from double-blinded randomized clinical trials. In these trials, rating scales for measuring psychopathology and side-effects are used. In- and exclusion criteria for these trials are often very stringent, and participants eligible for these trials often differ from the patients everyday clinical psychiatrists treat as substance abuse, somatic comorbidity, compulsory measures, etc are not allowed. 52
Effectiveness is defined as “the ability of an intervention to produce the desired beneficial effect in actual use” and consist of four domains: Efficacy, tolerability and safety, function and acceptability. 53 An example of effectiveness as outcome measure is time to any cause of discontinuation as used in the CATIE trial that compared several atypical antipsychotic drugs and one typical antipsychotic drug. 8 This outcome measure might treat some drugs unfairly because patients are more likely to drop-out early due to acute side-effects such as EPS or sedation whereas e.g. weight gain occur over time and is less likely to cause early discontinuation.
Meta-analysis suggests that some of the atypical antipsychotic drugs are more efficacious than others.13, 54 Besides clozapine, olanzapine, amisulpride, risperidone and zotepine seem to be more efficacious than typical and other atypical antipsychotic drugs. 13
Clozapine has shown superiority in treatment-resistant patients, but this unique effect has not been found in non-treatment-resistant patients. 55
Clinical recommendations
Antipsychotic drugs should ideally not be used until a proper physical examination has been performed, but this might not be possible due to lack of cooperation from the patient. For the typical antipsychotics, the dose-response relationship seems to be an inverted u-curve, 56 suggesting excessive high dosages cause side-effects such as sedation and EPS which often are misinterpreted as negative symptoms. Similar is not found with the atypical antipsychotics which might be due to lesser tendency to cause EPS. 57 Therefore, patients should always receive the lowest effective dosage in order to get the optimum treatment of their symptoms with as few side-effects as possible. The optimum dosage is differentiated with higher dosage in the acute phase, and after stabilization an effort to reduce the dosage in the maintenance phase should be done. Today, atypical antipsychotic drugs are considered first line drugs and typical antipsychotic drugs should be reserved for patients not responding to atypical antipsychotic drugs due to an increased risk of developing TD. 58 Even though, meta-analysis support some difference in efficacy between the atypical antipsychotic drugs, so far clinical response has been highly individual and unpredictable 59 suggesting that choice of antipsychotic drugs should be based on prior response to treatment, side-effect profile and the patient's preference. In case of non-or partial response more efficacious drugs, even with a less tolerable metabolic profile, should be used. Clozapine should be reserved for patients not responding to at least two antipsychotic drugs, or patients with schizophrenia having tardive dyskinesia or severe suicidal ideations. 60 In cases of persistent aggression in schizophrenia clozapine might be of value due to a special antiaggressive effect. 61
Depot formulations of antipsychotic drugs
Antipsychotic drug treatment is associated with a lower risk of relapse compared to placebo treatment, 62 but non-adherence to treatment is common when treating patients with schizophrenia. 63 Reasons for non-adherence are many, but cognitive difficulties or lack of a daily routine for medication are common.30,64 The clinical assessment of adherence is difficult and sometimes inaccurate, which can delay intervention. 63 When using long-acting injectable antipsychotic drugs adherence to treatment is known, and it is possible to intervene at an earlier stage, if the patient discontinues treatment. 64 The assortment of atypical long-acting injectable antipsychotic drugs is increasing, as drugs are in pipeline for marketing.
Time to onset of antipsychotic drug effect
It has been assumed that the antipsychotic effect occurred after several weeks of treatment. 65 The delay in treatment response was explained by the “depolarization block” theory; this theory arose from animal studies that showed continued firing from the dopamine neurons for three weeks after the antipsychotic drug treatment was initiated.66,67 Newer studies have shown an earlier effect,68–70 some with effect in the initial 24 hours. 68 Other studies have shown the greatest improvement in psychotic symptoms within the first two weeks compared to every two week period afterwards. 65 There is an important distinction to be made between onset of antipsychotic drug effect and full clinical effect of the drug. There is no doubt that time to full clinical effect is delayed weeks to months, but the initial effect occurs earlier, and is distinguishable from a pure sedative effect.65,68 Agitation in the acute phase of a psychotic breakthrough is common, and treatment thereof by either antipsychotic drugs or tranquilizers is important to minimize distress for the patient. 71
Switching Antipsychotic Drug Treatment
Switching antipsychotic drug treatment is common, e.g. shown in the CATIE trial, where all cause of discontinuation was the primary outcome. 8 The study was conducted under circumstances that resembled the everyday clinic, and showed that 74% of participants changed medication within 18 months. 8
The most common reasons for switching antipsychotic drug treatment are lack of efficacy or side-effects. Before switching a balance between the anticipated effects and side-effects should always be thoroughly discussed with the patient, 72 and the following should be considered: Method of switch, pharmacokinetics of the antipsychotic drugs, risks during change and adverse events in conjunction with switching treatment.
Switching can be done either abruptly by discontinuing the prior treatment and initiating the new treatment at the same time; gradual switch by tapering off prior drug before initiating the new drug or cross tapering by tapering off prior drug while initiating the new drug. No evidence supports using any specific method of switching. 73
Pharmacokinetic considerations
The half life of the drugs used in the switch is important for timing of the switch. If switching from a drug with a long half life, e.g. depot formulation or aripiprazole, a slow down tapering is not necessary, as the concentration decreases slowly. In contrast, e.g. quetiapine has a short half life, and therefore the new drug should be initiated and titrated up, before lowering the dosage of quetiapine. 74
Below are the different important receptor systems discussed in relation to switch of antipsychotic drug therapy. The general description of the receptor systems can be found in the mechanism of action paragraph.
Dopaminergic considerations
Switch from a drug with a high affinity for the dopaminergic receptor to a drug with a lower affinity encompasses a risk of rebound psychosis. 75 This is probably caused by dopamine hypersensitivity in the mesolimbic system where physiological levels of dopamine can cause psychosis because of lower dopaminergic antagonism. There is also a risk of rebound tardive dyskinesia that has been hidden by EPS on the previous drug, or caused by the hypersensitivity of dopamine receptors. 75 The occurrence of switch emergent EPS can be seen when switching from a drug with a low affinity for the dopamine receptor to a drug with a higher affinity.
Histaminergic considerations
The main concern when switching between drugs with different affinities for the histaminergic receptor is sedation or lack hereof. 75 Switching from a drug with high histaminergic receptor affinity to a drug with lower affinity, symptoms of histaminergic rebound can occur, e.g. insomnia. The opposite switch can result in massive sedation, and possibly lower adherence to treatment, if the patient isn't informed of the effect. Rebound insomnia usually resolves with time, but can be minimized with a slow cross tapering, or with a short period of treatment with tranquilizers, e.g. benzodiazepines. 75
Muscarinergic considerations
There is risk of muscarinergic rebound when switching from a drug with a high anticholinergic effect to a drug with a lower or no antagonistic effect on the muscarine receptor. The symptoms are nausea, vomiting, diaphoresis and sometimes insomnia. 75 Slow tapering off the medication usually resolves the problem, but when an abrupt discontinuation is necessary, e.g. agranulocytosis on clozapine treatment, substitution with anticholinergic drugs can minimize rebound symptoms. 74
Conclusions on switching
Ideally before initiating a change in antipsychotic drug treatment, the patient should be treated with the initial antipsychotic drug in clinical relevant dosages and for sufficient time to see improvement. The patient should in general be informed about the risks of adverse events and also the possible gains of a switch, as to give the patient adequate information to make an informed decision regarding treatment, but also to prepare the patient for possible adverse events.
The authors recommend the cross-titration method because this method probably has the lowest risk of relapse but the abrupt switch is necessary in cases of severe adverse events. The physician should consider the pharmacokinetics, receptor profiles of the drugs and reasons for changing medication before planning the switch. Atypical antipsychotic drugs receptor affinities and side-effect profiles can be seen in Figure 1.
Polypharmacy and Concomitant Medication
Little is known about the combination of antipsychotic drugs, and the use of concomitant medication such as benzodiazepines, antidepressant or anticonvulsants as described below. Antipsychotic polypharmacy is defined as the concurrent use of two or more antipsychotic drugs in a single patient.76,77 The use of antipsychotic polypharmacy is common even though the evidence is sparse and mostly indicates increased adverse effects, with no increased symptom control. 76
Polypharmacy is used in several other fields of medicine, e.g. treatment of hypertension, where drugs usually have different pharmacological mechanisms of action, e.g. beta blockers and thiazide diuretics for hypertension. The assumed dopamine D2 blocking mechanism behind antipsychotic drug action is universal to all antipsychotic drugs, although differences exist in receptor affinities for other receptors as described earlier. The theoretical reason for combining drugs to increase the antipsychotic effect is therefore limited. 77 Polypharmacy can be necessary in cases of, e.g. cross-titration and in the treatment of breakthrough psychosis. 76
If patients experiences antipsychotic drug induced hyperprolactinemia, and dose reduction or change of medication is impossible, concomitant treatment with aripiprazole should be considered as this has shown promise in reducing prolactin levels.78–80 The mechanism behind this is probably a dopaminergic agonistic effect on the pituitary gland by aripiprazole, a partial dopamine agonist.78,80,81
Studies combining antipsychotic drugs with each other have shown limited effect on the psychotic symptoms in general, and in some cases even increased the burden of side-effects. 77 The augmentation of antipsychotic drugs due to lack of efficacy should generally not be used until treatment with clozapine has been tried. 13
In cases of partial response to clozapine, augmentation of clozapine with anticonvulsants has shown some promise in improving psychotic symptoms,82,83 although the primary use of anticonvulsants in combination with clozapine is to lower risk of seizures during clozapine treatment. 84
The combination of clozapine and atypical antipsychotic drugs has not shown great promise in improving antipsychotic effects, although studies have been conducted on e.g. risperidone, amisulpride, and aripiprazole.85–90 A meta-analysis has shown a small but significant effect, but the clinical relevancy of this is doubtful. 91 The results of augmentation due to reduction of side-effects seem more promising.
The combination of antipsychotic drugs and benzodiazepines is used for treating anxiety and agitation even though evidence is sparse. 92 The difficulties in benzodiazepine drug treatment are well known, e.g. addiction, cognitive problems, sedation and withdrawal symptoms.92,93
Depression occurs in patients with schizophrenia, and treatment depends on the phase of the psychotic disease. The dose of antipsychotic drugs should be increased when dealing with psychotic patients. Antidepressants should be reserved for patients with psychotic symptoms under control. 94 One of the major problems with diagnosing depression in patients with schizophrenia is the existence of negative symptoms, e.g. anhedonia, decreased range of expressed emotion and lack of motivation, which can imitate depression. 95 The Calgary Depression Scale for Schizophrenia can help to diagnose depression in patients with schizophrenia differentiating depressive from negative symptoms. 95
Safety
Safety and side-effects
The side-effects of antipsychotic drugs are many but most side-effects can be linked to a single receptor whereas the mechanisms of others are more complex or remain unknown. Antipsychotic drugs are often used in patient populations where e.g. taking overdose, substance misuse and increased burden of cardiovascular risk factors are common. This increases the demands of monitoring safety when treating with antipsychotic drugs. Patients with schizophrenia have a reduced lifespan of approximately 15 years, even when controlling for suicide and accidental deaths, with cardiovascular disease as main cause of death. The exact contribution of medication remains unknown. 96
Cardiovascular and metabolic side-effects
From the very beginning of the antipsychotic era, antipsychotic drugs have been associated with sudden death. 97 Sudden death in otherwise healthy people is probably related to cardiac arrhythmia secondary to electrophysiological changes of ion channels in the heart. 98 Prolongation of the QT interval to more than 500 ms has been associated with an increased risk of developing the potential fatal cardiac arrhythmia Torsade de Pointes (TdP).99,100 However, this mechanism only accounts for a minor part of the increased mortality seen in patients with schizophrenia. 101 The antipsychotic-induced metabolic changes such as weight gain, increased risk of developing diabetes and dyslipidemia are far more important.102,103 Clozapine, olanzapine and quetiapine account for the antipsychotic drugs that cause the worst metabolic changes but results from studies in first-episode psychosis suggest that drugs prior considered as weight neutral such as ziprasidone are associated with a moderate weight gain.104,105 Most trials comparing antipsychotic drug effects on weight gain are limited by the fact that patients are receiving antipsychotic drugs at baseline, i.e. patients switched from a drug with high weight gain potentials to a drug with lower weight gain potentials would gain less weight or perhaps even lose weight on the new drug and vice versa. The mechanism of antipsychotic drug induced diabetes is probably related to decreased insulin sensitivity which is worsened further by weight gain and central adiposity. 106 For olanzapine and clozapine, a direct diabetogenic effect regardless of weight gain has been established. 102 Diabetic ketoacidosis can occur as an acute complication of antipsychotic treatment. 107 Olanzapine and clozapine seem to have the highest prevalence but most cases are reversible when discontinuing the offending drug. 108 Glycosylated hemoglobin (HbA1c) is often elevated in these patients, 107 and a decreased HDL and increased triglycerides are associated with an increased risk of insulin resistance. 38 It should be mentioned that estimating the contribution of the drug to metabolic status might be difficult due to other factors such as a sedentary lifestyle. 109
Dyslipidemia increases the risk of cardiovascular disease due to arteriosclerosis. Atypical antipsychotic drugs affect both triglyceride and cholesterols. 38 Results from the CATIE trials suggest that of the atypical antipsychotic drugs clozapine, olanzapine and quetiapine affect triglyceride and cholesterols the most. 8 Patients with antipsychotic-induced dyslipidemia might benefit from life-style interventions or switch to a more metabolic tolerable antipsychotic drug, but in many cases treatment with statins is necessary.
Several antipsychotic drugs function as antagonists at the α1 noradrenergic receptors causing venous vasodilatation and a drop in blood pressure. To maintain the same cardiac output, an increase in the heart rate, called reflex tachycardia, occurs. 110 Drugs with potent α1 antagonism cause orthostatic hypotension but tolerance to this side-effect usually develops after a few weeks and can be minimized by up-titrating the dose slowly. Especially, elderly and pregnant women are vulnerable to α1 induced side-effects. Increases in heart rate during treatment with antipsychotic drugs can also occur due to blocking of the cholinergic receptors. 110
Extrapyramidal side-effects
Extrapyramidal side-effects (EPS) occur due to blocking of more than 75% of the dopamine receptors in the striatum as described in the mechanism of action paragraph. Atypical antipsychotic drugs cause less EPS than typical antipsychotic drugs but a dose-dependent relationship exist meaning that virtually all antipsychotic drugs are capable of causing EPS, even the atypical antipsychotic drugs. 111 Clozapine and quetiapine have lower affinities for the dopamine receptors resulting in a rare association with EPS. 112 Monitoring of EPS is important because EPS has been associated with poor compliance, reduced quality of life, increased suicide rate and avoiding acute EPS symptoms decreases the risk of tardive dyskinesia. 113 EPS consists of the four following domains: Parkinsonism, dystonia, akathisia and dyskinesia. 113
The annual risk for developing tardive dyskinesia is around 5% for the typical antipsychotic drugs and around 1% for the atypical antipsychotic drugs. 58
Seizures
Antipsychotic drugs lower the seizure threshold which should be taken into account in patients at high risk for seizures, e.g. alcohol withdrawal and epilepsy. 120 Especially higher plasma levels of clozapine are associated with seizures whereas risperidone seems to be safer.121,122 The mechanism for antipsychotic drug induced seizures remains speculative, but involvement of muscarine and histamine receptors has been suggested. 120
Hyperprolactinemia
Secretion of prolactin from the pituitary gland is inhibited by dopamine, and due to the dopamine receptor blocking properties of antipsychotic drugs hyperprolactinemia can occur. The symptoms are amenorrhea, gynecomastia, galactorrhea, sexual dysfunction and osteoporosis. Symptoms of hyperprolactinemia should always be investigated and a probable cause found.
Increased prolactin levels have been associated with an increase in the risk of breast cancer. 123 The lipid solubility of antipsychotic drugs is important as high solubility facilitates easier diffusion over the blood-brain barrier. Antipsychotic drugs with low lipid solubility are given in higher dosages to reach clinical relevant intrathecal concentrations, and as the pituitary gland is located outside the blood-brain barrier higher concentrations of dopaminergic antagonistic drugs results in higher risk for hyperprolactinemia. 124 Especially risperidone and amisulpride are associated with symptomatic hyperprolactinemia due to their low lipid solubility and high dopamine affinity,125,126 but paliperidone has also been implicated, but is not as extensively studied yet. 127 Quetiapine and clozapine are not associated with hyperprolactinemia. 124 Aripiprazole has due to the high affinity for dopamine receptors and the partial dopamine agonist mechanism the ability to reverse antipsychotic induced hyperprolactinemia.78,128
Sedation
Several receptors e.g. muscarine M1, noradrenergic α-1, serotonin 5-HT2A/C, GABAA and histamine are involved in causing sedation. 129 However, antipsychotic drug sedation is mainly due to an antagonistic effect on the histamine receptor. 129 Antagonism at the dopamine receptors can elicit anhedonia and lack of motivation, mostly due to lack of reward and drive, but clinically it can be difficult to distinguish from sedation induced by antihistamine effects. 130 FDA gave a black box warning for parenteral olanzapine in combination with benzodiazepines due to deaths probably caused by excessive sedation. 131 Same precautions should be taken during titration of clozapine due to its hypotensive and sedative properties, or other drugs with similar receptor profiles. 132
Neuroleptic malignant syndrome
All antipsychotic drugs are capable of inducing neuroleptic malignant syndrome (NMS) which is a potential fatal complication. The syndrome consists of four symptoms: Hyperpyrexia, EPS, altered mental state and autonomic instability, but not all are necessarily present. 133 Especially for NMS involving atypical antipsychotics, EPS is not always present. 134 Laboratory findings include elevated creatinine kinase, leucocytosis and arterial blood gas acidosis. The exact mechanism for NMS is unknown but dopamine blockade is undoubtedly involved. 135 In case of NMS all antipsychotics drugs should be discontinued and in more severe cases treatment with bromocriptine and dantrolene is indicated. 135 Benzodiazepines can be used for treatment of mild to moderate cases, and in controlling agitation. ECT is also effective in treating NMS. 135
Serious clozapine related side-effect
Clozapine is associated with blood dyscrasias; approximately 0.8% develops agranulocytosis and 3% neutropenia which elucidate the need for hematological monitoring. 136 Most cases of agranulocytosis occur during the first six months of treatment and after a year the risk is similar to treatment with phenothiazines. 136 Olanzapine and quetiapine have also been associated with neutropenia, but do not seem to cause agranulocytosis.137,138
Besides the blood dyscrasias, clozapine is also associated with myocarditis and tachycardia. Myocarditis develops during the first months and symptoms are: Fever, tachycardia, dyspnea and chest pain. ECG might show ST-elevation, but the best way to diagnose myocarditis is to do troponin levels. 139 Cardiomyopathy is a long-term side-effect which seldom occurs until after six months of treatment and common symptoms are: Peripheral edema, tachycardia, exertional dyspnea and fatigue. Cardiomyopathy is detected through ECG changes and clinical symptoms, but should be confirmed by echocardiography. 140
Therapeutic drug monitoring (TDM) is recommended during clozapine treatment due to the large inter-individual variation in plasma levels and because a therapeutic threshold at 350-420 μg/L has been found. TDM in general has only a minor role in dose adjustment of antipsychotic drugs, because no concentration-response relationship has been found for most antipsychotic drugs. 141
Cognitive side-effects
Cognitive deficits are seen in patients with schizophrenia before the onset of psychosis and initiation of treatment,142,143 and can be linked to functional outcome, e.g. employment and compliance to treatment.30,144 These deficits can be exacerbated or improved by pharmacological interventions. Anticholinergic effect, either administered as an anticholinergic drug, e.g. biperiden, or as antagonistic effect on the muscarinergic receptors decreases cognitive function, especially memory and attention. 46
Sedation reduces the general arousal level, and a decreased or increased level of arousal can probably impair cognitive functioning as suggested in the Yerkes-Dodson law, which means that sedation, e.g. histaminergic antagonism, can improve cognition in hyperaroused patients and decrease cognitive functioning in others. 51
Other concomitant treatments can influence cognitive functioning, especially treatment with benzodiazepines. This has not yet been tested in patients with schizophrenia, but cognitive testing on patients with other chronic psychiatric disorders has shown significant decreases in cognitive functioning compared to controls, and a significant improvement after discontinuation.93,145,146
Atypical antipsychotic drugs in comparison with typical antipsychotic drugs show a slight advantage in cognitive improvement in favor of the atypical antipsychotic drugs, even though this finding is questioned by suboptimal study designs in studies of typical versus atypical antipsychotic drug effects on cognitive functioning. 147 This is further questioned by findings indicating that the difference between atypical and typical antipsychotic drugs is non-existing when treating with optimal dosages of both drugs.147,148
Metabolism of antipsychotic drugs
The most important metabolic pathway for most antipsychotic drugs is the cytochrome P450 (CYP450) system 149 that metabolizes drugs before they can be excreted through the kidneys, but other metabolic pathways like the aldehyde oxidases that is involved in the metabolism of ziprasidone exist. 149
The CYP450 system consists of over 50 different enzymes that metabolize a large number of compounds which allows the body to function under different conditions and adapt to new expositions. 150
The CYP450 enzymes primarily responsible for the metabolism of antipsychotic drugs are CYP1A2, CYP3A4 and CYP2D6.149,151,152 Most antipsychotic drugs are metabolized by several CYP450 enzymes, 151 but an inhibition or induction of the primary CYP450 enzyme can have clinical implications for the individual patient depending on the width of the therapeutic index of the involved drugs, the existence of additional metabolic pathways, patient's sensitivity to desired and adverse effects and the strength of the inhibition or induction. 149 In Figure 2, an overview of the most important substrates, inhibitors and inductors are depicted. Induction of the CYP enzymes is a process that can take up to several weeks before maximum effect occurs, as it is caused by an increased production of CYP450 enzymes. In contrast, inhibition occurs immediately and reaches maximum effect when the inhibiting drug reaches steady state. 149 As induction is caused by an increase in the CYP450 enzyme, the effect is also prolonged after the offending drug is discontinued, until the CYP450 enzyme again has reached its habitual activity. 149
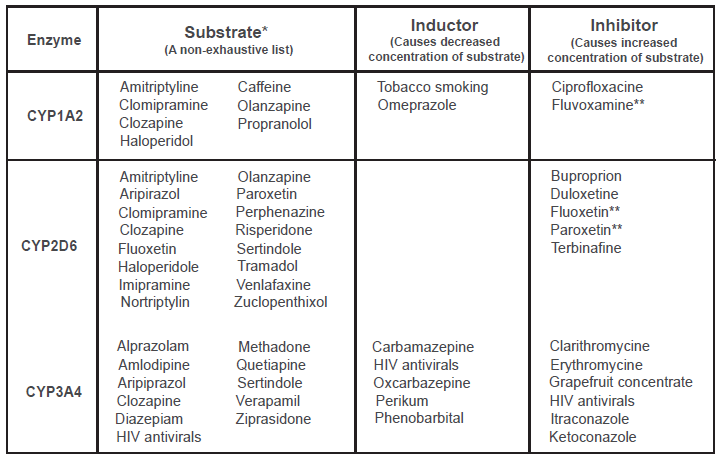
Amisulpride and paliperidone are not metabolized in the liver,149,152,153 but are excreted directly through the kidneys unchanged. This knowledge can be used when treating patients with severe liver disease. 152
Patient Preference
Treatment of patients with psychotic illness should not only focus on ameliorating psychotic symptoms as measured by scales, but a more holistic approach including the patient's subjective experience with the treatment, e.g. desired effects, side-effects, cultural acceptance of treatment, and pharmacological versus non-pharmacological treatment should be applied. 154 In the recent years, a switch from compliance to adherence in terminology and practice has occurred; the former is the physician taking the major role in deciding treatment and the latter is a patient-physician collaboration concerning treatment.154,155
Decision to prescribe a drug is a balance between perceived positive effects on one side and adverse effects on the other side. This should be discussed with the patient. The insight and awareness of symptoms and the understanding of treatment needs increase adherence whereas negative symptoms, cognitive deficits and drug misuse decrease adherence. 154 Insight and negative symptoms can be influenced by information regarding the disease. A continued discussion of pros and cons of treatment during the entire course of the illness is important. 1
The most common reported dissatisfactions with treatment are side-effects, lack of involvement in treatment planning, lack of information about effects and side-effects and lack of involvement of family members. 154 The attention on side-effects is important during any treatment and of course also during treatment with antipsychotic drugs. The method of inquiring about side-effects is of significant importance. 156 When using a standardized questionnaire with open questions compared to closed questions, a higher positive answer rate was found. 156
Not all side-effects are equally distressing to the patient, and the differences are also seen in between genders. In general, weight gain, depression, and insomnia are more distressing than cognitive side effects, sedation and sexual side-effects. 154 Females found weight gain more distressing than males, and males found sexual side-effects more distressing than females. 154 It is the patient's subjective feeling of effect compared to the feeling of adverse effects that tip towards either side resulting in better or worse adherence to treatment. 157
Adherence to treatment is important; it lays the foundation for clinical effectiveness. 154 It is of crucial importance that the patient's subjective feelings of desired effect and side-effects on treatment is noted and evaluated early and throughout the course of the illness as these are associated with adherence.154,156 This is important as low adherence is associated with risk of relapse, risk of admission, longer duration of admission and increased risk of suicide.31,158 Good adherence to treatment is in contrast associated with a better functional outcome for the patient. 159 The physician-rated symptom scales can be useful in the evaluating progress regarding symptoms, but must never stand alone in the clinical practice.
Conclusions
Advances in treatment of psychosis have occurred over the years including a plethora of newer pharmacological interventions. These have come with new challenges from side-effects, demands for monitoring and safety measures. Up to date psychopharmacological knowledge is therefore essential for the physician. There has been a shift towards greater patient involvement in treatment decisions, and there is increased focus on the patient's subjective feelings regarding treatment effects and side-effects, hopefully resulting in better attitudes towards and greater adherence to medication.
Disclosures
The authors report no conflicts of interest.
